b Gansu Key Laboratory of Polymer Materials, College of Chemistry and Chemical Engineering, Northwest Normal University, Lanzhou 730070, China;
c Gansu Engineering Laboratory of Applied Mycology, Hexi University, Zhangye 734000, China
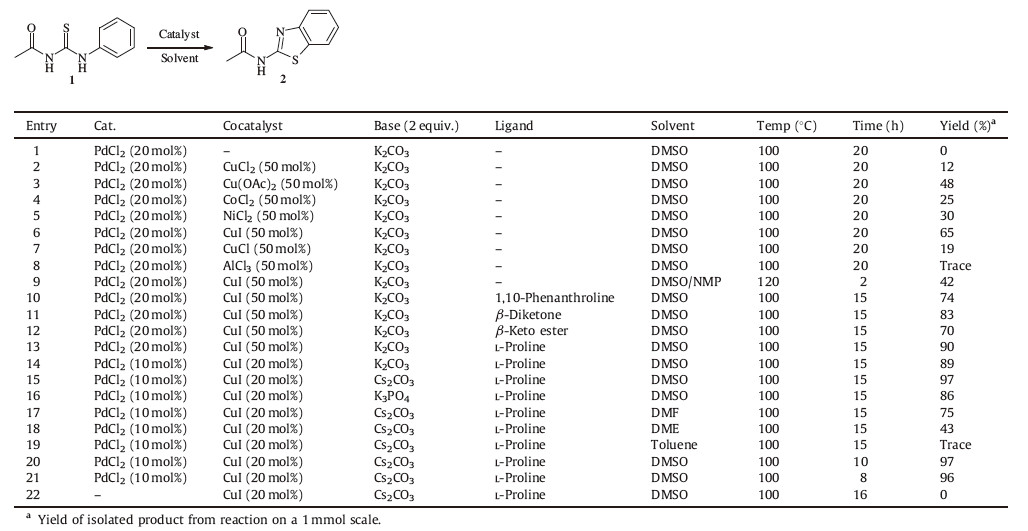
The benzothiazole moiety is an important scaffold due to its widespread occurrence in bioactive natural products,pharmaceuticals, organic optoelectronic materials,and ligands for phosphorescent complexes [1-4]. In particular,substituted Nbenzothiazol- 2-yl-amides are an important class of heterocyclic compounds that exhibit a wide range of biological properties [5-9] such as ubiquitin ligase inhibition [5],antitumor [6],antirotavirus infections [7],modulating the adenosine receptor [8, 9],and the nuclear hormone receptor [9]. For example,the N-benzothiazol-2- yl-cyclohexanecarboxamide,as a new anticancer drug,was selected as one of the most promising screening hit compounds (Fig. 1) [6]. The acylation reaction from 2-aminobenzothiazole,one of the classical methods for the preparation of these molecules [5, 6],is known for the limited diversity of the commercially available starting materials. Furthermore,the preparation of 2-aminobenzothiazole also required the use of the toxic bromine.
|
Download:
|
| Fig. 1.Structure of Sankyo investigational new drugs. | |
The past several years have witnessed the great progress in the development of the C-S bond formation promoted by transition metals,which can provide more efficient,practical,and straightforward approaches to valuable sulfur-containing compounds [10, 11]. However,these methods have been mainly focused on the ‘‘traditional’’ cross-coupling reactions of ArX (X = Cl,Br,I,OTf,and B(OH)2) and sulfides [12-39]. To achieve greener and more atomeconomic C-S bond formations,transition metal-catalyzed direct oxidative cross-coupling of C-H bonds and sulfides would be ideal [40-47].
