a Corresponding authors at:a College of Science, Behbahan Khatam Alanbia University of Technology, Behbahan, Iran;
b Department of Chemistry, Gachsaran Branch, Islamic Azad University, Gachsaran, Iran;
c Department of Chemistry, Omidieh Branch, Islamic Azad University, Omidieh, Iran
1. Introduction
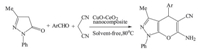
4H-Benzo[b]pyran derivatives are an important class of heterocyclic compounds having important pharmaceutical and biological activities. These compounds are used as anticancer, anticoagulant,diuretic,spasmolytic and antianaphylactic agents [1]. Therefore 4H-benzo[b]pyrans received significant amount of attention from pharmaceutical and organic chemistry communities. The commonly used method for the synthesis of 4Hbenzo[b]pyrans is the condensation of aldehyde,cinnamonitrile derivatives and dimedone in the presence of acidic or basic catalysts. Therefore,various methods have been developed for the synthesis of 4H-benzo[b]pyrans [2, 3, 4, 5, 6, 7, 8, 9, 10]. Also,these compounds can be prepared via multicomponent condensation reactions of aldehydes,cinnamonitrile derivatives and 3-methyl-1-phenyl-2- pyrazoline-5-one [11, 12, 13, 14, 15]. Recently,development of nano catalysts has emerged as a fertile field for research and innovation. In particular,nanocrystalline oxides have proven useful to chemists in the laboratory and industry due to the efficient activation of the adsorbed compounds and reaction rate enhancement,selectivity, easier work-up and recyclability of the supported catalysts and the eco-friendly green reaction conditions [16, 17, 18]. Also the practical applications of nanocomposite metal oxides as the catalysts in organic synthesis have increased due to their high catalytic activity because of the large surface area [19, 20, 21]. Recently,the catalytic application of CuO-CeO2 nanocomposite for the oxidation of CO in H2-rich streams was reported [22][22]. Although there is a report on the synthesis and catalytic properties of CuO-CeO2 catalyst,the applicability of CuO-CeO2 nanocomposite in organic reactions and synthesis of biologically important molecules is yet to be investigated. Therefore,we decided to prepare and use this catalyst in the organic synthesis. In continuation of our research program to develop efficient and green catalysts in organic synthesis [23, 24, 25, 26],herein we wish to report the applicability of CuO-CeO2 nanocomposite as a heterogeneous,green and recyclable catalyst for the synthesis of biologically important 4H-benzo[b]pyran derivatives via the one-pot three-component condensation of 3-methyl-1-phenyl-2- pyrazoline-5-one with aromatic aldehydes and malononitrile under solvent-free conditions (Scheme 1).
|
Download:
|
| Scheme 1 Synthesis of 4H-benzo[b]pyran derivatives catalyzed by CuO-CeO2 nanocomposite. | |
2. Experimental
All products were characterized by comparison of their spectroscopic data (1H NMR,IR) and physical properties with those reported in the literature. Chemicals were purchased from Fluka and Merck chemical companies. Yields refer to isolated pure products.
General procedure for the synthesis of 4H-benzo[b]pyran derivatives: A mixture of 3-methyl-1-phenyl-2-pyrazoline-5-one (1 mmol),malononitrile (1 mmol),aldehyde (1 mmol) and CuO- CeO2 (0.05 g),was heated in an oil bath (80 8C) for the appropriate period of time as shown on Table 1. After the completion of the reaction (monitored by TLC analysis),the resulting mixture was cooled,acetone (10 mL) was added and the catalyst was recovered by filtration to be reused subsequently. Evaporation of the solvent from the filtrate and recrystallization of the solid residue from hot ethanol afforded the pure products in high yields. The spectral and analytical data as follows:
Table 1,entry 6: IR (KBr,cm-1): υmax 3400,3300,3200,2900, 2200,1645,1400,1300,1000. 1H NMR (400 MHz,CDCl3): d 1.9 (s, 3H,CH3),4.8 (s,2H,NH2),5.3 (s,1H,CH),7.17 (d,1H,J = 8.4 Hz,Ar), 7.26 (s,1H,Ar),7.35 (t,1H,J = 7.2 Hz,Ar),7.46-7.51 (m,3H,Ar), 7.66 (d,2H,J = 8 Hz,Ar). 13C NMR (100 MHz,CDCl3): d: 12.77 (CH3), 33.48 (CH),61.89 (CN),97.57,118.64,121.27,126.97,128.04, 129.35,129.67,131.50,133.94,137.40,137.86,143.95,146.05, 158.87.
