2. 上海海洋大学 上海水产养殖工程技术研究中心, 上海 201306
20世纪60年代,随着坛紫菜(Pyropia haitanensis)的基础生物学、生活史、全人工采苗和人工栽培技术等研究取得成功,大规模栽培取得了巨大发展[1]。近十几年来,随着栽培规模和密度的增加,群体内近交繁殖严重,坛紫菜野生种种质退化,产量和品质均有明显的下降。另外,气候变暖对坛紫菜栽培也造成了较严重的影响[2],据近十几年来的调查,在坛紫菜壳孢子采苗期间及采苗后约1个月时,海水温度回升现象时常发生,即所谓的“小阳春”,轻者影响壳孢子的萌发和叶状体的生长,严重时会引起大规模的烂苗和掉苗,特别是2008年9—10月,在福建闽东和浙江温州等地持续的高温天气,造成坛紫菜栽培海区的温度出现回升,发生了大规模的烂菜,使菜农遭受巨大的经济损失[3]。研究[4]表明,到21世纪末,极端高温天气的出现频率和持续时间均会增加,且海水平均温度也会逐渐升高。因此,开展坛紫菜耐高温新品种(系)的培育,对该产业的可持续发展具有重要意义。近年来,利用遗传育种技术已培育出多个坛紫菜耐高温新品种(系),并在生产中推广应用,取得了良好的增产增收效果[2-3, 5-9]。
圆紫菜(Pyropia suborbiculata)属暖温性海藻,其叶状体呈圆形或肾脏形,在我国主要分布于青岛以南的黄海、东海和南海海域[10]。目前国内外学者[11-16]已对圆紫菜的生活史、藻体形态建成、色素突变体和分类标记等进行了研究,已有研究[17]表明,热带产的圆紫菜具有耐高温的潜力,但其耐高温性是否优于坛紫菜仍未知。
研究分别将圆紫菜野生品系(PS-WT)和坛紫菜栽培品系(PH-WT10)的壳孢子萌发体和叶状体置于23 ℃(常温)和30 ℃(高温)下培养,以探究两者的耐高温性差异,为今后开展圆紫菜遗传改良,选拔出可供生产上使用的长型品种奠定理论基础。
1 材料与方法 1.1 实验材料、壳孢子收集与叶状体培养本实验使用的圆紫菜野生品系于2016年采自广东省阳江市沿海(21°28′N~22°41′N, 111°16′E~112°21′ E)的自然礁石上,建立纯系后被命名为PS-WT[13];使用的坛紫菜栽培品系于2015年采自福建省福鼎市大员当沿海(27°06′ N, 120°18′ E)的坛紫菜栽培网帘,建立纯系后被命名为PH-WT10[18]。2个品系均以自由丝状体的形式被保存于实验室内,保存条件同文献[19]。培养液为添加MES培养基的灭菌海水[20]。
将少量(约0.02 g) 自由丝状体打碎至长约300 μm的小段后,接种到文蛤(Meretrix meretrix)壳表面,形成贝壳丝状体。暗光3 d后,将贝壳丝状体见光培养,培养条件:温度(23±0.5)℃,光强20 μmol photons/(m2·s),光周期10L ∶ 14D。14 d后,刷去贝壳上多余的丝状体,更换全部培养液,并将光强调至40 μmol photons/(m2·s)。待丝状体长满贝壳后,将其进行促熟处理,促熟条件:温度(29±0.5)℃,光强20 μmol photons/(m2·s),光周期8L ∶ 16D。数周后,将成熟较好的贝壳丝状体放入塑料杯(250 mL)中后置于(23±0.5)℃下进行充气培养,并放入3~6根棉线供壳孢子附着。待壳孢子附着后,将棉线置于充气瓶(250 mL)中进行充气培养,15 d后,将壳孢子萌发体刮下后继续充气培养,培养条件:温度(23±0.5)℃,光强60 μmol photons/(m2·s),光周期10L ∶ 14D,每5 d更换1/2培养液。
1.2 壳孢子萌发体的耐高温性测试将壳孢子液倒入铺有盖玻片的培养皿(Φ=9 cm)中,静置1 h后,将壳孢子萌发体分别置于(23±0.5)℃和(30±0.5)℃下培养,其他培养条件:光强60 μmol photons/(m2·s),光周期10L ∶ 14D,每5 d更换1/2事先已调节好温度的培养液。14 d后,统计壳孢子萌发体的存活率和假根发生率,以及含不同细胞数的萌发体百分率,并将附有壳孢子萌发体的盖玻片置于光学显微镜(Eclipse 90i,尼康)下拍照记录。每个温度组设置3个重复。