In our previous work,we have shown that N-benzothiazol-2-ylamides can be synthesized smoothly by Cu-catalyzed intramolecular cyclization of various substituted 1-acyl-3-(2-bromophenyl) thioureas [48]. This method can provide more diversiform Nbenzothiazol- 2-yl-amides through the carbon-heteroatom formation under relatively mild conditions and avoid the use of the toxic bromine. However,the drawback of this procedure is the limited diversity of the commercially available starting materials due to the use of substituted ortho-haloarylamines. In order to further extend the diversity of N-benzothiazol-2-yl-amides,we have recently demonstrated an efficient intramolecular cyclization of substituted 1-acetyl-3-(2-phenyl)thiourea catalyzed by iron through C-H functionalization [49]. This method can provide more diversiform N-benzothiazol-2-yl-amides under relatively mild conditions. However,the purification of the target compounds is challenging using the column chromatography or recrystallization,since it is inescapable to obtain 1-acetyl-3-phenylurea whose polarity is similar to that of 1-acetyl-3-(2-phenyl)thiourea. Recently,Doi’s group [46] reported a Pd-catalyzed synthesis of 2-substituted benzothiazoles via a C-H Functionalization reaction. Therefore,we envisioned that Pd-catalyzed cyclization of 1-acyl-3-(2-phenyl)- thiourea 1 would represent a viable method for the formation and purification of substituted N-benzothiazol-2-yl-amides 2 (Scheme 1).

|
Download:
|
| Scheme. 1.Pd-catalyzed cyclization of 1-acyl-3-(2-aryl)thiourea by C–H functionalizations directly without further purification. | |
All reagents were commercially available and used as supplied. Dimethyl sulfoxide (DMSO) was dried and distilled from calcium hydride. N,N-Dimethylformamide (DMF),toluene,DME and CH3CN were dried prior to use using standard methods. Unless otherwise stated,analytical grade solvents and commercially available reagents were used as received. Thin layer chromatography (TLC) employed glass 0.20 mm silica gel plates. Flash chromatography columns were packed with 200-300 mesh silica gel.
All new compounds were characterized by IR,1H NMR,13C NMR and HRMS. The known compounds were characterized by 1H NMR, 13C NMR and HRMS. The IR spectra were run on a Nicolete spectrometer (KBr). The 1H NMR and 13C NMR spectra were recorded on a BRUKER AVANCEIII 400 MHz spectrometer. The chemical shifts (d) were given in parts per million relative to an internal standard tetramethylsilane. High resolution mass spectra (HRMS) were measured with a Waters Micromass GCT instrument and accurate masses were reported for the molecular ion (M+). Melting points were determined on a Perkin-Elmer differential scanning calorimeter and the thermometer was uncorrected.
2.1. General procedure for the synthesis of 1-acyl-3-arylthioureas [49, 50]To a 25 mL round-bottom flask equipped with a magnetic stirring bar was added acyl chloride (10 mmol),NH4SCN (15 mmol) and CH2Cl2 (20 mL),followed by PEG-400 (0.1 mmol). The mixture was stirred for approximately 3 h at room temperature. Aromatic amine (10 mmol) was added to the mixture and stirred for another 2 h at room temperature. The solvent was removed under reduced pressure to give the resulting residue as a solid,which was washed with water three times,to give the crude product. The analytical samples were obtained by recrystallization from C2H5OH in good yields ([4TD$DIF]88%-98%).
2.2. General procedure for the synthesis of N-benzothiazol-2-ylamides by a Pd-catalysed C(sp2)-H functionalization reactionA round-bottom flask equipped with a stirring bar was charged with 1-acyl-3-arylthioureas (1 mmol),PdCl2 (10 mol%),CuI (20 mol%),Cs2CO3 (2 equiv.),and L-proline (20 mol%) in 5 mL of DMSO. The mixture was stirred at 100 ℃ for the indicated time in Table 2. After cooling to room temperature,the reaction mixture was extracted with ethyl acetate (10 mL × 3). The organic layers were combined,dried over Na2SO4 and concentrated under reduced pressure,and then purified by silica gel chromatography (acetone/petroleum ether = 1:4) to yield the desired product 2.
