1. Introduction
Polyfunctionalized 4H-pyranshaveauniqueroleinmedicinal chemistry due to their wide range of biological and pharmacological activities [1, 2]. These compounds are utilized as anticancer agents,anti-coagulants,spasmolytics,and anti-anaphylactics [3, 4]. 4H-Pyran derivatives containing heterocyclic rings are extensively used for their pharmacological activities [5, 6]. Furthermore,4-phenyl-4H-pyrans have been identified as potent and specific IKCa channel blockers [7]. In addition,a number of 2-amino-4H-pyrans are valuable photoactive materials [8].
On the other hand,multicomponent reactions (MCRs) are efficient methods in heterocyclic scaffolds for the construction of diverse chemical libraries of drug-like molecules [9]. Synthetic strategies consisting MCRs have established themselves as a useful tool for the rapid introduction and development of molecular diversity [10]. MCRs increase the efficiency by combining several operational steps without isolation of intermediates or changing the reaction conditions [11, 12]. Because of their convergence and productivity,the MCRs have attracted noticeable attention from the combinatorial chemistry community [13].
Previously,2-amino-4H-pyrans were produced by the cyclization of arylidenemalononitriles with β-dicarbonyl compounds in the presence of bases such as piperidine [14],triethylamine [15], magnesium oxide [16],Mg/La [17],PEG1000-DAIL/EM [18],and nano ZnO [19]. Furthermore,diethyl 2,6-dimethyl-4-aryl-4H-pyran-3,5-dicarboxylates was synthesized with a low yield, utilizing ZnCl2 as a catalyst,acetic anhydride as the solvent,and column chromatography as the purification method [20, 21].
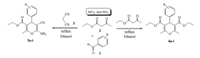
However,many of these methods require expensive reagents, acidic conditions,long reaction times,excess of reagents/catalyst,or toxic organic solvents and tedious workup,yet the yields are sometimes not ideal. In recent years,the use of heterogeneous catalysts has provided significant advantages in organic synthesis,for example,environmental compatibility,operational simplicity; also they are low cost,nontoxic,and easy to separate from the reaction mixtures [22, 23]. Furthermore,heterogeneous solid acids and especially those based on silica gel and other mesoporous high surface area support materials are beginning to show an influential role in the greening of fine and speciality chemicals manufacturing procedures [24, 25, 26]. In continuation of our recent work using heterogeneous solid acid nanoparticles in organic reactions [27, 28, 29], we decided to investigate nano silica supported tin (II) chloride (SnCl2/nano SiO2) as a green catalyst for the practical and environmentally benign one-pot multicomponent synthesis of diethyl 2,6-dimethyl-4-aryl-4H-pyran-3,5-dicarboxylates and 6-amino-5-cyano-2-methyl-4-aryl-4H-pyran-3-carboxylic acid ethylester (Scheme 1).
|
Download:
|
| Scheme 1. SnCl2/nano SiO2 catalyzed synthesis of polyfunctionalized 4H-pyrans. | |
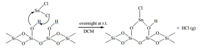
Nano silica gel supported tin (II) chloride was prepared according to the procedure reported in the literatures with some modification [30, 31]. In a typical procedure,tin (II) chloride dihydrate (0.28 g) was added to a suspension of nano particles of silica gel (3.075 g) in dichloromethane (DCM) (25.0 mL). The mixture was stirred at room temperature overnight. Then the solvent was removed under reduced pressure and the residue was heated at 100℃ under vacuum for 5 h to furnish SnCl2/nano SiO2. The prepared SnCl2/nano SiO2 has been structurally characterized by EDAX and SEM analysis.
2.2. General procedure for the preparation of diethyl 4-aryl-2,6-dimethl-4H-pyran-3,5-dicarboxylate (4a-i)A mixture of aldehyde (2 mmol),ethyl acetoacetate (4 mmol), and nano silica supported tin (II) chloride (0.15 mol%) was refluxed in ethanol (10 mL) for the appropriate time (monitored by TLC analysis). The hot reaction mixture was filtered to separate the catalyst and filtrate was solidified after cooling to room temperature. Then,ice water was added to mixture. The organic phase was extracted with dichloromethane,washed with water and dried with Na2SO4. The solvent was removed under reduced pressure. The crude products were purified by recrystallization (ethanol and water) with sufficient purity for spectral analysis.
