2. 上海海洋大学 农业农村部淡水水产种质资源重点实验室, 上海 201306;
3. 上海海洋大学 上海水产养殖工程技术研究中心, 上海 201306;
4. 上海海洋大学 水产科学国家级实验教学示范中心, 上海 201306
中华绒螯蟹(Eriocheir sinensis,以下简称河蟹)是一种洄游性的甲壳类动物,广泛分布于我国东部沿海地区[1],是重要的经济蟹类。众所周知,自然条件下野生河蟹的生活史较为复杂,表现为:河蟹生殖蜕壳之前的大部分时间都在淡水河流和湖泊中生活,而生殖蜕壳后的河蟹则成群结队地向下游河口区或浅海处进行洄游[2-4],在降海洄游过程中河蟹性腺逐步发育,最终在半咸水中发育成熟并进行交配、产卵和孵化[5-6],随后幼体孵化后在河口半咸水中发育为大眼幼体后开始溯河洄游到淡水中进行生长发育[6]。研究表明,长江水系野生河蟹种群具有优异的养殖性能和经济价值[7-8],但迄今为止,对其成蟹降海洄游所知甚少,故研究其洄游规律具有一定的理论意义和现实意义。
脂类是重要的营养成分之一,对于河蟹的生长发育十分重要[9-10]。肝胰腺是甲壳动物最主要的营养物质消化吸收和储存器官,其营养状况可以间接反映甲壳动物饵料的营养价值[11-12]。脂类是肝胰腺中储存的主要能源物质和性腺发育的必需营养物质,对河蟹降海洄游和性腺发育起着重要作用[13-14]。此外,河蟹作为广盐性蟹类中高渗调节的代表种类[15],盐度是河蟹洄游过程中的主要环境因子,对甲壳动物脂质代谢存在一定的影响[16-17],且甲壳动物细胞膜上的脂肪酸组成对其渗透压调节起着重要作用[18-21]。先前的研究表明,长江野生河蟹每年11月洄游到江苏镇江江段性腺发育已经基本成熟,翌年3—4月在长江口崇明岛东侧的产卵场中发现的河蟹雌体已经报卵[22],雄体已经有过交配行为[5-6]。因此,在野生河蟹降海洄游过程中,采集性腺发育基本成熟(没有交配和产卵)的河蟹群体和产卵场中已经交配和抱卵的群体,比较这两种群体的肝胰腺指数、性腺指数、总脂含量和脂肪酸组成,对于理解河蟹的降海洄游、营养状况、渗透压调节与脂类营养的关系具有一定指导价值。迄今为止,尚未见长江野生河蟹洄游过程中肝胰腺指数、性腺指数及其脂类营养组成的报道。鉴于此,本文首先比较了长江水系野生淡水与半咸水成蟹的肝胰腺指数和性腺指数,并进一步测定和比较了肝胰腺和肌肉的总脂含量和脂肪酸组成,旨在为进一步理解长江野生河蟹的降海洄游规律和生态生理适应机制提供基础资料。
1 材料与方法 1.1 实验用蟹和解剖2015年11月在江苏镇江附近的长江干流(32.11°N, 119.27°E)捕捞降海洄游的淡水中的野生河蟹,长江河口区的半咸水中的河蟹(雌蟹为抱卵蟹)于2016年4月初捕捞自崇明岛东侧的河蟹产卵场(31.62°N, 121.40°E),所有实验用蟹均由当地渔民采用蟹笼或小型底拖网捕捞而得,淡水的盐度为0,河口区半咸水的盐度为15。随机挑选体质量接近的淡水河蟹与半咸水河蟹雌雄各10只用于后续实验,淡水河蟹雌雄个体的平均规格分别为(109.02 ± 1.45)g和(135.06 ± 2.43)g,半咸水河蟹雌雄个体的平均规格分别为(112.49±2.83)g和(133.55±3.11)g。所有实验用蟹活体运输至上海海洋大学甲壳动物营养繁殖实验室用于后续实验。用干毛巾擦干河蟹体表面水分后,用电子天平(JY1002,上海浦春计量仪器有限公司,精确度=0.01 g)测量体质量,并采用游标卡尺(M150,上海精美量具厂,准确度=0.02 mm)测量其甲壳长和甲壳宽。活体解剖所有个体,取出全部肝胰腺和性腺并准确称量,用于计算肝胰腺指数(hepatosomatic index, HSI)和性腺指数(gonadosomatic index, GSI)。河蟹躯体经冷冻处理后,精刮出大部分肌肉,随后将肝胰腺、性腺和肌肉样品保存于-40 ℃冰箱,用于后续的营养成分测定。HSI和GSI的计算公式如下:
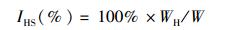 (1)
(1)
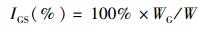 (2)
(2)
式中:IHS为肝胰腺指数;IGS为性腺指数;WH为肝胰腺质量,g;WG为性腺质量,g;W为体质量,g。