Table 1,entry 10: IR (KBr,cm-1): υmax 3400,3300,3100,2200, 1645,1400,1300,1000. 1H NMR (500 MHz,DMSO-d6): d 1.79 (s, 3H),4.55 (s,1H),6.71 (d,2H,J = 7.82 Hz),7.03 (d,2H,J = 7.81 Hz), 7.2 (s,2H),7.31 (t,1H,J = 7.03 Hz),7.48 (t,2H,J = 7.31 Hz),7.77 (d, 2H,J = 7.74 Hz),9.32 (s,1H). 13C NMR (125 MHz,DMSO-d6): d 13.4 (CH3),37.5 (CH),57.9 (CN),98.5,110.8,119.6,120.6,120.9,127.1, 129.8,130.1,133.5,138.3,144.9,145.9,150.0,160.
Table 1,entry 11: (KBr,cm-1): υmax 3089,3082,2200,1673, 1590,1515,1390,1250,1120,1070,1030. 1H NMR (500 MHz, DMSO-d6): d 1.77 (s,3H),4.83 (s,1H),7.30-7.33 (m,3H),7.47-7.50 (m,4H),7.78 (d,2H,J = 8.5 Hz),7.82 (d,2H,J = 8.0 Hz). 13C NMR (125 MHz,DMSO-d6): d 13.4,37.5,57.9,98.5,110.8,119.6,120.6, 120.9,127.1,129.8,130.1,133.5,138.3,144.9,145.9,150.0,160.5. Table 1,entry 12: White solid,mp: 198-200 8C; (KBr,cm-1): υmax 3300,3120,2900,2220,1650,1520,1250,1120. 1H NMR (500 MHz,DMSO-d6): d 1.78 (s,6H),4.7 (s,2H),7.14-7.18 (m,6H), 7.29-7.35 (m,4H),7.48 (t,4H,J = 8.0 Hz),7.76 (d,4H,J = 7.5 Hz). Elem. Anal. Found: C 70.84,H 4.57,N 19.41,(calcd. for C34H26N8O2: C 70.75,H 4.53,N 19.36).
Table 1,entry 13: White solid,mp: 157-159 8C; (KBr,cm-1): υmax 3400,3300,3100,2800,2200,1645-1400,1300,1000. 1H NMR (400 MHz,CDCl3): d 1.89 (s,3H,CH3),4.81 (s,2H,NH2),4.86 (s,1H,CH),7.35 (t,2H,J = 8 Hz,Ar),7.52 (m,4H,Ar),7.7 (d,2H, J = 7.6 Hz,Ar),7.77 (s,1H,Ar),7.85 (d,2H,J = 8.4 Hz,Ar). 13C NMR (100 MHz,CDCl3): d 12.97 (Me),37.67 (CH),60.46 (CN),98.23, 119.21,121.2,125.66,126,126.33,126.7,126.8,127.76,128, 128.96,129.33,132.92,133.34,137.58,139.28,143.9,146.58, 158.33. Elem. Anal. Found: C 82.34,H 5.24,N 8.05 (calcd. for C24H18N2O: C 82.26,H 5.18,N 7.99).
