The increasing demand for propene in the international market, as well as the general attempt to transform cheap and abundant feedstocks and by-products of oil refining processes into more valuable products,have recently resulted in considerable research interest in the oxidative dehydrogenation (ODH) of propane. A large variety of unsupported and supported oxide catalysts have been proposed for this reaction,with the most widely studied ones being based on vanadia or molybdena [1]. Though scarcely studied, the nickel- and cobalt-based oxide systems have also been reported to be active and selective recently [2, 3, 4, 5, 6, 7, 8]. Davies et al. found that nanocrystalline cobalt oxide could activate propane, yielding propene at room temperature and atmospheric pressure [6]. However,the conversion of propane was very low (around 1%) and decreased quickly. The magnesium-cobalt phosphates CoxMg3--x(PO4)2 were investigated in the ODH of ethane and propane [7]. Over CoMg2(PO4)2,a propane conversion of 33% with a propene selectivity of 43.8% were obtained at 550 ℃. Recently, Mitran et al. performed the ODH of propane over Co(x)MgAlO mixed oxides obtained from LDH precursors [8]. With Co(5)MgAlO and Co(7)MgAlO catalysts,the best yields for propene of about 10% were attained for the reaction performed at 600 ℃.
In this paper,we reported the catalytic performance of a novel catalyst,P-modified nanosized cobalt oxide,which is proven highly effective for the ODH of propane.
The phosphorus modified cobalt oxide catalysts,which is denoted as P(x)CoO (x refers to the P/Co atomic ratio and varies between 0 and 0.1),were prepared by a modified sol-gel method using citric acid as a ligand. Typically,the solution containing the appropriate amounts of cobalt nitrate and ammonium phosphate were added dropwise to a stoichiometric amount of citric acid solution to generate a completely homogeneous and transparent solution. The mixed solution was then subjected to slow evaporation at 70 ℃ until a highly viscous residue formed. After drying at 110 ℃ for 24 h,a gel precursor was developed,and then calcinated at 400 ℃ for 4 h in a muffle furnace.
The specific surface areas of the catalysts were measured by BET technique with N2 as the adsorbate. X-ray powder diffraction (XRD) analysis was performed using a Panalytical X’pert PRO power diffractometer with Cu Kα radiation. Transmission electron microscopy (TEM) images were taken from a FEI Tecnai-F30 FEG instrument with an acceleration voltage of 300 kV. The FT-IR spectra were obtained at room temperature using a Nicolet Avatar 330 infrared spectrometer with a spectral resolution of 4 cm-1.
The catalytic reaction of propane ODH was carried out in a fixed bed flow reactor under atmospheric pressure. For each sample tested,0.1 g of catalyst was mixed with an equal amount of quartz particles and loaded in a quartz tubular reactor (5 mm i.d.). The feed consisted of propane and the mixture of oxygen and nitrogen (C3H8/O2/N2 = 1.2/1/8),with the GHSV of 30,000 mL/(g h). The reactants and products were analyzed using an on-line gas chromatograph equipped with squalene-impregnated Al2O3 and carbon molecular sieve columns and a thermal conductivity detector (TCD).
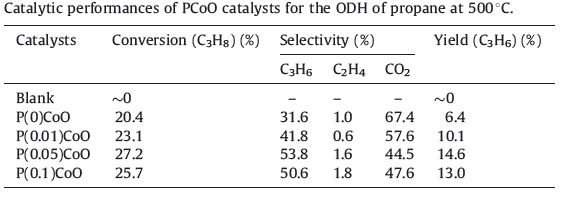
Table 1 summarizes the results of propane ODH over different catalysts at 500 ℃. Under the reaction conditions,non-catalytic auto-oxidation of propane in the empty reactor is negligible. Pure cobalt oxide shows a moderate activity for propane oxidation,but producing mainly CO2. With the addition of phosphorus,the modified catalyst represents a significant improvement in its catalytic performance,especially in propene selectivity. Both propane conversion and propene selectivity attain their maximal values when P content is increased to P/Co = 0.05,beyond which the conversion and the selectivity seem to decline.
