b National Research Council of Canada, Vancouver V6T 1W5, Canada
Electrochemical capacitors,also called supercapacitors,are considered potential energy storage devices due to their high power density,excellent reversibility,and long cycle life,and thus have attracted extensive research investigations in recent years [1, 2, 3]. A variety of electrode materials have been explored,such as carbon materials (e.g.,activated carbon black,carbon nanotubes, and carbon aerogel) [4, 5],transition metal oxides [6, 7, 8],and their salts (e.g.,RuO2 [9],PbO2 [10],MnO2 [11, 12],NiO [13],Co3O4 [14], NaxMnO2 [15],and KxMnO2 [16]),and conducting polymers.
As a new type of thermoelectric material,NaCo2O4 has generated extensive interest because of its large thermoelectric power,low electrical resistivity,and thermal conductivity [17], and has been widely used in energy conversion and electronic devices. Some reports show that NaCo2O4 has a typical bronze oretype layered structure,consisting of Na+ and CoO2 blocks alternately stacking along the c-axis to form layers [18, 19]. As this layered structure is advantageous for the material’s electron transmission and stability,NaCo2O4 could be applied in electrochemical capacitors to yield excellent electrochemical performance and good cyclic stability. However,to the best of our knowledge,NaCo2O4 has only been studied as a new thermoelectric material but not as a supercapacitor material.
In this study,we synthesize a sub-micron-scaled NaCo2O4 sample via a simple solid-state reaction,and then use this NaCo2O4 as the electrode material for supercapacitors. We also discuss the composition,morphology,and electrochemical capacitive behaviors of the synthesized NaCo2O4 sample. 2. Experimental 2.1. Synthesis of NaCo2O4 materials
The CoSO4 7H2O,Na2CO3,NaOH,and graphite used were of analytical grade,while the acetylene black and PTFE were of battery grade.
100 mL of CoSO4 solution (1.0 mol/L) was added to 60 mL of NaOH solution (4 mol/L) and the mixture was stirred for 30 min at 60℃ to yield a brown precipitate. Co3O4 particles were then obtained by filtration followed by washing and drying for 12 h at 100℃. NaCo2O4 was synthesized by ball-milling mixtures of Co3O4 and Na2CO3 (at a molar ratio of 2:3) for 2 h,followed by heating in an air atmosphere at 800℃ for 10 h. 2.2. Sample characterizations
The obtained particles were characterized on a Bruker D8 Advance powder X-ray diffractometer (XRD) with monochromatized Cu Kα radiation (λ= 0.154056 nm). The morphology of the NaCo2O4 was investigated with a scanning electron microscope (SEM) (LEO 1430VP,Germany). 2.3. Electrochemical measurements
NaCo2O4 (70 wt.%),graphite (15 wt.%),and acetylene black (10 wt.%) were mixed thoroughly with a certain amount of 5 wt.% PTFE aqueous solution to obtain a homogeneous slurry possessing adequate rheological properties. The slurry was scrape-coated onto a stainless steel mesh with a surface area of 1 cm2 and dried at 80℃ for 12 h. Subsequently,the pasted electrodes were pressed at 10 MPa for 5 min to ensure electrical contact between the mesh and the active material. The total weight of the active material in the electrode was controlled at about 20 mg. Symmetric supercapacitors were assembled face to face using two identical electrodes with the same mass of active materials. The electrolyte used was a diaphragm paper with 6 mol/L NaOH.
Cyclic voltammetry (CV) testing was performed using a threeelectrode cell,in which a Ni-grid and a Hg/HgO electrode were used as the counter and reference electrodes,respectively. Cyclic voltammograms (CVs) were recorded between 0.15 V and 0.65 V at a scanning rate of 20 mV/s. Galvanostatic charge/discharge cycling was performed in a two-electrode system with identical electrodes having the same active electrode materials (i.e.,Type I symmetric supercapacitor). A constant current density of 50 mA/g was employed for charging/discharging the cell in a voltage range of 0.25-0.7 V. 3. Results and discussion 3.1. Crystal structure analysis
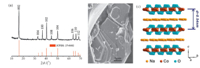
Fig. 1(a) shows the XRD pattern of the synthesized NaCo2O4. The characteristic peaks of the prepared sample are well in accordance with JCPDS-27-0682. No original oxides or other impurity phases were detected in these patterns,indicating singlephase NaCo2O4,which has a hexagonal layered structure in space group P6322. Peak (002) is found at 2θ= 16.2°and corresponds to the interlayer space (0.54 nm),as shown in Fig. 1(c). According to the Scherrer equation (D = Kλ/βcosθ),the average c-stacking height,calculated from (002) reflection,was 27.86 nm for layered NaCo2O4. Therefore,we could reasonably estimate that the average layer number of NaCo2O4 is about 52,indicating that crystal particles mainly grow along c-axis.
