Recently, considerable attention has been focused on pyrazolines and substituted pyrazolines due to their interesting biological activities. They possess anti-microbial [1], anti-fungal [2], antidepressant [3], anti-convulsant [4], anti-inflammatory [5], etc. properties. Moreover, N-acetyl pyrazoline derivatives exhibit important pharmaceutical profiles [6, 7, 8]. Hence, the pyrazoline framework represents an interesting template for combinatorial [9] as well as medicinal chemistry [10, 11]. On the other hand, 1,2,3-triazoles have received much attention due to their interesting bioactivity profile such as anti-biotic, anti-fungal [12], anti-cancer [13], anti-HIV [14], anti-microbial [15], etc. properties. Also, they serve as potential chemotherapeutic agents for various diseases [16].
In connection with the above and in continuation of our earlier work on environmentally benign, green synthesis of 1,2,3-triazolyl chalcone hybrids [17], we decided to attempt the synthesis of molecules containing both of the two pharmacologically active moieties mentioned above in a single frame. Thus, we disclose the synthesis of 1,2,3-triazolyl-N-acylpyrazoline hybrids from easily accessible 1,2,3-triazolyl chalcones, the details of which are presented vide infra.
Chalcones, belonging to the flavonoids family, are convenient synthons for the synthesis of five [18], six [19] and seven membered [20] heterocyclic compounds. With regard to pyrazoline derivatives, several methods have been employed for their synthesis, including the condensation of chalcones with hydrazine, hydrazine derivatives [21, 22, 23, 24] and thiosemicarbazide [25] under acidic [21, 22] or basic [25] conditions, and the cycloaddition of nitrilimines, generated in situ from the corresponding hydrazonoyl halides by the action of a suitable base, to carbon-carbon double bonds of a dipolarophile [26, 27, 28, 29]. Hence, considerable interest has been focused on the synthesis of pyrazolines from chalcones.
Typical procedure: To a solution of (E)-1-(1-benzyl-5-methyl- 1H-1,2,3-triazol-4-yl)-3-(4-methylphenyl)prop-2-en-1-one [18] (1e, 1.0 equiv.) and hydrazine hydrate (2.0 equiv.) in acetic acid (5 mL) was refluxed for 3 h. Then, the reaction mixture was poured onto ice-water to afford the 1,2,3-triazolyl-N-acetylpyrazoline hybrid (2e) in 96% yield as a white solid, which was filtered and recrystallized from ethanol.
1-(3-(1-Benzyl-5-methyl-1H-1,2,3-triazol-4-yl)-5-(4-methylphenyl)- 4,5-dihydro-1H-pyrazol-1-yl)ethanone (2e): Mp: 150- 151 ℃; 1H NMR (300 MHz, CDCl3): δ 7.11-7.37 (m, 9H, ArH), 5.54 (s, 2H, NCH2), 5.50 (dd, 1H, J = 4.8 & 12.0 Hz, CH), 3.87(dd, 1H, J = 12.0 & 18.6 Hz, CH2), 3.44 (dd, 1H, J = 4.8 & 18.6 Hz, CH2), 2.51(s, 3H, COCH3), 2.32 (s, 3H, CH3), 2.29 (s, 3H, ArCH3); 13C NMR (75 MHz, CDCl3): δ 168.46, 149.47, 138.68, 138.24, 137.19, 134.25, 132.80, 129.41, 129.08, 128.53, 127.25, 125.65, 58.69, 51.95, 43.25, 21.79, 21.00, 9.41; Mass (ES/MS): m/z 396.58 [M+Na]+; Anal. Calcd. for C22H23N5O: C 70.76, H 6.21, N 18.75. Found: C 70.70, H 6.22, N 18.78.
Perusal of literature indicates a lack of reports on the synthesis of N-acylpyrazolines linked with a 1,2,3-triazole core. In this regard, we herein report synthesis of novel 1,2,3-triazolyl-Nacylpyrazoline hybrids in a facile and efficient manner starting from chalcones [17].
