b Division of Functional Materials and Nano-Devices, Ningbo Institute of MaterialTechnology and Engineering, Chinese Academy of Science, Ningbo 315201, China ;
c University of Chinese Academy of Sciences, Beijing 100039, ChinaUniversity of Chinese Academy of Sciences, Beijing 100039, China
With the advent of more miniaturized wearable/portable electronics, micro-electro mechanical systems (MEMS), epidermal and implantable medical devices, and wireless sensor networks, it is essential to develop high-performance miniaturized and flexible electrochemical storage devices able to match these electronic components [1-3]. Because of the advantages of supercapacitors (SCs) such as high output power capability, fast chargingdischarging kinetics, long cycling life and good safety performance [4-7], micro supercapacitors (MSCs) are considered to be promising candidates for energy storage toward micro-scale, flexible, light-weight, sustainable and renewable power sources to build self-powered micro/nanodevices and systems [8-10]. Moreover, on-chip MSCs in an interdigitated planar form can shorten the ion diffusion length and decrease the resistance of electrolytes, resulting in a high power density, a fast charge/discharge rate and high-frequency response capability, which could be applied in some special applications, such as for ac line-filtering [11, 12]. Flexible all-solid-state MSCs without using a liquid electrolyte can be directly employed as embedded energy-storage devices for portable and wearable microelectronics systems [13, 14]. In order to fully meet the growing market demands toward fast-response, compact design and integration with other devices, it is necessary to exploit new electrode materials of MSCs with higher volumetric capacitance and fast frequency response capability, regardless of the device architecture and electrolyte selection [8, 15].
MXenes (Mn+1XnTx:M = Ti, V, Nb, etc.; X = C, N; n = 1-3; Tx is the functional termination group), a novel family of two-dimensional (2D) metal carbides, which can be prepared by the selective removal of the atomically thin aluminum layers from the ternary transition metal carbide (MAX phase) structure [16], have already proved to be promising candidates for electrode materials in Li-ion batteries [17, 18], Na-ion batteries [19, 20], Na-ion hybrid capacitors [21] and SCs [22, 23] with volumetric capacitance exceeding most of carbon materials. In addition, MXenes have been recognized as one of the efficient electrode materials because the flexible interlayer space and large interstitial space can accommodate more and ions at high charge/discharge rate, which would be beneficial to demonstrate good rate and frequency response capability [24, 25]. Moreover, the MXene film obtained by vacuum-assisted filtration or DC magnetron sputtering could further extend the application scope, such as applying as transparent conductive films [26, 27]. However, to our best knowledge, the MXene film used in fabricating MSCs is not reported to date.
In this work, the MXene films were firstly produced through vacuum-filtrating and subsequent controlled transferring onto various substrates including polyethylene terephthalate (PET), silicon oxide film and titanium plate. And the MXene film in 1 mol L-1 H2SO4 under a three-electrode system demonstrates a good rate and fast charge/discharge capabilities. On this basis, we construct MXene-based flexible all-solid-state symmetric MSCs on PET substrate by micro-fabrication process using polyvinyl alcohol (PVA)/H2SO4 as electrolyte for the first time. As a consequence, the MSC exhibits an excellent rate performance with the scan rate of up to 1000 Vs-1 as well as the fast frequency response (t0 = 0.5 ms), and provides a large volumetric capacitance of 1.44 F cm-3 at the current density of 0.288 A cm-3.
2. Experimental 2.1. Synthesis of Ti3C2Tx MXeneThe MAX phase used as precursor for MXene synthesis herein— Ti3AlC2—was prepared according to the reported method [22]. After that, concentrated HCl was diluted with distilled water (18 MΩ cm-1) to obtain a 6 mol L-1 solution (30 mL total), then 1.98 g of LiF was added to this solution. And the mixture was stirred for more than 5 min to dissolve thoroughly the salt. Subsequently, 1 g of Ti3AlC2powderswas addedslowly into themixture solutionto avoid overheating. The exfoliating processwas conducted at 40 ℃ for45 h, after which the mixture was washed through at least 5 cycles of distilled water addition, centrifugation (3500 rpm, 5min for each cycle), and decanting, until the supernatant reached a pH of approximately 6. To obtain the exfoliated Ti3C2Tx nanosheets, asmade Ti3C2Tx flakes were dispersed in distilled water (0.4 g MXene per 0.1 L of water) and deaerated with Ar. Then the mixture was sonicated for 1 h and followed by centrifuge (3500 rpmfor 1 h) and the supernatantwas collected. The concentration of the supernatant of exfoliated MXene was about 2 mg mL-1.
