文章信息
- 马忠娜, 周波蓉, 宋雅楠, 张苗, 夏伟
- Ma Zhongna, Zhou Borong, Song Yanan, Zhang Miao, Xia Wei
- PYCR1在结直肠癌诊断和随访中的价值分析
- Value of PYCR1 in diagnosis and follow-up of colorectal cancer
- 实用肿瘤杂志, 2026, 41(2): 147-153
- Journal of Practical Oncology, 2026, 41(2): 147-153
基金项目
- 国家自然科学基金(82204845); 上海市科学技术委员会科技创新行动计划(22ZR1447900); 上海市浦东新区卫生健康委员会学科建设项目(PWZxk2022-04)
-
通信作者
- 夏伟,Email:awingxia@163.com
-
文章历史
- 收稿日期:2025-01-27
2. 湖南省肿瘤医院放射物理技术部, 湖南 长沙 410013;
3. 上海市浦东新区公利医院核医学科, 上海 200135;
4. 上海中医药大学附属第七人民医院临床研究管理办公室, 上海 200137
2. Radiotherapy Technology Department, Hunan Cancer Hospital, Changsha 410013, China;
3. Department of Nuclear Medicine, Gongli Hospital of Shanghai Pudong New Area, Shanghai 200135, China;
4. Clinical Research Unit, the Seventh People's Hospital of Shanghai University of Traditional Chinese Medicine, Shanghai 200137, China
2022年全球结直肠癌发病率和死亡率分别居癌症第3位和第2位[1]。近年来,我国结直肠癌发病率和死亡率均呈上升趋势。结直肠癌早期症状不明显,多数患者确诊时已为中晚期,且治疗后康复情况迥异[2-3]。虽然癌胚抗原(carcinoembryonic antigen, CEA)和糖类抗原19-9(carbohydrate antigen 19-9, CA19-9)已用于结直肠癌的临床诊断和随访,但诊断的敏感度和特异度均不高[4]。
吡咯啉-5羧酸还原酶1(pyrroline-5-carboxylate reductase 1, PYCR1)基因位于17号染色体q25.3,编码319个氨基酸组成的蛋白质[5]。PYCR1是谷氨酸合成脯氨酸途径中的关键酶,通过催化吡咯啉-5磷酸还原为脯氨酸并伴随烟酰胺腺嘌呤二核苷酸磷酸的氧化,在保护细胞内氨基酸代谢、氧化应激和线粒体的完整性中起重要作用[6-9]。PYCR1在多种恶性肿瘤中高表达,参与调控肿瘤细胞能量代谢和信号传递,与肿瘤细胞增殖、迁移和侵袭等恶性生物学特征密切相关,为潜在肿瘤标志物[10]。PYCR1过表达促进结直肠癌细胞的增殖与迁移[11-12]。但目前关于PYCR1与结直肠癌的研究多聚焦于组织和细胞层面,因此本研究分析结直肠腺癌患者血清PYCR1水平,比较PYCR1和CEA联合检测与CEA和CA19-9联合检测对结直肠癌诊断和术后复发的预测效能,以期评估PYCR1作为血清生物学标志物的可行性,为临床诊断和随访提供依据。
1 资料与方法 1.1 生物信息学分析2024年6月采用基因表达谱交互分析(Gene Expression Profiling Interactive Analysis, GEPIA)数据库(http://gepia2.cancer-pku.cn/)获取癌症基因组图谱(The Cancer Genome Atlas, TCGA)和基因型组织表达(Genotype-Tissue Expression, GTEx)数据库中的RNA-seq表达数据,分析在结直肠腺癌组织中PYCR1的表达情况[13]。
