b Department of Chemistry, Sanandaj Branch, Islamic Azad University, Sanandaj 720, Iran
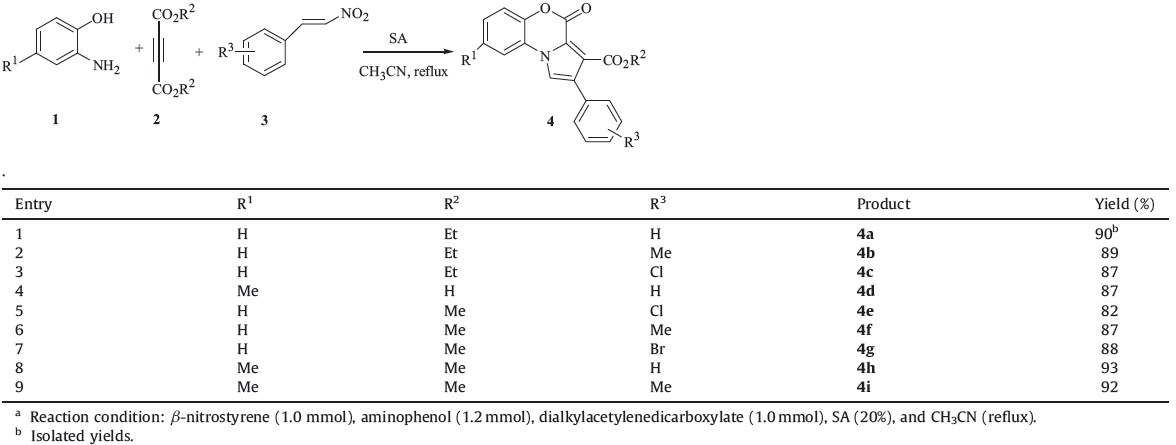
During the last two decades, multicomponent reactions have been exploited by combinatorial chemists as a source of appendage diversity. One-pot multicomponent reactions are often resourceeffective and environmentally acceptable and, thus, greener as compared to multistep reactions. They can offer significant advantages over conventional single-step syntheses by reducing reaction time and saving money, energy, and raw materials, thus resulting in both economical and environmental benefits. At the same time, diversity can be achieved by building up libraries by varying each component [1]. 1,4-Benzoxazine ring systems are found in a broad range of natural and synthetic biologically active molecules [2, 3, 4, 5, 6, 7, 8, 9]. Recently, pharmacological evaluation and structure- activity relationships of some of simple or fused pyrrolo-1,4- benzoxazines have been reported [7, 8, 9]. Although pyrrolo-1,4- benzoxazines play important roles in drug discovery as biologically active compounds, the available strategies for the synthesis of these compounds are limited. So far, only a few methods have been described for the assembly of these tricyclic compounds, which have two common disadvantages: (i) The substituents to diversify products are limited. For example, it is difficult to incorporate diverse substituents on a pyrrole ring. (ii) The products were obtained in low yields because of the multistep reactions required or multiple byproducts incurred [6, 7, 8, 9]. Sulfamic acid (H2N3O3H, SA), a common inorganic acid, is nonvolatile, noncorrosive, stable, water resistance and incapable of forming metal complexes, making it an outstanding alternative to metal catalysts in different areas of organic synthesis as an efficient and green reagent [10]. As part of our current studies [11, 12, 13, 14] on the development of new routes to heterocyclic system, we report here a one-pot catalytic reaction involving 2-aminophenols 1, dialkylacetylenedicarboxylates 2, and nitrostyrene derivatives 3 in the presence of SA, which leads to pyrrolo-1,4-benzoxazine derivatives in high yields (Scheme 1).
|
Download:
|
| Scheme 1.Three-component synthesis of pyrrolo-1,4-benzoxazine derivatives 4. | |
Commercially available materials were used without further purifications. β-Nitrostyrene was synthesized according to the reported methods [15]. Melting points were measured on an Electrothermal 9100 apparatus. Mass spectra were recorded on an Agilent-5975C inert XL MSD mass spectrometer operating at an ionization potential of 70 eV. Elemental analyses were performed using a Heraeus CHN-O - Rapid analyzer. 1H NMR and 13C NMR spectra were measured (in CDCl3 solution) with a Bruker DRX-250 Avance spectrometer at 250.1 MHz, and 62.9 MHz, respectively. IR spectra were recorded on a Shimadzu IR-460 spectrometer.