2.2. Synthesis of MXene films5 mL of as-made supernatant dispersion was added to water to dilute to 50 mL, and then take 2 mL of diluted dispersion out in order to obtain the MXene film. The films were produced similarly as our previously report [28]. Briefly described as follows: the suspension was vacuum-filtrated using a Millipore filter (50 mmin diameter and 0.45 μm in pore size). The membrane with captured hybrid film was then cut into size of choice, wetted with deionized water, and transferred onto PET, silicon oxide film and titanium plate substrates, respectively, using isopropanol to remove trapped air between the MXene film and the substrate. After that, the cellulose filter membrane was dissolved using acetone (successively pure baths) to leave a hybrid thin film on these substrates. The film was then rinsed with methanol and dried at room temperature.
2.3. Characterization and electrochemical measurementsField-emission scanning electron microscope (SEM; JSM- 6701F), Transmission electron microscope (TEM; JEOL 2100 FEG), X-ray photoelectron spectrometer (ESCALAB250xi) and X-ray diffraction (XRD; TTR-III) were employed to characterize the electrode materials.
Electrochemical properties of MXene films were carried out in the three electrode system in 1 mol L-1 H2SO4 electrolyte, and a platinum sheet and an Ag/AgCl electrode were used as the counter and the reference electrodes, respectively. Electrochemical tests of MSCs were performed in PVA-H2SO4 gel-electrolyte. The PVA- H2SO4 gel-electrolyte was prepared by the following steps: 6 g of H2SO4 was added to 60 mL DI water, and then 6 g of PVA powder was added. The resulting mixture was heated to 85 ℃ for 2 h under vigorous stirring. Electrochemical tests of SCs, including cyclic voltammetry (CV), galvanostatic charge/discharge (GCD), and electrochemical impedance spectroscopy (EIS) were carried out using an electrochemical workstation (CHI660D, Shanghai, China).
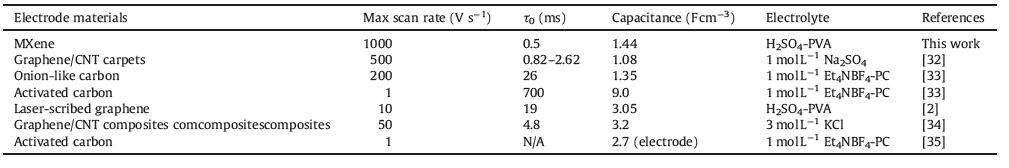
The areal capacitance (CA) and volumetric capacitance (Cv) were calculated from the GCD curves based on Eq. (1):
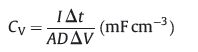
|
(1) |
The volumetric energy density (E) and power density (P) were defined according to Eqs. (2) and (3), respectively:
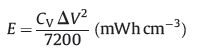
|
(2) |
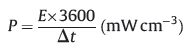
|
(3) |
where I is the current density (mA), Δt is the discharge time (s), ΔV is the potential window of the discharging (V), A is the geometric surface of MSC, D is the thickness (cm) of MXene film (520 nm).
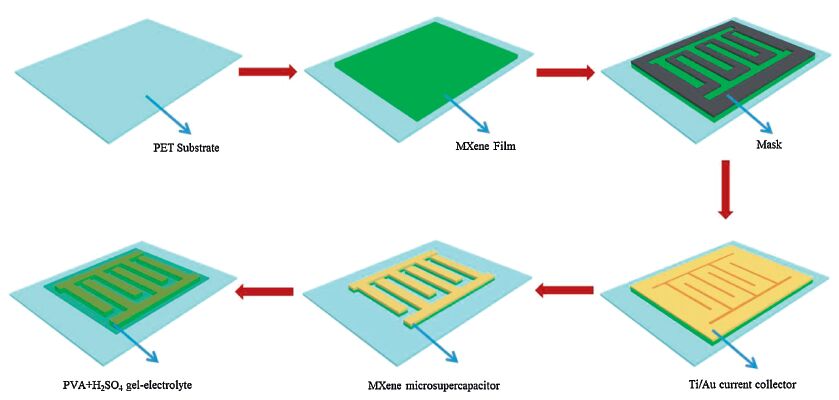
3. Results and discussionThe preparation process of on-chip MSCs with MXene films was illustrated in Fig. 1. Firstly, the clean PET film with 200 μm thick was employed as the flexible substrate. Subsequently, the MXene films were fabricated through vacuum-filtrating and then transferred onto PET substrate. The interdigital finger mask was placed on the top of MXene film, and Ti/Au (20/50 nm) layer acted as the current collector with an interdigitated pattern was deposited using film-plating machines (ZHD-3″, Beijing Technol Science Co., ). Thereafter, the patterns of the MXene microelectrodes on PET substrate were created by oxidative etching of the exposed MXene film in H2O2 diluted solution (10 wt%) for 10 s, and then rinsed with deionized water. Finally, the PVA + H2SO4 gel-electrolyte was coated on the microelectrodes to form a flexible all-solid-state MSC. The MSC was made up of 32 in-plane interdigital microelectrodes (16 positive and 16 negative microelectrodes). Each microelectrode was 200 μm in width and 10 mm in length, and the distance between adjacent microelectrodes was 200 μm, and the overall dimension of microelectrode was 1 cm × 2 cm. In our system, the apparent surface area of the electrode was calculated to be 0.35 cm2.