1.3 叶状体的回温测试随机选取在(23±0.5)℃下培养30 d的叶状体各20株,每10株为1组置于充气瓶(1 L)中进行充气培养,培养温度分别为(23±0.5)℃和(30±0.5)℃,其他培养条件:光强60 μmol photons/(m2·s),光周期10L ∶ 14D,每5 d更换1/2事先已调节好温度的培养液。10 d后,拍摄藻体的形态和细胞显微照片,并采用超便携式调制叶绿素荧光仪Mini-PAM(Walz,Effeltrich)测定叶状体中部的PS Ⅱ最大光量子产量(Fv/Fm),测量方法同于参考文献[21]。上述培养均在培养箱(MIR-554,三洋)内完成,实测昼夜培养液中的温差为±0.5 ℃。
1.4 数据统计分析实验数据用Excel 2010进行常规计算后,采用SPSS 22.0软件进行独立样本t检验。统计值用平均值±标准差(Mean±SD)描述,统计结果P<0.05时存在显著差异,P<0.01时存在极显著差异。
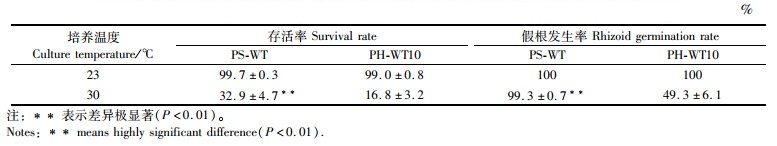
2 结果 2.1 壳孢子萌发体的存活率和假根发生率如表 1所示:在23 ℃组中,PS-WT和PH-WT10的壳孢子萌发体的存活率和假根发生率均超过99%,两者差异不显著(P>0.05);但在30 ℃组中,PS-WT的存活率和假根发生率分别为32.9%和99.3%,PH-WT10的存活率和假根发生率仅为16.8%和49.3%,两者差异极显著(P < 0.01)。
|
表 1 圆紫菜(PS-WT)和坛紫菜(PH-WT10)的壳孢子萌发体在不同温度下培养14 d后的存活率和假根发生率 Tab.1 Rates of survival and rhizoid germination of conchospore germlings of P. suborbiculata (PS-WT) and P. haitanensis (PH-WT10) after being cultured at different temperature for 14 days |
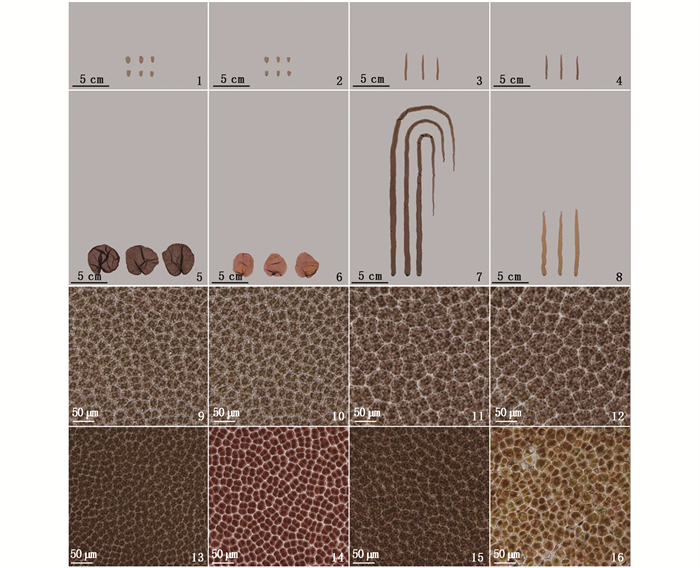
与23 ℃组相比,30 ℃组的PS-WT的壳孢子萌发体除细胞分裂速度减慢,颜色变红外,其他均无异常(图版Ⅰ-1, 2);而30 ℃组的PH-WT10的壳孢子萌发体其细胞分裂速度极慢,颜色变浅,色素体呈凝缩状(图版Ⅰ-3, 4)。

|
1, 2. 分别为在23和30 ℃下培养的PS-WT的壳孢子萌发体;3, 4. 分别为在23和30 ℃下培养的PH-WT10的壳孢子萌发体。 1, 2. Conchospore germlings of PS-WT cultured at 23 and 30 ℃, respectively; 3, 4. Conchospore germlings of PH-WT10 cultured at 23 and 30 ℃, respectively. 图版 Ⅰ 圆紫菜(PS-WT)和坛紫菜(PH-WT10)的壳孢子萌发体在不同温度下培养14 d后的形态 Plate Ⅰ Morphology of conchospore germlings of P. suorbiculata (PS-WT) and P. haitanensis (PH-WT10) after being cultured at different temperature for 14 days |
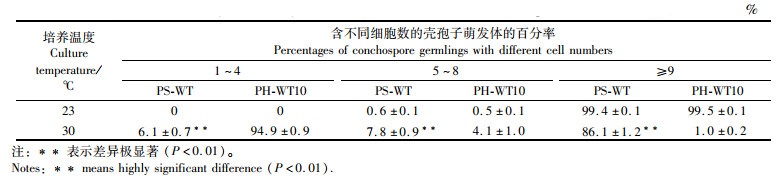
在23 ℃组中,含9个以上细胞的壳孢子萌发体的百分率,PS-WT和PH-WT10分别为99.4%和99.5%(表 2)。在30 ℃组中,PS-WT的壳孢子萌发体,其含9个以上细胞的百分率最高,为86.1%,其次为细胞数5~8和1~4个,分别占7.8%和6.1%。在PH-WT10中,含9个以上细胞的壳孢子萌发体只占总数的1.0%,而细胞数1~4个的百分率最高(94.9%),其次为细胞数5~8个(4.1%)。
|
表 2 圆紫菜(PS-WT)和坛紫菜(PH-WT10)的壳孢子萌发体在不同温度下培养14 d后含不同细胞数的个体百分率 Tab.2 Percentages of conchospore germlings with different cell numbers of P. suborbiculata (PS-WT) and P. haitanensis (PH-WT10) after being cultured at different temperature for 14 days |
