b State Key Laboratory of Pollution Control and Resource Reuse & School of the Environment, Nanjing 210023, China
In recent years,the discharge ofwastewater containingCu2+ from electroplating,machinery and electronic industries have increased rapidly,which causes serious environmental problems [1]. In addition to coagulation,electric treatment and membrane,adsorption is an alternative technique for the removal of heavy metal ions from water [2, 3]. Although traditional granular ion-exchange and chelating resins have been developed for the treatment of heavy metal pollution,there still exist some shortcomings to be improved. For example,traditional granular resins need to be packed in columns and run under high pressure,which causes resins brittle. In addition,traditional granular resins cannot be added into the river or lake directly because of their difficult separation and recovery from the water. Thus,traditional granular resins could not be used to manage water pollution accidents on site.
In present work,we synthesize a novel spongy adsorbent with PVA (polyvinyl alcohol) sponge for the removal of Cu2+ from water effectively. The synthetic method and preparation process are simple. Compared to the traditional heavy metal capture agents, this new spongy adsorbent has higher mechanical strength and toughness,and it can also be separated from water more easily after adsorption. The spongy adsorbent was characterized by elementary analysis,SEM and FT-IR spectroscopy. XPS and FT-IR spectra were also used to clarify the adsorption mechanism of Cu2+ on PVA-MA-HH. Furthermore,the adsorption isotherm and desorption of Cu2+ on the new chelating sponge were studied. 2. Experimental
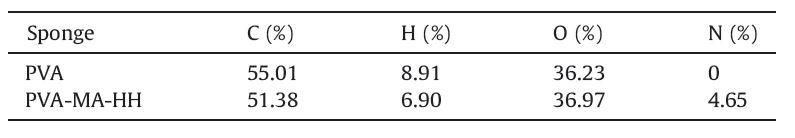
PVA sponge (Dontai Sponge Products Company,China) was cut into cubes with each side length of 7 mm. The PVA cubes were then extracted with acetone for 10 h in a Soxhlet apparatus and dried under vacuum at 323 K for 10 h. The synthesis process was shown in Scheme 1.
|
Download:
|
| Scheme 1. Preparation process of PVA-MA-HH. | |
The graft polymerization of methacrylate (MA) onto PVA was carried out by free radical polymerization. 2.5 g of PVA was soaked in 200 mL distilled water in a three-necked flask. Then the combined initiator system (BPO,Tween-80 and toluene) was added in the flask,and the mixture was vigorously shaken for 10 min. The monomer (MA) was added in,and the reaction mixture was allowed to stir for 6 h at 357 K. The products (PVA-MA) were filtered and washed with distilled water. The grafted polymer was dried at 313 K to a constant weight.
PVA-MA was added into 100 mL of 3% (v/v) hydroxylamine solution,and the pH value was adjusted to 12.5 by adding a sodium hydroxide solution (10%). Then the mixture was stirred for 12 h at 318 K. The final product named as PVA-MA-HH was filtered, washed with distilled water,and dried at 313 K to a constant weight. Then PVA-MA-HH was characterized by element analysis, FT-IR,SEM and XPS.
Adsorption isotherm study of Cu2+ on PVA-MA-HH was conducted according to a method reported in the literature [4]. The concentration of Cu2+ (ce) was determined using an atomic adsorption spectrophotometer. Thus,qe (mg/g),the adsorption capacity was calculated according to Eq. (1):

where V is the volume of solution (L),Wis the weight of dry sponge (g) and c0 is the initial concentration of Cu2+ in the solution.
For desorption studies,0.100 g of PVA-MA-HH was loaded with Cu2+ using a 100 mL Cu2+ (about 300 mg/L) solution at 293 K with contact time of 4 h. Cu2+-loaded PVA-MA-HH was collected and washed with distilled water. The sponge was then agitated with 20 mL of 0.1 mol/L EDTA and 1 mol/L HCl solution. The final concentration of the metal ion in the aqueous phase was determined. The desorption efficiency of Cu2+ from PVA-MA-HH was calculated from the amount of Cu2+ adsorbed on PVA-MA-HH and the final concentration of Cu2+ in the desorption medium. 3. Results and discussion
As shown in Table 1,nitrogen content of PVA-MA-HH increases after the graft polymerization and chemical modification, which indicates the existence of hydroxamic acid groups in PVA-MA-HH.
| Table 1 Results of E.A. of the sponge. |
The porous structure of PVA and PVA-MA-HH can be observed in the scanning electron microscopy (Fig. 1). But compared with PVA,PVA-MA-HH surface was more rough, which could be explained by the graft polymers and modified function groups.