|
Download:
|
| Figure 1. Schematic illustration of the fabrication process of on-chip MSCs with MXene films. | |
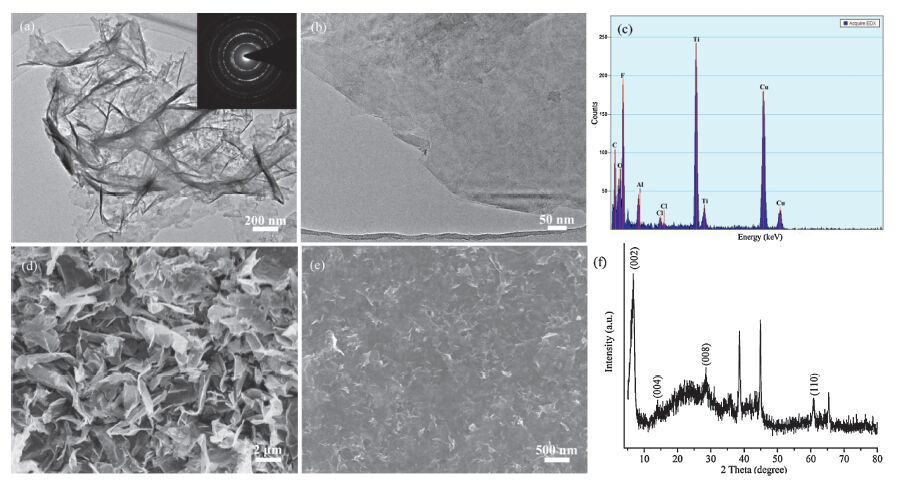
The morphology and structure characterizations of exfoliated MXene were shown in Fig. 2. TEM and SEM images show asprepared exfoliated MXene presenting ultrathin nanosheets with a lateral dimension of 1-5 mm, and the 2D MXene nanosheets were beneficial to electron transport. The selected area electron diffraction (SAED) pattern of the exfoliated MXene indicated a typical polycrystalline nature (inset of Fig. 2a). The energydispersive X-ray (EDX) spectrum of detected Ti, C, O, F and Al originating from the exfoliated MXene, and the Cl element could be attributed to the contamination introduced during the preparation or exfoliation process. The MXene film had a homogeneously and flat surface as shown in Fig. 2e, which was conducive to constructing the microelectrodes through up-bottom approach. The XRD analysis (Fig. 2f) also verified the structure of exfoliated MXene, corresponding to the (0 0 2), (0 0 4), (0 0 8) and (1 1 0) crystal faces at around 6.6°, 13.8°, 28.3° and 60.5°, respectively, and the rest diffraction peaks can be ascribed to Ti3AlC2, that was the precursor of MXene.