N-(4-Ethylbenzo[d]thiazol-2-yl)acetamide (2f): A gray solid (80% yield); mp: 264-268 ℃; IR (cm-1): 3169.9,2990.1,2359.9, 1661.1,1550.4; 1H NMR (400 MHz,CDCl3): δ 9.42 (s,1H),7.67 (dd, 1H,J = 6.3,2.9 Hz),7.27 (dd,2H,J = 4.4,1.9 Hz),3.04 (q,2H, J = 7.6 Hz),2.28 (s,3H),1.34 (t,3H,J = 7.6 Hz); 13C NMR (100 MHz, CDCl3): δ 171.64(s),156.91 (s),146.45 (s),136.81 (s),131.98 (s), 125.25 (s),124.22 (s),118.92 (s),25.36 (s),23.51 (s),14.79 (s); HRMS calcd. for C11H12N2OS [M]+: 220.0670; found [5TD$DIF]200.0678.
N-(6-Fluorobenzo[d]thiazol-2-yl)acetamide (2 g): A white solid (94% yield); mp: 224-231 ℃; IR (cm-1): 3207.8,3071.0,2983.9, 2360.4,1689.2; 1H NMR (400 MHz,CDCl3): δ 7.70 (dd,1H,J = 8.9, 4.6 Hz),7.53 (dd,1H,J = 8.0,2.5 Hz),7.19 (td,1H,J = 8.9,2.6 Hz), 2.31 (s,3H); 13C NMR (100 MHz,CDCl3): δ 168.33 (s),160.93 (s), 158.50 (s),121.30 (d,J = 9.1 Hz),114.75 (s),108.09 (s),107.82 (s), 23.46 (s); HRMS calcd. for C9H7FN2OS [M]+: 210.0263; found 210.0256.
3. Results and discussionWhile not commercially available,benzothioureas are stable and easily synthesized [50, 51] from inexpensive starting materials in high yields on a multigram scale. Following Scheme 2,the synthesis of benzothioureas can be achieved in a straightforward manner starting from inexpensive aryl acid chloride and arylamines. Aryl acid chloride was treated with ammonium sulfocyanide in the presence of PEG-400 in CH2Cl2,followed by the addition of arylamines,to obtain 1-arylacyl-3-phenylthiourea in good to excellent yields. This intermediate can be used directly without further purifications.

|
Download:
|
| Scheme. 2.The synthesis of 1-acyl-3-arylthioureas. | |
In a preliminary experiment,we investigated the intramolecular C-S bond formation of 1-acetyl-3-phenylthiourea utilizing PdCl2 (20%) and a mild base (K2CO3,2 equiv.) in DMSO for 20 h at 100 ℃ (Table 1,entry 1). However,the reaction almost failed to take place. Subsequently,we screened several metal salts as cocatalysts, including AlCl3,CuCl2,Cu(OAc)2,CoCl2,NiCl2,FeCl3,CuI, and CuCl,and found that the addition of CuI considerably enhanced this reaction (Table 1,entries 2-8). However,the desired yield was still not obtained. Surprisingly,when Doi’s condition was used,the yield was still very low (42%) (Table 1,entry 9). Generally,the choice of the ligands is important for the reaction catalyzed by the metal,which prompted us to explore the effect of several bidentate ligands. We carried out the reaction of 1-acetyl-3-phenylthiourea by screening these ligands,such as 1,10-phenanthroline,β-keto esters,β-diketones,and L-proline. (Table 1,entries 10-13),and we were pleased to find that the use of these ligands can notably improve the yield of the product under the same conditions,and that L-proline proved to be the best among an array of ligands tested (Table 1,entry 14). When the amount of CuI and PdCl2 was decreased to 20 mol% and 10 mol%,respectively,the catalytic activity was maintained (Table 1,entry 14). Furthermore,we also investigated other bases (Cs2CO3 and K3PO4) (Table 1,entries 15- 16),solvents (DMF,DME,and toluene) (Table 1,entries 17-19) and reaction time (Table 1,entries 20-21). When only CuI was used in this cyclization,no reaction can take place (Table 1,entry 22). Thus, the optimized reaction conditions are as the follows: substrate (1 mmol),PdCl2 (10 mol%),CuI (20 mol%),Cs2CO3 (2 equiv.), L-proline (20 mol%) in DMSO (4 mL) within 8 h at 100 ℃.
