2. 上海海洋大学 水产科学国家级实验教学示范中心, 上海 201306;
3. 上海海洋大学 海洋生物科学国际联合研究中心, 中国科学技术部, 上海 201306;
4. 上海海洋大学 水产种质资源发掘与利用教育部重点实验室, 上海 201306
侧线系统是鱼类和两栖类体表一种重要的感觉系统,有感觉水流、水压、水温、微弱电场变化的功能[1-3],对鱼类的摄食、避敌、生殖、集群、洄游等行为有着非常重要的作用[4-7]。斑马鱼的侧线系统由一系列分散的神经丘构成,包括前侧线系统(ALL)和后侧线系统(PLL)[8]。侧线系统起源于头部神经基板中的侧线基板[9]。Six1/2,Six4/5和Eya1基因可作为头部神经基板和侧线神经丘前体细胞的标记基因[9]。在小鼠内耳毛细胞分化中Eya1-Six1通过激活Atoh1a表达促进毛细胞的分化[10],Eya1和Six1/2在斑马鱼内耳和神经丘发育的过程中也是必需的[11-12]。近期的研究表明:侧线系统的发育受到Wnt等信号的调控[13],但具体的调控机制尚未完全阐明。
Wnt信号通路是一条非常保守的信号转导途径,广泛存在于所有动物中,对胚胎的早期发育、组织器官发生、组织再生以及其他生理过程起着非常重要的作用[14]。Wnt信号也参与后侧线原基的迁移和次级神经丘形成[15-16]。Wnt8是经典Wnt信号途径的一类配体,可通过β-catenin调控下游基因转录。Wnt8参与胚胎背-腹轴的建立,促进侧中胚层的发育[17]。Dkk(Dickkopf)是Wnt信号通路中重要的拮抗因子之一,能够特异地抑制经典Wnt信号通路[18]。本研究将利用转基因斑马鱼操控经典Wnt信号,研究Wnt信号对侧线基板和神经丘前体细胞的影响。
1 材料与方法 1.1 实验材料本实验所用AB品系斑马鱼,hsp:wnt8a-egfp和hsp:dkk1-egfp转基因斑马鱼[19]均养在循环养殖系统中(上海海圣生物设备有限公司)。养殖系统水温维持在28.5 ℃,按14 h/10 h的光暗周期,每天早晚各喂食一次卤虫。胚胎饲养于灭菌后的胚胎水(63 mg/L CaSO4、10 mg/L MgSO4、4 mg/L KCl、1.1 mg/L NaH2PO4、0.01 mg/L亚甲基蓝)中,胚胎发育分期参照KIMMEL等[20]。YO-PRO-1购自Life Technology,NBT和BCIP购自Roche。
1.2 热激处理胚胎AB品系斑马鱼分别与hsp:wnt8a-egfp和hsp:dkk1-egfp转基因斑马鱼杂交。将所得的部分胚胎在5 hpf热激1 h,发现胚胎分别表现出头部化和尾部化的缺陷表型,这表明hsp:wnt8a和hsp:dkk1两个转基因鱼热激处理后确实过表达了Wt8a和Dkk1蛋白。当胚胎发育至17 hpf(hour-post fertilization)时,将胚胎等分为实验组和对照组,各54条。实验组胚胎39 ℃热激1 h,然后放回28.5 ℃培养箱继续培养;对照组胚胎持续培养在28.5 ℃温箱中。
1.3 YO-PRO-1染色用新鲜的胚胎水稀释配制2 μmol/L YO-PRO-1染色工作液,将发育至3 dpf的胚胎放入染色液中,避光染色1 h。染色后的胚胎,先用胚胎水清洗两遍,然后转移至MS-222(0.1 mg/mL)中麻醉5 min。将其转移到平底的96孔板中,每个孔中放入一个胚胎,仅留少量胚胎水,并使胚胎侧向铺于板底以便观察。在荧光倒置显微镜(Axio Observer Z1)下进行神经丘和毛细胞计数并拍照。
1.4 碱性磷酸酶染色胚胎经4% PFA(用PBS配制)室温固定1 h,然后用PBS洗涤3次,再用3% H2O2室温强光下使胚胎脱色完全。用PBS洗涤胚胎5 min,再用显色缓冲液洗涤胚胎3次,每次5 min。最后将胚胎浸没于含NBT和BCIP的显色液中,避光染色10~15 min。待胚胎显色完成后,用4% PFA终止显色反应,再用PBS洗涤胚胎2~3次。最后将胚胎浸于100%甘油中用于观察拍照。
1.5 整体原位杂交利用NCBI网站上Primer-BLAST设计Six2b和Eya1特异性引物(包含部分编码序列),见表 1。以斑马鱼30 hpf胚胎cDNA为模板,PCR扩增得到目的基因的片段,纯化并连接pBSK+载体,转化后送至生工生物工程(上海)有限公司测序鉴定,得到阳性克隆。以含有Six2b和Eya1特异性片段的质粒载体线性化后作为模板,使用T7 RNA聚合酶合成带地高辛标记的反义RNA探针。具体的原位杂交和显色过程参照文献[21]进行。