General procedure for the preparation of compounds 4a-j:Take 4a for example. In a round bottom flask equipped with a magnetic stirrer, diethyl acetylenedicarboxylate (1 mmol) and 2-amino phenol (1.2 mmol) in CH3CN (3 mL) were charged and the mixture was stirred vigorously at room temperature. Then, β-nitrostyrene (1 mmol) and SA (20 mol%) were added to the mixture and the mixture was refluxed for 24 h open to air until TLC analysis indicated the total consumption of the starting materials. Upon completion, the reaction mixture was cooled to room temperature and then poured into 3 mL of water. The solid product was removed by filtration and purified by recrystallization from 95% ethanol to afford the pure title compounds.
Methyl 4-oxo-2-phenyl-4H-benzo[b]pyrrolo[1,2-d][1, 4]oxazine- 3-carboxylic acid (4a): Gray powder, yield 0.30 g (90%); mp: 181-183 ℃. IR (KBr) (υmax/cm-1): 1732 (C=O of ester), 1740 (C=O of ester), 1250 (C-O of ester), 1410, 1606 (Ar). 1H NMR (CDCl3, 250.1 MHz): δ 1.31 (t, 3H, 3JHH = 6.9 Hz, CH3), 4.41 (q, 2H, 3JHH = 6.9 Hz, OCH2), 7.33-7.68 (m, 10H, 10CH aromatic). 13C NMR (CDCl3, 62.9 MHz): δ 13.9 (CH3), 61.9 (OCH2), 114.5 (C), 115.8 (C), 118.3 (CH), 121.5 (CH), 122.5 (CH), 125.1 (C), 127.1 (CH), 127.8 (CH), 127.9 (C), 128.0 (CH), 128.7 (2CH), 129.2 (2CH), 132.0 (C), 143.0 (C), 152.0 (C=O), 164.7 (C=O). Anal. calcd. for C20H15NO4 (333.35): C, 72.06; H, 4.54; N, 4.20; O, 19.20. Found: C, 72.12; H, 4.25; N, 4.56.
Methyl 4-oxo-2-(p-tolyl)-4H-benzo[b]pyrrolo[1,2-d][1, 4]oxazine- 3-carboxylate (4b): White powder; yield 0.31 g (89%); mp: 173-175 ℃. IR (KBr) (υmax/cm-1): 1731 (C=O of ester), 1740 (C=O of ester), 1241 (C-O of ester), 1417, 1607 (Ar). 1H NMR (CDCl3, 250.1 MHz): δ 1.35 (t, 3H, 3JHH = 7.0 Hz, CH2 CH3), 2.12 (s, 3H, CH3), 4.37 (q, 2H, 3JHH = 7.0 Hz, OCH2), 7.18-7.34 (m, 9H, 9CH aromatic). 13C NMR (CDCl3, 62.9 MHz): δ 13.9 (CH2CH3), 21.2 (CH3), 62.0 (OCH2), 114.4 (C), 115.5 (C), 118.4 (CH), 121.6 (CH), 122.4 (CH), 125.0 (C), 127.1 (CH), 127.6 (2C), 129.0 (CH), 129.2 (2CH), 129.4 (2CH), 137.8 (C), 143.0 (C), 152.1 (C=O), 164.8 (C=O). MS (m/z): 347 (M+, 100), 302 (74), 274 (42). Anal. calcd. for C21H17NO4 (347.37): C, 72.61; H, 4.93; N, 4.03. Found: C, 72.40; H, 5.00; N, 4.26.