|
Download:
|
| Fig. 1.(a) XRD pattern,(b) SEM image,and (c) schematic illustration of the microstructure of the obtained NaCo2O4. | |
NaCo2O4 consists of Na+ and CoO2 blocks alternately stacking along the c-axis to form a layered structure; thus,the physical properties of NaCo2O4 are highly two-dimensional,as shown in Fig. 1(b). The unit cell of NaCo2O4 is built up with twisting sheets of edge-sharing CoO6 octahedrons,where Co is located in the centre of the octahedron and O at the six vertexes,and two octahedrons share one edge,forming traditional calcium titanate with a perovskite structure [20]. Na+ provides electrons for storage in the electrical layer and stabilizes the crystal structure. 3.2. Electrochemical performance
Fig. 2(a) shows the CV of the NaCo2O4 electrode within the first 100 cycles in 6 mol/L NaOH aqueous solution at a scan rate of 20 mV/s. It can be seen that the CVs are rectangular with only a pair of very weak redox peaks. As shown in Fig. 2(b) and (c),the weak redox peaks appear at 0.51 V and 0.37 V (vs. Hg/HgO). These redox peaks are similar to those of NaxMnO2,which can be definitivelyascribed to the intercalation/deintercalation of Na+ into and from the solid lattice [11, 21, 22, 23]. Moreover,in Fig. 2(a),the symmetrical CV curve of the NaCo2O4 electrode with respect to the zero-current axis means that the Faradic reactions of NaCo2O4 are reversible. In Fig. 2(a) we can also see that the CV curves of the NaCo2O4 electrode are very stable during the 100 cycles,indicating excellent electrochemical stability over the potential range of 0.15-0.65 V. This demonstrates that the layered structure of NaCo2O4 remains stable during the charge/discharge process.

|
Download:
|
| Fig. 2.CV of NaCo2O4 electrode in 6 mol/L NaOH aqueous solution at a scan rate of 20 mV/s. | |
Fig. 3(a) illustrates the typical charge/discharge cycles for the NaCo2O4 electrodes at a current density of 50 mA/g. During both charge/discharge processes,the potential behavior shows good linear activity,and the charge/discharge curves present similar symmetrical triangular waves. These results demonstrate that the NaCo2O4 electrodes exhibit a good response for an electrochemical capacitor.

|
Download:
|
| Fig. 3.(a) Charge/discharge curves and (b) cycling behavior of the NaCo2O4 electrodes at a current density of 50 mA/g. | |
The specific capacity of the NaCo2O4 electrode can be calculated from the charge/discharge curves,according to the following Eq. (1):

Fig. 3(b) shows the excellent cycling behaviors of the symmetric NaCo2O4/NaCo2O4 supercapacitor in 6 mol/L NaOH electrolyte at 50 mA/g current density between 0.25 V and 0.7 V. A decrease in specific capacitance of only 9% was observed in the initial 500 cycles at a current density of 50 mA/g,indicating that the structure of layered NaCo2O4 is very stable during the long-term intercalation/ deintercalation of Na+ ions. In addition,the coulombic efficiency of the capacitor is almost 100%,except during the initial several cycles. As the electrode material is not fully activated at the beginning,the specific capacity is lower for the first few cycles, after which the active substance and the electrolyte are in full contact and the electrode material is activated,resulting in an increased capacitance. The small grain of the NaCo2O4 material also contributes to its excellent cycling performance. 4. Conclusion
A new supercapacitor material,sub-micron-scaled NaCo2O4, was prepared via a solid-phase sintering method using Co3O4 and Na2CO3 as the starting materials. XRD and SEM show that the synthesized NaCo2O4 is single-phased with a hexagonal layered structure. The specific discharge capacity of the NaCo2O4 electrode was 337 F/g in a 6 mol/L NaOH solution between 0.15 V and 0.65 V at 50 mA/g current density. This demonstrates that the synthesized NaCo2O4 possesses a favorable electrochemical reversibility and excellent supercapacitor properties. The cycling performance of the synthesized NaCo2O4 is also admirable. It is concluded that NaCo2O4,a thermoelectric material,is a promising supercapacitor material. Acknowledgment
This work was financially supported by the Natural Science Foundation of China (No. 21071046) under approval. I
| [1] | A.S. Aricò1, P. Bruce, B. Scrosati, J.M. Tarascon, W. Schalkwijk, Nanostructured materials for advanced energy conversion and storage devices, Nat. Mater. 4 (2005) 366-377. |