|
Download:
|
| Fig. 1. SEM photos of sponge (a) PVA and (b) PVA-MA-HH. | |
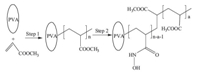
As shown in Fig. 2,the new chelating sponge PVA-MA-HH have distinctive absorption peaks compared with PVA: The peak in the range of 1720-1760 cm-1 is related to the ‘C55O’ in the ester groups,and the peak near 1580 cm-1 is due to ‘-N-H’ groups [5]. These observations indicate that ester and amino groups have been successfully grafted and modified on the PVA sponge.
|
Download:
|
| Fig. 2. FT-IR spectra of sponge. | |
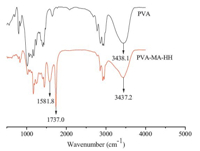
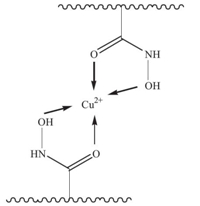
It was reported that chelating interactions could occur between heavy metal ions and the hydroxy groups [6]. As shown in Fig. 3, the ‘-O-H’ bond of PVA-MA-HH shifts from 3437.2 cm-1 to 3445.4 cm-1 after adsorption of Cu2+,which suggests that chelating interactions occur between the ‘-O-H’ groups and Cu2+ during the adsorption process. The adsorption mechanism of Cu2+ by PVA-MA-HH is shown in Scheme 2 [7, 8].
|
Download:
|
| Fig. 3. FT-IR spectra of PVA-MA-HH before and after adsorption of Cu2+. | |
|
Download:
|
| Scheme 2. Adsorption mechanism of Cu2+. | |
The XPS spectrum of PVA-MA-HH after adsorption of Cu2+ is
shown in Fig. 4. The peak at 934.4 eV indicates that the ‘Cu’ is
chelated by an ‘O’ [9],which is consistent with the adsorption
mechanism of Cu on PVA-MA-HH shown in Scheme 2.
The isothermal equilibrium adsorption data are fitted by the
Langmuir and Freundlich equations [3, 4]:
As shown in Table 2,both 0.1 mol/L EDTA and 1 mol/L HCl
solution can effectively desorb the Cu2+ from PVA-MA-HH.
A new chelating sponge modified with hydroxamic acid
groups was prepared with a graft polymer of PVA (polyvinyl
alcohol). The equilibrium adsorption capacity of PVA-MA-HH for
Cu2+ was about 73.23 mg/g. The Freundlich model could fit the
adsorption isotherm well,which indicated a multilayer adsorption
and the adsorption energy negatively correlated with the
surface coverage. The high value of ‘KF’ indicated a strong
adsorption force between PVA-MA-HH and Cu2+. Both 0.1mol/L
EDTA and 1 mol/L HCl solution could desorb Cu2+ from PVA-MAHH
effectively.
Acknowledgments
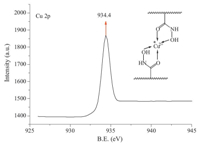
Fig. 4. XPS spectra of PVA-MA-HH after adsorption.


where qe is the equilibrium adsorption capacity (mg/g),ce is the
equilibrium concentration (mg/L). qm represents the monolayer
saturation adsorption capacity of the adsorbate and b the relative
constant. KF and n are characteristic constants. The constants
evaluated by application of isotherm equation are shown in Fig. 5.
Freundlich model is reliable because the correlation factor (R2) is
larger than 0.99,which indicates a multilayer adsorption and the
adsorption energy negatively correlates with the surface coverage.
The high value of ‘KF’ indicates a strong adsorption force between
PVA-MA-HH and Cu2+.
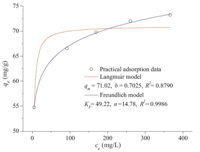
Fig. 5. Adsorption isotherm of Cu2+ at 293 K (pH 4.8).
![]()
Table 2
Data of adsorption-desorption process.
We acknowledge generous support provided by the National Science Funds for Young Scientists (No. 51008152),Jiangsu Province Science Funds for the Integration of Production-studyresearch (No. BY2012154) and Jiangsu Province Environmental Production Research Funds (No. 2012006).
| [1] | X.W. Lu, L.J. Wang, K. Lei, J. Huang, Y.X. Zhai, Contamination assessment of copper, lead, zinc, manganese and nickel in street dust of Baoji,NWChina, J. Hazard. Mater. 161 (2009) 1058-1062. |
| [2] | C.Y. Chen, S.Y. Chen, Adsorption properties of a chelating resin containing hydroxy groupand iminodiacetic acid for copper ions, J. Appl. Polym. Sci. 94 (2004) 2123-2130. |
| [3] | J.N. Wang, L. Xu, C. Cheng, Y. Meng, A.M. Li, Preparation of new chelating fiber with waste PET as adsorbent for fast removal of Cu2+ and Ni2+ from water: kinetic and equilibrium adsorption studies, Chem. Eng. J. 193-194 (2012) 31-38. |
| [4] | J.N. Wang, L. Xu, Y. Meng, et al., Adsorption of Cu2+ on new hyper-cross-linked polystyrene adsorbent: batch and column studies, Chem. Eng. J. 178 (2011) 108-114. |
| [5] | P. Taddei, P. Monti, G. Freddi, et al., Binding of Co(Ⅱ) and Cu(Ⅱ) cations to chemically modified wool fibres: an IR investigation, J. Mol. Struct. 650 (2003) 105-113. |
| [6] | Teaching and Researching Section of Beijing Normal University, Inorganic Chemistry, Academic Press, Beijing, 2005p. 708. |
| [7] | Teaching and Researching Section of Beijing Normal University, Inorganic Chemistry, Academic Press, Beijing, 2005p. 710. |
| [8] | E. Leporati, Thermodynamic and spectrophotometric investigation of complex formation between hydrogen, cobalt(Ⅱ), nickel(Ⅱ), and copper(Ⅱ) ions and 2-amino-n-hydroxypropanamide in aqueous solution, J. Chem. Soc., Dalton Trans. 7 (1989) 1299-1309. |
| [9] | N.S. Mcintyre, M.G. Cook, X-ray photoelectron studies on some oxides and hydroxides of cobalt, nickel, and copper, Anal. Chem. 47 (1975) 2208-2213. |