Methyl 2-(4-chlorophenyl)-4-oxo-4H-benzo[b]pyrrolo[1,2- d][1, 4]oxazine-3-carboxylate (4c): White powder; yield 0.32 g (87%); mp = 193-195 ℃. IR (KBr) (υmax/cm-1): 1726 (C=O of ester), 1736 (C=O of ester), 1245 (C-O of ester), 1416, 1609 (Ar). 1H NMR (CDCl3, 250.1 MHz): δ 1.29 (t, 3H, 3JHH = 6.7 Hz, CH3), 4.39 (q, 2H, 3JHH = 6.7 Hz, OCH2), 7.26-7.67 (m, 9H, 9CH aromatic). 13C NMR (CDCl3, 62.9 MHz): δ 13.9 (CH3), 62.0 (OCH2), 114.4 (C), 115.5 (C), 115.9 (C), 118.4 (CH), 121.5 (CH), 122.4 (CH), 125.1 (CH), 127.3 (C), 128.1 (CH), 128.9 (2CH), 129.1 (2CH), 130.6 (C), 134.0 (C), 143.1 (C), 151.8 (C=O), 164.4 (C=O). MS (m/z): 367 (M+, 100), 322 (83), 295 (44). Anal. calcd. for C20H14ClNO4 (367.79): C, 65.31; H, 3.84; N, 3.81. Found: C, 65.20; H, 4.00; N, 3.66.
Methyl 4-oxo-2-phenyl-4H-benzo[b]pyrrolo[1,2-d][1, 4]oxazine- 3-carboxylate (4d): Gray powder; yield 0.28 g (87%); mp: 200-202 ℃. IR (KBr) (υmax/cm-1): 1719 (C=O of ester), 1725 (C=O of ester), 1248 (C-O of ester), 1411, 1623 (Ar). 1H NMR: (CDCl3, 250.1 MHz): δ 3.92 (s, 3H, OCH3), 7.26-7.69 (m, 10H, 10CH aromatic).13C NMR (CDCl3, 62.9 MHz) δ 52.8 (OCH3), 114.4 (C), 115.6 (C), 118.4 (CH), 121.6 (CH), 122.0 (CH), 125.1 (C), 127.2 (CH), 127.7 (CH), 127.9 (C), 128.1 (CH), 128.8 (2CH), 129.3 (2CH), 132.0 (C), 143.1 (C), 152.0 (C=O), 164.7 (C=O). MS (m/z): 319 (M+, 98), 288 (100), 260 (75). Anal. calcd. for C19H13NO4 (319.32): C, 71.47; H, 4.10; N, 4.39. Found: C, 71.30; H, 4.00; N, 4.56.
Methyl 2-(4-chlorophenyl)-4-oxo-4H-benzo[b]pyrrolo[1,2- d][1, 4]oxazine-3-carboxylate (4e): Gray powder; yield 0.29 g (82%); mp: 198-200 ℃. IR (KBr) (υmax/cm-1): 1725 (C=O of ester), 1740 (C=O of ester), 1250 (C-O of ester), 1412, 1607 (Ar). 1H NMR (CDCl3, 250.1 MHz): δ 3.64 (s, 3H, OCH3), 7.26-7.67 (m, 9H, 9CH aromatic). 13C NMR (CDCl3, 62.9 MHz): δ 52.9 (OCH3), 114.5 (C), 115.7 (C), 115.9 (C), 118.4 (CH), 121.4 (CH), 121.8 (CH), 125.1 (CH), 127.4 (C), 128.1 (CH), 129.0 (2CH), 129.1 (2CH), 130.5 (C), 134.0 (C), 143.1 (C), 151.9 (C=O), 164.9 (C=O). Anal. calcd. for C19H12ClNO4 (353.76): C, 64.51; H, 3.42; N, 3.96. Found: C, 64.62; H, 3.50; N, 3.87.
Methyl 4-oxo-2-(p-tolyl)-4H-benzo[b]pyrrolo[1,2-d][1, 4]oxazine- 3-carboxylate (4f): Gray powder; yield 0.29 g (87%); mp: 175-177 ℃. IR (KBr) (υmax/cm-1): 1730 (C=O of ester), 1740 (C=O of ester), 1242 (C-O of ester), 1414, 1606 (Ar). 1H NMR (CDCl3, 250.1 MHz): δ 2.36 (s, 3H, CH3), 3.92 (s, 3H, OCH3), 7.20-7.65 (m, 9H, 9CH aromatic). 13C NMR (CDCl3, 62.9 MHz): δ 21.2 (CH3), 52.9 (OCH3), 114.5 (C), 115.6 (C), 118.4 (CH), 121.6 (CH), 122.4 (CH), 125.1 (C), 127.1 (CH), 127.5 (2C), 129.0 (CH), 129.2 (2CH), 129.5 (2CH), 137.8 (C), 143.0 (C), 152.1 (C=O), 165.3 (C=O). Anal. calcd. for C20H15NO4 (333.35): C, 72.06; H, 4.54; N, 4.20. Found: C, 72.20; H, 4.31; N, 4.16.