|
Download:
|
| Figure 2. (a, b) TEM images of exfoliated MXene with low and high magnifications. Inset is the corresponding SAED pattern. (c) EDX spectrum of exfoliated MXene, and the Cu element originated from copper mesh. (d) SEM image of exfoliated MXene. (e) SEM image of MXene film on the PET substrate. (f) XRD pattern of exfoliated MXene. | |
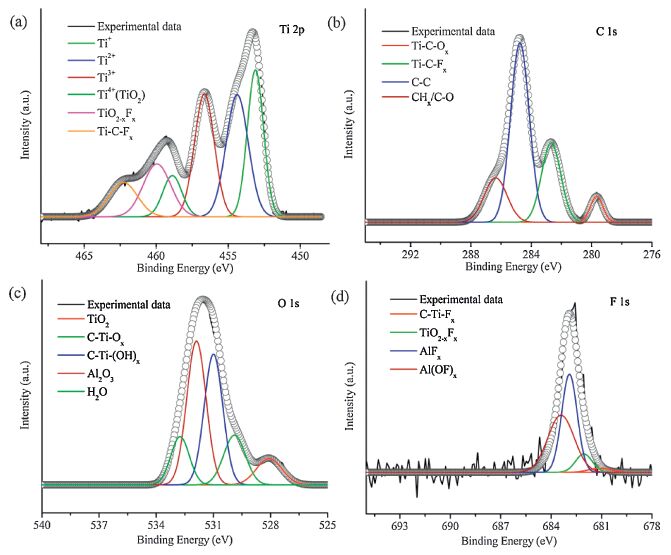
XPS investigations were conducted to reveal the functional groups and chemical environment on the surface of the samples. As can be seen from Fig. 3a, the Ti 2p spectrum of MXene film can be fitted into six main constituent peaks, namely four different valence states bond of Ti element, TiO2-xFx bond at 460.1 eV and Ti-C-Fx bond at 462.3 eV, respectively [29]. And the presence of Ti- C and Ti-O bonds indicated the formation of Ti3C2(OH)2 [30]. The C 1s region (Fig. 3b) of as-made MXene film was fit by four peaks. The peaks at 279.7, 282.7, 284.8 and 286.4 eV corresponded to Ti-C-O, Ti-C-Fx, C-C and CHx/C-O bonds, respectively [31]. The well-fitted regions of O 1s and F 1s also certified the stability of the composition when producing the MXene films except for containing some precursor Ti3AlC2 in the films according to Fig. 3c and d. Additionally, a large number of oxygen-containing functional groups on the MXene film surfaces could facilitate the ions of the electrolyte to fast immerse and diffuse into the electrode bulk.
|
Download:
|
| Figure 3. (a) Ti 2p, (b) C 1s, (c) O 1s and (d) F 1s XPS spectra of MXene film (black solid lines are experimental data, the dark gray hollow lines are fitted results). | |
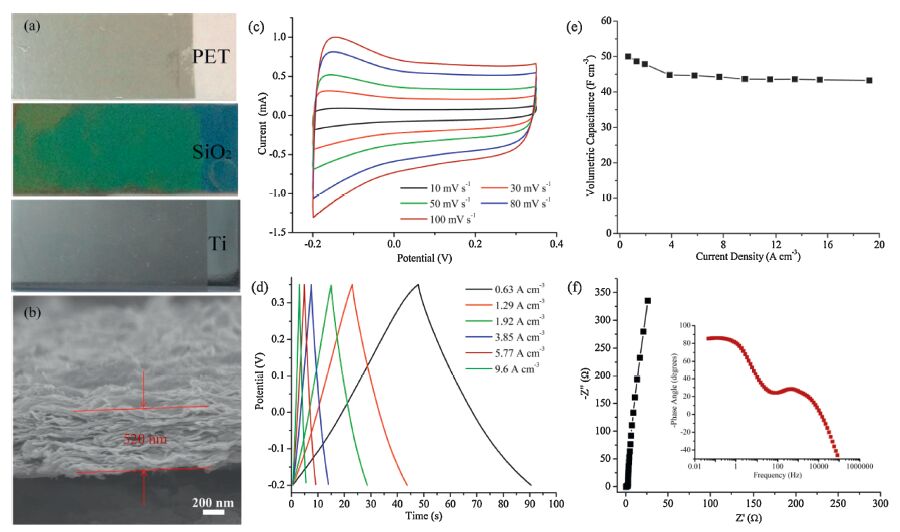
As shown in Fig. 4a, the MXene films prepared by vacuumfiltrating could be transferred onto various flat substrates, such as PET, silicon oxide film and titanium plate with the size dimension of 1 cm - 3 cm, which would expand the application field of MXene films, for example, the MXene films on silicon oxide substrate could construct integrated circuit a chip when integrating the MCSs with silicon-based electronics. The thickness of the MXene film was 520 nm on the basis of the cross section SEM image (Fig. 4b). The electrochemical properties of the MXene film were evaluated in 1 mol L-1 H2SO4 prior to investigating electrochemical performance of the MXene-based MSCs. The CV curves (Fig. 4c) of the MXene film electrode exhibited a typical rectangular shape when the scan rate ranged from 10 mV s-1 to100 mV s-1, indicating fast charge/discharge capability and good diffusion of electrolytes in the electrode material. The GCD curves (Fig. 4d) presented a good reversibility of charge storage and small voltage drop, which could be attributed to high electric conductivity of MXene films [22]. As can be seen from Fig. 4e, the volumetric capacitance (49.6 F cm-3) of MXene film electrode at the current density of 0.63A cm-3 retained about 87% (43.2 F cm-3) when the current density was up to 19.2 A cm-3, suggesting a better rate capability. The EIS plot was almost perpendicular to the real axis in the low-frequency region (Fig. 4f), indicating an ideal capacitive behavior. And the equivalent series resistance (Rs) was about 0.5 Ω. The plot of impedance phase angle versus frequency demonstrated a fast frequency response characteristic.
