Dihydropyrimidinones (DHPMs) and their derivatives are important classes of compounds in the field of pharmaceuticals and exhibit significant biological properties such as antiviral, antihypertensive and antibacterial as well as anti-HIV and antitumor activities [1]. Some of them are also successfully used as calcium channel blockers and α-1a-antagonists [2, 3, 4, 5, 6].
Because of their importance and wide range of applications, different types of methods are reported for the preparation of DHPMs which of them the Biginelli reaction is the most important ones.
This reaction is based on a one-pot condensation reaction of β-dicarbonyl compounds with aldehydes and urea or thiourea in the presence of an acid catalyst [7]. The method although is useful but suffer from the use of HCl.
In continuation of this study different types of catalysts are reported for the promotion of the Biginelli reaction which of them silica-bonded S-sulfonic acid (SBSSA), sulfonated carbon materials (SCM), bentonite/PS-SO3H, MCM-41-R-SO3H [8, 9, 10, 11], zeolites [12], HClO4-SiO2 [13], Yb(OTf)2, Cu(OTf)2, Zn(OTf)2, and Bi(OTf)2 [14] and different types of ionic liquids [15, 16, 17], melamine trisulfonic acid (MTSA), ZrOCl2⋅8H2O, silica triflate, Fe(HSO4)3, trichloro isocyanuric acid (TCCA) [18, 19, 20, 21, 22] are examples.
Although, these methods are an improvement many of them suffer from disadvantages such as harsh reaction conditions, use of harmful organic solvents, long reaction times, tedious work-up procedure, expensive and moisture sensitive reagents, strongly acidic conditions, unsatisfactory yields, non-recoverability of the catalyst and environmental pollution.
Therefore, introduction of an inexpensive and efficient reagent for this reaction under milder and greener conditions is still in demand.
Chemicals were purchased from Fluka, Merck and Aldrich Chemical Companies. All yields refer to the isolated products. The purity determination of the substrate and reaction monitoring were accompanied by thin-layer chromatography (TLC) on a silicagel polygram SILG/UV 254 plates. Products were separated and purified by different chromatographic techniques and were identified by the comparison of their IR, NMR and refractive index with those reported for the authentic samples. All yields refer to the isolated products.
The IR spectra were run on a Perkin-Elmer bio-spectrometer. The 1H NMR (300 MHz) was run on a Bruker Avance DPX-300 FTNMR spectrometer (δ in ppm). Melting points were recorded on a Büchi B-545 apparatus in open capillary tubes.
A mixture of the aldehyde (1 mmol), 1,3-dicarbonyl compound (1 mmol), urea (or thiourea) (1.2 mmol) and NSPVPC (20 mg) was heated in an oil-bath (100 ℃) under solvent-free conditions. After completion of the reaction (monitored by TLC) the reaction mixture was cooled to room temperature, EtOH (5 mL) was added and filtered to separate the catalyst. Then the crude product was recrystallized from EtOH to give the pure product. Spectroscopic data for the selected products are as follows:
Ethyl-6-methyl-2-oxo-4-phenyl-1,2,3,4-tetrahydropyrimidine-5- carboxylate: IR (KBr, cm-1): υ 3245, 3118, 2978, 1725, 1701, 1649; 1H NMR (300 MHz, DMSO-d6): δ 1.06 (t, 3H, J = 6.84 Hz), 2.24 (s, 3H), 3.94 (q, 2H, J = 6.75 Hz), 5.13 (d, 1H, J = 3.06 Hz), 7.21-7.37 (m, 5H), 7.37 (brs, 1H), 9.19 (brs, 1H).
4-(4-Methoxyphenyl)-6-phenyl-pyrimidin-2(1H)-one: IR (KBr, cm-1): y 3436, 3247, 3113, 2929, 1724, 1705, 1649; 1H NMR (300 MHz, DMSOd6): δ 1.10 (t, 3H, J = 7.08 Hz), 2.17 (s, 3H), 3.71 (s, 3H), 3.96 (q, 2H, J = 7.14 Hz), 5.07 (d, 1H, J = 2.97 Hz), 6.85 (d, 2H, J = 8.49 Hz), 7.12 (d, 2H, J = 8.55 Hz), 7.67 (brs, 1H), 9.15 (brs, 1H).