在23 ℃组中,PS-WT的藻体呈圆形或肾脏形,PH-WT10的藻体呈细长形,两者的细胞均呈深棕褐色,具典型的星状色素体。在30 ℃组中,PS-WT的叶状体除生长减慢,细胞颜色变红外,其他均无异常,未出现死亡细胞,而PH-WT10的叶状体其生长显著变慢,细胞颜色变淡,色素体由星状变成弥散状,出现较多的死亡细胞(图版Ⅱ)。
|
1, 2. 分别为在23 ℃下培养30 d的PS-WT的叶状体形态; 3, 4. 分别为在23 ℃下培养30 d的PH-WT10的叶状体形态; 5, 6. 分别为再在23和30 ℃下培养10 d的PS-WT的叶状体形态; 7, 8. 分别为再在23和30 ℃下培养10 d的PH-WT10的叶状体形态; 9, 10. 分别为在23 ℃下培养30 d的PS-WT的细胞显微照片; 11, 12. 分别为在23 ℃下培养30 d的PH-WT10的细胞显微照片; 13, 14. 分别为再在23和30 ℃下培养10 d的PS-WT的细胞显微照片; 15, 16. 分别为再在23和30 ℃下培养10 d的PH-WT10的细胞显微照片。 1, 2. Morphology of the 30-day-old PS-WT blades cultured at 23 ℃; 3, 4. Morphology of the 30-day-old PH-WT10 blades cultured at 23 ℃; 5, 6. Morphology of the PS-WT blades cultured at 23 and 30 ℃ for another 10 days respectively; 7, 8. Morphology of the PH-WT10 blades cultured at 23 and 30 ℃ for another 10 days respectively; 9, 10. Cell micrograph of the 30-day-old PS-WT blades cultured at 23 ℃; 11, 12. Cell micrograph of the 30-day-old PH-WT10 blades cultured at 23 ℃; 13, 14. Cell micrograph of the PS-WT blades cultured at 23 and 30 ℃ for another 10 days respectively; 15, 16. Cell micrograph of the PH-WT10 blades cultured at 23 and 30 ℃ for another 10 days respectively. 图版 Ⅱ 在23 ℃下培养30 d的圆紫菜(PS-WT)和坛紫菜(PH-WT10)的叶状体以及再在不同温度下培养10 d的藻体形态和生长及细胞显微照片 Plate Ⅱ Morphology, growth and cell micrograph of the 30-day-old blades (at 23 ℃) of P. suorbiculata (PS-WT) and P. haitanensis (PH-WT10) and after being cultured at different temperature for another 10 days |
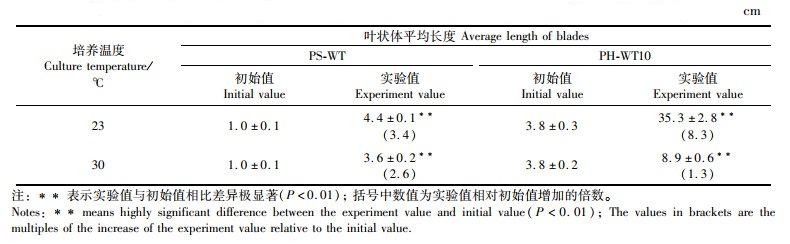
如表 3所示:在23 ℃下培养30 d后,PS-WT和PH-WT10的叶状体平均长度为1.0和3.8 cm(初始值);在23和30 ℃下再培养10 d后,PS-WT的叶状体长度(实验值)分别增加了3.4和2.6倍,而PH-WT10分别增加了8.3和1.3倍。
|
表 3 在23 ℃下培养30 d圆紫菜(PS-WT)和坛紫菜(PH-WT10)的叶状体再在不同温度下培养10 d后的叶状体平均体长 Tab.3 Average length of the 30-day-old blades (at 23 ℃) of P. suorbiculata (PS-WT) and P. haitanensis (PH-WT10) after being cultured at different temperatures for another 10 days |
与实验开始时的初始值相比:在23 ℃组中,PS-WT和PH-WT10的实验值无显著差异(P>0.05),但在30 ℃组中,PS-WT的实验值增加了1.8%,而PH-WT10却下降了34.5%,两者差异极显著(P < 0.01)。见表 4。