|
表 1 实验所使用的引物序列 Tab.1 Primer sequences used for all the experiments |
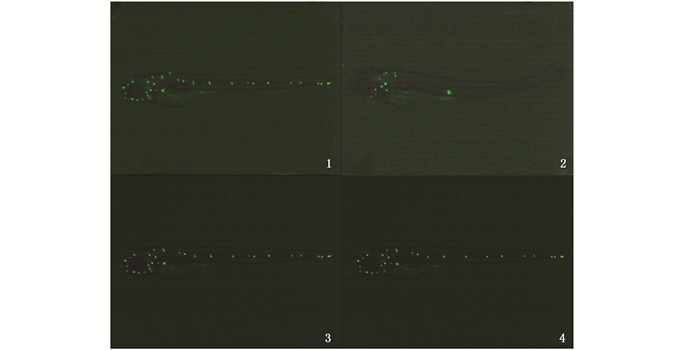
YO-PRO-1是一种细胞核染料,能特异性进入毛细胞。在17 hpf(侧线基板形成期)对转基因斑马鱼hsp:dkk1-egfp热激处理1 h,待胚胎发育至3 dpf时,用YO-PRO-1染色后,荧光显微镜下对斑马鱼后侧线系统神经丘的毛细胞进行计数,同时以AB野生型斑马鱼作为阴性对照。结果显示:未进行热激处理的hsp:dkk1-egfp转基因斑马鱼后侧线系统神经丘数目为8.75±0.68,神经丘毛细胞数为20.56 ± 2.39;热激处理后hsp:dkk1-egfp转基因斑马鱼中后侧线系统中未检测到毛细胞,而热激后的AB鱼与未热激的AB鱼毛细胞数量无显著变化,可见热激处理本身对毛细胞的损伤有限(图版Ⅰ、表 2)。这表明经过热激处理诱导Dkk1过表达,导致斑马鱼后侧线神经丘的毛细胞无法正常发育,即侧线基板形成期的Wnt信号对斑马鱼后侧线神经丘毛细胞的发育是必需的。
|
图版Ⅰ Dkk1过表达对神经丘毛细胞的影响 Plate1 The effect of Dkk1 overexpression on hair cell in neuromast 1.未经热激的hsp:dkk1-egfp转基因斑马鱼,神经丘毛细胞的YO-PRO-1染色;2.于17 hpf热激诱导Dkk1过表达后,神经丘毛细胞的YO-PRO-1染色; 3.未经热激的野生型斑马鱼AB,神经丘毛细胞YO-PRO-1染色;4.于17 hpf热激后,神经丘毛细胞YO-PRO-1染色 1. hsp: dkk1-egfp transgenic zebrafish without heating shock, the hair cells of neuromast were stained with YO-PRO-1; 2. The hair cells of neuromast of hsp: dkk1-egfp transgenic zebrafish heated at 17 hpf were stained by YO-PRO-1; 3. AB wild type zebrafish without heating shock, the hair cells of neuromast were stained with YO-PRO-1; 4. The hair cells of neuromast of AB wild type zebrafish heated at 17 hpf were stained by YO-PRO-1 |

|
表 2 后侧线系统神经丘毛细胞数目 Tab.2 Hair cells in neuromasts of the posterior lateral line |
碱性磷酸酶是多潜能干细胞的重要标记,也可以用于标记斑马鱼神经丘的前体细胞[22]。
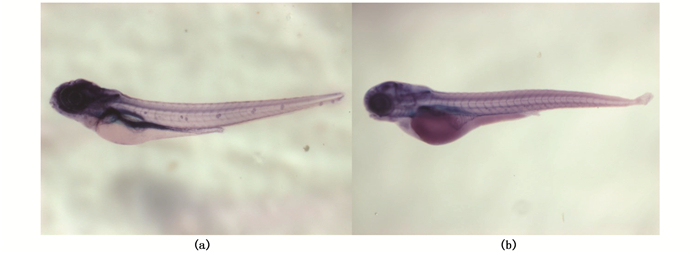
在胚胎发育至17 hpf时,热激处理诱导Dkk1过表达,待仔鱼发育至3 dpf时,进行碱性磷酸酶染色。结果显示:未经热激的hsp:dkk1-egfp转基因斑马鱼神经丘处出现碱性磷酸酶信号;Dkk1过表达后,斑马鱼侧线神经丘中的碱性磷酸酶信号减弱,且神经丘数量减少(图 1)。这表明:Wnt信号是斑马鱼侧线神经丘的前体细胞形成所必需的。
|
图 1 Dkk1过表达对神经丘前体细胞的影响
Fig. 1 The effect of Dkk1 overexpression on the precursor cells of neuromasts
(a)未经热激的hsp:dkk1-egfp转基因斑马鱼,神经丘的碱性磷酸酶染色;(b)于17 hpf热激诱导Dkk1过表达后,神经丘的碱性磷酸酶染色 (a)hsp: dkk1-egfp transgenic zebrafish without heating shock, the neuromast were stained with NBT/BCIP; (b)The hair cells of neuromast of hsp: dkk1-egfp transgenic zebrafish heated at 17 hpf were stained with NBT/BCIP |