| Table 1 Synthesis of 4H-benzo[b]pyran derivatives catalyzed by CuO-CeO2 nanocomposite. |
The CuO-CeO2 nanocomposite catalyst was prepared by a coprecipitation method. Emission scanning electron microscopy (ESEM),X-ray diffraction (XRD) and energy dispersive spectroscopy (EDS) were used to characterize the catalyst. XRD pattern of fresh CuO-CeO2 nanocomposite calcined at 450 8C is illustrated. The main reflection placed at 2u range of 15-808 indicates a cubic fluorite structure with highest intensity at (1 1 1) plane that belongs to the XRD standard JCPDS card number of 43-1002. According to the pattern of pure CeO2 (not shown in the figure), only the diffraction peaks of ceria crystallites were detected and there were no peaks related to copper oxide nanoparticles. It is hypothesized that fine dispersion and/or amorphous structure of CuO with low content on the surface of ceria clusters is responsible for the absence of copper oxide peaks in the XRD pattern. However, there are some differences between the values of unit cell parameters for pure CeO2 and those for CuO-CeO2 due to lattice shrinkage in the surface ceria structure. The calculations showed that the unit cell parameters were 5.45A˚ and 5.42A˚ for CeO2 and CuO-CeO2,respectively. The observed shrinkage may be a consequence of partial incorporation of CuO into the CeO2 lattice structure that has been reported for co-precipitation synthesis. The calculated average crystal size of the ceria in the catalyst based on dominant peak at 2u of 28.58 using Debye-Scherrer equation was 7.5 nm. The BET specific surface area was determined to be 131 m2/g,which confirmed the estimated value for the average nanoparticle size of the catalyst measured by the XRD test. Fig. 1 presents the ESEM micrograph of the catalyst at two different magnifications to study the morphology and evaluate the nanocluster size of the catalyst. It seems that 7 nm sized particles calculated based on the BET surface area,are agglomerated to approximately 33 nm sized clusters and produced bigger nanostructured CuO-CeO2 catalyst. EDS analysis and atomic absorption showed the presence of about 5.8 wt.% CuO in the CuO-CeO2 catalyst.

|
Download:
|
| Fig. 1.ESEM image of the CuO-CeO2 nanocomposite catalyst at two different magnifications. | |
The catalyst showed very good catalytic activity. This might be due to its small particle size,which provides a large surface area for reactant adsorption and therefore high catalytic activity. For the optimization of the reaction conditions,the reaction of benzaldehyde, malononitrile and 3-methyl-1-phenyl-2-pyrazoline-5-one were chosen as a model and their behavior was studied under various conditions. The best results were achieved by carrying out the reaction of benzaldehyde,3-methyl-1-phenyl-2-pyrazoline-5- one and malononitrile (with a 1:1:1 molar ratio) in the presence of 0.05 g of CuO-CeO2 nanocomposite at 80 8C for 12 min undersolvent-free conditions (Table 1,entry 1). Using these optimized conditions,the reaction of various aromatic aldehydes,containing electron-donating and electron-withdrawing groups,was studied.
The solid products were easily isolated by simple filtration and recrystallized from hot ethanol. All known products were identified by comparison of the melting points and the analytical data (IR,NMR) with those reported for authentic samples. Also,it should be mentioned that products were obtained without byproducts in good to high yields with short reaction times. In organic reactions,when a heterogeneous catalyst is used,the recyclability of the catalyst is very important. In order to investigate the recyclability of the CuO-CeO2 nanocomposite, the reaction of benzaldehyde,malononitrile and 3-methyl-1- phenyl-2-pyrazoline-5-one,was selected again as a model. In this experiment,after the completion of the reaction,the catalyst was washed with hot acetone,dried and stored for another consecutive reaction run. The recycled catalyst has been examined in the next run and the desired products were obtained in good to high yields. This process was repeated for 8 times and no appreciable decrease in yield was observed (Table 2).
| Table 2 Recyclability study of CuO-CeO2 nanocomposite. |
Finally,in order to evaluate the efficiency and superiority of our catalyst,we began to run the reaction between benzaldehyde, malononitrile and 3-methyl-1-phenyl-2-pyrazoline-5-one in the presence of pure CuO or CeO2 nanoparticles under the same conditions. The obtained results showed that in the presence of pure CuO or CeO2,recyclability of the catalyst decreased,much longer reaction time was required,and the yields were lower. 4. Conclusion
In conclusion,we have developed an efficient green procedure for the one-pot synthesis of 4H-benzo[b]pyran derivatives using recyclable CuO-CeO2 nanocomposite under solvent-free conditions. CuO-CeO2 nanocomposite was easy to handle and can be recovered simply by filtration. It remains active for at least 8 runs without a significant decrease of the yields. Moreover,high yields, short reaction time,ease of work-up and the minimal amount of waste generated will make the present method an important addition to the available methodologies for the synthesis of 4Hbenzo[ b]pyran derivatives. Acknowledgment
We are thankful to the Behbahan Khatam Alanbia University of Technology for the partial support of this work. Appendix A. Supplementary data
1001-8417/$ - see front matter ⓒ 2013 Jalal Albadi. Published by Elsevier B.V. on behalf of Chinese Chemical Society. All rights reserved.