Very recently we have reported the preparation of N-sulfonic acid poly(4-vinylpyridinum) chloride (NSPVPC) and its application in different types of organic reactions [23, 24, 25].
Herein, and in continuation of these studies we wish to report the applicability of this reagent in the promotion of the synthesis of DHPMs under solvent-free conditions.
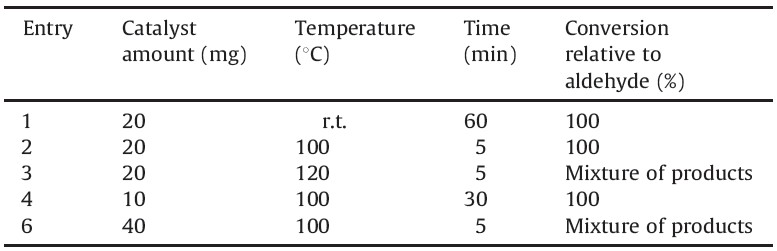
To optimize the reaction conditions the reaction of 4- nitrobenzaldehyde, ethyl acetoacetate and urea was studied in different conditions in the presence of different amounts of NSPVPC (Table 1). The obtained results showed that the reaction using 20 mg of the catalyst at 100 ℃ proceeded with highest yield in shorter reaction times (Table 1, entry 2).
| Table 1 Effects of temperature and the amount of catalyst on the Biginelli reaction under solvent-free conditions. |
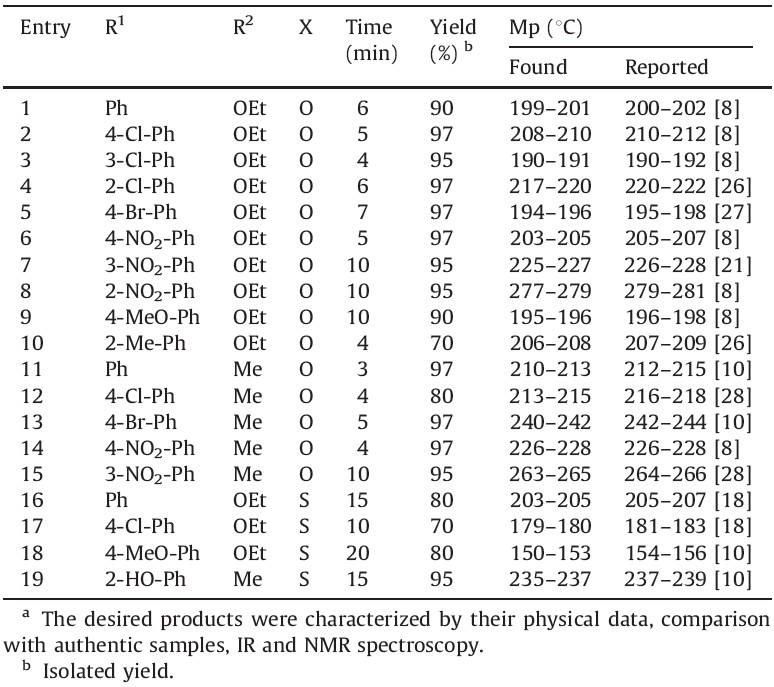
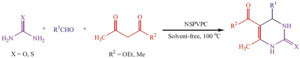
To assess the efficiency of NSPVPC in the preparation of 3,4- dihydropyrimidin-2(1H)-ones derivatives, other aromatic aldehydes were also reacted under the optimal reaction conditions to furnish the corresponding products in good to high yields in very short reaction times (Scheme 1). It is clear from Table 2.