1.2 临床资料收集2022年2月至2023年12月就诊于上海中医药大学附属第七人民医院的133例血清样本及其相关资料,其中健康体检者20例,结直肠腺瘤患者31例,结直肠腺癌术前患者28例,结直肠腺癌术后无复发患者30例,结直肠腺癌术后复发患者24例。各组患者一般资料比较,差异均无统计学意义(均P > 0.05,表 1)。纳入标准:(1)健康体检者体检健康且肿瘤指标检测值在正常范围内;(2)结直肠腺瘤患者经肠镜活检或手术切除病理确诊为结直肠腺瘤;(3)结直肠腺癌术前患者术前经病理活检证实为结直肠腺癌或术后病理确诊为结直肠腺癌;(4)结直肠腺癌术后无复发患者在接受结直肠腺癌根治术1个月以后,研究期间随访未发现复发;(5)结直肠腺癌术后复发患者为结直肠腺癌根治术术后复发或复发伴有转移者;(6)自愿参加研究。排除标准:(1)合并严重肝肾功能损伤或机体内环境紊乱;(2)合并其他原发性恶性肿瘤。本研究经上海中医药大学附属第七人民医院伦理委员会审核通过(审批号:2022-7th-HIRB-025),研究对象均知情同意。
| 临床特征 | 健康体检者(n=20) | 结直肠腺瘤(n=31) | 结直肠腺癌术前(n=28) | 结直肠腺癌术后无复发(n=30) | 结直肠腺癌术后复发(n=24) | F/χ2值 | P值 |
| 性别 | 3.497 | 0.478 | |||||
| 男性 | 9(45.0) | 19(61.3) | 17(60.7) | 21(70.0) | 16(66.7) | ||
| 女性 | 11(55.0) | 12(38.7) | 11(39.3) | 9(30.0) | 8(33.3) | ||
| 年龄(岁,x±s,范围) | 64.20±4.56(57~70) | 63.65±10.43(37~85) | 68.68±9.08(48~83) | 63.17±9.53(43~76) | 62.50±9.92(47~82) | 1.966 | 0.104 |
| 高血压 | 2.772 | 0.428 | |||||
| 有 | / | 19(61.3) | 14(50.0) | 12(40.0) | 12(50.0) | ||
| 无 | / | 12(38.7) | 14(50.0) | 18(60.0) | 12(50.0) | ||
| 糖尿病 | 1.268 | 0.737 | |||||
| 有 | / | 5(16.1) | 4(14.3) | 3(10.0) | 5(20.8) | ||
| 无 | / | 26(83.9) | 24(85.7) | 27(90.0) | 19(79.2) |
采用人PYCR1酶联免疫吸附试验(enzyme-linked immunosorbent assay, ELISA)检测试剂盒(货号EH11556,武汉菲恩生物科技有限公司)检测血清PYCR1表达水平。采用全自动酶标仪(型号GloMax-Multi,美国Promega公司)检测450 nm波长处各孔吸光度值,绘制标准曲线,计算各组血清样本PYCR1蛋白表达水平。
1.4 统计学分析应用SPSS 25.0软件进行统计学分析。应用GraphPad Prism 9软件绘图。计量资料两组间比较采用t检验或Mann-Whitney U检验,多组间比较采用单因素方差分析。计数资料组间比较采用χ2检验。PYCR1表达与血液肿瘤标志物水平的相关性分析采用Spearman相关分析。绘制受试者工作特征(receiver operating charateristic, ROC)曲线,获得曲线下面积(area under the curve, AUC),选择最大约登指数时的值作为PYCR1水平的最佳截断值。采用ROC曲线分析比较PYCR1、CEA和CA19-9指标单独和联合检测对结直肠癌诊断和术后复发的相关预测效能(CEA≥5 ng/mL为阳性,CA19-9≥27 U/mL为阳性。指标联合检测时,≥1个指标阳性视为联合检测阳性)。以P < 0.05为差异具有统计学意义。