| Table 1 Intramolecular cyclization of 1-acetyl-3-phenylthiourea: optimization of the catalytic condition. |
In response to this encouraging result,we used a range of substituted 1-acetyl-3-(phenyl)thioureas to investigate the scope and limitation of this reaction. The corresponding products were obtained in excellent yields (88%-98%). The results obtained under the optimized conditions are listed in Table 2. Initially,the substituents of phenyl were screened. The results demonstrate that little effect of the substituted groups on the benzene ring was observed for this transformation. Furthermore,substituents at different positions of the phenyl ring do not significantly affect the efficiency (Table 2,entries 1-8). It is noteworthy that the halosubstituted benzenes survived leading to halo-substituted products,which can be used for further transformations (Table 2, entries 2,7,8 and 11). In order to make the new Sankyo investigational drugs,the R group was selected as a cyclohexyl to give the corresponding products (Table 2,entries 10-12).
| Table 2 The synthesis of N-benzothiazol-2-yl-amides.a |
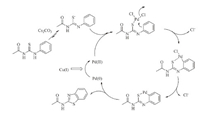
Although extensive studies on reaction mechanism have not yet been carried out,the proposed mechanism can be proposed according to the similar palladium-catalyzed processes [51] (Scheme 3). 1-Acetyl-3-(phenyl)thiourea was converted to the thioenolate in the presence of Cs2CO3. Pre-association of the sulphur atom in the thioenolate to Pd(OAc)2 facilitates the orthopalladation process with the concomitant release of chloride ion. The formation of the six-membered palladacycle and the subsequent reductive elimination leads to N-benzothiazol-2-yl-amide and Pd(0). The Pd(0) species are reoxidized to Pd(II) by CuI,thus completing the catalytic cycle.
|
Download:
|
| Scheme. 3.Postulated reaction mechanism. | |
In conclusion,we have achieved an efficient intramolecular cyclization of substituted 1-acetyl-3-(2-phenyl) thioureas catalyzed by palladium(II) catalysts through C(sp2)-H functionalization. This method can provide more diversiform N-benzothiazol-2- yl-amides efficiently and quickly in high yields under relatively mild conditions. The combination of the generality with respect to the substrate scope and facile accessibility to the starting materials may generate numerous synthetic possibilities. Further mechanistic analysis of these reactions will be the subject of future work.
AcknowledgmentsThis work was supported by the National Natural Science Foundation of China (Nos. 21462016,21262010),Natural Science Foundation of Gansu Province and the Advanced Research Fund of Jinchuan Group Co.,Ltd.