|
Download:
|
| Scheme 1.NSPVPC catalyzed synthesis of 3,4-dihydropyrimidin-2(1H)-ones and -thiones. | |
| Table 2 Preparation of 3,4-dihydropyrimidin-2(1H)-ones and -thiones using NSPVPC as the catalyst under solvent-free conditions.a |
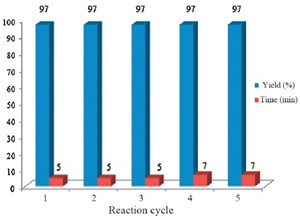
In continue, we decided to study the catalytic activity of the recycled catalyst for the synthesis of DHPMs. After the separation of the product, the catalyst was washed with EtOAc and acetone and derived at 50 ℃. As shown in Fig. 1, NSPVPC can be recycled at least five times without significant decrease in its activity. It should be note that the IR spectra of the recovered catalyst was the same as the catalyst used for the first run and also pH analysis of the recovered catalyst showed nearly the same loading of H+ as the freshly prepared catalyst. These observations suggest that the nature of the catalyst remains intact after each run and leaching of the acid species is not occurred during the course of the reaction.
|
Download:
|
| Fig. 1.Recyclability of the catalyst. | |
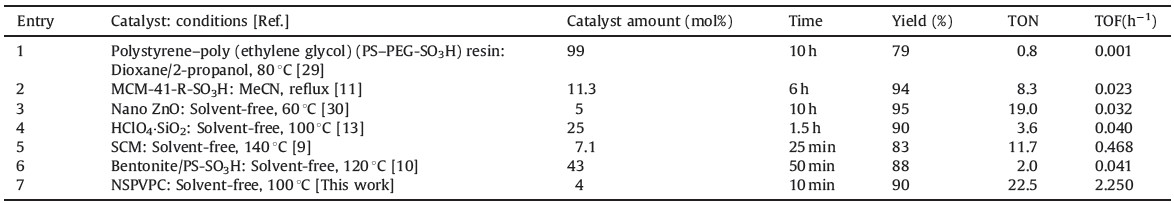
| Table 3 Comparison of the results of the reaction of ethyl acetoacetate, urea with 4-OMePhCHO using NSPVPC with those obtained by reported catalysts. |
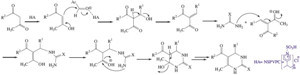
A plausible mechanism for the Biginelli reaction in the presence of NSPVPC is shown in Scheme 2.
|
Download:
|
| Scheme 2.Proposed mechanism of the reaction. | |
In summary, we have introduced NSPVPC as a highly efficient catalyst for the synthesis of 3,4-dihydropyrimidin-2(1H)-ones and -thiones via the one-pot multi-component condensation reactions. This method in accompanied will several advantages such as: ease of preparation and handling of the catalyst, simplicity and easy work-up, high reaction rates and excellent yields. Also, the recovered catalyst can be reused several times without significant decrease in its activity.
The authors are thankful to the University of Guilan Research Council for the partial support of this work.