2 结果 2.1 生物信息学分析GEPIA数据库比较275例结肠腺癌组织与349例正常结肠组织及92例直肠腺癌组织与328例正常直肠组织的PYCR1表达显示,结肠腺癌和直肠腺癌组织中PYCR1表达均升高(均P < 0.05,图 1)。
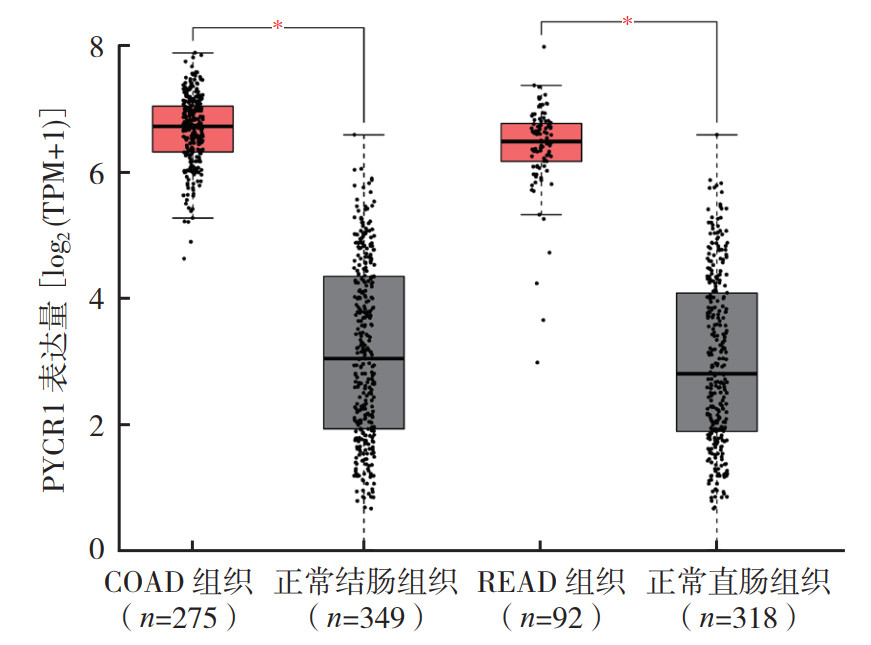
|
| 注 PYCR1:吡咯啉-5羧酸还原酶1(pyrroline-5-carboxylate reductase 1);TPM:每百万转录本数(transcripts per million);COAD:结肠腺癌(colon adenocarcinoma);READ:直肠腺癌(rectum adenocarcinoma);*P < 0.05 图 1 GEPIA数据库分析结直肠腺癌中PYCR1表达水平 Fig.1 PYCR1 expression in colorectal adenocarcinoma tissues in the GEPIA database |
与健康体检者比较,结直肠腺瘤患者、结直肠腺癌术前患者和结直肠腺癌术后复发患者的血清PYCR1表达水平均升高(均P < 0.01,图 2A),结直肠腺癌术后无复发患者的血清PYCR1表达水平差异无统计学意义(P > 0.05)。与结直肠腺癌术前患者比较,结直肠腺瘤患者和结直肠腺癌术后无复发患者血清PYCR1表达均降低(均P < 0.05,图 2A~2B)。与结直肠腺癌术后无复发患者比较,结直肠腺癌术后复发患者的血清PYCR1表达水平升高(P < 0.01)。结直肠腺癌术前和术后无复发患者中,血清PYCR1在结肠腺癌和直肠腺癌中的表达水平比较,差异均无统计学意义(均P > 0.05,图 2C)。血清PYCR1水平在结直肠肿瘤患者中高表达,且随结直肠肿瘤的发展呈升高趋势,可能是潜在的结直肠肿瘤分子标志物。
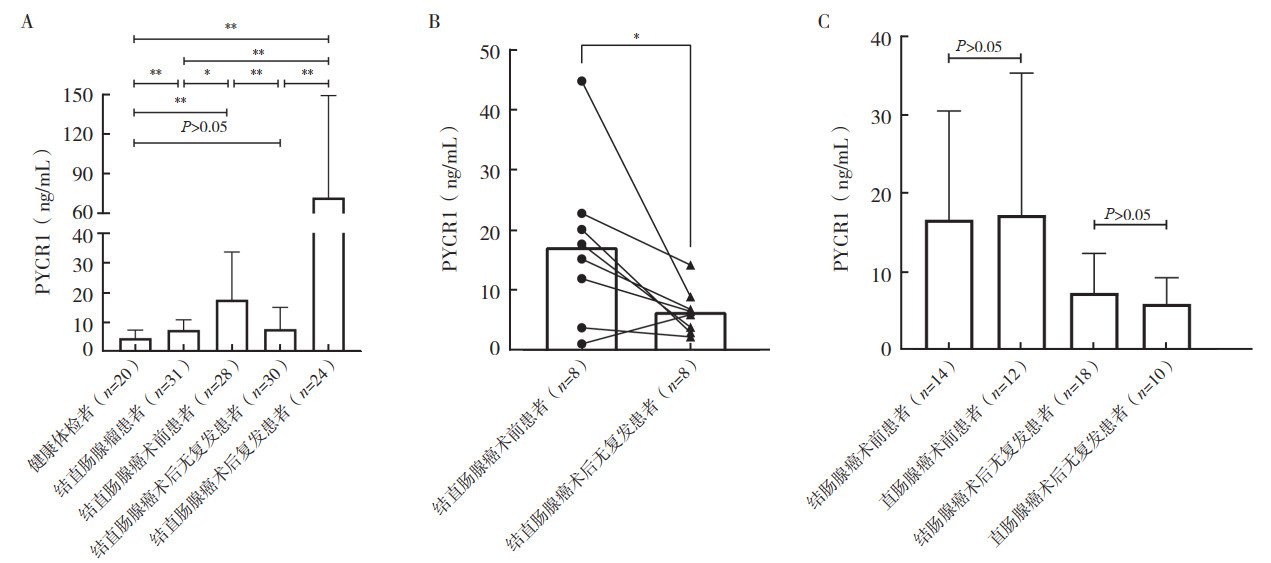
|
| 注 A:健康体检者、结直肠腺瘤患者、结直肠癌术前患者、结直肠癌术后无复发患者和结直肠癌术后复发患者的血清PYCR1表达水平比较;B:结直肠腺癌术前和术后无复发配对样本的血清PYCR1表达水平比较;C:结肠腺癌和直肠腺癌术前和术后无复发患者血清PYCR1表达水平比较;PYCR1:吡咯啉-5羧酸还原酶1(pyrroline-5-carboxylate reductase 1);*P < 0.05;**P < 0.01 图 2 健康体检者、结直肠腺瘤患者、结直肠腺癌术前患者、结直肠腺癌术后无复发患者和结直肠腺癌术后复发患者的血清PYCR1表达水平比较 Fig.2 Comparison of serum PYCR1 levels in healthy subjects, colorectal adenoma patients, pre-operative colorectal adenocarcinoma patients, post-operative colorectal adenocarcinoma patients without recurrence, and post-operative colorectal adenocarcinoma patients with recurrence |
分析结直肠腺癌术前患者和结直肠腺癌术后复发患者数据,以血清PYCR1表达水平的中位数20.03 ng/mL作为临界值进行分组分析。结果显示,血清PYCR1表达在肿瘤大小和远处转移方面比较,差异均具有统计学意义(均P < 0.05,表 2)。
| 临床特征 | 例数 | 血清PYCR1水平 | χ2值 | P值 | |
| < 20.03 ng/mL | ≥20.03 ng/mL | ||||
| 性别 | 0.083 | 0.773 | |||
| 男性 | 33 | 17(51.5) | 16(48.5) | ||
| 女性 | 19 | 9(47.4) | 10(52.6) | ||
| 年龄 | 0 | 1 | |||
| < 60岁 | 16 | 8(50.0) | 8(50.0) | ||
| ≥60岁 | 36 | 18(50.0) | 18(50.0) | ||