|
Download:
|
| Figure 4. (a) The digital photographs of the MXene films were transferred onto various substrates including PET (upside), silicon oxide film (middle) and titanium plate (bottom).(b) The cross section SEM image of MXene film, and the thickness was 520 nm. (c, d) CV and GCD curves of MXene film on titanium plate using 1 mol L-1 H2SO4 as electrolyte in a three electrode system. (e) The volumetric capacitance as a function of the current density. (f) EIS plots measured at a frequency range of 0.05 kHz to 100 kHz, inset: plot of impedance phase angle versus frequency. | |
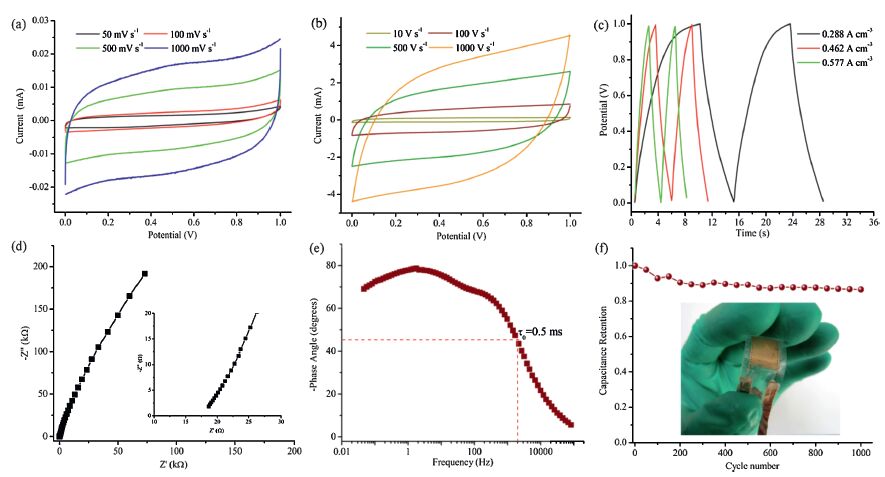
CV, GCD, EIS and cycling stability were recognized as the principal methods for evaluating the electrochemical performances of SCs. As shown in Fig. 5a (from 50 mV s-1 to 1000 mV s-1) and Fig. 5b (from 10 V s-1 to 1000 V s-1), the CV curves of the MXene-based symmetric MSC demonstrated a rapid current response on voltage reversal at the end potential, and had a nearly rectangular CV shape even at a very high scan rate of 1000 V s-1, indicating ultrafast charging and discharging capability and instantaneous power capability. It is important to note that a scan rate of 1000 V s-1 was among the highest value reported for MSCs. The volumetric capacitance of the MSC was 1.44 F cm-3 , with a volumetric energy density of 0.2 mWh cm-3 (with a specific power of 144mWcm-3 at this point) at the current density of 0.288 A cm-3 (Fig. 5c). And such volumetric capacitance value was substantially higher than that of reported MSC devices (Table 1), such as 3-dimensional graphene carbon nanotube carpet-based MSC (1.08 F cm-3 ) [32], onion-like carbon-based MSC (1.35 F cm-3) [33], and activated carbon electrode (2.7 F cm-3 ) [35]. As can be seen from Fig. 5d, the EIS plot did not show a semicircle at the high frequency region, suggesting an almost negligible charge-transfer resistance (Rct) owing to good electrical conductivity of MXene film and strong adhesion between the MXene electrode and the current collector. The characteristic frequency (f0) was the frequency at a phase angle of 458 at which the value of resistive was identical with the value of capacitive impedance. And the corresponding relaxation time constant (t0 = 1/f0) was the minimum time needed to discharge all the electrical energy from the SCs with an efficiency of greater than 50%. The characteristic frequency of the MXene-based MSC (Fig. 5e) was about 2000 Hz (relaxation time constant t0 = 0.5 ms), demonstrating ultrafast frequency response. And the relaxation time constant of 0.5 ms was much shorter than that of MSCs based on onion-like carbon (26 ms) and activated carbon (700 