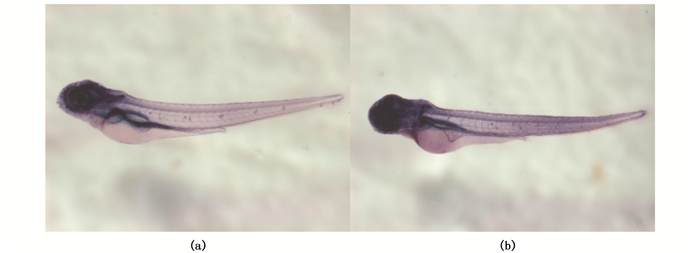
为检测Wnt信号对斑马鱼侧线神经丘前体细胞形成是否具有充分性作用,在胚胎发育至17 hpf时,热激处理诱导Wnt8a过表达,于3 dpf时取仔鱼利用碱性磷酸酶染色检测神经丘前体细胞情况。结果显示:未经热激的hsp:wnt8a-egfp转基因斑马鱼(对照组)神经丘处检测到较强的碱性磷酸酶信号;经热激的hsp:wnt8a-egfp转基因斑马鱼侧线神经丘中碱性磷酸酶信号相对于对照组显著减弱(图 2)。由此可见,过表达Wnt8a并不能促进斑马鱼神经丘前体细胞的形成,反而抑制前体细胞的形成。
|
图 2 Wnt8a过表达对神经丘前体细胞的影响
Fig. 2 The effects of Wnt8a overexpression on the precursor cells of neuromasts
(a)未经热激的hsp:wnt8a-egfp转基因斑马鱼,神经丘前体细胞的碱性磷酸酶染色;(b)热激诱导Wnt8a过表达后,神经丘前体细胞的碱性磷酸酶染色 (a)hsp: wnt8a-egfp transgenic zebrafish without heating shock, the neuromast were stained with NBT/BCIP; (b)The hair cells of neuromast of hsp: wnt8a-egfp transgenic zebrafish heated at 17 hpf were stained with NBT/BCIP |
既然Wnt信号参与斑马鱼侧线神经丘前体细胞的调控,Six2b和Eya1表达于神经基板和神经丘的前体细胞,参与毛细胞前体细胞的形成。因此,推测Wnt信号是否通过对Six2b和Eya1基因的表达调控参与侧线发育。通过整体原位杂交技术检测了在Wnt8a和Dkk1过表达后,斑马鱼胚胎中侧线神经丘前体细胞标记基因Six2b和Eya1在斑马鱼神经丘中表达量的变化。实验结果显示:未经热激的hsp:dkk1-egfp转基因斑马鱼胚胎中,Six2b和Eya1集中在28 hpf和36 hpf时神经丘和迁移的侧线原基中表达,48 hpf时Six2b和Eya1在神经丘中表达;热激诱导Dkk1的过表达后,在28 hpf、36 hpf、48hpf时未检测到Six2b和Eya1在迁移的侧线原基和神经丘处的表达(图版Ⅱ)。结果表明Dkk1过表达抑制Six2b和Eya1在斑马鱼迁移的侧线原基和神经丘处表达。
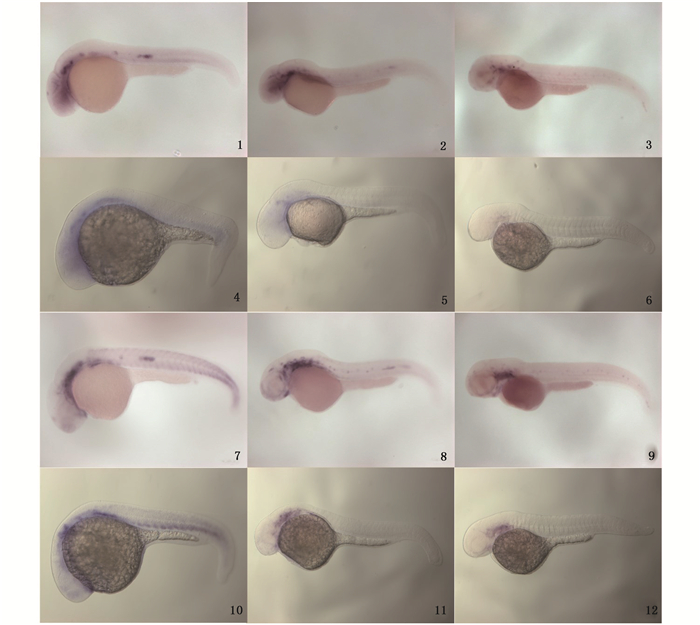
|
图版Ⅱ 整体原位杂交分析毛细胞相关基因Six2b和Eya1在hsp:dkk1-egfp转基因斑马鱼胚胎不同发育阶段的表达 Plate2 The expression pattern of hair cell related genes Six2b and Eya1 in different stages of transgenic zebrafish hsp:dkk1-egfp 1-3.未经热激的hsp:dkk1-egfp转基因斑马鱼,Six2b基因分别在28 hpf、36 hpf、48 hpf时的表达;4-6.于17 hpf热激诱导Dkk1过表达后,Six2b基因分别在28 hpf、36 hpf、48 hpf时的表达;7-9.未经热激的hsp:dkk1-egfp转基因斑马鱼,Eya1基因分别在28 hpf、36 hpf、48 hpf时的表达;10-12.于17 hpf热激诱导Dkk1过表达后,Eya1基因分别在28 hpf、36 hpf、48 hpf时的表达 1-3. hsp:dkk1-egfp transgenic zebrafish without heating shock, the expression pattern of gene Six2b at the stage of 28 hpf, 36 hpf and 48 hpf, respectively; 4-6. The expression pattern of gene Six2b at the stage of 28 hpf, 36 hpf and 48 hpf respectively in hsp:dkk1-egfp transgenic zebrafish heated at 17 hpf; 7-9. hsp:dkk1-egfp transgenic zebrafish without heating shock, the expression pattern of gene Eya1 at the stage of 28 hpf, 36 hpf and 48 hpf, respectively; 10-12. The expression pattern of gene Eya1 at the stage of 28 hpf, 36 hpf and 48 hpf respectively in hsp:dkk1-egfp transgenic zebrafish heated at 17 hpf |