| 肿瘤大小 | 4.923 | 0.047 | |||
| < 5 cm | 11 | 10(90.9) | 1(9.1) | ||
| ≥5 cm | 7 | 3(42.9) | 4(57.1) | ||
| 淋巴结转移 | 1.237 | 0.266 | |||
| 无 | 18 | 11(61.1) | 7(38.9) | ||
| 有 | 21 | 8(38.1) | 13(61.9) | ||
| 远处转移 | 9.641 | 0.002 | |||
| 无 | 26 | 19(73.1) | 7(26.9) | ||
| 有 | 24 | 7(29.2) | 17(70.8) | ||
| 注 PYCR1:吡咯啉-5羧酸还原酶1(pyrroline-5-carboxylate reductase 1) | |||||
以健康体检者和结直肠腺癌术前患者的血清PYCR1、CEA和CA19-9表达水平绘制ROC曲线,分析血清PYCR1水平对结直肠癌的潜在诊断价值(表 3)。血清PYCR1水平诊断结直肠癌的截断值为9.66 ng/mL。PYCR1和CEA联合检测的特异度为100%,敏感度为62.96%,阳性预测值为100%,阴性预测值为62.96%,敏感度、阴性预测值和AUC均优于CEA和CA19-9联合检测。
| 检测指标 | AUC | 95% CI | P值 | 特异度(%) | 敏感度(%) | 误诊率(%) | 漏诊率(%) | 阳性预测值(%) | 阴性预测值(%) |
| PYCR1 | 0.811 | 0.689~0.933 | < 0.01 | 100 | 57.14 | 0 | 42.86 | 100 | 62.50 |
| CEA | 0.861 | 0.751~0.970 | < 0.01 | 100 | 40.74 | 0 | 59.26 | 100 | 51.52 |
| CA19-9 | 0.708 | 0.537~0.880 | 0.030 | 100 | 16.67 | 0 | 83.33 | 100 | 42.86 |
| CEA+PYCR1 | 0.871 | 0.766~0.977 | < 0.01 | 100 | 62.96 | 0 | 37.04 | 100 | 62.96 |
| CEA+CA19-9 | 0.819 | 0.685~0.954 | 0.01 | 100 | 41.67 | 0 | 58.33 | 100 | 51.72 |
| 注 AUC:曲线下面积(area under the curve);PYCR1:吡咯啉-5羧酸还原酶1(pyrroline-5-carboxylate reductase 1);CEA:癌胚抗原(carcinoembryonic antigen);CA19-9:糖类抗原19-9(carbohydrate antigen 19-9) | |||||||||
以结直肠腺瘤和结直肠腺癌术前患者的血清PYCR1、CEA和CA19-9表达作ROC曲线,分析血清PYCR1水平对结直肠腺瘤和结直肠腺癌的鉴别诊断价值(表 4),血清PYCR1水平的截断值为11.66 ng/mL。PYCR1和CEA联合检测的特异度为100%,敏感度为59.26%,阳性预测值为100%,阴性预测值为64.52%,敏感度和阳性预测值均优于CEA和CA19-9联合检测,但AUC劣于CEA和CA19-9联合检测。
| 检测指标 | AUC | 95% CI | P值 | 特异度(%) | 敏感度(%) | 误诊率(%) | 漏诊率(%) | 阳性预测值(%) | 阴性预测值(%) |
| PYCR1 | 0.687 | 0.540~0.835 | 0.014 | 93.55 | 53.57 | 6.45 | 46.43 | 88.24 | 69.05 |
| CEA | 0.745 | 0.605~0.886 | 0.004 | 100 | 40.74 | 0 | 59.25 | 100 | 55.56 |