| [1] | R.S. Keri, M.R. Patil, S.A. Patil, S. Budagumpi, A comprehensive review in current developments of benzothiazole-based molecules in medicinal chemistry, Eur. J. Med. Chem. 89 (2015) 207-251. |
| [2] | A. Rouf, C. Tanyeli, Bioactive thiazole and benzothiazole derivatives, Eur. J. Med. Chem.97 (2015) 911-927. |
| [3] | A.G. DiKundar, G.K. Dutta, T.N. Guru Row, S. Patil, Polymorphism in opto-electronic materials with a benzothiazole-fluorene core: a consequence of high conformational flexibility of p-conjugated backbone and alkyl side chains, Cryst. Growth Des. 11 (2011) 1615-1622. |
| [4] | T. Girihar, W. Cho, Y.H. Kim, et al., A systematic identification of efficiency enrichment between thiazole and benzothiazole based yellow iridium(III) complexes, J. Mater. Chem.2 (2014) 9398-9405. |
| [5] | F. Parlati, U.V. Ramesh, R.P. Singh, et al., Benzothiazole and thiazole 5,5-bipyridine compositions and their use as ubiquitin ligase inhibitors, PCT Int. Appl. WO 2005037845, 2005. |
| [6] | M. Yoshida, I. Hayakawa, N. Hayashi, et al., Synthesis and biological evaluation of benzothiazole derivatives as potent antitumor agents, Bioorg. Med. Chem. Lett. 15 (2005) 3328-3332. |
| [7] | T.R. Bailey, D.C. Pevear, Benzothiazol compounds, compositions and methods for treatment and prophylaxis of rotavirus infections and associated diseases, PCT Int. Appl. WO 2004078115, 2004. |
| [8] | A. Alanine, A. Flohr, A.K. Miller, R.D. Norcross, C. Riemer, Benzothlazole derivatives, PCT Int. Appl. WO 2001097786, 2001. |
| [9] | S. Kerwin, L.H. Hurley, M.R. De, Compounds and method for providing pharmacologically active preparation and uses thereof, PCT Int. Appl. WO9748694, 1997. |
| [10] | H. Liu, X. Jiang, Transfer of sulfur: from simple to diverse, Chem. Asian J. 8 (2013) 2546-2563. |
| [11] | D.V. Partyka, Transmetalation of unsaturated carbon nucleophiles from boroncontaining species to the mid to late block metals of relevance to catalytic C-X coupling reactions (X = C, F, N, O, Pb, S, Se, Te), Chem. Rev.111 (2011) 1529-1595. |
| [12] | H. Yu, M. Zhang, Y. Li, Copper-catalyzed synthesis of benzo[b]thiophenes and benzothiazoles using thiocarboxylic acids as a coupling partner, J. Org. Chem. 78 (2013) 8898-8903. |
| [13] | H. Deng, Z. Li, F. Ke, X. Zhou, Cu-catalyzed three-component synthesis of substituted benzothiazoles in water, Chem. Eur. J.18 (2012) 4840-4843. |
| [14] | H.J. Xu, Y.Q. Zhao, T. Feng, Y.S. Feng, Chan-lam-type S-arylation of thiols with boronic acids at room temperature, J. Org. Chem.77 (2012) 2878-2884. |
| [15] | D.J.C. Prasad, G. Sekar, Cu-catalyzed one-pot synthesis of unsymmetrical diaryl thioethers by coupling of aryl halides using a thiol precursor, Org. Lett. 13 (2011) 1008-1011. |
| [16] | L.L. Sun, C.L. Deng, R.Y. Tang, X.G. Zhang, CuI/TMEDA-catalyzed annulation of 2- bromo alkynylbenzenes with Na2S: synthesis of benzo[b]thiophenes, J. Org. Chem.76 (2011) 7546-7550. |
| [17] | H.L. Kao, C.F. Lee, Efficient copper-catalyzed s-vinylation of thiols with vinyl halides, Org. Lett. 13 (2011) 5204-5207. |
| [18] | F. Ke, Y. Qu, Z. Jiang, et al., An efficient copper-catalyzed carbon-sulfur bond formation protocol in water, Org. Lett.13 (2011) 454-457. |