对照组即未经热激的hsp:wnt8a-egfp转基因斑马鱼中,28 hpf和36 hpf时Six2b和Eya1在神经丘和迁移的侧线原基中表达,48 hpf时Six2b和Eya1在神经丘中表达;热激诱导Wnt8a的过表达后,在28 hpf、36 hpf、48 hpf时侧线原基和神经丘处未检测到Six2b和Eya1的表达(图版Ⅲ)。结果表明Wnt8a过表达抑制Six2b和Eya1在斑马鱼迁移的侧线原基和神经丘处表达。
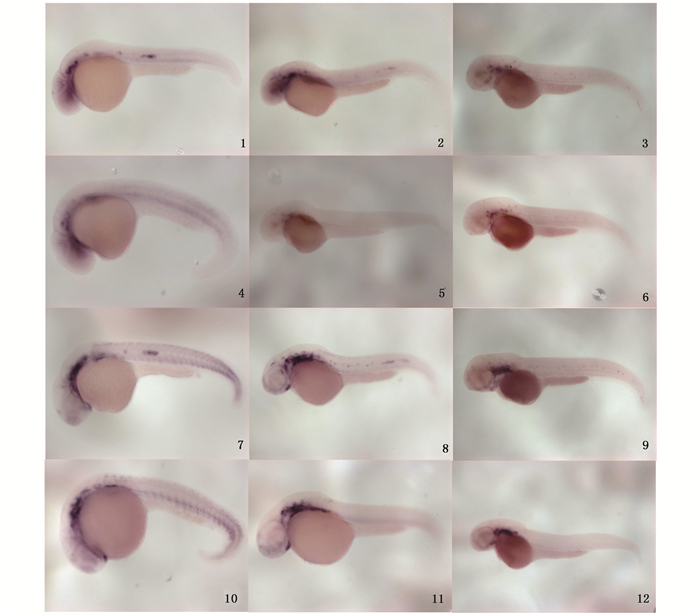
|
图版Ⅲ 整体原位杂交分析毛细胞相关基因Six2b和Eya1在hsp:wnt8a-egfp转基因斑马鱼胚胎不同发育阶段的表达 Plate3 The expression pattern of hair cell related genes Six2b and Eya1 in different stages of transgenic zebrafish hsp:wnt8a-egfp 1-3.未经热激的hsp:wnt8a-egfp转基因斑马鱼,Six2b基因分别在28 hpf、36 hpf、48 hpf时的表达;4-6.于17 hpf热激诱导Dkk1过表达后,Six2b基因分别在28 hpf、36 hpf、48 hpf时的表达;7-9.未经热激的hsp:wnt8a-egfp转基因斑马鱼,Eya1基因分别在28 hpf、36 hpf、48 hpf时的表达;10-12.于17 hpf热激诱导Dkk1过表达后,Eya1基因分别在28 hpf、36 hpf、48 hpf时的表达 1-3. hsp:wnt8a-egfp transgenic zebrafish without heating shock, the expression pattern of gene Six2b at the stage of 28 hpf, 36 hpf and 48 hpf, respectively; 4-6. The expression pattern of gene Six2b at the stage of 28 hpf, 36 hpf and 48 hpf respectively in hsp:wnt8a-egfp transgenic zebrafish heated at 17hpf; 7-9. hsp:wnt8a-egfp transgenic zebrafish without heating shock, the expression pattern of gene Eya1 at the stage of 28 hpf, 36 hpf and 48 hpf, respectively; 10-12. The expression pattern of gene Eya1 at the stage of 28 hpf, 36 hpf and 48 hpf respectively in hsp:wnt8a-egfp transgenic zebrafish heated at 17 hpf |
在侧线基板形成期抑制Wnt信号(Dkk1过表达)导致神经丘发育受阻。碱性磷酸酶染色,结合Six2b和Eya1的表达变化表明:Wnt信号可能通过抑制神经丘前体细胞中的Eya1和Six2b表达,使前体细胞无法正常发育,进而导致神经丘发育缺陷。此发现与斑马鱼Lef1突变体(经典Wnt信号的直接靶基因)中的表型类似(后侧线原基迁移受阻,缺少尾部神经丘),且Lef1突变体的侧线缺陷表型也是由后侧线原基中增殖细胞数量下降产生的[15, 23]。这表明Wnt信号确实是侧线神经丘前体细胞正常发育所必需。