| CA19-9 | 0.528 | 0.328~0.728 | 0.102 | 100 | 16.67 | 0 | 83.33 | 100 | 31.03 |
| CEA+PYCR1 | 0.769 | 0.631~0.906 | 0.002 | 100 | 59.26 | 0 | 40.74 | 100 | 64.52 |
| CEA+CA19-9 | 0.806 | 0.657~0.954 | 0.008 | 100 | 41.67 | 0 | 58.33 | 100 | 39.13 |
| 注 AUC:曲线下面积(area under the curve);PYCR1:吡咯啉-5羧酸还原酶1(pyrroline-5-carboxylate reductase 1);CEA:癌胚抗原(carcinoembryonic antigen);CA19-9:糖类抗原19-9(carbohydrate antigen 19-9) | |||||||||
以结直肠腺癌术后无复发患者和结直肠腺癌术后复发患者的血清PYCR1、CEA和CA19-9表达水平绘制ROC曲线,分析血清PYCR1水平对结直肠腺癌术后复发的预测价值(表 5),PYCR1水平截断值为12.64 ng/mL。PYCR1和CEA联合检测的特异度为80.00%,敏感度为85.71%,阳性预测值为75.00%,阴性预测值为88.89%,均优于CEA和CA19-9联合检测。
| 检测指标 | AUC | 95% CI | P值 | 特异度(%) | 敏感度(%) | 误诊率(%) | 漏诊率(%) | 阳性预测值(%) | 阴性预测值(%) |
| PYCR1 | 0.899 | 0.804~0.993 | < 0.01 | 90.00 | 83.33 | 10.00 | 16.67 | 86.96 | 87.10 |
| CEA | 0.933 | 0.863~1.000 | < 0.01 | 90.00 | 80.95 | 10.00 | 19.05 | 85.00 | 87.10 |
| CA19-9 | 0.727 | 0.570~0.884 | 0.006 | 89.66 | 54.55 | 10.34 | 45.45 | 80.00 | 72.22 |
| CEA+PYCR1 | 0.919 | 0.832~1.000 | < 0.01 | 80.00 | 85.71 | 20.00 | 14.29 | 75.00 | 88.89 |
| CEA+CA19-9 | 0.921 | 0.840~1.000 | < 0.01 | 79.31 | 80.95 | 20.69 | 19.05 | 73.91 | 85.19 |
| 注 AUC:曲线下面积(area under the curve);PYCR1:吡咯啉-5羧酸还原酶1(pyrroline-5-carboxylate reductase 1);CEA:癌胚抗原(carcinoembryonic antigen);CA19-9:糖类抗原19-9(carbohydrate antigen 19-9) | |||||||||
分析52例结直肠腺癌术前患者和结直肠腺癌术后复发患者血清中的PYCR1和肿瘤标志物水平显示,血清中的PYCR1和肿瘤标志物水平显示,PYCR1水平与CEA呈强正相关(r=0.832,P < 0.01),与CA19-9(r=0.632)、CA50(r=0.587)和CA242(r=0.574)呈中等正相关(均P < 0.01),与AFP(r=0.298,P=0.038)、CA125(r=0.435,P < 0.01)和CA72-4(r=0.380,P < 0.01)呈弱正相关,与CA15-3无相关性(r=0.232,P=0.135)。
3 讨论多数结直肠癌患者按正常、腺瘤至腺癌发展。Ⅰ期结直肠癌患者5年生存率可 > 90%,而Ⅳ期患者则 < 15%[14-15]。复发是结直肠癌预后不良的主要表现。10%~20%的Ⅱ期和30%~40%的Ⅲ期结直肠癌患者会复发,增加死亡风险[16]。PYCR1作为线粒体合成脯氨酸的关键酶,在肿瘤细胞增殖、侵袭、转移、耐药和改善肿瘤微环境等生物过程中发挥作用,与肿瘤发生、发展和预后不良密切相关,是潜在的肿瘤标志物和治疗靶点[5, 10, 17-18]。