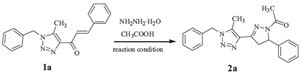
For the purpose of optimization, synthesis of N-acetylpyrazoline (3a) was attempted (Scheme 1) under different conditions viz. (i) reaction through conventional heating (ii) reaction under microwave irradiation. The details are presented below.
|
Download:
|
| Scheme 1.Optimization for the synthesis of 1,2,3-triazole-N-acetylpyrzoline hybrid (3a). | |
| Table 1 Optimization for the synthesis of 1,2,3-triazole-N-acetylpyrzoline hybrid (3a). |
|
Download:
|
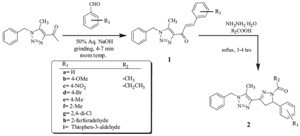
| Scheme 2.Synthesis of 1,2,3-triazolyl-N-acetyl/N-propionylpyrzolines (2). | |
| Table 2 Synthesis of 1,2,3-triazolyl-N-acetyl/N-propionylpyrazoline hybrids (2a–p). |
|
Download:
|
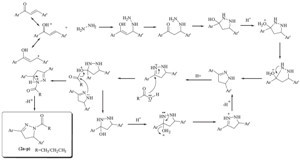
| Scheme 3.A feasible mechanism for N-acylatedpyrazolines. | |
In conclusion, we have achieved an efficient and facile synthesis of a library of hitherto novel 1,2,3-triazolyl-N-acetyl/N-propionylpyrazoline hybrids in excellent yields via easily accessible chalcones.
The authors wish to thank IRHPA, DST for providing 300 MHz NMR facility and UGC (BSR)-India for giving financial support as JRF & SRF.
| [1] | M. Alam, S.A.A. Nami, M. Parveen, et al., Microwave assisted synthesis and in silico screening of steroidal pyrazolines, Chin. Chem. Lett. 23 (2012) 1039-1042. |
| [2] | S.S. Korgaokar, P.H. Patil, M.T. Shah, H.H. Parekh, Studies on pyrazolines: preparation and antimicrobial activity of 3-(3'(p-chlorophenylsulphonamidophenyl)-5-aryl-acetylpyrazolines, Indian J. Pharm. Sci. 58 (1996) 222-225. |
| [3] | P.Y. Rajendra, R.A. Lakshmana, et al., Synthesis and antidepressant activity of some 1,3,5-triphenyl-2-pyrazolines and 3-(2''-hydroxy naphthalen-1''-yl)-1,5-diphenyl-2-pyrazolines, Bioorg. Med. Chem. Lett. 15 (2005) 5030-5034. |
| [4] | Z. Ozdemir, H.B. Kandilici, et al., Synthesis and studies on antidepressant and anticonvulsant activities of some 3-(2-furyl)-pyrazoline derivatives, Eur. J. Med. Chem. 42 (2007) 373-379. |
| [5] | R.H. Udupi, A.S. Kushnoor, A.R. Bhat, Synthesis and biological evaluation of certain pyrazoline derivative of 2-(6-methoxynaphthyl)-propionic acid, Indian J. Heterocycl. Chem. 8 (1998) 63-66. |
| [6] | A.K. Rana, S.B. Lade, S. Sorathia, et al., Synthesis, characterization and antimicrobial evaluation of some new N-acetyl pyrazoline derivatives from substituted furan-2-carbaldehyde, Der Chem. Sin. 4 (2012) 965-969. |
| [7] | A.M. Fahmy, K.M. Hassa, A.A. Khalaf, R.A. Ahmed, Synthesis of some new betalactams, 4-thiazolidinones and pyrazolines, Indian J. Chem. 26 (1987) 884-887. |
| [8] | B. Holla, P.M. Shivarama, M.K. Akberali, Shivananda, Studies on arylfuran derivatives: part X. Synthesis and antibacterial properties of arylfuryl-delta (2)-pyrazolines, Farmaco 55 (2000) 256-263. |
| [9] | P. Brooking, A. Doran, P. Grimsey, et al., Split-Split. A multiple synthesiser approach to efficient automated parallel synthesis, Tetrahedron Lett. 40 (1999) 1405-1408. |
| [10] | J. Regan, S. Breitfelder, P. Cirillo, T. Gilmore, et al., Pyrazole urea-based inhibitors of p38 MAP kinase: from lead compound to clinical candidate, J. Med. Chem. 45 (2002) 2994-3008. |
| [11] | P. Pevarello, M.G. Brasca, R. Amici, P.G. Orsini, et al., 3-Aminopyrazole inhibitors of CDK2/cyclin A as antitumor agents. 1. Lead finding, J. Med. Chem. 47 (2004) 3367-3380. |