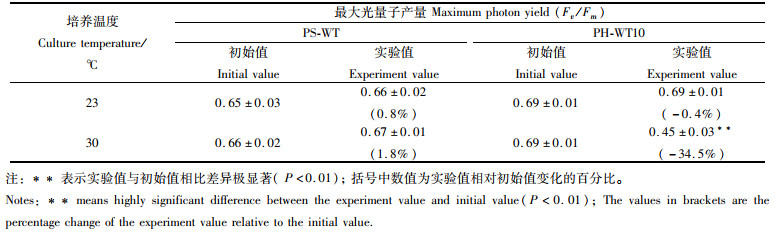
|
表 4 在23 ℃下培养30 d的圆紫菜(PS-WT)和坛紫菜(PH-WT10)的叶状体再在不同温度下培养10 d的最大光量子产量(Fv/Fm) Tab.4 Maximum photon yield of the 30-day-old blades (at 23 ℃) of P. suborbiculata (PS-WT) and P. haitanensis (PH-WT10) after being cultured at different temperature for another 10 days |
圆紫菜是一种广温性红藻,地理分布区域较广。MONOTILLA等[17]发现采自低纬度区域的圆紫菜其叶状体在30 ℃下可存活数周,与其他采自较高纬度的圆紫菜相比,具有更强的耐高温性。因此,本研究以采自中国南方沿海(低纬度)的圆紫菜为材料,比较分析其与坛紫菜的高温耐受性。
在生产栽培中,坛紫菜壳孢子采苗的适宜温度为25~27 ℃,在采苗后的几天内,若海水温度回升幅度较大,会严重影响壳孢子萌发体的存活与生长[22]。吕峰等[3]研究发现,坛紫菜野生品系的壳孢子萌发体在30 ℃下培养15 d后,其存活率会降低至6.7%。研究发现,在30 ℃培养下,PS-WT的壳孢子萌发体的存活率明显高于PH-WT10,且存活的个体其细胞形态正常,分裂速度较快,而PH-WT10存活的个体其细胞色素体凝缩,分裂速度极慢,这可能与高温破坏细胞的光合系统有关[23]。紫菜的假根来源于壳孢子第一次细胞分裂时产生的两个极性细胞之一,发育正常的假根一般短而发达,呈透明状,附着在网帘上使叶状体不会因风浪和水流等外力的冲击而脱离[24]。有研究[25]表明,高温会抑制坛紫菜壳孢子萌发体的假根形成。30 ℃下存活的PS-WT的壳孢子萌发体基本均可形成假根,而PH-WT10仅有约半数可形成假根。上述结果初步证实,PS-WT的壳孢子萌发体的耐高温性明显优于PH-WT10。
在坛紫菜幼苗阶段,水温回升轻者阻碍幼苗生长,严重时会引起大规模的烂苗和掉苗[5]。相比常温,在30 ℃下培养的PS-WT的叶状体其生长仅轻微减慢,而PH-WT10的叶状体生长显著变慢,且藻体出现小的烂洞。有研究[26]表明,植物在长时间或较高温度的热胁迫下,其细胞内细胞骨架解聚,原生质体结构解体,并最终死亡。经高温培养10 d后,PS-WT的细胞除颜色变红外,其他均无异常,未出现死亡细胞,而PH-WT10的细胞颜色变淡,色素体由星状变成弥散状,出现较多的死亡细胞,表明它受30 ℃高温的影响相对较大。Fv/Fm是PSⅡ最大光化学量子产量,反映植物PSⅡ原初光能转化效率,正常条件下,Fv/Fm值很稳定,但当植物受到逆境胁迫时,Fv/Fm值会明显下降[27]。黄林彬等[28]研究发现,随着培养温度的升高(22、24和25 ℃),条斑紫菜叶状体的Fv/Fm值逐渐降低。陈伟洲等[29]研究发现,相比常温,高温(29 ℃)下培养的坛紫菜叶状体其Fv/Fm明显降低。因此,Fv/Fm可作为鉴定紫菜叶状体对高温耐受性的指标。研究发现,叶状体经高温培养10 d后,PS-WT的Fv/Fm值增加了1.8%,而PH-WT10却下降了34.5%。上述结果初步证实,PS-WT的叶状体的耐高温性明显优于PH-WT10。
YANG等[12]研究发现,与坛紫菜一样,圆紫菜的壳孢子前几次分裂均为横分裂,但随着藻体继续发育,它的梢部会大量且持续地放散单孢子,最终导致其形态为圆形或肾脏形,在生产上不利于对其进行收割。张聪等[30]发现Pyropia chauhanii的野生品系(PC-WT)可放散单孢子,但它的诱变品系(PC-Y1)不放散单孢子;YAN等[31]采用人工诱变技术,从条斑紫菜少量放散单孢子的品系(WT)中分离出了可大量放散的品系,表明控制紫菜单孢子放散的基因可被人工所诱变。因此,今后可对圆紫菜进行诱变处理,选育出藻体细长且耐高温的新品种用于生产。
| [1] |
朱建一, 严兴洪, 丁兰平, 等. 中国紫菜原色图集[M]. 北京: 中国农业出版社, 2016. ZHU J Y, YAN X H, DING L P, et al. Color atlas of Chinese laver[M]. Beijing: China Agriculture Press, 2016. |
| [2] |