已有研究从组织和细胞水平证实,在结直肠腺瘤向结直肠腺癌演进过程中,PYCR1表达呈上升趋势,是癌前病变结直肠腺瘤向结直肠癌转化的潜在因素[12, 19-20]。PYCR1高表达增强肿瘤细胞的迁移与侵袭能力,在结直肠癌进展与转移中扮演重要角色[11, 21]。目前尚无PYCR1在结直肠癌血清学方面的相关研究。本研究从血清学角度揭示PYCR1表达随结直肠疾病进展(正常、结直肠腺瘤至结直肠腺癌)呈上升趋势,且与结直肠癌的肿瘤大小和远处转移存在关联。结直肠癌患者术后,血清PYCR1恢复正常水平,而在复发患者中表达水平升高。相较于传统组织活检,血清检查具有侵袭性小和可重复检测等特点[22]。这为PYCR1成为结直肠癌诊断和随访的血清标志物提供有力依据。
本研究通过计算阳性预测值和阴性预测值等指标,深入探讨血清PYCR1水平在结直肠癌诊断和术后随访中的临床应用价值。在结直肠癌诊断方面,与CEA和CA19-9联合检测比较,血清PYCR1和CEA联合检测的阴性预测值提高11.24%,漏诊率降低21.30%,说明PYCR1和CEA联合检测可以提升识别结直肠癌的能力。在结直肠腺瘤和腺癌鉴别诊断方面,与CEA和CA19-9联合检测比较,血清PYCR1和CEA联合检测的阴性预测值提高25.39%,漏诊率降低17.59%,说明PYCR1和CEA联合检测可以更加准确地鉴别良恶性肿瘤,并采取治疗措施防止病情恶化。在结直肠癌术后随访监测方面,与CEA和CA19-9联合检测比较,血清PYCR1和CEA联合检测的阳性和阴性预测值分别提高1.09%和3.70%,漏诊率降低4.76%,说明PYCR1和CEA联合检测可以减少不必要的医疗支出,减轻患者及其家庭的经济和心理负担。以上数据表明PYCR1联合CEA检测在结直肠癌诊断和术后随访中具有重要价值。
综上所述,血清PYCR1表达水平随结直肠疾病进展呈上升趋势,与肿瘤恶性进展和不良预后密切相关。PYCR1作为潜在的血清肿瘤标志物,无论对结直肠癌诊断、良恶性肿瘤鉴别诊断还是结直肠癌术后复发随访,都具有一定的临床价值,有望提高结直肠癌诊断准确性,同时更加精准地监测肿瘤进展和结直肠癌复发高危人群的风险分级管理。在本研究的基础上,后续将开展多中心研究,同时进一步探索PYCR1在结直肠癌发生和发展中的作用和机制,为PYCR1应用于临床提供更多的理论依据和研究数据。
利益冲突 所有作者声明无利益冲突
| [1] |
Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022:GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2024, 74(3): 229-263. |
| [2] |
王慧, 刘超, 章春雪, 等. 精胺氧化酶靶向调控硫氧还蛋白还原酶1促进结肠癌恶性进展[J]. 实用肿瘤杂志, 2024, 39(4): 317-332. DOI:10.13267/j.cnki.syzlzz.2024.049 |
| [3] |
中华人民共和国国家卫生健康委员会, 中华医学会肿瘤学分会. 中国结直肠癌诊疗规范(2023年版)[J]. 中华外科杂志, 2023, 61(8): 617-644. |
| [4] |
栗宇, 佟赫, 阴建, 等. 血清CEA、CA19-9和CA242检测在中国人群结直肠癌机会性筛查中应用的荟萃分析[J]. 中华健康管理学杂志, 2024, 10(8): 601-608. |
| [5] |
Bogner AN, Stiers KM, Tanner JJ. Structure, biochemistry, and gene expression patterns of the proline biosynthetic enzyme pyrroline-5-carboxylate reductase (PYCR), an emerging cancer therapy target[J]. Amino Acids, 2021, 53(12): 1817-1834. DOI:10.1007/s00726-021-02999-5 |