2.3. General procedure for the preparation of ethyl-6-amino-5-cyano-4-aryl-2-methyl-4H-pyran-3-carboxylate(5a-i)A mixture of aldehyde (2 mmol),malononitrile (2 mmol),ethyl acetoacetate (2 mmol),and nano silica supported tin (II) chloride (0.15 mol%) was refluxed in ethanol (5 mL) for the appropriate time (monitored by TLC analysis). The hot reaction mixture was filtered to separate the catalyst and filtrate was solidified after cooling to room temperature. The solid product was consecutively washed with hexane (2 mL),ethyl acetate (2×0.5 mL),and dried.
Diethyl 4-(3-methoxyphenyl)-2,6-dimethl-4H-pyran-3,5-dicarboxylate (4h): Yellow crystal; 85%; mp: 91-94℃,FT-IR (KBr, cm-1):ν 3020 (C-H),1682 (C=O);1H NMR (400 MHz,CDCl3):δ 1.42 (t,3H,J= 6.8 Hz),1.72 (s,3H),3.81 (s,2H),3.90 (s,1H),4.10 (q, 2H,J= 6.8 Hz),6.58-7.17 (CH aromatic);13C NMR (400 MHz, CDCl3):δ 14.5,17.6,48.1,52.8,64.2,106.4,112.5,116.7,120.7, 128.8,141.1,155.6,162.3,166.9. Anal. calcd. for C20H23O6:C, 66.85; H,6.40. Found: C,66.79; H,6.48.
Ethyl-6-amino-5-cyano-4-(4-bromophenyl)-2-methyl-4H-pyran-3-carboxylate (5e): Orange crystal; 95%; mp: 264-265℃,FT-IR (KBr,cm-1 ):ν 3401,3319 (NH2),2210 (CN),1692(C=O);1H NMR (400 MHz,DMSO-d6):δ 1.10 (t,3H,J= 7.2 Hz),2.28 (s,3H),2.48 (s, 1H),3.94 (q,2H,J= 7.2 Hz),4.27 (s,NH2),6.93-7.4 (CH aromatic);13C NMR (400 MHz,DMSO-d6):δ 15.3,18.6,36.6,60.9,63.4,111.5, 116.8,120.6,133.9,135.8,156.7,159.1,160.3,165.9. Anal. calcd. for C16H16O4N2: C,53.03; H,4.14; N,7.73. Found: C,52.91; H,4.10; N,7.80.
Ethyl-6-amino-5-cyano-4-(3-methoxyphenyl)-2-methyl-4H-pyran-3-carboxylate (5h):Yellow crystal; 83%,mp: 264-265℃,FT-IR (KBr,cm-1):ν 3405,3315 (NH2),2206 (CN),1706 (C=O);1H NMR (400 MHz,DMSO-d6):δ 1.61 (t,3H,J= 6.8 Hz),2.12 (s,3H),3.83 (s, 3H),3.97 (s,1H),4.10 (s,NH2),4.45 (q,2H,J= 6.8 Hz),6.51-7.85 (CH aromatic);13C NMR (400 MHz,DMSO-d6):δ 11.2,17.2,40.1, 57.9,61.1,63.8,105.5,111.2,117.9,119.8,123.4,130.7,141.8, 158.2,159.9,162.5,170.2. Anal. calcd. for C17H18O4N2: C,64.96; H, 5.73; N,8.91. Found: C,64.87; H,5.66; N,8.97.
3. Results and discussionAccording to literature reports,utilities of the heterogeneous solid-supported materials as new environmentally friendly catalysts for the synthesis of biologically active molecules have increased. Hence,we describe a simple and efficient method for the synthesis of polyfunctionalized 4H-pyrans using the environmental friendly and reusable nano silica supported tin (II) chloride as a catalyst herein. The catalyst was prepared by reacting of tin (II) chloride with silica under favorable conditions (Scheme 2). In order to study the morphology and dimension of SnCl2/nano SiO2,SEM image of SnCl2/nano SiO2 was obtained and shown in Fig. 1.
|
Download:
|
| Scheme 2. Reaction of SnCl2 with silicagel. | |
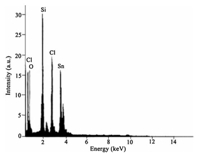
These results show that the size of commercial silicagel and synthesized SnCl2/nano SiO2 are till about 20 nm (Fig. 1). In the EDAX pattern of SnCl2/nano SiO2(Fig. 2),Sn,Cl,Si and O elements are indicated. This analysis is detecting that SnCl2 is supported in nano silicagel.