陈昌生, 纪德华, 谢潮添, 等. 坛紫菜耐高温品系选育及经济性状的初步研究[J]. 海洋学报, 2008, 30(5): 100-106. CHEN C S, JI D H, XIE C T, et al. Preliminary study on selecting the high temperature resistance strains and economic traits of Porphyra haitanensis[J]. Acta Oceanologica Sinica, 2008, 30(5): 100-106. DOI:10.3321/j.issn:0253-4193.2008.05.014 |
| [3] |
吕峰, 严兴洪, 刘长军, 等. 坛紫菜耐高温品系的选育与海区中试[J]. 上海海洋大学学报, 2010, 19(4): 457-462. LYU F, YAN X H, LIU C J, et al. Selection of a high-temperature tolerant strain of Porphyra haitanensis and its cultivation in sea area[J]. Journal of Shanghai Ocean University, 2010, 19(4): 457-462. |
| [4] |
GUO X J, HUANG J B, LUO Y, et al. Projection of heat waves over China for eight different global warming targets using 12 CMIP5 models[J]. Theoretical and Applied Climatology, 2017, 128(3/4): 507-522. |
| [5] |
梁艳, 徐燕, 陈昌生, 等. 坛紫菜优质新品系(Q-1)主要经济性状的研究[J]. 渔业科学进展, 2009, 30(4): 108-116. LIANG Y, XU Y, CHEN C S, et al. Research on the main economic traits of the new strain of high quality Porphyra haitanensis[J]. Progress in Fishery Sciences, 2009, 30(4): 108-116. |
| [6] |
纪德华, 谢潮添, 陈昌生, 等. 野生坛紫菜的选育及经济性状的研究[J]. 海洋学报, 2008, 30(1): 140-146. JI D H, XIE C T, CHEN C S, et al. The selective breeding of wild Porphyra haitanensis and its economical character[J]. Acta Oceanologica Sinica, 2008, 30(1): 140-146. DOI:10.3321/j.issn:0253-4193.2008.01.020 |
| [7] |
严兴洪, 马少玉. 坛紫菜抗高温品系的筛选[J]. 水产学报, 2007, 31(1): 112-119. YAN X H, MA S Y. Selection of a high-temperature resistant strain of Porphyra haitanensis (Rhodophyta)[J]. Journal of Fisheries of China, 2007, 31(1): 112-119. |
| [8] |
王长青, 严兴洪, 黄林彬, 等. 坛紫菜优良品系"申福2号"的特性分析与海区中试[J]. 水产学报, 2011, 35(11): 1658-1667. WANG C Q, YAN X H, HUANG L B, et al. Characterization of an improved strain (SF-2) of Porphyra haitanensis (Bangiales, Rhodophyta) and its pilot cultivation in mariculture farm[J]. Journal of Fisheries of China, 2011, 35(11): 1658-1667. |