| [1] | C.O. Kappe, 100 Years of the Biginelli dihydropyrimidine synthesis, Tetrahedron 49 (1993) 6963-6963. |
| [2] | C.O. Kappe, Biologically active dihydripyrimidones of the Bignelli-type. A literature survey, Eur. J. Med. Chem. 35 (2000) 1043-1052. |
| [3] | C.O. Kappe, D. Kummar, R.S. Varma, Microwave-assisted high-speed parallel synthesis of 4-aryl-3,4-dihydropyrimidin-2(1H)-ones using a solventless Biginelli condensation protocol, Synthesis (1999) 1799-1803. |
| [4] | K.S. Atwal, G.C. Rovnyak, S.D. Kimball, et al., Dihydropyrimidine calcium channel blockers. 2. 3-substituted-4-aryl-1,4-dihydro-6-methyl-5-pyrimidine carboxylic acid esters as potent mimics of dihydropyridines, J. Med. Chem. 33 (1990) 2629-2635. |
| [5] | C.O. Kappe, O.V. Shishkin, G. Uray, P. Verdino, X-ray structure, conformational analysis, enantioseparation and determination of absolute configuration of the mitotic kinesin Eg5 inhibitor monastrol, Tetrahedron 56 (2000) 1859-1862. |
| [6] | G.C. Rovynal, S.D. Kimball, B. Beyer, et al., Calcium entry blockers and activators: conformational and structural determinants of dihydropyrimidine calcium channel modulators, J. Med. Chem. 38 (1995) 119-129. |
| [7] | P. Biginelli, Aldehyde-urea derivatives of aceto-and oxaloacetic acids, Gazz. Chim. Ital. 23 (1893) 360-413. |
| [8] | M. Tajbakhsh, Y. Ranjbar, A. Masuodi, S. Khaksar, A simple and environmentally benign protocol for Biginelli reactions catalyzed by silica-bonded S-sulfonic acid, Chin. J. Catal. 33 (2012) 1542-1545. |
| [9] | M. Moghaddas, A. Davoodnia, M.M. Heravi, N. Tavakoli-Hoseini, Sulfonated carbon catalyzed Biginelli reaction for one-pot synthesis of 3,4-dihydropyrimidin-2(1H)-ones and -thiones, Chin. J. Catal. 33 (2012) 706-710. |
| [10] | R. Javad Kalbasi, A.R. Massah, B. Daneshvarnejad, Preparation and characterization of bentonite/PS-SO3H nanocomposites as an efficient acid catalyst for the Biginelli reaction, Appl. Clay Sci. 55 (2012) 1-9. |
| [11] | G.H. Mahdavinia, H. Sepehrian, MCM-41 anchored sulfonic acid (MCM-41-RSO3H): a mild, reusable and highly efficient heterogeneous catalyst for the Biginelli reaction, Chin. Chem. Lett. 19 (2008) 1435-1439. |
| [12] | S.R. Mistry, K.C. Maheria, Synthesis of diarylpyrimidinones (DAPMs) using large pore zeolites, J. Mol. Catal. A: Chem. 355 (2012) 210-215. |
| [13] | S.R. Narahari, B.R. Reguri, O. Gudaparthi, K. Mukkanti, Synthesis of dihydropyrimidinones via Biginelli multi-component reaction, Tetrahedron Lett. 53 (2012) 1543-1545. |
| [14] | (a) A. Dondoni, A. Massi, Parallel synthesis of dihydropyrimidinones using Yb(Ⅲ)-resin and polymer-supported scavengers under solvent-free conditions. A green chemistry approach to the Biginelli reaction, Tetrahedron Lett. 42 (2001) 7975-7978; (b) A.S. Paraskar, G.K. Dewkar, A. Sudalai, Cu(OTf)2: a reusable catalyst for highyield synthesis of 3,4-dihydropyrimidin-2(1H)-ones, Tetrahedron Lett. 44 (2003) 3305-3308; (c) H. Xu, Y.G. Wang, A rapid and efficient Biginelli reaction catalyzed by zinc triflate, Chin. J. Chem. 21 (2003) 327-331; (d) R. Varala, M.M. Alam, S.R. Adapa, Bismuth triflate catalyzed one-pot synthesis of 3,4-dihydropyrimidin-2 (1H)-ones: an improved protocol for the Biginelli reaction, Synth. Lett. (2003) 67-70. |