|
Download:
|
| Fig. 1. SEM photograph of (a) nano-SiO2 and (b) SnCl2/nano SiO2. | |
|
Download:
|
| Fig. 2. The EDAX spectrum of SnCl2/nano SiO2. | |
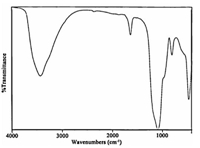
To identify the structure of this catalyst,we studied IR spectra of SnCl2/nano SiO2(Fig. 3). In the IR spectrum of SnCl2/nano SiO2,the Sn-Cl,Sn-O,Si-OH and Si-O-Si resonances were observed in 1559,563,3347 and 1061 cm-1,respectively. Based on these results,we suggest the following structure for SnCl2/nano SiO2.
|
Download:
|
| Fig. 3. The IR spectrum of SnCl2/nano SiO2. | |
We would like to report the preparation of polyfunctionalized 4H-pyran using reactions involving arylaldehydes,ethylacetoacetate,and malononitrile (Scheme 1). We chose the reaction of benzaldehyde 1,ethylacetoacetate 2 and malononitrile 3 as a model system for the optimization study.
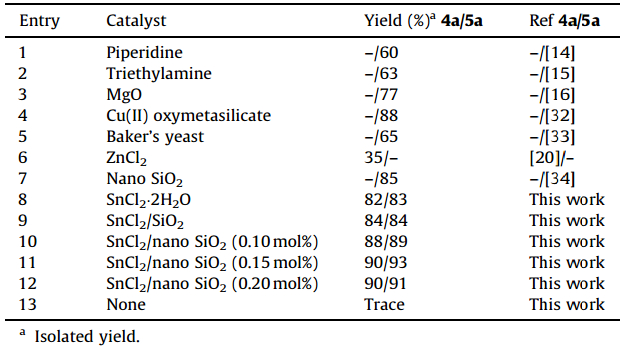
Initially,a series of comparative experiments were performed to compare the effectiveness of SnCl2/nano SiO2 versus other catalysts in the formation of polyfunctionalized 4H-pyran. In some cases,data for other catalysts were taken from the literature. The results are shown in Table 1. SnCl2/nano SiO2 was assessed to be the most efficient of all tested catalysts,in terms of reaction time and yield (Table 1,entry 11). As shown in Table 1,the yield for products 4a and 5a is high in the presence of SnCl2/SiO2 as a catalyst (Table 1,entry 9). Also it indicates the yields of products in the presence of SnCl2/nano SiO2(Table 1,entry 10) are higher than those catalyzed by SnCl2/SiO2. We believe that the heterogeneous nanoparticles have higher surface-to-volume ratio,which provides a larger number of active sites per unit area in comparison to their heterogeneous counterparts. Also,as shown in Table 1 no product was obtained in the absence of the catalyst (Table 1,entry 13).
| Table 1 Literature results for the synthesis of 4a and 5a with various catalysts. |
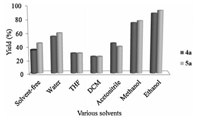
In the next step,the synthesis of 4a and 5a was separately carried out in different solvents such as water,ethanol,methanol, tetrahydrofuran (THF),acetonitrile and dichloromethane (DCM) under stirring and reflux. The results are summarized in Fig. 4. In the presence of water the products were obtained in low yield 55%-60%.
|
Download:
|
| Fig. 4. Effect of solvents on the activity of SnCl2/nano SiO2 catalyst. | |
Unfortunately,when the reactions were carried out in THF and DCM,the desired product was only obtained in 30%,25% yields respectively. Obviously,the protic solvents such as ethanol were more suitable than aprotic solvents (Fig. 4). In solvent-free conditions the reaction did not give the desired product.