1.2 总脂和脂肪酸测定参考FOLCH等[23]的方法,采用V(氯仿):V(甲醇)=2:1溶液提取样品中总脂并测定其含量。根据MORRISON等[24]的方法,采用14%的三氟化硼-甲醇溶液对以上总脂样品进行甲脂化处理,然后采用Agilent 7890B-5977A气相色谱-质谱联用仪(GC-MS)进行脂肪酸分析。色谱柱为Omegawax 320毛细管柱(30 m×0.25 mm×0.25 μm; Supelco, Billefonte, PA, USA),进样口温度为240 ℃,辅助加热器温度为245 ℃。升温程序:由40 ℃以10 ℃/min速率升至170 ℃,再以2 ℃/min速率升至220 ℃,保留1 min,最后以2 ℃/min速率升至230 ℃,保留5 min。根据脂肪酸混标出峰时间对样品的脂肪酸进行定性,然后采用峰面积归一化法对脂肪酸进行相对定量。
1.3 数据分析采用SPSS 20.0软件对数据进行统计分析,数据以平均值±标准误(Mean±SE)表示。采用Levene法进行方差齐性检验,当数据不满足齐性方差时对百分比数据进行反正弦或者平方根处理,用独立样本t检验(independent samples t-test)进行显著性分析,P < 0.05为差异显著,P < 0.01为差异极显著。
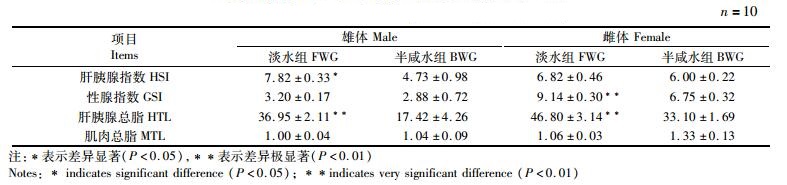
2 结果 2.1 野生长江淡水与半咸水河蟹组织系数和总脂含量的比较野生长江淡水与半咸水河蟹的组织系数和总脂含量见表 1。对雄体而言,淡水组河蟹的肝胰腺指数和肝胰腺总脂含量均显著高于半咸水组(P < 0.05),但两组间雄蟹的性腺指数和肌肉总脂含量均无显著性差异(P> 0.05)。对雌体而言,性腺指数和肝胰腺总脂含量均在淡水组较高(P < 0.05),而两者间的肝胰腺指数和肌肉总脂含量无显著性差异(P> 0.05)。
|
表 1 野生长江淡水与半咸水河蟹组织系数和总脂含量的比较(%湿质量) Tab.1 Tissue indices and total lipid content of wild E. sinensis between freshwater and brackish water in the Yangtze River(% wet mass) |
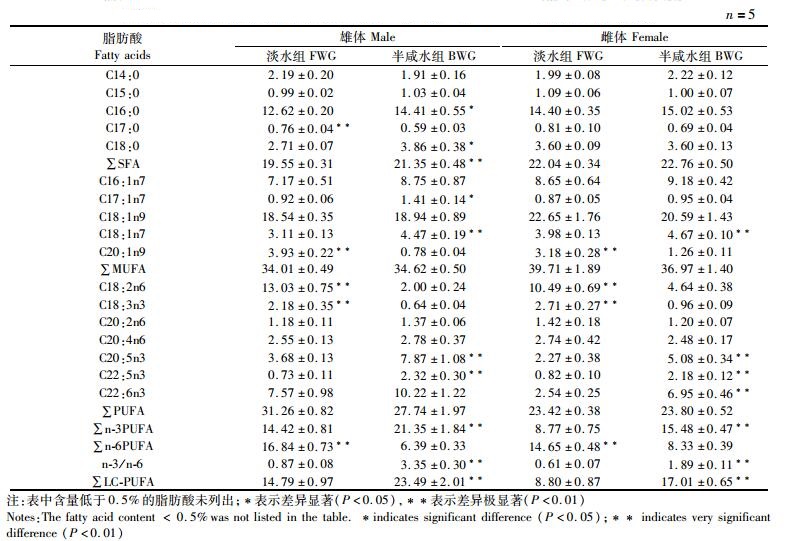
野生长江淡水与半咸水河蟹肝胰腺的脂肪酸含量见表 2。半咸水组雄蟹肝胰腺中的C16:0、C18:0和总饱和脂肪酸(total saturated fatty acids, ∑SFA)含量显著高于淡水组(P < 0.05);半咸水组雄蟹肝胰腺中的C17:1n7和C18:1n7含量较高(P < 0.05),但两组间总单不饱和脂肪酸(total monounsaturated fatty acids, ∑MUFA)含量无显著性差异(P>0.05);淡水组肝胰腺中的C18:2n6、C18:3n3和总n-6多不饱和脂肪酸(total n-6 polyunsaturated fatty acids, ∑n-6 PUFA)含量显著高于半咸水组(P < 0.05),而C20:5n3、C22:5n3、总n-3多不饱和脂肪酸(total n-3 polyunsaturated fatty acids, ∑n-3 PUFA)和长链多不饱和脂肪酸(total long chain polyunsaturated fatty acids, ∑LC-PUFA)含量及∑n-3 PUFA/∑n-6PUFA比值均在半咸水组较高(P < 0.05)。对雌蟹而言,淡水组和半咸水组肝胰腺中的∑SFA含量无显著性差异(P>0.05);淡水组肝胰腺中的C20:1n9含量显著高于半咸水组,而半咸水组中C18:1n7的含量较高(P < 0.05),但是两组间的∑MUFA含量无显著性差异(P>0.05);C18:2n6、C18:3n3和∑n-6 PUFA含量均在淡水组较高(P < 0.05),半咸水组肝胰腺中的C20:5n3、C22:5n3、C22:6n3、∑n-3 PUFA和∑LC-PUFA含量及∑n-3 PUFA/∑n-6 PUFA的值显著高于淡水组(P < 0.05)。
|
表 2 野生长江淡水与半咸水河蟹肝胰腺脂肪酸含量的比较(%总脂肪酸) Tab.2 Comparison of the fatty acid composition in the hepatopancreas of wild E. sinensis between freshwater and brackish water in the Yangtze River (% total fatty acids) |