| [19] | D. Chen, Z.J. Wang, W. Bao, Copper-catalyzed cascade syntheses of 2H-benzo[ b][1,4]thiazin-3(4H)-ones and quinoxalin-2(1H)-ones through capturing S and N atom respectively from AcSH and TsNH2, J. Org. Chem. 75 (2010) 5768- 5771. |
| [20] | C.L. Li, X.G. Zhang, R.Y. Tang, P. Zhong, J.H. Li, Copper-catalyzed thiolation annulations of 1,4-dihalides with sulfides leading to 2-trifluoromethyl benzothiophenes and benzothiazoles, J. Org. Chem.75 (2010) 7037-7040. |
| [21] | W. You, X. Yan, Q. Liao, C. Xi, Cu-catalyzed double s-alkenylation of potassium sulfide: a highly efficient method for the synthesis of various thiophenes, Org. Lett. 12 (2010) 3930-3933. |
| [22] | Y. Jiang, S. Xie, Y. Qin, X. Zhang, D. Ma, A general and efficient approach to aryl thiols: CuI-catalyzed coupling of aryl lodides with sulfur and subsequent reduction, Org. Lett. 11 (2009) 5250-5253. |
| [23] | S. Murru, P. Mondal, R. Yella, B.K. Patel, Copper(I)-catalyzed cascade synthesis of 2-substituted 1,3-benzothiazoles: direct access to benzothiazolones, Eur. J. Org. Chem. (2009) 5406-5413. |
| [24] | S. Murru, H. Ghosh, S.K. Sahoo, B.K. Patel, Intra- and intermolecular C-S bond formation using a single catalytic system: first direct access to arylthiobenzothiazoles, Org. Lett. 11 (2009) 4254-4257. |
| [25] | C.G. Bates, P. Saejueng, M.Q. Doherty, D. Venkataraman, Copper-catalyzed synthesis of vinyl sulfides 6 (2004) 5005-5008. |
| [26] | Z. Qiao, H. Liu, X. Xiao, et al., Efficient access to 1,4-benzothiazine: palladiumcatalyzed double C-S bond formation using Na2S2O3 as sulfurating reagent, Org. Lett. 15 (2013) 2594-2597. |
| [27] | M. Kuhn, F.C. Falk, J. Paradies, Palladium-catalyzed C-S coupling: access to thioethers, benzo[b]thiophenes, and thieno[3,2-b]thiophenes, Org. Lett.13 (2011) 4100-4103. |
| [28] | C.C. Eichman, J.P. Stambuli, Zinc-mediated palladium-catalyzed formation of carbon-sulfur bonds, J. Org. Chem. 74 (2009) 4005-4008. |
| [29] | T. Dahl, C.W. Tornoe, B. Bang-Andersen, P. Nielsen, M. Jorgensen, Palladiumcatalyzed three-component approach to promazine with formation of one carbon- sulfur and two carbon-nitrogen bonds, Angew. Chem. Int. Ed. 47 (2008) 1726-1728. |
| [30] | J.Y. Lee, P.H. Lee, Palladium-catalyzed carbon-sulfur cross-coupling reactions with indium tri(organothiolate) and its application to sequential one-pot processes, J. Org. Chem.73 (2008) 7413-7426. |
| [31] | M.A. Fernandez-Rodroeguez, Q. Shen, J.F. Hartwig, A general and long-lived catalyst for the palladium-catalyzed coupling of aryl halides with thiols, J. Am. Chem. Soc. 128 (2006) 2180-2181. |
| [32] | M. Iwasaki, M. Iyanaga, Y. Tsuchiya, et al., Palladium-catalyzed direct thiolation of aryl C-H bonds with disulfides, Chem. Eur. J. 20 (2014) 2459-2462. |
| [33] | X.B. Xu, J. Liu, J.J. Zhang, Y.W. Wang, Y. Peng, Nickel-mediated inter- and intramolecular C-S coupling of thiols and thioacetates with aryl Iodides at room temperature, Org. Lett. 15 (2013) 550-553. |
| [34] | N. Sakai, T. Miyazaki, T. Sakamoto, et al., Single-step thioetherification by indiumcatalyzed reductive coupling of carboxylic acids with thiols, Org. Lett.14 (2012) 4366-4369. |