| [12] | X.L. Wang, K. Wan, C.H. Zhou, Synthesis of novel sulfanilamide-derived 1,2,3-triazoles and their evaluation for antibacterial and antifungal activities, Eur. J. Med. Chem. 45 (2010) 4631-4639. |
| [13] | W.T. Li, W.H. Wu, C.H. Tang, R. Tai, S.T. Chen, One-pot tandem copper-catalyzed library synthesis of 1-thiazolyl-1,2,3-triazoles as anticancer agents, ACS Comb. Sci. 13 (2011) 72-78. |
| [14] | R. Alvarez, S. Velazquez, A. San-Felix, et al., 1,2,3-Triazole-[2',5'-bis-O-(tertbutyldimethylsilyl-b-D-ribofuranosyl]-3'-spiro-5''-(4''-amino-1'',2''-oxathiole-2'',2''-dioxide) (TSAO) analogues. Synthesis and anti-HIV-1 activity, J. Med. Chem. 37 (1994) 4185-4194. |
| [15] | Xu Zhao, Bo Wei Lu, Jun Rui Lu, et al., Design, synthesis and antimicrobial activities of 1,2,3-triazole derivatives, Chin. Chem. Lett. 23 (2012) 933-935. |
| [16] | S. Wang, Q. Wang, Y. Wang, L. Liu, et al., Novel anthraquinone derivatives: synthesis via click chemistry approach and their induction of apoptosis in BGC gastric cancer cells via reactive oxygen species (ROS)-dependent mitochondrial pathway, Bioorg. Med. Chem. Lett. 18 (2008) 6505-6508. |
| [17] | P. Shanmugavelan, M. Sathishkumar, S. Nagarajan, et al., A facile synthesis of 1, 2,3-triazolyl indole hybrids via SbCl3-catalysed Michael addition of indoles to 1,2,3-triazolyl chalcones, J. Chem. Sci. 124 (2012) 941-950. |
| [18] | B.J. Gaede, L.L. Mc Dermott, Novel perfluoroalkyl-substituted pyrazoles. 1. Hydroxypyrazoles, J. Heterocycl. Chem. 30 (1993) 49-54. |
| [19] | K. Shibata, I. Katsuyama, H. Izoe, et al., Synthesis of 4,6-di substituted 2-methyl pyridines and their 3-carboxamides, J. Heterocycl. Chem. 30 (1993) 277-281. |
| [20] | X. Jiaxi, C. Wang, Q. Zhang, Synthesis of 1-3,3a,5-tetraaryl-3a,4,5,6-tetrahydro-3H-1,2,4-trizolo[4,3-a][1,5] benzodiazepines, Heteroatom Chem. 6 (2001) 557-559. |
| [21] | D. Azarifar, H. Ghasemnejad, Microwave-assisted synthesis of some 3,5-arylated 2-pyrazolines, Molecules 8 (2003) 642-648. |
| [22] | D. Azarifar, M. Shaebanzadeh, Synthesis and characterization of new 3,5-dinaphthyl substituted 2-pyrazolines and study of their antimicrobial activity, Molecules 7 (2002) 885-895. |
| [23] | M.A. Ali, M.S.A.A. Siddiqui, Synthesis, structural activity relationship and antitubercular activity of novel pyrazoline derivatives, Eur. J. Med. Chem. 42 (2007) 268-275. |
| [24] | M. Amir, H. Kumar, S.A. Khan, Synthesis and pharmacological evaluation of pyrazoline derivatives as new anti-inflammatory and analgesic agents, Bioorg. Med. Chem. Lett. 18 (2008) 918-922. |
| [25] | M. Kidwai, S. Kukreja, R. Thakur, K2CO3-mediated regioselective synthesis of isoxazoles and pyrazolines, Lett. Org. Chem. 3 (2006) 135-139. |
| [26] | H.M. Hassaneen, A.S. Shawali, N.M. Elwaln, et al., Synthesis and cycloaddition reactions of C-(2-naphthoyl)-N-arylmethanohydrazonylpyridinium bromides, Arch. Pharm. Res. 15 (1992) 292-297. |
| [27] | H.M. Hassaneen, R.H. Hilal, N.M. Elwan, et al., The regioselectivity in the formation of pyrazolines and pyrazoles from nitrilimines, Heterocycl. Chem. 21 (1984) 1013-1016. |
| [28] | H.M. Hassaneen, H.A.H. Mousa, A.S. Shawali, Chemistry of C-heteroarylnitrilimines. Synthesis and cycloaddition reactions of N-phenyl-C-(2-thienyl)nitriliminem, J. Heterocycl. Chem. 24 (1987) 1665-1668. |
| [29] | H.M. Hassaneen, H.A.H. Mousa, N.M. Abed, A.S. Shawali, Chemistry of C-heteroarylhydrazidoyl halides. Synthesis and reactions of N-(p-Nitrophenyl)-C-(2-thienyl)-formohydrazidoyl halides, Heterocycles 27 (1988) 695-706. |