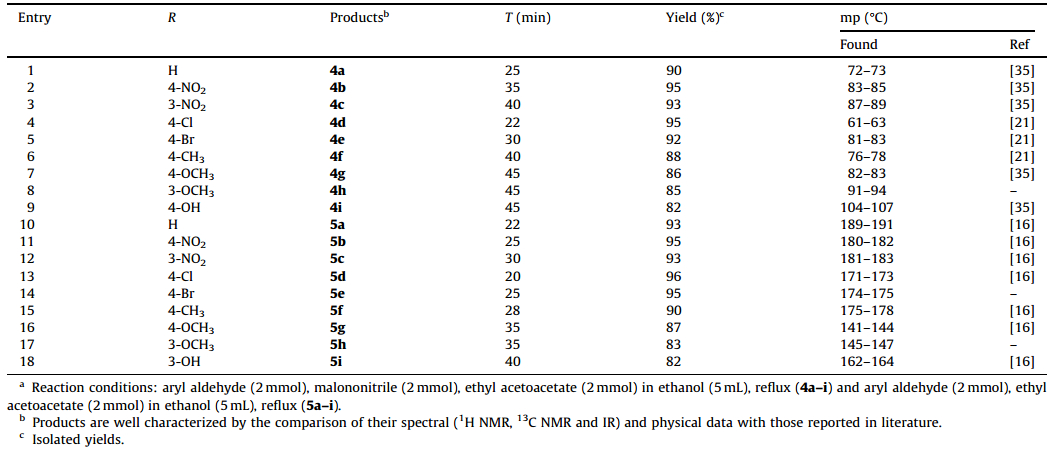
In continuation of our research,we used a diverse of aldehydes to investigate these three-component reactions under the optimized conditions. We observed various aldehydes could be introduced with high efficiency and produced products in high yields. Also aldehydes bearing electron-withdrawing group such as NO2,Cl in the para-position reacted very smoothly,while reactants with electron-donating group decreased both the rate of reaction and yield of the corresponding products as shown in Table 2.
| Table 2 Synthesis of polyfunctionalized 4H-pyrans catalyzed by SnCl2/nano SiO2(0.15 mol%)a. |
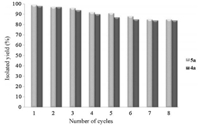
As an environmentally-friendly methodology,the reuse and recovery of the SnCl2/nano SiO2 are highly desirable. In order to study the reusability of nano silicagel supported tin (II) chloride as a catalyst,eight batches of the experiments were carried out for the preparation of products 4a and 5a (Fig. 5). The catalyst was filtered off and washed with an excess of ethanol and reused in a new reaction.
|
Download:
|
| Fig. 5. Repeated use of SnCl2/nano SiO2 for the synthesis of 4H-pyran derivetives 4a and 5a. | |
The characterization of the fresh SnCl2/nano SiO2 and after eight times of reuse showed the same particle size by SEM (Fig. 6). Interestingly,the shape and size of the nanoparticles remained unchanged before and after reaction. We believe that,this is also the possible reason for the extreme stability of the SnCl2/nano SiO2 presented herein.

|
Download:
|
|
Fig. 6. SEM images of SnCl2/nano SiO2 before use (a) and after eight times reuse (b). |
|
This paper describes SnCl2/nano SiO2 as a heterogeneous,easy to prepare,inexpensive,and efficient catalyst for the synthesis of polyfunctionalized 4H-pyrans. The distinguished advantages of this procedure are novelty,operational simplicity,high yields and the ease of isolation. The products were obtained in excellent yields and the reaction times were significantly shorter than the available methods. The present approach demonstrates a simple and effective method using SnCl2/nano SiO2 as novel nano-scale materials.
AcknowledgmentThe authors are grateful to University of Kashan for supporting this work by Grant No. 159196/XV.