Methyl 2-(3-bromophenyl)-4-oxo-4H-benzo[b]pyrrolo[1,2- d][1, 4]oxazine-3-carboxylate (4g): Gray powder; yield 0.35 g (88%); mp: 140-142 ℃. IR (KBr) (υmax/cm-1): 1723 (C=O of ester), 1732 (C=O of ester), 1250 (C-O of ester), 1424, 1593 (Ar). 1H NMR (CDCl3, 250.1 MHz): δ 3.93 (s, 3H, OCH3), 7.26-7.61 (m, 9H, 9CH aromatic). 13C NMR (CDCl3, 62.9 MHz): δ 52.9 (OCH3), 114.5 (C), 115.9 (C), 116.0 (C), 118.4 (CH), 121.4 (CH), 122.7 (CH), 125.2 (CH), 126.4 (C), 127.4 (CH), 127.8 (2CH), 130.3 (2CH), 130.7 (C), 130.9 (C), 143.0 (C), 151.9 (C=O), 164.8 (C=O). Anal. calcd. for C19H12BrNO4 (398.22): C, 57.31; H, 3.04; N, 3.52. Found: C, 57.20; H, 3.12; N, 3.61.
Methyl 8-methyl-4-oxo-2-phenyl-4H-benzo[b]pyrrolo[1,2- d][1, 4]oxazine-3-carboxylate (4h): Gray powder; yield 0.31 g (93%); mp: 177-179 ℃. IR (KBr) (υmax/cm-1): 1720 (C=O of ester), 1730 (C=O of ester), 1250 (C-O of ester), 1424, 1615 (Ar). 1H NMR (CDCl3, 250.1 MHz): δ 2.20 (s, 3H, CH3), 3.92 (s, 3H, OCH3), 7.12-7.67 (m, 9H, 9CH aromatic). 13C NMR (CDCl3, 62.9 MHz): d 21.1 (CH3), 52.9 (OCH3), 114.6 (C), 115.5 (C), 115.8 (CH), 118.1 (C), 121.1 (CH), 121.8 (CH), 127.7 (CH), 127.9 (2CH), 128.8 (C), 128.9 (CH), 129.2 (2CH), 132.1 (C), 135.2 (C), 141.0 (C), 152.3 (C=O), 165.3 (C=O). Anal. calcd. for C20H15NO4 (333.35): C, 72.06; H, 4.54; N, 4.20. Found: C, 72.20; H, 4.40; N, 4.56.
Methyl 8-methyl-4-oxo-2-(p-tolyl)-4H-benzo[b]pyrrolo[1,2- d][1, 4]oxazine-3-carboxylate (4i): White powder; yield 0.32 g (92%); mp: 203-205 ℃. IR (KBr) (υmax/cm-1): 1735 (C=O), 1740 (C=O), 1251 (C-O). 1H NMR (CDCl3, 250.1 MHz): δ 2.35 (s, 6H, 2CH3), 3.92 (s, 3H, OCH3), 7.12-7.62 (m, 8H, ℃H aromatic). 13C NMR (CDCl3, 62.9 MHz): δ 21.1 (CH3), 21.2 (CH3), 52.8 (OCH3), 114.5 (C), 115.3 (C), 115.7 (CH), 117.9 (C), 121.1 (CH), 121.7 (CH), 127.4 (CH), 127.8 (2CH), 129.1 (C), 129.2 (CH), 129.5 (2CH), 135.2 (C), 137.7 (C), 140.9 (C), 152.3 (C=O), 165.4 (C=O). Anal. calcd. for C21H17NO4 (347.37): C, 72.61; H, 4.93; N, 4.03. Found: C, 72.40; H, 4.81; N, 4.36.