对雄蟹肌肉中的脂肪酸组成而言,淡水组肌肉中的C17:0、C18:0和∑SFA含量显著高于半咸水组(P < 0.05),但两组间的∑MUFA含量无显著性差异(P>0.05);淡水组肌肉中的C18:2n6、C18:3n3、C20:2n6和∑n-6 PUFA含量显著高于半咸水组(P < 0.05),而C20:5n3、C22:5n3、∑n-3 PUFA和∑LC-PUFA含量及∑n-3 PUFA/∑n-6 PUFA的值均在半咸水组较高(P < 0.05)。在雌蟹中,淡水组肌肉中的C17:0、C18:0和∑SFA含量显著高于半咸水组(P < 0.05);半咸水组肌肉中的C16:1n7、C18:1n9、C18:1n7和∑MUFA的含量显著高于淡水组(P < 0.05);淡水组雌蟹肌肉中的C18:3n3、C20:2n6、C20:4n6和∑n-6 PUFA含量较高(P < 0.05),而C20:5n3、∑n-3PUFA和∑LC-PUFA含量及∑n-3 PUFA/∑n-6 PUFA值均在半咸水组较高(P < 0.05,表 3)。
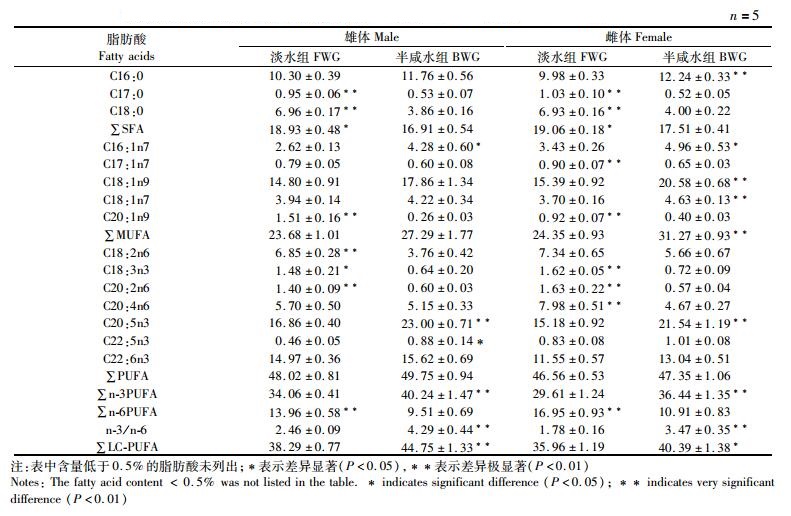
|
表 3 野生长江淡水与半咸水河蟹肌肉脂肪酸含量的比较(%总脂肪酸) Tab.3 Comparison of fatty acid composition in the muscle of wild E. sinensis between freshwater and brackish water in the Yangtze River (% total fatty acids) |
河蟹是一种广盐性的甲壳动物,具有很强的渗透压调节能力[25-26],生殖蜕壳后通常向河口区或浅海进行降海洄游,在河口区完成交配、产卵、孵化等[27],盐度可以促进河蟹的性腺发育和交配[28-29]。本研究结果表明淡水组雄体的肝胰腺指数及其总脂含量均高于半咸水组,这可能是因为雄体洄游过程中肝胰腺中大量脂肪被消耗,故肝胰腺指数下降较多。淡水组和半咸水组雌体的肝胰腺指数并没有显著差异,推测是因为本次采样的镇江江段的河蟹雌体卵巢发育已经接近成熟(卵巢指数为9.14%),雌体在长江中降海洄游和卵巢发育过程中已经消耗了肝胰腺中的大量脂类来提供能量,或者将肝胰腺中的脂类转运到卵巢中满足卵巢对脂类的需要[9, 14],从而导致肝胰腺指数已经下降到6%左右。河蟹肝胰腺中的脂类以甘油三酯为主,甘油三酯是甲壳动物的主要能源物质[9],因此,在河蟹雌体降河洄游过程中肝胰腺中的总脂含量有所下降,可能是肝胰腺中的甘油三酯被用于氧化供能所导致的。
咸水中河蟹雄体的性腺指数低于淡水组,这可能是因为雄体在咸水中交配后,雄体交配过程中将精荚输送到雌体的纳精囊内[6],导致性腺指数下降。半咸水组雌体性腺指数低于淡水组,是因为雌体已经完成第一次排卵,第一次排卵后的卵巢指数通常在1%~2%[30],然而雌体在抱卵过程中,卵巢开始第二次发育,故本研究中半咸水组的卵巢指数可达6.75%[31]。无论雌体还是雄体,各组河蟹肌肉中总脂含量均无显著差异,这是因为肌肉中的常规生化组成通常比较稳定,才能维持肌肉正常的生理功能[32]。
3.2 河蟹洄游过程中脂肪酸含量的变化先前研究表明,水体盐度的变化会影响甲壳动物组织中的脂肪酸组成,如锯缘青蟹(Scylla serrata)肌肉和肝胰腺中的长链多不饱和脂肪酸(LC-PUFA)和总n-3多不饱和脂肪酸的(∑n-3 PUFA)含量随水体盐度的升高呈下降趋势。而较高盐度亦可显著降低河蟹肝胰腺中EPA(C20:5n3)和DHA(C22:6n3)的含量[33]。研究表明DHA和EPA不仅是机体维持细胞结构和功能的重要脂肪酸,且是河蟹性腺发育中卵巢中储存的主要多不饱和脂肪酸,在其产卵、受精和孵化等过程中起着重要作用[34-35]。本研究中半咸水组河蟹肝胰腺中的EPA、DHA和∑n-3 PUFA的含量均显著高于淡水组,其可能原因有两点:(1)河蟹洄游过程中,肝胰腺中的饱和及单不饱和脂肪酸被作为能源物质而利用,因此肝胰腺中的EPA、DHA和∑n-3 PUFA相对总脂肪酸而言,其相对百分比例呈现增加趋势;(2)水体盐度对河蟹肝胰腺中的脂质代谢存在一定的影响,随着水体盐度提高,肝胰腺中的EPA、DHA和∑n-3 PUFA被相对保留下来。此外,本研究中淡水组河蟹肌肉的EPA和DHA均显著低于半咸水组,而C18:2n6含量相对较高,这可能是肌肉中的脂肪酸PUFA组成存在一定的补偿效应,组织中DHA和EPA百分含量上升,可能会导致∑n-6PUFA百分含量下降。先前研究表明,提高了组织中的n-3 LC-PUFA百分含量有利于提高甲壳动物的渗透压调节能力[19, 29],这可能是半咸水组肝胰腺和肌肉中n-3 LC-PUFA百分含量较高的重要原因之一。