低等脊椎动物的侧线系统与内耳和鳃上结构均发育起源于后部基板[9]。在两栖类中,发现Wnt信号可促进后部基板标记基因的表达[24],在斑马鱼中尽管Wnt信号是耳基板诱导所必需的,但是在鸡胚中异位表达Wnt不能充分诱导耳基板形成[25-26]。这和在斑马鱼侧线系统中观察的结果相似,在基板形成期异位表达Wnt8a,神经丘前体细胞,以及Eya1和Six2b基因的表达均受到抑制。推测在头部神经基板的诱导形成过程中,受到多个不同胚胎结构发出的动态信号,综合作用于基板区。而单纯异位表达Wnt信号,并不能特异性增强侧线基板中的Wnt信号,而是造成所有胚胎组织的Wnt信号均上调,因此造成侧线的发育受到抑制。
| [1] | BAKER C V H, BRONNER-FRASER M. Vertebrate cranial placodes I. Embryonic induction[J]. Developmental Biology, 2001, 232(1): 1–61. DOI:10.1006/dbio.2001.0156 |
| [2] | WILKENS L A, HOFMANN M H. The paddlefish rostrum as an electrosensory organ:A novel adaptation for plankton feeding[J]. Bio Science, 2007, 57(5): 399–407. |
| [3] | GOULET J, ENGELMANN J, CHAGNAUD B P, et al. Object localization through the lateral line system of fish:theory and experiment[J]. Journal of Comparative Physiology A, 2008, 194(1): 1–17. |
| [4] | MOGDANS J, GEISEN S. Responses of the goldfish head lateral line to moving objects[J]. Journal of Comparative Physiology A, 2009, 195(2): 151–165. DOI:10.1007/s00359-008-0394-3 |
| [5] | NORTHCUTT R G. Distribution and innervation of lateral line organs in the axolotl[J]. The Journal of Comparative Neurology, 1992, 325(1): 95–123. |
| [6] | CHENG H, HUANG S Q, HEATWOLE H. Ampullary organs, pit organs, and neuromasts of the Chinese giant salamander, Andrias davidianus[J]. Journal of Morphology, 1995, 226(2): 149–157. DOI:10.1002/(ISSN)1097-4687 |
| [7] | SONG J K, YANH Y, POPPERA N. Damage and recovery of hair cells in fish canal (but not superficial) neuromasts after gentamicin exposure[J]. Hearing Research, 1995, 91(1/2): 63–71. |
| [8] | GHYSEN A, DAMBLY-CHAUDIÈRE C. The lateral line microcosmos[J]. Genes & Development, 2007, 21(17): 2118–2130. |
| [9] | SCHOSSER G. Making senses: development of vertebrate cranial placodes[M]. JEON K W. International Review of Cell and Molecular Biology. Amsterdam: Elsevier, 2010: 129-234. |