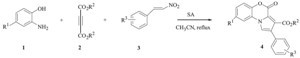
First, we studied the reaction of 2-amino phenol, diethyl acetylenedicarboxylate and β-nitrostyrene in order to optimize the reaction conditions with respect to temperature, time, and the molar ratio of SA to the substrates. We found that 20 mol% of SA was sufficient to obtain the corresponding pyrrolo-1,4-benzoxazine 4a in 90% yield within 24 h in CH3CN under reflux conditions. The reaction was run without SA as catalyst and we did not obtain any product. We also examined other solvents such as methanol, 1,2-dichloroethane, tetrahydrofuran and toluene. When methanol, toluene, tetrahydrofuran or 1,2-dichloroethane was employed as solvents under reflux conditions, approximately the same results as in acetonitrile were obtained. With the optimized conditions in hand, the three-component reaction of 2-aminophenols, dialkylacetylenedicarboxylates and β-nitrostyrene derivatives for the synthesis of pyrrolo-1,4-benzoxazines were studied. The results are summarized in Table 1. The yields were not sensitive to the substrates employed. Nearly all reactions give similar isolated yields, and the reaction is clean with no side products observed. The 1H NMR and 13C NMR spectra of the crude products clearly indicated the formation of pyrrolo-1,4-benzoxazine 4a-i. Products other than 4 could not be detected by NMR spectroscopy.
| Table 1 Synthesis of novel pyrrolo-1,4-benzoxazines.a |
The structures of compounds 4a-i were deduced from their elemental analyses data, IR, 1H NMR and 13C NMR spectra. For example, the 1H NMR spectrum of 4d exhibited one singlet recognized as arising from the methoxy group (δ 3.92) protons. The aromatic moiety gave rise to characteristic signals in the aromatic region of the spectrum. The IR spectrum of 4d exhibited absorption bands due to carbonyl groups of esters at 1719 cm-1 and 1725 cm-1. The mass spectrum of 4d displayed molecular ion peak atm/z of 319. The 1H NMR decoupled 13CNMR spectrum of 4d showed 17 distinct resonances that confirm the proposed structure.
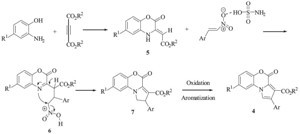
Although we have not experimentally established the mechanism of this reaction, a possible explanation is proposed in Scheme 2. On the basis of well-established chemistry of o-aminophenols and dimethyl acetylenedicarboxylate [16] reaction between 2-amino phenol and dialkyl acetylenedicarboxylate affords benzoxazine 5. Compound 5 possesses enamine characters [17] and thus readily reacts with β-nitrostyrene, which is activated by SA to generate the intermediate 6. The subsequent cyclization of intermediate 6 followed by an intramolecular substitution of the protonated nitro group leads to pyrrole precursors 7. The airmediated oxidative dehydrogenation of 7 results in the formation of pyrrolo-1,4-benzoxazines 4.
|
Download:
|
| Scheme 2.Possible mechanism for the synthesis of pyrrolo-1,4-benzoxazines derivatives 4. | |
In summary, the reaction involving 2-aminophenols, dialkylacetylenedicarboxylates and nitrostyrene derivatives in CH3CN provides a simple method for the synthesis of novel pyrrolo-1,4- benzoxazine derivatives of synthetic and pharmacological interest. The simplicity of the present procedure makes it an interesting alternative to other approaches. It has been noticed so far, that the modifications on the pyrrole moiety in pyrrolo-1,4-benzoxazines displayed valuable biological activity and these modifications can be utilized to develop potentially active agents in the future. Thus, the exploration of the pyrrole modifications needs to continue.