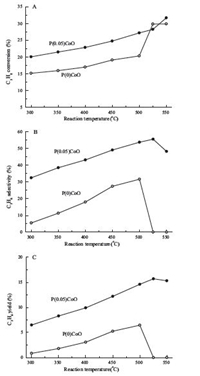
Fig. 1 shows the results of propane ODH over P(0)CoO and P(0.05)Co catalysts at different temperatures. Compared to pure cobalt oxide,the P-modified sample presents higher conversion of propane and better selectivity to propene,thus resulting in an enhanced propene yield,in the entire range of temperatures investigated. The best performance of P(0.05)Co catalyst was achieved at 525 ℃,where propene selectivity amounts to 55.6% at a propane conversion of 28.3%. To the best of our knowledge,this is the highest propene selectivity combined with a reasonably high propane conversion obtained over cobalt-based catalysts reported thus far. The stability of P(0.05)Co was also examined at 500 ℃. It was found that the catalyst gave the propane conversion of ~28% and propene selectivity of 53% without any loss in activity and selectivity over a running time of 15 h examined,indicating the high stability of the P-modified catalyst.
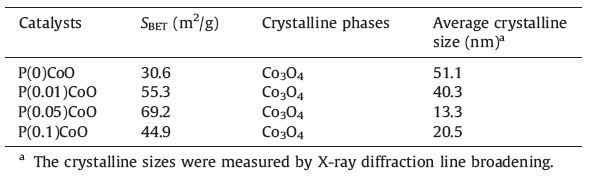
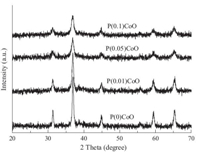
The XRD patterns of PCoO catalysts with different P contents are illustrated in Fig. 2. For all catalysts with or without phosphorus, the only diffraction peaks appearing at 31.3°,36.9°,45.0°,59.5° and 65.4° are assigned to those of Co3O4 [9]. No peaks due to Pcontaining phases could be found,indicating that the P species are amorphous,or well dispersed. However,all peaks become broader with the addition of phosphorus,demonstrating that the crystalline size of Co3O4 was reduced by P modification. The XRD results and the surface areas of the catalysts are summarized in Table 2. It can be seen that the P-modified samples show higher surface areas and smaller crystallites by XRD than do pure cobalt oxide. Moreover,PCoO catalysts present quite different crystalline sizes, strongly depending on their composition. Among the catalysts investigated,P(0.05)CoO exhibits the highest surface area and the smallest crystalline size.
The morphology of the catalysts was observed by TEM and shown in Fig. 3. The influences of phosphorus on particle size and morphology of Co3O4 is manifest. The pure Co3O4 catalyst is composed of nano particles with sizes of 40-50 nm,which are much larger than those of P-modified samples. The particle sizes of the samples decrease in the order of P(0)CoO > P(0.01)- P(0.01)CoO > P(0.1)CoO > P(0.05)CoO,in good agreement with XRD results described above. Such a trend,however,is just opposite to the sequence of propane conversion as shown in Table 1. This observation clearly indicates the catalytic activity is closely related to the particle size of the catalyst. Generally, nanocatalytic materials with a high surface area should generate better catalytic performance due to their numerous edges and corners which are conventionally considered as active sites for adsorption of reactants.
| Table 1 Catalytic performances of PCoO catalysts for the ODH of propane at 500 ℃. |
|
Download:
|
|
Fig. 1. Catalytic performance as a function of temperature for P(0)CoO and P(0.05)CoO catalysts. (A) C3H8 conversion; (B) C3H6 selectivity; (C) C3H6 yield. |
|
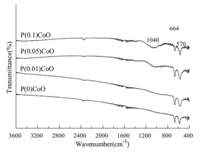
The FT-IR spectra of PCoO catalysts are shown in Fig. 4. All of the catalysts display two distinctive bands at 570 cm -1 and 664 cm -1 originating from Co3O4 [10]. With the addition of phosphorus to the catalyst,a new band appears at around 1040 cm-1 and its intensity is enhanced with increasing P content. This band can be assigned to PO43 - [11]. The phosphate radical related species in the catalyst is highly dispersed and undetectable by XRD. Clearly,the anion PO43 - plays an important role in modifying the structure and catalytic properties of the catalyst. Previous publications have reported that SO42- can act as a grain refiner and/or growth inhibitor,thus decreasing the crystalline size of cerium hydroxide/ oxide doped with SO42- [12]. Here,PO43 - probably plays a similar role in the case of PCoO catalysts. Another effect of PO43 - is that it serves as an agent for isolating the active sites,which has been proposed for CrP/TiO2 [13] and phosphorous-modified graphitic mesoporous carbon [14]. For selective oxidation,the site isolation of the catalyst is very important to achieve a high selectivity [15]. Moreover,other properties,including reducibility and the surface acid-base property of the catalyst,may also be modified by phosphorus. Regarding these,detailed characterization and further study are under way.