| [10] | AHMED M, WONG E Y M, SUN J B, et al. Eya1-Six1 interaction is sufficient to induce hair cell fate in the cochlea by activating Atoh1 expression in cooperation with Sox2[J]. Developmental Cell, 2012, 22(2): 377–390. DOI:10.1016/j.devcel.2011.12.006 |
| [11] | KOZLOWSKI D J, WHITFIELD T T, HUKRIEDE N A, et al. The zebrafish dog-eared mutation disrupts eya1, a gene required for cell survival and differentiation in the inner ear and lateral line[J]. Developmental Biology, 2005, 277(1): 27–41. |
| [12] | SCHLOSSER G, AWTRY T, BRUGMANN S A, et al. Eya1 and Six1 promote neurogenesis in the cranial placodes in a SoxB1-dependent fashion[J]. Developmental Biology, 2008, 320(1): 199–214. DOI:10.1016/j.ydbio.2008.05.523 |
| [13] | THOMAS E D, CRUZ I A, HAILEY D W, et al. There and back again:development and regeneration of the zebrafish lateral line system[J]. Wiley Interdisciplinary Reviews:Developmental Biology, 2015, 4(1): 1–16. DOI:10.1002/wdev.2015.4.issue-1 |
| [14] | LOGAN C Y, NUSSE R. The Wnt signaling pathway in development and disease[J]. Annual Review of Cell andDevelopmental Biology, 2004, 20: 781–810. DOI:10.1146/annurev.cellbio.20.010403.113126 |
| [15] | MCGRAW H F, DRERUP C M, CULBERTSON M D, et al. Lef1 is required for progenitor cell identity in the zebrafish lateral line primordium[J]. Development, 2011, 138(18): 3921–3930. DOI:10.1242/dev.062554 |
| [16] | WADA H, GHYSEN A, ASAKAWA K, et al. Wnt/Dkk negative feedback regulates sensory organ size in zebrafish[J]. Current Biology, 2013, 23(16): 1559–1565. DOI:10.1016/j.cub.2013.06.035 |
| [17] | BAKER K D, RAMEL M C, LEKVEN A C. A direct role for Wnt8 in ventrolateral mesoderm patterning[J]. Developmental Dynamics, 2010, 239(11): 2828–2836. DOI:10.1002/dvdy.v239:11 |
| [18] | NIEHRS C. Function and biological roles of the Dickkopf family of Wntmodulators[J]. Oncogene, 2006, 25(57): 7469–7481. DOI:10.1038/sj.onc.1210054 |
| [19] | STOICK-COOPER C L, WEIDINGER G, RIEHLE K J, et al. Distinct Wnt signaling pathways have opposing roles in appendage regeneration[J]. Development, 2007, 134(3): 479–489. |