| [1] | M.H. Elnagdi, H.A. Elfaham, G.E.H. Elgemeie, Utility of α,β-unsaturated nitrirles in heterocyclic synthesis, Heterocycles 20 (1983) 519-550. |
| [2] | S. Goldmann, J. Stoltefus, 1,4-Dihydropyridines: effects of chirality and conformation on the calcium antagonist and calcium agonist activities, Angew. Chem. Int. Ed. Engl. 30 (1991) 1559-1578. |
| [3] | L.L. Andreani, E. Lapi, Aspects and orientations of modern pharmacognosy, Boll. Chim. Farm. 99 (1960) 583-586. |
| [4] | L. Bonsignore, G. Loy, D. Secci, A. Calignano, Synthesis and pharmacological activity of 2-oxo-(2H) 1-benzopyran-3-carboxamide derivatives, Eur. J. Med. Chem. 28 (1993) 517-520. |
| [5] | G.R. Green, J.M. Evans, A.K. Vong, Pyrans and their benzo derivatives synthesis, in: A.R. Katritsky, C. Rees, E.F.V. Scriven (Eds.), Comprehensive Heterocyclic Chemistry Ⅱ, Pergamon Press, Oxford, 1995, p. 469. |
| [6] | A. Sánchez, F. Hernández, P.C. Cruz, et al., Infrared irradiation-assisted multicomponent synthesis of 2-amino-3-cyano-4H-pyran derivatives, J. Mex. Chem. Soc. 56 (2012) 121-127. |
| [7] | K. Urbahns, E. Horvath, J.P. Stasch, F. Mauler, 4-Phenyl-4H-pyrans as IKCa channel blockers, Bioorg. Med. Chem. Lett. 13 (2003) 2637-2639. |
| [8] | I. Devi, P.J. Bhuyan, Sodium bromide catalysed one-pot synthesis of tetrahydrobenzo[ b]pyrans via a three-component cyclocondensation under microwave irradiation and solvent free conditions, Tetrahedron Lett. 45 (2004) 8625-8627. |
| [9] | A. Domling, Recent developments in isocyanide based multicomponent reactions in applied chemistry, Chem. Rev. 106 (2006) 17-89. |
| [10] | D.J. Ramon, M. Yus, Asymmetric multicomponent reactions (AMCRs): the new frontier, Angew. Chem. Int. Ed. 44 (2005) 1602-1634. |
| [11] | C. Ma, Y. Yang, Thiazolium-mediated multicomponent reactions: a facile synthesis of 3-aminofuran derivatives, Org. Lett. 7 (2005) 1343-1345. |
| [12] | E. Soleimani, M.M. Khodaei, N. Batooie, M. Baghbanzadeh, Water-prompted synthesis of alkyl nitrile derivatives via Knoevenagel condensation and Michael addition reaction, Green Chem. 13 (2011) 566-569. |
| [13] | G. Evano, N. Blanchard, M. Toumi, New copper-mediated coupling reactions and their applications in natural products and designed biomolecules synthesis, Chem. Rev. 108 (2008) 3054-3131. |
| [14] | N. Martin, C. Pascual, C. Seoane, J.L. Soto, The use of some activated nitriles in heterocyclic syntheses, Heterocycles 26 (1987) 2811-2816. |
| [15] | S.E. Zayed, E.I. AbouElmaged, S.A. Metwally, M.H. Elnagdi, Reactions of sixmembered heterocyclic b-enaminonitriles with electrophilic reagents, Collect. Czech. Chem. Commun. 56 (1991) 2175-2182. |
| [16] | D. Kumar, R.V. Buchi, S. Sharad, U. Dube, S. Kapur, A facile one-pot green synthesis and antibacterial activity of 2-amino-4H-pyrans and 2-amino-5-oxo-5,6,7,8-tetrahydro-4H-chromenes, Eur. J. Med. Chem. 44 (2009) 3805-3809. |
| [17] | N. SeshuBabu, N. Pasha, K.T. VenkateswaraRao, P.S. Sai Prasad, N. Lingaiah, A heterogeneous strong basic Mg/La mixed oxide catalyst for efficient synthesis of polyfunctionalized pyrans, Tetrahedron Lett. 49 (2008) 2730-2733. |
| [18] | D. Fang, J.M. Yang, H.B. Zhang, C.M. Jiao, Synthesis of 4H-pyrans catalyzed by thermol-regulated PEG1000-based ionic liquid/EM, J. Ind. Eng. Chem. 17 (2011) 386-388. |