| [6] |
Christensen EM, Patel SM, Korasick DA, et al. Resolving the cofactor-binding site in the proline biosynthetic enzyme human pyrroline-5-carboxylate reductase 1[J]. J Biol Chem, 2017, 292(17): 7233-7243. DOI:10.1074/jbc.M117.780288 |
| [7] |
Westbrook RL, Bridges E, Roberts J, et al. Proline synthesis through PYCR1 is required to support cancer cell proliferation and survival in oxygen-limiting conditions[J]. Cell Rep, 2022, 38(5): 110320. DOI:10.1016/j.celrep.2022.110320 |
| [8] |
Hollinshead KER, Munford H, Eales KL, et al. Oncogenic IDH1 mutations promote enhanced proline synthesis through PYCR1 to support the maintenance of mitochondrial redox homeostasis[J]. Cell Rep, 2018, 22(12): 3107-3114. DOI:10.1016/j.celrep.2018.02.084 |
| [9] |
Schwörer S, Berisa M, Violante S, et al. Proline biosynthesis is a vent for TGFβ-induced mitochondrial redox stress[J]. EMBO J, 2020, 39(8): e103334. DOI:10.15252/embj.2019103334 |
| [10] |
Xue WH, Wu K, Guo XN, et al. The pan-cancer landscape of glutamate and glutamine metabolism: a comprehensive bioinformatic analysis across 32 solid cancer types[J]. Biochim Biophys Acta Mol Basis Dis, 2024, 1870(2): 166982. DOI:10.1016/j.bbadis.2023.166982 |
| [11] |
秦曦, 姜政. PYCR1在结肠癌中的表达及对细胞的增殖、侵袭及迁移的影响[J]. 第三军医大学学报, 2019, 41(20): 1961-1968. |
| [12] |
Alaqbi SS, Burke L, Guterman I, et al. Increased mitochondrial proline metabolism sustains proliferation and survival of colorectal cancer cells[J]. PLoS One, 2022, 17(2): e0262364. DOI:10.1371/journal.pone.0262364 |
| [13] |
Tang ZF, Li CW, Kang BX, et al. GEPIA a web server for cancer and normal gene expression profiling and interactive analyses[J]. Nucleic Acids Res, 2017, 45(W1): W98-W102. DOI:10.1093/nar/gkx247 |