| [1] | A. Dömling, Recent developments in isocyanide based multicomponent reactions in applied chemistry, Chem. Rev. 106 (2006) 17-89. |
| [2] | B. Achari, S.B. Mandal, P.K. Dutta, C. Chowdhury, Perspectives on 1,4-benzodioxins, 1,4-benzoxazines and their 2,3-dihydro derivatives, Synlett (2004) 2449-2467. |
| [3] | J.H. Wu, F.R. Chang, K. Hayashi, et al., Antitumor agents. part 218: cappamensin a, a new in vitro anticancer principle, from capparis sikkimensis, Biorg. Med. Chem. Lett. 13 (2003) 2223-2225. |
| [4] | J. Bergman, S. Rosenzweig-Lipson, R.D. Spealman, Differential effects of dopamine D1 and D2 receptor agonists on schedule-controlled behavior of squirrel monkeys, J. Pharmacol. Exp. Ther. 273 (1995) 40-48. |
| [5] | P.J. Rybczynski, R.E. Zeck, D.W. Combs, et al., Benzoxazinones as PPARγ agonists. Part 1: SAR of three aromatic regions, Biorg. Med. Chem. Lett. 13 (2003) 2359-2362. |
| [6] | G. Campiani, A. Garofalo, I. Fiorini, et al., Pyrrolo[2,1-c][1,4]benzothiazines: synthesis, structure-activity relationships, molecular modeling studies, and cardiovascular activity, J. Med. Chem. 38 (1995) 4393-4410. |
| [7] | D.S. Snyder, L. Tradtrantip, C. Yao, M.J. Kurth, A.S. Verkman, Potent, metabolically stable benzopyrimido-pyrrolo-oxazine-dione (BPO) CFTR inhibitors for polycystic kidney disease, J. Med. Chem. 54 (2011) 5468-5477. |
| [8] | X.Q. Xie, X.W. Han, J.Z. Chen, M. Eissenstat, A. Makriyannis, High-resolution NMR and computer modeling studies of the cannabimimetic aminoalkylindole prototype WIN-55212-2, J. Med. Chem. 42 (1999) 4021-4027. |
| [9] | S. Kurhade, R.D. Kaduskar, B. Dave, et al., Synthesis of a novel tetracyclic azaindolo[ 2,1-c][1,4]benzoxazine ring system, Tetrahedron Lett. 52 (2011) 1874-1877. |
| [10] | A. Santra, G. Guchhait, A.K. Misra, Efficient acylation and sulfation of carbohydrates using sulfamic acid, a mild, eco-friendly catalyst under organic solvent-free conditions, Green Chem. 13 (2011) 1345-1351. |
| [11] | I. Yavari, A. Mirzaei, L. Moradi, A. Mokhtarporiani-Sanandaj, Efficient synthesis of trialkyl (E)-3-{3-Oxo-2-3,4-dihydro-2-(1H)-quinoxalinylidene}prop-1-ene-1,2,3-tricarboxylates, Synth. Commun. 37 (2007) 1195-1200. |
| [12] | I. Yavari, L. Moradi, Synthesis of isoxazoles through the reaction of activated acetylenes and alkyl 2-nitroethanoates in the presence of triphenylphosphine, Tetrahedron Lett. 47 (2006) 1627-1629. |
| [13] | I. Yavari, M. Piltan, L. Moradi, Synthesis of pyrrolo[2,1-a]isoquinolines from activated acetylenes, benzoylnitromethanes, and isoquinoline, Tetrahedron 65 (2009) 2067-2071. |
| [14] | M. Piltan, I. Yavari, L. Moradi, S.A. Zarei, PEG-Mediated catalyst-free expeditious synthesis of functionalized benzene/biaryl and fluoren-9-one derivatives from activated acetylenes and 1,3-diones, J. Korean Chem. Soc. 56 (2012) 316-321. |
| [15] | J. Christoffers, H. Frey, A. Rosiak, in: B. Pleitker (Ed.), Iron Catalysis in Organic Chemistry, Wiley-VCH, Weinheim, 2008, pp. 217-244. |
| [16] | N. Kawahara, T. Shimamori, T. Itoh, H. Ogura, Thermal reactions of o-aminophenols and dimethyl actylenedicarboxylate, Heterocycles 24 (1986) 2803-2807. |
| [17] | I. Yavari, A. Mirzaei, L. Moradi, Efficient synthesis of dialkyl (2Z)-2-[(E)-1-Aryl-2-(3-arylquinoxalin-2-yl)ethenyl]but-2-enedioates, Helv. Chim. Acta 89 (2006) 2825-2829. |