| [20] | KIMMEL C B, BALLARD W W, KIMMEL S R, et al. Stages of embryonic development of the zebrafish[J]. Developmental Dynamics, 1995, 203(3): 253–310. DOI:10.1002/aja.1002030302 |
| [21] | THISSE C, THISSE B. High-resolution in situ hybridization to whole-mount zebrafishembryos[J]. Nature Protocol, 2007, 3(1): 59–69. |
| [22] | VILLABLANCA E J, RENUCCI A, SAPÈDE D, et al. Control of cell migration in the zebrafish lateral line:implication of the gene "tumour-associated calcium signal transducer" tacstd[J]. Developmental Dynamics, 2006, 235(6): 1578–1588. DOI:10.1002/(ISSN)1097-0177 |
| [23] | VALDIVIA L E, YOUNG R M, HAWKINS T A, et al. Lef1-dependent Wnt/β-catenin signalling drives the proliferative engine that maintains tissue homeostasis during lateral line development[J]. Development, 2011, 138(18): 3931–3941. DOI:10.1242/dev.062695 |
| [24] | PARK B Y, SAINT-JEANNET J P. Hindbrain-derived Wnt and Fgf signals cooperate to specify the oticplacode in Xenopus[J]. Developmental Biology, 2008, 324(1): 108–121. |
| [25] | FRETER S, MUTA Y, MAK S S, et al. Progressive restriction of otic fate:the role of FGF and Wnt in resolving inner ear potential[J]. Development, 2008, 135(20): 3415–3424. DOI:10.1242/dev.026674 |
| [26] | PHILLIPS B T, STORCH E M, LEKVEN A C, et al. A direct role for Fgf but not Wnt in oticplacode induction[J]. Development, 2004, 131(4): 923–931. |
2. National Demonstration Center for Experimental Fisheries Science Education, Shanghai Ocean University, Shanghai 201306, China;
3. International Research Center for Marine Biosciences at Shanghai Ocean University, Ministry of Science and Technology, Shanghai 201306, China;
4. Key Laboratory of Exploration and Utilization of Aquatic Genetic Resources, Shanghai Ocean University, Ministry of Education, Shanghai 201306, China
 2018,
Vol. 27
2018,
Vol. 27