本研究比较野生河蟹降海洄游过程中淡水组和半咸水组河蟹的性腺指数、肝胰腺指数和脂类组成,可以为进一步研究长江野生河蟹的降海洄游规律和生态生理适应机制提供参考资料,但是尚不清楚造成这些变化的具体原因,下一步研究需要室内单因素和多因素调控实验,进一步探讨降海洄游、水体盐度和交配产卵等因素对河蟹脂质代谢的影响,从而解析河蟹降海洄游过程中脂类组成变化的具体原因,探讨河蟹降海洄游和脂质代谢的关系。
| [1] |
SUI L Y, ZHANG F M, WANG X M, et al. Genetic diversity and population structure of the Chinese mitten crab Eriocheir sinensis in its native range[J]. Marine Biology, 2009, 156(8): 1573-1583. |
| [2] |
CHENG Y X, WU X G, YANG X Z, et al. Current trends in hatchery techniques and stock enhancement for Chinese mitten crab, Eriocheir japonica sinensis[J]. Reviews in Fisheries Science, 2008, 16(1/3): 377-384. |
| [3] |
DITTEL A I, EPIFANIO C E. Invasion biology of the Chinese mitten crab Eriochier sinensis:a brief review[J]. Journal of Experimental Marine Biology and Ecology, 2009, 374(2): 79-92. |
| [4] |
HERBORG L M, BENTLEY M G, CLARE A S, et al. Mating behaviour and chemical communication in the invasive Chinese mitten crab Eriocheir sinensis[J]. Journal of Experimental Marine Biology and Ecology, 2006, 329(1): 1-10. |
| [5] |
张列士. 河蟹生活史的研究及蟹苗的捕捞[J]. 水产科技情报, 1973, 1(2): 5-21. ZHANG L S. Study on the life history of Chinese mitten crab and the fishing of juveniles[J]. Fisheries Science and Technology Information, 1973, 1(2): 5-21. |
| [6] |
张列士, 李军. 河蟹增养殖技术[M]. 北京: 金盾出版社, 2002: 5-248. ZHANG L S, LI J. Hatchery technology of Erincheir sinensis[M]. Beijing: Jindun Press, 2002: 5-248. |
| [7] |
WU X G, ZHAO H L, JIANG X D, et al. Comparison of culture performance and gonadal development of wild-caught Chinese mitten crab Eriocheir sinensis juveniles from three major river populations[J]. Fisheries Science, 2018, 84(6): 924-937. |
| [8] |