| [14] |
Siegel RL, Wagle NS, Cercek A, et al. Colorectal cancer statistics, 2023[J]. CA, 2023, 73(3): 233-254. |
| [15] |
中华医学会肿瘤学分会早诊早治学组, 丁克峰, 徐忠法, 等. 中国结直肠癌早诊早治专家共识(2023版)[J]. 中华医学杂志, 2023, 103(48): 3896-3908. |
| [16] |
Nors J, Iversen LH, Erichsen R, et al. Incidence of recurrence and time to recurrence in stage Ⅰ to Ⅲ colorectal cancer: a nationwide Danish cohort study[J]. JAMA Oncol, 2024, 10(1): 54-62. DOI:10.1001/jamaoncol.2023.5098 |
| [17] |
D'Aniello C, Patriarca EJ, Phang JM, et al. Proline metabolism in tumor growth and metastatic progression[J]. Front Oncol, 2020, 10: 776. DOI:10.3389/fonc.2020.00776 |
| [18] |
Li Y, Xu JH, Bao PC, et al. Survival and clinicopathological significance of PYCR1 expression in cancer: a meta-analysis[J]. Front Oncol, 2022, 12: 985613. DOI:10.3389/fonc.2022.985613 |
| [19] |
Zheng K, Sha NN, Hou GF, et al. IGF1R-phosphorylated PYCR1 facilitates ELK4 transcriptional activity and sustains tumor growth under hypoxia[J]. Nat Commun, 2023, 14(1): 6117. DOI:10.1038/s41467-023-41658-z |
| [20] |
Roth U, Razawi H, Hommer J, et al. Differential expression proteomics of human colorectal cancer based on a syngeneic cellular model for the progression of adenoma to carcinoma[J]. Proteomics, 2010, 10(2): 194-202. DOI:10.1002/pmic.200900614 |
| [21] |
Yan K, Xu X, Wu T, et al. Knockdown of PYCR1 inhibits proliferation, drug resistance and EMT in colorectal cancer cells by regulating STAT3-mediated p38 MAPK and NF-κB signalling pathway[J]. Biochem Biophys Res Commun, 2019, 520(2): 486-491. DOI:10.1016/j.bbrc.2019.10.059 |
| [22] |
中国临床肿瘤学会CSCO结直肠癌专家委员会, 袁瑛, 张苏展. 结直肠癌分子标志物临床检测中国专家共识[J]. 中华胃肠外科杂志, 2021, 24(3): 191-197. |
 2026, Vol. 41
2026, Vol. 41