| Table 2 BET surface areas and XRD results of PCoO catalysts with different P contents. |
|
Download:
|
| Fig. 2. XRD patterns of PCoO catalysts with different P contents. | |

|
Download:
|
| Fig. 3. TEM images of (a) P(0)CoO,(b) P(0.01)CoO,(c) P(0.05)CoO and (d) P(0.1)CoO catalysts. | |
|
Download:
|
| Fig. 4. FT-IR spectra of PCoO catalysts with different P contents. | |
In summary,nanosized phosphorus-modified cobalt oxide catalysts have been synthesized by the sol-gel method and proved to be efficient for oxidative dehydrogenation of propane to propene. Compared to the pure cobalt oxide,the P-modified samples exhibit much smaller crystallite sizes,which is responsible for their higher catalytic activities. Moreover,the highly dispersed phosphate in PCoO catalysts may act as an agent for the isolation of active sites,thus yielding better selectivity toward propene than the P-free catalyst.
This project was supported by the National Basic Research Program of China (Nos. 2010CB732303 and 2013CB933102),the National Natural Science Foundation of China (Nos. 21073148 and 21033006) and the Program for Innovative Research Team of the Ministry of Education of China (No. IRT1036).
| [1] | F. Cavani, N. Ballarini, A. Cericola, Oxidative dehydrogenation of ethane and propane: how far from commercial implementation, Catal. Today 127(2007) 113-131. |
| [2] | Y.M. Liu, L.C. Wang, M. Chen, et al., Highly selective Ce-Ni-O catalysts for efficient low temperature oxidative dehydrogenation of propane, Catal. Lett. 130(2009) 350-354. |
| [3] | Q.H. Zhang, C.J. Cao, T. Xu, et al., NiO-polyoxometalate nanocomposites as efficient catalysts for the oxidative dehydrogenation of propane and isobutane, Chem. Commun.(2009) 2376-2378. |
| [4] | J.H. Li, C.C. Wang, C.J. Huang, W.Z. Weng, H.L. Wan, Low temperature catalytic performance of nanosized CeNbNiO mixed oxide for oxidative dehydrogenation of propane to propene, Catal. Lett. 137(2010) 81-87. |
| [5] | J.H. Li, C.C. Wang, C.J. Huang, et al., Mesoporous nickel oxides as effective catalysts for oxidative dehydrogenation of propane to propene, Appl. Catal. A: Gen. 382(2010) 99-105. |
| [6] | T.E. Davies, T. García, B. Solsona, S.H. Taylor, Nanocrystalline cobalt oxide: a catalyst for selective alkane oxidation under ambient conditions, Chem. Commun.(2006) 3417-3419. |
| [7] | A. Aaddane, M. Kacimi, M. Ziyad, Oxidative dehydrogenation of ethane and propane over magnesium-cobalt phosphates CoxMg3-x(PO4)2, Catal. Lett. 73(2001) 47-53. |
| [8] | G. Mitran, T. Cacciaguerra, S. Loridant, D. Tichit, I.C. Marcu, Oxidative dehydrogenation of propane over cobalt-containing mixed oxides obtained from LDH precursors, Appl. Catal. A: Gen. 417(2012) 153-162. |
| [9] | B. Jongsomjit, J. Panpranot, J.G. Goodwin, Co-support compound formation in alumina-supported cobalt catalysts, J. Catal. 204(2001) 98-109. |
| [10] | S.G. Christoskova, M. Stoyanova, M. Georgieva, D. Mehandjiev, Preparation and characterization of a higher cobalt oxide, Mater. Chem. Phys. 60(1999) 39-43. |
| [11] | I. Matković, N. Maltar-Strmečki, V. Babić-Ivančić, M. Dutour Sikirić, V. Noethig-Laslo, Characterisation of β-tricalcium phosphate-based bone substitute materials by electron paramagnetic resonance spectroscopy, Radiat. Phys. Chem. 81(2012) 1621-1628. |
| [12] | Y. Kobayashi, Y. Fujiwara, Effect of SO4-2 on the corrosion behavior of ceriumbased conversion coatings on galvanized steel, Electrochim. Acta 51(2006) 4236-4242. |
| [13] | J. El-Idrissi, M. Kacimi, F. Bozon-Verduraz, M. Ziyad, Oxidative dehydrogenation of ethane over Cr/TiO2 modified by phosphorus, Catal. Lett. 56(1998) 221-225. |
| [14] | V. Schwartz, H. Xie, H.M. Meyer Ⅲ, S.H. Overbury, C.D. Liang, Oxidative dehydrogenation of isobutane on phosphorous-modified graphitic mesoporous carbon, Carbon 49(2011) 659-668. |
| [15] | R.K. Grasselli, Fundamental principles of selective heterogeneous oxidation catalysis, Top. Catal. 21(2002) 79-88. |