| [35] | Y. Yang, W. Hou, L. Qin, et al., Rhodium-catalyzed directed sulfenylation of arene C-H bonds, Chem. Eur. J.20 (2014) 416-420. |
| [36] | Y.Y. Lin, Y.J. Wang, C.H. Lin, J.H. Cheng, C.F. Lee, Synthesis of alkenyl sulfides through the iron-catalyzed cross-coupling reaction of vinyl Halides with thiols, J. Org. Chem.77 (2012) 6100-6106. |
| [37] | V.P. Reddy, K. Swapna, A.V. Kumar, K.R. Rao, Indium-catalyzed C-S cross-coupling of aryl halides with thiols, J. Org. Chem.74 (2009) 3189-3191. |
| [38] | M. Arisawa, T. Suzuki, T. Ishikawa, M. Yamaguchi, Rhodium-catalyzed substitution reaction of aryl fluorides with disulfides: p-orientation in the polyarylthiolation of polyfluorobenzenes, J. Am. Chem. Soc.130 (2008) 12214-12215. |
| [39] | Y.C. Wong, T.T. Jayanth, C.H. Cheng, Cobalt-catalyzed aryl-sulfur bond formation, Org. Lett.8 (2006) 5613-5616. |
| [40] | H. Wang, L. Wang, J. Shang, et al., Fe-catalysed oxidative C-H functionalization/C- S bond formation, Chem. Commun.48 (2012) 76-78. |
| [41] | Y. Cheng, J. Yang, Y. Qu, P. Li, Aerobic visible-light photoredox radical C-H functionalization: catalytic synthesis of 2-substituted benzothiazoles, Org. Lett.14 (2012) 98-101. |
| [42] | F. Jia, Z.P. Li, Iron-catalyzed/mediated oxidative transformation of C-H bonds, Org. Chem. Front. 1 (2014) 194-214. |
| [43] | L. Chu, X. Yue, F.L. Qing, Cu(II)-mediated methylthiolation of aryl C-H bonds with DMSO, Org. Lett. 12 (2010) 1644-1647. |
| [44] | X. Zhang, W. Zeng, Y. Yang, H. Huang, Y. Liang, Copper-catalyzed double C-S bonds formation via different paths: synthesis of benzothiazoles from N-benzyl- 2-iodoaniline and potassium sulfide, Org. Lett. 16 (2014) 876-879. |
| [45] | K. Inamoto, Y. Arai, K. Hiroya, T. Doi, Palladium-catalysed direct synthesis of benzo[b]thiophenes from thioenols, Chem. Commun. 43 (2008) 5529- 5531. |
| [46] | K. Inamoto, C. Hasegawa, K. Hiroya, T. Doi, Palladium-catalyzed synthesis of 2- substituted benzothiazoles via a C-H functionalization/intramolecular C-S bond formation process, Org. Lett.10 (2008) 5147-5150. |
| [47] | J.K. Wang, F. Peng, J.L. Jiang, et al., Synthesis of N-benzothiazol-2-yl-amides by a copper-catalyzed intramolecular cyclization process, Tetrahedron Lett. 49 (2008) 467-470. |
| [48] | J.K. Wang, Y.X. Zong, Y.X. Zhang, et al., Synthesis of N-benzothiazol-2-yl-amides by a Fe-catalyzed oxidative C(sp2)-H functionalization, Synlett 25 (2014) 2143- 2148. |
| [49] | X.C. Wang, Z. Li, Y. Tu, Y.X. Da, Synthesis of N-(5-aryloxymethyl-1,3,4-thiadiazol- 2-yl)-N'-(5-aryl-2-furoyl)-thioureas under phase transfer catalysis, Synth. Commun.32 (2002) 1113-1119. |
| [50] | Z. Li, X.C. Wang, Synthesis of 1-aryloxyacetyl-4-(4-nitrobenzoyl)-thiosemicarabzides under phase transfer catalysis and microwave irradiation, Synth. Commun.32 (2002) 3087-3092. |
| [51] | K. Inamoto, T. Saito, M. Katsuno, T. Sakamoto, K. Hiroya, Palladium-catalyzed C-H activation/intramolecular amination reaction: a new route to 3-aryl/alkylindazoles, Org. Lett. 9 (2007) 2931-2934. |
 2015, Vol.26
2015, Vol.26