| [9] |
宋武林. 坛紫菜新品种"申福1号"和"申福2号"耐高温性中试研究[J]. 上海海洋大学学报, 2016, 25(4): 522-527. SONG W L. Study on high-temperature-resisitance of the new varieties "Shenfu No. 1" and "Shenfu No. 2" of Porphyra haitanensis by cultivation at sea area[J]. Journal of Shanghai Ocean University, 2016, 25(4): 522-527. |
| [10] |
YANG L E, ZHOU W, HU C M, et al. A molecular phylogeny of the bladed Bangiales (Rhodophyta) in China provides insights into biodiversity and biogeography of the genus Pyropia[J]. Molecular Phylogenetics and Evolution, 2018, 120: 94-102. DOI:10.1016/j.ympev.2017.11.009 |
| [11] |
费勤娇, 丁洪昌, 张鹏, 等. 基于转录组信息的坛紫菜EST-SSR标记的开发及新品系SW-81的种质鉴定[J]. 上海海洋大学学报, 2021, 30(3): 464-474. FEI Q J, DING H C, ZHANG P, et al. Development of EST-SSR marker based on transcriptome and the research of germplasm identification for the new strain (SW-81) of Pyropia haitanensis[J]. Journal of Shanghai Ocean University, 2021, 30(3): 464-474. |
| [12] |
YANG X W, DING H C, YAN X H. Genetic analysis of the period of meiosis and morphogenesis of the gametophytic blades in Pyropia suborbiculata (Bangiales, Rhodophyta)[J]. Journal of Applied Phycology, 2019, 31(6): 4061-4068. |
| [13] |
赵爽, 丁洪昌, 刘长军, 等. 圆紫菜人工色素突变体的诱导与分离[J]. 海洋学报, 2019, 41(2): 114-122. ZHAO S, DING H C, LIU C J, et al. Induction and isolation of pigmentation mutants in Pyropia suborbiculata Kjellman (Bangiales, Rhodophyta)[J]. Haiyang Xuebao, 2019, 41(2): 114-122. |
| [14] |
BROOM J E, NELSON W A, YARISH C, et al. A reassessment of the taxonomic status of Porphyra suborbiculata, Porphyra carolinensis and Porphyra lilliputiana (Bangiales, Rhodophyta) based on molecular and morphological data[J]. European Journal of Phycology, 2002, 37(2): 227-235. |
| [15] |
ABE M, KOBAYASHI M, FUJIYOSHI E, et al. Use of PCR-RFLP for the discrimination of Japanese Porphyra and Pyropia species (Bangiales, Rhodophyta)[J]. Journal of Applied Phycology, 2013, 25(1): 225-232. |