何杰, 吴旭干, 姜晓东, 等. 野生和人工繁育大眼幼体在成蟹阶段的养殖性能比较[J]. 上海海洋大学学报, 2015, 24(1): 60-67. HE J, WU X G, JIANG X D, et al. Comparison of the culture performance of wild-caught and artifical breeding Chinese mitten crab megalopae reared in the grow-out ponds during the adult Eriocheir sinensis culture stage[J]. Journal of Shanghai Ocean University, 2015, 24(1): 60-67. |
| [9] |
成永旭, 堵南山, 赖伟. 不同阶段中华绒螯蟹肝胰腺的脂类及脂肪酸组成[J]. 动物学报, 1998, 44(4): 420-429. CHENG Y X, DU N S, LAI W. Lipid composition in hepatopancreas of Chinese mitten crab Eriocheir sinensis at different stages[J]. Acta Zoologica Sinica, 1998, 44(4): 420-429. |
| [10] |
WU X G, WANG Z K, CHENG Y X, et al. Effects of dietary phospholipids and highly unsaturated fatty acids on the precocity, survival, growth and hepatic lipid composition of juvenile Chinese mitten crab, Eriocheir sinensis(H. Milne-Edwards)[J]. Aquaculture Research, 2011, 42(3): 457-468. |
| [11] |
VOGT G, STORCH V, QUINITIO E T, et al. Pascual FP. Midgut gland as monitor organ for the nutritional value of diets in Penaeus monodon(Decapoda)[J]. Aquaculture, 1985, 48(1): 1-12. |
| [12] |
姚桂桂, 吴旭干, 成永旭, 等. 东海三疣梭子蟹雌体不同生理阶段肝胰腺的生化组成与其组织学结构的关系[J]. 海洋学报, 2008, 30(6): 122-131. YAO G G, WU X G, CHENG Y X, et al. The changes of histology and main biochemical compositionin the hepatopancreas at the different physiological stages of Portunus trituberculatus in East China Sea[J]. Acta Oceanologica Sinica, 2008, 30(6): 122-131. |
| [13] |
滕炜鸣, 吴旭干, 成永旭, 等. 莱茵种群和长江种群子一代中华绒螯蟹性腺发育及相关生物学指数变化的比较[J]. 上海水产大学学报, 2008, 17(1): 65-71. TENG W M, WU X G, CHENG Y X, et al. A comparative study on some biological index changes concerned with gonad development between two population of the Chinese mitten crab (Eriocheir sinensis):Rhine andYangtze[J]. Journal of Shanghai Fisheries University, 2008, 17(1): 65-71. |
| [14] |
WEN X B, CHEN L Q, AI C X, et al. Variation in lipid composition of Chinese mitten-handed crab, Eriocheir sinensis during ovarian maturation[J]. Comparative Biochemistry and Physiology Part B:Biochemistry and Molecular Biology, 2001, 130(1): 95-104. |