ms) [33], laser-scribed graphene (19 ms) [2], reduced graphene oxide and carbon nanotube composites (4.8 ms) [34] and electrochemically exfoliated graphene (1-2.9 ms) [12]. The possible reasons for the excellent CV performance and ultrafast frequency response of the MXene-based MSC were as follows. First, the design of interdigitated planar form can shorten the ion diffusion length and decrease the resistance of electrolytes. Second, MXene materials, serving as layered 2D metal carbides, were conductive to electron conduction and rapid diffusion of the electrolyte ions. Finally, a large number of oxygen-containing functional groups on the MXene film surfaces could facilitate the ions of the electrolyte to fast immerse and diffuse into the electrode bulk. Fig. 5f shows the cycle life of the MSC with a certain bending angle, finding that the flexible all-solid-state MSC revealed a better cycling stability with 87% capacitance retention after 1000 cycles and a stable electrochemical performance during bending.
|
|
Table 1 Comparison of the electrochemical performance of various MSCs built using different materials. |
|
Download:
|
| Figure 5. (a, b) CV curves of the MXene-based symmetric MSC on at different scan rates using PVA + H2SO4 as gel electrolyte. (c) GCD curves of the MXene-based symmetric MSC at different current densities. (d) EIS plot of the MXene-based symmetric MSC; inset showing enlarged plots in high-frequency region. (e) Corresponding plot of impedance phase angle versus frequency. (f) The cycling stability of the MXene-based symmetric MSC on the basis of CV curves measured at 100 mV s-1 when bending. | |
4. Conclusion
In summary, we firstly fabricate the MXene films on various substrates including flexible PET by vacuum-filtrating and subsequent controlled transferring. Based on this, MXene-based flexible all-solid-state symmetric MSCs on PET substrate are constructed through micro-fabrication process using polyvinyl alcohol (PVA)/ H2SO4 as electrolyte. As a result, the MSC demonstrates an outstanding rate performance with the scan rate of up to 1000 V s-1 as well as an ultrafast frequency response (t0 = 0.5 ms), and delivers a large volumetric capacitance of 1.44 F cm-3 and a high volumetric energy density (0.2 mWh cm-3) at the current density of 0.288 A cm-3 . In addition, the MSC exhibits a better cycling stability with 87% capacitance retention after 1000 cycles. And our research results presented here provide a rational design strategy for constructing with a high volumetric capacitance and fast frequency response capability.
AcknowledgmentThis work was supported by the National Nature Science Foundations of China (Nos. 21573265 and 51501208).