| [2] | P. Hall, M. Mirzaeian, S. Fletcher, et al., Energy storage in electrochemical capacitors: designing functional materials to improve performance, Energy Environ. Sci. 3 (2010) 1238-1251. |
| [3] | P. Simon, Y. Gogotsi, Materials for electrochemical capacitors, Nat. Mater. 7 (2008) 845-854. |
| [4] | M.G. Sullivan, R. Kötz, O. Haas, Thick active layers of electrochemically modified glassy carbon. Electrochemical impedance studies, J. Electroehem. Soc. 147 (2000) 308-317. |
| [5] | R. Salige, U. Fischer, C. Herta, J. Fricke, High surface area carbon aerogels for supercapacitors, J. Non-Cryst. Solids 225 (1998) 81-85. |
| [6] | C.C. Hu, W.C. Chen, K.H. Chang, How to achieve maximum utilization of hydrous ruthenium oxide for supercapacitors, J. Electrochem. Soc. 151 (2004) A281-A290. |
| [7] | C. Liu, F. Li, L.P. Ma, H.M. Cheng, Advanced materials for energy storage, Adv. Mater. 22 (2010) E28-E62. |
| [8] | K.T. Lee, C.B. Tsai, W.H. Ho, N.L. Wu, Superabsorbent polymer binder for achieving MnO2 supercapacitors of greatly enhanced capacitance density, Electrochem. Commun. 12 (2010) 886-889. |
| [9] | X.H. Xia, J.P. Tu, X.L. Wang, C.D. Gu, X.B. Zhao, Hierarchically porous NiO film grown by chemical bath deposition via a colloidal crystal template as an electrochemical pseudocapacitor material, J. Mater. Chem. 21 (2011) 671-679. |
| [10] | W. Zhang, Y.H. Qu, L.J. Guo, Performance of PbO2/activated carbonhybrid supercapacitor with carbon foam substrate, Chin. Chem. Lett. 23 (2012) 623-626. |
| [11] | Q.T. Qua, Y. Shi, S. Tian, et al., A new cheap asymmetric aqueous supercapacitor: activated carbon//NaMnO2, J. Power Sources 194 (2009) 1222-1225. |
| [12] | L.R. Wang, F. Ran, Y.T. Tan, L. Zhao, L.B. Kong, L. Kαng, Coral reel-like polyanaline manotubes prepared by a reactive template of manganese oxide for supercapacitor, Chin. Chem. Lett. 22 (2011) 964-968. |
| [13] | Q.T. Qu, L. Li, S. Tian, et al., A cheap asymmetric supercapacitor with high energy at high power: activated carbon//K0.27MnO2 ·0.6H2O, J. Power Sources 195 (2010) 2789-2794. |
| [14] | J.P. Zheng, P.J. Cygan, T.R. Jow, Hydrous ruthenium oxide as an electrode material for electrochemical capacitors, J. Electrochem. Soc. 142 (1995) 2699-2703. |
| [15] | V. Gupta, N. Miura, Electrochemically deposited polyaniline nanowire's network a high-performance electrode material for redox supercapacitor, Electrochem. Solid State Lett. 8 (2005) A630-A632. |
| [16] | V. Gupta, N. Miura, Influence of the microstructure on the supercapacitive behavior of polyaniline/single-wall carbon nanotube composites, J. Power Sources 157 (2006) 616-620. |
| [17] | I. Terasaki, Y. Sasago, K. Uchinokura, Large thermoelectric power in NaCo2O4 single crystals, Phys. Rev. B 56 (1997) 12685-12687. |
| [18] | C. Fouassier, G. Matejka, J.M. Reau, P. Hagenmuller, Sur de nouveaux bronzes oxygéné s de formule NaxCoO2 (χ1). Le système cobalt-oxygène-sodium, J. Solid State Chem. 6 (1973) 532-537. |
| [19] | M. Jansen, R. Hoppe, Notiz zur Kenntnis der Oxocobaltate des Natriums, Z. Anorg. Allg. Chem. 408 (1974) 104-106. |
| [20] | I. Terasaki, Transport properties and electronic states of the thermoelectric oxide NaCo2O4, Phys. Rev. B 328 (2003) 63-67. |
| [21] | L. Athouël, F. Moser, R. Dugas, et al., Variation of the MnO2 birnessite structure upon charge/discharge in an electrochemical supercapacitor electrode in aqueous Na2SO4 electrolyte, J. Phys. Chem. C 112 (2008) 7270-7277. |
| [22] | A. Caballero, L. Hernán, J. Morales, L. Sánchez, J. Santos, Ion-exchange properties of P2-NaxMnO2: evidence of the retention of the layer structure based on chemical reactivity data and electrochemical measurements of lithium cells, J. Solid State Chem. 174 (2003) 365-371. |
| [23] | O.A. Shlyakhtin, A.M. Skundin, S.J. Yoon, Y.J. Oh, Ni-Mn hydroxides as new high power electrode materials for supercapacitor applications, Mater. Lett. 63 (2009) 109-112. |
| [24] | C. Portet, P.L. Taberna, P. Simon, C. Laberty-Robert, Modification of Al current collector surface by sol-gel deposit for carbon-carbon supercapacitor applications, Electrochim. Acta 49 (2004) 905-912. |
| [25] | X. Zhang, P. Yu, H.T. Zhang, et al., Rapid hydrothermal synthesis of hierarchical nanostructures assembled from ultrathin birnessite-type MnO2 nanosheets for supercapacitor applications, Electrochim. Acta 89 (2013) 523-529. |
| [26] | Z.C. Li, H.L. Bao, X.Y. Miao, X.H. Chen, A facile route to growth of γ-MnOOH nanorods and electrochemical capacitance properties, J. Colloid Interf. Sci. 357 (2011) 286-291. |