http://dx.doi.org/10.1016/j.cclet.2013.05.031
| [1] | K. Singh, J. Singh, H. Singh, A synthetic entry into fused pyran derivatives through carbon transfer reactions of 1,3-oxazinanes and oxazolidines with carbon nucleophiles, Tetrahedron 52(1996) 14273-14280. |
| [2] | S. Rathod, B. Arbad, M. Lande, Preparation, characterization, and catalytic application of a nanosized Ce1MgxZr1-xO2 solid heterogeneous catalyst for the synthesis of tetrahydrobenzo[b]pyran derivatives, Chin. J. Catal. 31(2010) 631-636. |
| [3] | M. Seifi, H. Sheibani, High surface area MgO as a highly effective heterogeneous base catalyst for three-component synthesis of tetrahydrobenzopyran and 3,4-dihydropyrano[c]chromene derivatives in aqueous media, Catal. Lett. 126(2008) 275-279. |
| [4] | D. Shi, J. Mou, Q. Zhuang, X. Wang, One-pot synthesis of 2-amino-4-aryl-5-oxo-5,6,7,8-tetrahydro-4H-1-benzopyran-3-carbonitriles in aqueous media, J. Chem. Res.(2004) 821-823. |
| [5] | D.M. Pore, K.A. Undale, B.B. Dongare, U.V. Desai, Potassium phosphate catalyzed a rapid three-component synthesis of tetrahydrobenzo[b]pyran at ambient temperature, Catal. Lett. 132(2009) 104-108. |
| [6] | A. Mobinikhaledi, M.A. Bodaghi Fard, Tetrabutylammonium bromide in water as a green media for the synthesis of pyrano[2,3-d]pyrimidinone and tetrahydrobenzo[ b]pyran derivatives, Acta. Chim. Slov. 57(2010) 931-935. |
| [7] | G. Sabitha, K. Arundhathi, K. Sudhakar, B.S. Sastry, J.S. Yadav, Cerium(Ⅲ) chloridecatalyzed one-pot synthesis of tetrahydrobenzo[b]pyrans, Synth. Commun. 39(2009) 433-442. |
| [8] | X.Z. Lian, Y. Huang, Y.Q. Li, W.J. Zheng, A green synthesis of tetrahydrobenzo[b]-pyran derivatives through three-component condensation using N-methylimidazole as organocatalyst, Monatshefte für Chemie 139(2008) 129-131. |
| [9] | H.A. Oskooie, M.M. Heravi, N. Karimi, M. Ebrahim Zadeh, Caro'a acid-silica gel: an efficient and versatile catalyst for the one-pot synthesis of tetrahydrobenzo[b]-pyran derivatives, Synth. Commun. 41(2011) 436-440. |
| [10] | S. Rathod, B. Arbad, M. Lande, Preparation, pharacterization, and catalytic application of a nanosized Ce1MgxZr1-xO2 solid heterogeneous catalyst for the synthesis of tetrahydrobenzo[b]pyran derivatives, Chin. J. Catal. 31(2010) 631-636. |
| [11] | X.S. Wang, D.Q. Shi, S.J. Tu, C.S. Yao, Y.C. Wang, Synthesis and crystal structure of 3-methyl-5-cyano-6-amino-1-phenyl-4-(3,4-dimethoxyphenyl)-1,4-dihydropyrano[ 2,3-c]pyrazole, Chin. J. Struct. Chem. 22(2003) 331-335. |
| [12] | S.B. Guo, S.X. Wang, J.T. Li, D,L-Proline-catalyzed one-pot synthesis of pyrans and pyrano[2,3-c]pyrazole derivatives by a grinding method under solvent-free conditions, Synth. Commun. 37(2007) 2111-2120. |
| [13] | T.S. Jin, A.Q. Wang, Z.L. Cheng, J.S. Zhang, T.S. Li, A clean and simple synthesis of 6-amino-4-aryl-5-cyano-3-methyl-1-phenyl-1,4-dihydropyrano[2,3-c]pyrazole in water, Synth. Commun. 35(2005) 137-143. |