| [1] | P. Huang, C. Lethien, S. Pinaud, et al. On-chip and free standing elastic carbon films for micro-supercapacitors. Science 351 (2016) 691–695. DOI:10.1126/science.aad3345 |
| [2] | M.F. El-Kady, R.B. Kaner. Scalable fabrication of high-power graphene microsupercapacitors for flexible and on-chip energy storage. Nat. Commun. 4 (2013) 1475. DOI:10.1038/ncomms2446 |
| [3] | B.S. Shen, J.W. Lang, R.S. Guo, X. Zhang, X.B. Yan. Engineering the electrochemical capacitive properties of microsupercapacitors based on graphene quantum dots/MnO2 using ionic liquid gel electrolytes. ACS Appl. Mater. Interfaces 7 (2015) 25378–25389. DOI:10.1021/acsami.5b07909 |
| [4] | C. Zhong, Y.D. Deng, W.B. Hu, et al. A review of electrolyte materials and compositions for electrochemical supercapacitors. Chem. Soc. Rev 44 (2015) 7484–7539. DOI:10.1039/C5CS00303B |
| [5] | M.J. Shi, S.Z. Kou, B.S. Shen, et al. Improving the performance of all-solid-state supercapacitors by modifying ionic liquid gel electrolytes with graphene nanosheets prepared by arc-discharge. Chin. Chem. Lett. 25 (2014) 859–864. DOI:10.1016/j.cclet.2014.04.010 |
| [6] | L. Kang, S.X. Sun, L.B. Kong, J.W. Lang, Y.C. Luo. Investigating metal-organic framework as a new pseudo-capacitive material for supercapacitors. Chin. Chem. Lett. 25 (2014) 957–961. DOI:10.1016/j.cclet.2014.05.032 |
| [7] | J.J. Cai, L.B. Kong, J. Zhang, Y.C. Luo, L. Kang. A novel polyaniline/mesoporous carbon nano-composite electrode for asymmetric supercapacitor. Chin. Chem. Lett 21 (2012) 1509–1512. |
| [8] | Z.S. Wu, K. Parvez, S. Li, et al. Alternating stacked graphene-conducting polymer compact films with ultrahigh areal and volumetric capacitances for high-energy micro-supercapacitors. Adv. Mater. 27 (2015) 4054–4061. DOI:10.1002/adma.201501643 |
| [9] | W.W. Liu, X.B. Yan, J.T. Chen, Y.Q. Feng, Q.J. Xue. Novel and high-performance asymmetric micro-supercapacitors based on graphene quantum dots and polyaniline nanofibers. Nanoscale 5 (2013) 6053–6062. DOI:10.1039/c3nr01139a |
| [10] | W.W. Liu, C.X. Lu, H.L. Li, et al. Paper-based all-solid-state flexible micro-supercapacitors with ultra-high rate and rapid frequency response capabilities. J. Mater. Chem. A 4 (2016) 3754–3764. DOI:10.1039/C6TA00159A |
| [11] | W.W. Liu, Y.Q. Feng, X.B. Yan, J.T. Chen, Q.J. Xue. Superior micro-supercapacitors based on graphene quantum dots. Adv. Funct. Mater. 23 (2013) 4111–4122. DOI:10.1002/adfm.v23.33 |
| [12] | Z.S. Wu, Z.Y. Liu, K. Parvez, X.L. Feng, K. Mü llen. Ultrathin printable graphene supercapacitors with AC line-filtering performance. Adv. Mater. 27 (2015) 3669–3675. DOI:10.1002/adma.v27.24 |
| [13] | Z.Y. Liu, Z.S. Wu, S. Yang, et al. Ultraflexible in-plane micro-supercapacitors by direct printing of solution-processable electrochemically exfoliated graphene. Adv. Mater. 28 (2016) 2217–2222. DOI:10.1002/adma.201505304 |
| [14] | Z.Q. Niu, L. Zhang, L.L. Liu, et al. All-solid-state flexible ultrathin micro-supercapacitors based on graphene. Adv. Mater. 25 (2013) 4035–4042. DOI:10.1002/adma.v25.29 |
| [15] | W. Gao, N. Singh, L. Song, et al. Direct laser writing of micro-supercapacitors on hydrated graphite oxide films. Nat. Nanotechnol. 6 (2011) 496–500. DOI:10.1038/nnano.2011.110 |
| [16] | M.R. Lukatskaya, S.M. Bak, X.Q. Yu, et al. Probing the mechanism of high capacitance in 2D titanium carbide using in situ X-ray absorption spectroscopy. Adv. Energy Mater. 5 (2015) 1500589. DOI:10.1002/aenm.201500589 |
| [17] | M. Naguib, J. Halim, J. Lu, et al. New two-dimensional niobium and vanadium carbides as promising materials for Li-ion batteries. J. Am. Chem. Soc. 135 (2013) 15966–15969. DOI:10.1021/ja405735d |