| [16] |
郭文霞, 丁洪昌, 严兴洪. 光强和温度对圆紫菜叶状体体细胞发育分化的影响[J]. 上海海洋大学学报, 2020, 29(2): 209-217. GUO W X, DING H C, YAN X H. The effects of light intensity and temperature on the development and differentiation of single somatic cells isolated from the gametophytic blades in Pyropia suborbiculata (Bangiales, Rhodophyta)[J]. Journal of Shanghai Ocean University, 2020, 29(2): 209-217. |
| [17] |
MONOTILLA W D, NOTOYA M. Morphological and physiological responses of Porphyra suborbiculata Kjellman (Bangiales, Rhodophyta) blades from five localities[J]. Botanica Marina, 2004, 47(4): 323-334. |
| [18] |
DING H C, WANG T G, ZHANG P, et al. High-temperature resistance of the thin-blade strain of Pyropia haitanensis and its pilot cultivation in mariculture farm[J]. Journal of Applied Phycology, 2020, 32(4): 2261-2270. |
| [19] |
YAN X H, ARUGA Y. Induction of pigmentation mutants by treatment of monospore germlings with NNG in Porphyra yezoensis Ueda (Bangiales, Rhodophyta)[J]. Algae, 1997, 12(1): 39-54. |
| [20] |
王素娟, 张小平, 徐志东, 等. 坛紫菜营养细胞和原生质体培养的研究Ⅰ[J]. 海洋与湖沼, 1986, 17(3): 217-221. WANG S J, ZHANG X P, XU Z D, et al. A study on the cultivation of the vegetative cells and protoplasts of P. haitanensis Ⅰ[J]. Oceanologia et Limnologia Sinica, 1986, 17(3): 217-221. |
| [21] |
檀应华, 黄林彬, 严兴洪. 坛紫菜耐低盐品系的选育与特性分析[J]. 海洋与湖沼, 2014, 45(3): 504-512. TAN Y H, HUANG L B, YAN X H. Selection and characterization of a low-salinity tolerant strain in Pyropia haitanensis (Bangiales, Rhodophyta)[J]. Oceanologia et Limnologia Sinica, 2014, 45(3): 504-512. |
| [22] |
福建省水产局. 坛紫菜人工养殖[M]. 福州: 福建人民出版社, 1979: 1-101. Fujian Fisheries Bureau. Artificial cultivation of Pyropia haitanensis[M]. Fuzhou: Fujian People's Publishing House, 1979: 1-101. |
| [23] |
SATOH K, YAMANE Y, EMI T, et al. Effects of high temperatures on photosynthetic systems: Fluorescence Fo increases in Cyanobacteria[M]//Photosynthesis: Mechanisms and Effects. Budapest, Hungary: Springer, 1998: 2469-2472.