| [15] |
冯广朋, 卢俊, 庄平, 等. 盐度对中华绒螯蟹雌性亲蟹渗透压调节和酶活性的影响[J]. 海洋渔业, 2013, 35(4): 468-473. FENG G P, LU J, ZHUANG P, et al. Effects of salinity on osmo-ionic regulation and enzyme activities in mature female Eriocheir sinensis[J]. Marine Fisheries, 2013, 35(4): 468-473. |
| [16] |
LONG X W, WU X G, ZHAO L, et al. Effects of salinity on gonadal development, osmoregulation and metabolism of adult male Chinese mitten crab, Eriocheir sinensis[J]. PLoS One, 2017, 12(6): e0179036. |
| [17] |
CHEN K, LI E C, GAN L, et al. Growth and lipid metabolism of the Pacific White Shrimp Litopenaeus vannamei at different salinities[J]. Journal of Shellfish Research, 2014, 33(3): 825-832. |
| [18] |
SUI L Y, WILLE M, CHENG Y X, et al. The effect of dietary n-3 HUFA levels and DHA/EPA ratios on growth, survival and osmotic stress tolerance of Chinese mitten crab Eriocheir sinensis larvae[J]. Aquaculture, 2007, 273(1): 139-150. |
| [19] |
ROMANO N, WU X G, ZENG C S, et al. Growth, osmoregulatory responses and changes to the lipid and fatty acid composition of organs from the mud crab, Scylla serrata, over a broad salinity range[J]. Marine Biology Research, 2014, 10(5): 460-471. |
| [20] |
赵磊, 龙晓文, 吴旭干, 等. 水体盐度对中华绒螯蟹成体雄蟹渗透压调节和生理代谢的影响[J]. 水生生物学报, 2016, 40(1): 27-34. ZHAO L, LONG X W, WU X G, et al. Effects of water salinity on osmoregulation and physiological metabolism of adult male Chinese mitten crab Eriocheir sinensis[J]. Acta Hydrobiologica Sinica, 2016, 40(1): 27-34. |
| [21] |
ZHANG D Z, QI T T, LIU J, et al. Adaptively differential expression analysis in gill of Chinese mitten crabs (Eriocheir japonica sinensis) associated with salinity changes[J]. International Journal of Biological Macromolecules, 2018, 120: 2242-2246. |
| [22] |
GENG Z, FENG G P, ZHAO F, et al. Distribution of larval crabs (Eriocheir sinensis) and relationship with oceanographic features in the Yangtze Estuary[J]. Ecoscience, 2018, 25(2): 125-134. |