| [15] | R.W. Zheng, X.X. Wang, H. Xu, J.X. Du, Brønsted acidic ionic liquid: An efficient and reusable catalyst for the synthesis of 3,4-dihydropyrimidin-2(1H)-ones, Synth. Commun. 36 (2006) 1503-1513. |
| [16] | M. Li, W.S. Guo, L.R. Wen, Y.F. Li, H.Z. Yang, One-pot synthesis of Biginelli and Hantzsch products catalyzed by non-toxic ionic liquid (BMImSac) and structural determination of two products, J. Mol. Catal. A 258 (2006) 133-138. |
| [17] | J.J. Peng, Y.Q. Deng, Ionic liquids catalyzed Biginelli reaction under solvent-free conditions, Tetrahedron Lett. 42 (2001) 5917-5919. |
| [18] | F. Shirini, M.A. Zolfigol, J. Albadi, Melamine trisulfonic acid: a new, efficient and recyclable catalyst for the synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones in the absence of solvent, Chin. Chem. Lett. 22 (2011) 318-321. |
| [19] | F. Shirini, M.A. Zolfigol, E. Mollarazi, ZrOCl2.8H2O as an efficient reagent for the solvent-free synthesis of 3,4-dihydropyrimidin-2-(1H)-ones, Synth. Commun. 36 (2006) 2307-2310. |
| [20] | F. Shirini, K. Marjani, H.T. Nahzomi, Silica triflate as an efficient catalyst for the solvent-free synthesis of3,4-dihydropyrimidin-2(1H)-ones,Arkivoc 1 (2007)51-57. |
| [21] | F. Shirini, M.A. Zolfigol, A.R. Abri, Fe(HSO4)3 as an efficient catalyst for the preparation of 3,4-dihydropyrimidin-2(1H)-ones in solution and under solvent-free conditions, J. Iran. Chem. Soc. 5 (2008) 96-99. |
| [22] | F. Shirini, M.A. Zolfigol, E. Mollarazi, An efficient method for the synthesis of 3,4-dihydropyrimidin-2(1H)-ones (DHPMs) using trichloroisocyanuric acid (TCCA) under solvent-free conditions, Lett. Org. Chem. 2 (2005) 718-720. |
| [23] | F. Shirini, O.G. Jolodar, Introduction of N-sulfonic acid poly(4-vinylpyridinum) chloride as an efficient and reusable catalyst for the chemoselective 1,1-diacetate protection and deprotection of aldehydes, J. Mol. Catal. A: Chem. 356 (2012) 61-69. |
| [24] | F. Shirini, N.G. Khaligh, O.G. Jolodar, N-Sulfonic acid poly(4-vinylpyridinium) chloride: an efficient and reusable solid acid catalyst in N-Boc protection of amines, J. Iran. Chem. Soc. 10 (2013) 181-188. |
| [25] | F. Shirini, N.G. Khaligh, O.G. Jolodar, An efficient and practical synthesis of bis(indolyl)methanes catalyzed by N-sulfonic acid poly(4-vinylpyridinium) chloride, Dyes Pigments 98 (2013) 290-296. |
| [26] | R. Tayebee, M.M. Amini, M. Ghadamgahi, M. Armaghan, H5PW10V2O40/Pip-SBA-15: a novel reusable organic-inorganic hybrid material as potent Lewis acid catalyst for one-pot solvent-free synthesis of 3,4-dihydropyrimidinones, J. Mol. Catal. A: Chem. 366 (2013) 266-274. |
| [27] | H. Khabazzadeh, E. Tavakolinejad Kermani, T. Jazinizadeh, An efficient synthesis of 3,4-dihydropyrimidin-2(1H)-ones catalyzed by molte[Et3NH][HSO4], Arabian J. Chem. 5 (2012) 485-488. |
| [28] | A.R. Hajipour, M. Seddighi, Pyridinium-based Brønsted acidic ionic liquid as a highly efficient catalyst for one-pot synthesis of dihydropyrimidinones, Synth. Commun. 42 (2012) 227-235. |
| [29] | Z.J. Quan, Y.X. Da, Z. Zhang, X.C. Wang, PS-PEG-SO3H as an efficient catalyst for 3,4-dihydropyrimidones via Biginelli reaction, Catal. Commun. 10 (2009) 1146-1148. |
| [30] | F. Tamaddon, S. Moradi, Controllable selectivity in Biginelli and Hantzsch reactions using nanoZnO as a structure base catalyst, J. Mol. Catal. A Chem. 370 (2013) 117-122. |