|
| [24] |
何培民, 张泽宇, 张学成, 等. 海藻栽培学[M]. 北京: 科学出版社, 2018. HE P M, ZHANG Z Y, ZHANG X C, et al. Seaweed cultivation[M]. Beijing: Science Press, 2018. |
| [25] |
DING H C, FEI Q J, ZHANG P, et al. Isolation and characterization of a heat-resistant strain with high yield of Pyropia haitanensis induced by ultraviolet ray[J]. Aquaculture, 2020, 521: 735050. |
| [26] |
WAHID A, GELANI S, ASHRAF M, et al. Heat tolerance in plants: an overview[J]. Environmental and Experimental Botany, 2007, 61(3): 199-223. |
| [27] |
MORANT-MANCEAU A, PRADIER E, TREMBLIN G. Osmotic adjustment, gas exchanges and chlorophyll fluorescence of a hexaploid triticale and its parental species under salt stress[J]. Journal of Plant Physiology, 2004, 161(1): 25-33. |
| [28] |
黄林彬, 黄文, 严兴洪. 条斑紫菜两个耐高温品系的耐低盐特性[J]. 水产学报, 2020, 44(2): 222-233. HUANG L B, HUANG W, YAN X H. Low-salinity tolerance of two high-temperature resistant strains in Pyropia yezoensis[J]. Journal of Fisheries of China, 2020, 44(2): 222-233. |
| [29] |
陈伟洲, 许俊宾, 吴文婷, 等. 三种紫菜叶状体对高温胁迫的生理响应[J]. 热带海洋学报, 2015, 34(1): 49-55. CHEN W Z, XU J B, WU W T, et al. Physiological responses of three species of Pyropia thallus to high temperature stress[J]. Journal of Tropical Oceanography, 2015, 34(1): 49-55. |
| [30] |
张聪, 严兴洪. Pyropia churharii新品系的分离与特性分析[J]. 水产学报, 2014, 38(9): 1457-1465. ZHANG C, YAN X H. Isolation and characterization of new strains of Pyropia churharii (Bangiales, Rhodophyta)[J]. Journal of Fisheries of China, 2014, 38(9): 1457-1465. |
| [31] |
YAN X H, FUJITA Y, ARUGA Y. High monospore-producing mutants obtained by treatment with MNNG in Porphyra yezoensis Ueda (Bangiales, Rhodophyta)[J]. Hydrobiologia, 2004, 512(1): 133-140. |
2. Shanghai Engineering Research Center of Aquaculture, Shanghai Ocean University, Shanghai 201306, China
 2023,
Vol. 32
2023,
Vol. 32