| [14] | A. Hasaninejad, M. Shekouhy, N. Golzar, A. Zare, M.M. Doroodmand, Silica bonded n-propyl-4-aza-1-azoniabicyclo[2.2.2]octane chloride(SB-DABCO): a highly efficient, reusable and new heterogeneous catalyst for the synthesis of 4H-benzo[b]-pyran derivatives, Appl. Catal. A: Gen. 402(2011) 11-22. |
| [15] | D. Shi, J. Mou, Q. Zhuang, et al., Three-component one-pot synthesis of 1,4-dihydropyrano[2,3-c]pyrazole derivatives in aqueous media, Synth. Commun. 34(2004) 4557-4563. |
| [16] | S.F. Yin, B.Q. Xu, S.J. Wang, C.F. Ng, C.T. Au, Magnesia-Carbon nanotubes(MgOCNTs) nanocomposite: novel support of Ru catalyst for the generation of COx-free hydrogen from ammonia, Catal. Lett. 96(2004) 113-116. |
| [17] | M.T. Drexler, M.D. Amiridis, Kinetic investigation of the heterogeneous synthesis of flavanone over MgO, Catal. Lett. 79(2002) 175-181. |
| [18] | H. Mehrabi, M. Kazemi-Mireki, An efficient and recyclable nanocatalyst for the rapid and green synthesis of 3,4-dihydropyrano[c]chromenes, Chin. Chem. Lett. 22(2011) 1419-1422. |
| [19] | S. Samantaray, D.K. Pradhan, G. Hota, B.G. Mishra, Catalytic application of CeO2-CaO nanocomposite oxide synthesized using amorphous citrate process toward the aqueous phase one pot synthesis of 2-amino-2-chromenes, Chem. Eng. J. 193(2012) 1-9. |
| [20] | S. Li, W. Zhang, M.H. So, et al., One-pot solvothermal synthesis of Pd/Fe3O4 nanocomposite and its magnetically recyclable and efficient catalysis for Suzuki reactions, J. Mol. Catal. A: Chem. 359(2012) 81-87. |
| [21] | I. Mohammadzadeh, H. Sheibani, A convenient one-pot synthesis of new chromeno[3,4-c]chromene and chromeno[3,4-c]pyridine derivatives in the presence of high surface area of magnesium oxide, Chin. Chem. Lett. 23(2012) 1327-1330. |
| [22] | A. Razeghi, A. Khodadadi, H. Ziaie-Azad, Y. Mortazavi, Activity enhancement of Cu-doped ceria by reductive regeneration of CuO-CeO2 catalyst for preferential oxidation of CO in H2-rich streams, Chem. Eng. J. 164(2010) 214-220. |
| [23] | J. Albadi, M. Kehavarz, F. Shirini, M. Vafaie-nezhad, Copper iodide nanoparticles on poly(4-vinyl pyridine): a new and efficient catalyst for multicomponent click synthesis of 1,4-disubstituted-1,2,3-triazoles in water, Catal. Commun. 27(2012) 17-20. |
| [24] | J. Albadi, N. Iravani, M. Khoshakhlagh, A new, green and recyclable poly(4-vinylpyridine)-supported copper iodide nanoparticles catalyst for the synthesis of aryl-14H-dibenzo[a-o]xanthenes, Iran J. Catal. 2(2012) 85-89. |
| [25] | J. Albadi, N. Iravani, F. Shirini, F. Dehghan, A green recyclable poly(4-vinylpyridine)-supported copper iodide nanoparticle catalyst for the chemoselective synthesis of pentaerythritol diacetals from aromatic aldehydes, J. Chem. Res.(2012) 610-612. |
| [26] | J. Albadi, M. Keshavarz, M. Abedini, M. Khoshakhlagh, Copper iodide nanoparticles on poly(4-vinyl pyridine): a new and efficient catalyst for the synthesis of 1,8-dioxoocatahydroxanthenes under solvent-free conditions, J. Chem. Sci. 125(2013) 295. |