| [19] | P. Bhattacharyya, K. Pradhan, S. Paul, A.R. Das, Nano crystalline ZnO catalyzed one pot multicomponent reaction for an easy access of fully decorated 4H-pyran scaffolds and its rearrangement to 2-pyridone nucleus in aqueous media, Tetrahedron Lett. 53 (2012) 4687-4691. |
| [20] | J. Wolinsky, H.S. Haue, Substituted γ-pyrans, J. Org. Chem. 34 (1969) 3169. |
| [21] | J.C. Wilson, G.S. Mcgrath, S.A. Srinivasan, 4H-pyran charge control agents for electrostatographic toners and developers, US Patent, 6 221 550 (2001). |
| [22] | P. Salehi, M. Dabiri, M.A. Zolfigol, M.A. Bodaghi Fard, Silica sulfuric acid: an efficient and reusable catalyst for the one-pot synthesis of 3, 4-dihydropyrimidin-2(1H)-ones, Tetrahedron Lett. 44 (2003) 2889-2891. |
| [23] | M. Moghaddas, A. Davoodnia, M. Heravi, N. Tavakoli-Hoseini, Sulfonated carbon catalyzed Biginelli reaction for one-pot synthesis of 3,4-dihydropyrimidin-2(1H)-ones and-thiones, Chin. J. Catal. 33 (2012) 706-710. |
| [24] | D.C. Sherrington, A.P. Kybett, Supported Catalysts and Their Applications, The Royal Society of Chemistry, Cambridge, 2001p. 196. |
| [25] | L. Huang, Z. Wang, T.P. Ang, J. Tan, P.K. Wong, A novel SiO2 supported Pd metal catalyst for the Heck reaction, Catal. Lett. 112 (2006) 219-225. |
| [26] | V. Siddaiah, G.M. Basha, R. Srinuvasarao, S.K. Yadav, HClO4-SiO2: an efficient reusable catalyst for the synthesis of 3,4,5-trisubstituted 1,2,4-triazoles under solvent-free conditions, Catal. Lett. 141 (2011) 1511-1520. |
| [27] | A. Ziarati, J. Safaei-Ghomi, S. Rohani, Pseudo five-component process for the synthesis of functionalized tricarboxamides using CuI nanoparticles as reusable catalyst, Chin. Chem. Lett. 24 (2013) 195-198. |
| [28] | J. Safaei-Ghomi, M.A. Ghasemzadeh, Zinc oxide nanoparticles: a highly efficient and readily recyclable catalyst for the synthesis of xanthenes, Chin. Chem. Lett. 23 (2012) 1225-1229. |
| [29] | A. Ziarati, J. Safaei-Ghomi, S. Rohani, Sonochemically synthesis of pyrazolones using reusable catalyst CuI nanoparticles that was prepared by sonication, Ultrason. Sonochem. 20 (2013) 1069-1075. |
| [30] | H.R. Darabi, K. Aghapoor, F. Mohsenzadeh, et al., Heterogeneous SnCl2/SiO2 versus homogeneous SnCl2 acid catalysis in the benzo[N,N]-heterocyclic condensation, Bull. Korean Chem. Soc. 32 (2011) 213-218. |
| [31] | B.F. Mirjalili, A. Bamoniri, A. Akbari, One-pot synthesis of 3,4-dihydropyrimidin-2(1H)-ones (thiones) promoted by nano-BF3 SiO2, J. Iran Chem. Soc. 8 (2011) S135-S140. |
| [32] | M.M. Heravi, Y.S. Beheshtiha, Z. Pirnia, S. Sadjadi, M. Adibi, One-pot, threecomponent synthesis of 4H-pyrans using Cu(Ⅱ) oxymetasilicate, Synth. Commun. 39 (2009) 3663-3667. |
| [33] | U.R. Pratap, D.V. Jawale, P.D. Netankar, R.A. Mane, Baker's yeast catalyzed one-pot three-component synthesis of polyfunctionalized 4H-pyrans, Tetrahedron Lett. 52 (2011) 5817-5819. |
| [34] | S. Banerjee, Al Horn, H. Khatri, G. Sereda, A green one-pot multicomponent synthesis of 4H-pyrans and polysubstituted aniline derivatives of biological, pharmacological, and optical applications using silica nanoparticles as reusable catalyst, Tetrahedron Lett. 52 (2011) 1878-1881. |
| [35] | K. Urbahns, H.G. Heine, B. Junge, et al., Substituted 4H-pyrans with a modulating effect on calcium channels, EP Patents, 0758648 (1997). |