| [23] |
FOLCH J, LEES M, SLOANE STANLEY G H. A simple method for the isolation and purification of total lipides from animal tissues[J]. Journal of Biological Chemistry, 1957, 226(1): 497-509. |
| [24] |
MORRISON W R, SMITH L M. Preparation of fatty acid methyl esters and dimethylacetals from lipids with boron fluoride-methanol[J]. Journal of Lipid Research, 1964, 5: 600-608. |
| [25] |
庄平, 贾小燕, 冯广朋, 等. 不同盐度条件下中华绒螯蟹亲蟹行为及血淋巴生理变化[J]. 生态学杂志, 2012, 31(8): 1997-2003. ZHUANG P, JIA X Y, FENG G P, et al. Variations of behavior and haemolymph physiology of female parent Chinese crab (Eriocheir sinensis) under different water salinities[J]. Chinese Journal of Ecology, 2012, 31(8): 1997-2003. |
| [26] |
WANG Z F, BAI Y Z, ZHANG D Z, et al. Adaptive evolution of osmoregulatory-related genes provides insight into salinity adaptation in Chinese mitten crab, Eriocheir sinensis[J]. Genetica, 2018, 146(3): 303-311. |
| [27] |
堵南山. 中华绒螯蟹的洄游[J]. 水产科技情报, 2004, 31(2): 56-57. DU N S. Migration of Chinese mitten-handed crab Eriocheir sinensis[J]. Fisheries Science and Technology Information, 2004, 31(2): 56-57. |
| [28] |
吴旭干, 赵亚婷, 何杰, 等. 低盐度海水和淡水对中华绒螯蟹性腺发育及交配行为的影响[J]. 动物学杂志, 2013, 48(4): 555-561. WU X G, ZHAO Y T, HE J, et al. Effects of brackish water and fresh water on gonadal development and mating behavior in adult Chinese mitten crab[J]. Chinese Journal of Zoology, 2013, 48(4): 555-561. |
| [29] |
LONG X W, WU X G, ZHAO L, et al. Physiological responses and ovarian development of female Chinese mitten crab Eriocheir sinensis subjected to different salinity conditions[J]. Frontiers in Physiology, 2018, 8: 1072. |
| [30] |
LIU Z J, WU X G, CHENG Y X, et al. Ovarian re-maturation following the first spawning in the Chinese mitten crab, Eriocheir sinensis(H. Milne-Edwards)[J]. Aquaculture Research, 2011, 42(3): 417-426. |
| [31] |