| [18] | Z.Y. Lin, D.F. Sun, Q. Huang, et al. Carbon nanofiber bridged two-dimensional titanium carbide as a superior anode for lithium-ion batteries. J. Mater. Chem. A 3 (2015) 14096–14100. DOI:10.1039/C5TA01855B |
| [19] | D.Q. Er, J.W. Li, M. Naguib, Y. Gogotsi, V.B. Shenoy. Ti3C2 MXene as a high capacity electrode material for metal (Li, Na, K, Ca) ion batteries. ACS Appl. Mater. Interfaces 6 (2014) 11173–11179. DOI:10.1021/am501144q |
| [20] | Y. Dall'Agnese, P.L. Taberna, Y. Gogotsi, P. Simon. Two-dimensional vanadium carbide (MXene) as positive electrode for sodium-ion capacitors. J. Phys. Chem. Lett. 6 (2015) 2305–2309. DOI:10.1021/acs.jpclett.5b00868 |
| [21] | X.F. Wang, S. Kajiyama, H. Iinuma, et al. Pseudocapacitance of MXene nanosheets for high-power sodium-ion hybrid capacitors. Nat. Commun. 6 (2015) 6544. DOI:10.1038/ncomms7544 |
| [22] | M. Ghidiu, M.R. Lukatskaya, M.Q. Zhao, Y. Gogotsi, M.W. Barsoum. Conductive two-dimensional titanium carbide ‘clay’ with high volumetric capacitance. Nature 516 (2014) 78–81. |
| [23] | M.Q. Zhao, C.E. Ren, Z. Ling, et al. Flexible MXene/carbon nanotube composite paper with high volumetric capacitance. Adv. Mater. 27 (2015) 339–345. DOI:10.1002/adma.v27.2 |
| [24] | Y. Xie, Y. Dall'Agnese, M. Naguib, et al. Prediction and characterization of MXene nanosheet anodes for non-lithium-ion batteries. ACS Nano 8 (2014) 9606–9615. DOI:10.1021/nn503921j |
| [25] | S. Kajiyama, L. Szabova, K. Sodeyama, et al. Sodium-ion intercalation mechanism in MXene nanosheets. ACS Nano 10 (2016) 3334–3341. DOI:10.1021/acsnano.5b06958 |
| [26] | Z. Ling, C.E. Ren, M.Q. Zhao, et al. Flexible and conductive MXene films and nanocomposites with high capacitance. Proc. Natl. Acad. Sci. U.S.A. 111 (2014) 16676–16681. DOI:10.1073/pnas.1414215111 |
| [27] | J. Halim, M.R. Lukatskaya, K.M. Cook, et al. Transparent conductive two-dimensional titanium carbide epitaxial thin films. Chem. Mater. 26 (2014) 2374–2381. DOI:10.1021/cm500641a |
| [28] | X. Zhang, X.B. Yan, J.T. Chen, J.P. Zhao. Large-size graphene microsheets as a protective layer for transparent conductive silver nanowire film heaters. Carbon 69 (2014) 437–443. DOI:10.1016/j.carbon.2013.12.046 |
| [29] | J. Halim, K.M. Cook, M. Naguib, et al. X-ray photoelectron spectroscopy of select multi-layered transition metal carbides (MXenes). Appl. Surf. Sci 362 (2016) 406–417. DOI:10.1016/j.apsusc.2015.11.089 |
| [30] | M. Naguib, M. Kurtoglu, V. Presser, et al. Two-dimensional nanocrystals produced by exfoliation of Ti3AlC2. Adv. Mater. 23 (2011) 4248–4253. DOI:10.1002/adma.201102306 |
| [31] | S. Myhra, J.A.A. Crossley, M.W. Barsoum. Crystal-chemistry of the Ti3AlC2 and Ti4AlN3 layered carbide/nitride phases-characterization by XPS. J. Phys. Chem. Solids 62 (2001) 811–817. DOI:10.1016/S0022-3697(00)00268-7 |
| [32] | J. Lin, C.G. Zhang, Z. Yan, et al. 3-Dimensional graphene carbon nanotube carpetbased microsupercapacitors with high electrochemical performance. Nano Lett 13 (2013) 72–78. DOI:10.1021/nl3034976 |
| [33] | D. Pech, M. Brunet, H. Durou, et al. Ultrahigh-power micrometre-sized supercapacitors based on onion-like carbon. Nat. Nanotechnol. 5 (2010) 651–654. DOI:10.1038/nnano.2010.162 |
| [34] | M. Beidaghi, C.L. Wang. Micro-supercapacitors based on interdigital electrodes of reduced graphene oxide and carbon nanotube composites with ultrahigh power handling performance. Adv. Funct. Mater. 22 (2012) 4501–4510. DOI:10.1002/adfm.v22.21 |
| [35] | D. Pech, M. Brunet, P.L. Taberna, et al. Elaboration of a microstructured inkjetprinted carbonelectrochemical capacitor. J. Power Sources 195 (2010) 1266–1269. DOI:10.1016/j.jpowsour.2009.08.085 |
 2016, Vol. 27
2016, Vol. 27