于智勇, 吴旭干, 常国亮, 等. 中华绒螯蟹第二次卵巢发育期间卵巢和肝胰腺中主要生化成分的变化[J]. 水生生物学报, 2007, 31(6): 799-806. YU Z Y, WU X G, CHANG G L, et al. Changes in the main biochemical composition in ovaries and Hepatopancreas of Chinese mitten crab, Eriocheir sinensis(H. Milne-Edwards) during the second ovarian development[J]. Changes in the main biochemical composition in ovaries and Hepatopancreas of Chinese mitten crab, Eriocheir sinensis(H. Milne-Edwards) during the second ovarian development, 2007, 31(6): 799-806. |
| [32] |
WU X G, CHANG G L, CHENG Y X, et al. Effects of dietary phospholipid and highly unsaturated fatty acid on the gonadal development, tissue proximate composition, lipid class and fatty acid composition of precocious Chinese mitten crab, Eriocheir sinensis[J]. Aquaculture Nutrition, 2010, 16(1): 25-36. |
| [33] |
王帅, 吴旭干, 陶宁萍, 等. 盐度调控对中华绒螯蟹肝胰腺主要营养品质的影响[J]. 现代食品科技, 2015, 31(12): 318-324. WANG S, WU X G, TAO N P, et al. Effects of water salinity on the nutritional quality of Eriocheir sinensis hepatopancreas[J]. Modern Food Science and Technology, 2015, 31(12): 318-324. |
| [34] |
WU X G, CHENG Y X, SUI L Y, et al. Biochemical composition of pond-reared and lake-stocked Chinese mitten crab Eriocheir sinensis(H. Milne-Edwards)[J]. Aquaculture Research, 2007, 38(14): 1459-1467. |
| [35] |
SUI L Y, SUN H X, WU X G, et al. Effect of dietary HUFA on tissue fatty acid composition and reproductive performance of Chinese mitten crab Eriocheir sinensis(H. Milne-Edwards) broodstock[J]. Aquaculture International, 2011, 19(2): 269-282. |
2. Key Laboratory of Freshwater Aquatic Genetic Resources, Ministry of Agriculture and Rural Affairs, Shanghai Ocean University, Shanghai 201306, China;
3. Shanghai Engineering Research Center of Aquaculture, Shanghai Ocean University, Shanghai 201306, China;
4. National Demonstration Centre for Experimental Fisheries Science Education, Shanghai Ocean University, Shanghai 201306, China
 2020,
Vol. 29
2020,
Vol. 29


