2. 上海海洋大学 省部共建水产种质资源发掘与利用教育部重点实验室, 上海 201306
紫菜属(Pyropia)[1]在系统分类学上隶属于红藻门(Rhodophyta)、原红藻纲(Protoflorideae)、红毛菜目(Bangiales),红毛菜科(Bangiaceae),广泛分布于寒带至亚热带的潮间带[2]。紫菜属中的坛紫菜(Pyropia haitanensis)和条斑紫菜(Pyropia yezoensis)的经济价值较高,已在我国被大规模栽培,分别被栽培于长江南和长江北的沿海。
条斑紫菜藻体较薄,可被加工成附加值较高的菜饼,其年产量虽然只约占我国紫菜年总产量的25%,但产值却与占年总产量75%的坛紫菜相当[3]。但它不耐高温,不适合在我国南方沿海栽培。坛紫菜的高温耐受性较好,但它的藻体较厚,只能被加工成泡汤用的低价值菜饼。为了选育出藻体薄且又适合在南方高水温沿海栽培的紫菜品种,我们从印度引进了Pyropia chauhanii。该紫菜的叶状体较薄,适宜的生长温度几乎与坛紫菜相当[4],但它的一大缺陷是野生型品系的叶状体会放散大量单孢子,致使藻体无法长大[5]。为此,张聪和严兴洪[5]利用人工诱变技术从野生型品系的后代中分离出生长快、品质优良且不放单孢子的新品系(PC-Y1)。紫菜叶状体释放单孢子是一种无性繁殖方式,若放散量太大,会降低叶状体的生长速度,并降低产量,但如果放散量适量,对增加栽培网帘上的苗种数、提高产量和品质等均有重要意义[6-8],王素娟等[9]利用条斑紫菜可放散单孢子的生物学特性,在栽培于海上的紫菜网上进行单孢子二次采苗试验,获得了成功。本文对P. chauhanii野生型品系(PC-WT)的叶状体单孢子放散规律进行了较系统的研究,并对PC-Y1品系是否真不放散单孢子也作了进一步的验证。
1 材料与方法 1.1 实验材料P. chauhanii野生型品系(PC-WT),是从采自印度半岛的天然叶状体上分离出来的,新品系(PC-Y1)是PC-WT的单孢子苗经60Co-γ射线诱变后筛选获得的[5]。两个品系均以自由丝状体的形式被保存于实验室内,保存方法见参考文献[10]。
1.2 叶状体的培养取适量每个品系的自由丝状体,分别经粉碎机打碎后移植于干净的贝壳表面,培养为贝壳丝状体,贝壳丝状体的培养方法同文献[11]。数周后,待贝壳表面长出膨大藻丝,取1个贝壳于塑料杯中,加入适量的灭菌培养液[12]和数根棉线。待棉线附着一定数量的壳孢子后,将它们移入充气瓶中充气培养,培养条件:温度为(22±1)℃,光照密度为60 μmol photons/ (m2·s),光周期为10L∶14D,每5天更换1/2的培养液。
1.3 圆盘体培养与单孢子放散观察实验用打孔器(Φ=2.5 mm)分别从日龄为40、45和50 d 的PC-WT和PC-Y1品系的叶状体的梢部、中部和基部取30个圆盘体,每10个一组,放入3个已加入200 mL培养液的塑料杯中充气培养,培养条件:温度为(22±1)℃,光照密度为60 μmol photons/ (m2·s),光周期为10L∶14D。每天更换一次塑料杯,每隔2天拍照记录圆盘体的形态。在每天换下的塑料杯中重新倒入200 mL培养液继续充气培养,培养条件同上。2周后,将附着在塑料杯及充气管上的单孢子苗刮下,统计它们的数量,即得圆盘体的单孢子放散量。
2 结果 2.1 圆盘体的生长与单孢子放散随着培养天数的增加,PC-WT品系的圆盘体开始放散单孢子,其边缘逐渐呈现缺刻,变得不完整,有的开始解体。圆盘体出现缺刻的时间和解体程度,与原来所处的藻体部位和培养时间长短有关。边缘缺刻出现的时间,来自梢部的圆盘体最早,中部次之,基部最晚。当日龄40 d 的圆盘体培养至第6天,梢部和中部的部分圆盘体开始解体,至第10天,梢部的圆盘体只剩下约20%,中部的圆盘体还剩约60%,基部的圆盘体均出现了缺刻,但无解体(图版Ⅰ,1-3)。日龄45 d 的圆盘体,培养至第10天,梢部的圆盘体的细胞全部变成单孢子放散出来,只留下圆盘体的白色组织胶质与细胞壁骨架;中部的圆盘体单孢子放散也很厉害,第10天只剩下约30%的圆盘体,其余在单孢子放散后解体了;基部的圆盘体也有约10%已解体(图版Ⅰ,4-6)。日龄50 d 的圆盘体,培养至第4天,梢部圆盘体的细胞已全部放散,至第6天圆盘体解体消失;培养至第10天,中部的圆盘体也只剩下组织胶质和细胞壁骨架,但基部圆盘体有的放散单孢子后发生解体,有的不放散单孢子,其直径明显增加(图版Ⅰ,7-9)。

|
图 图版Ⅰ Pyropia chauhanii的野生型品系(PC-WT)和诱变品系(PC-Y1)不同日龄和不同部位的叶状体圆盘体的生长情况
Fig. 图版Ⅰ Growth of discs from the different ages and different parts of the gametophytic blades of wild-type strain (PC-WT) and induced strain(PC-Y1)in Pyropia chauhanii
1~3,4~6,7~9. 分别为日龄40、45和50 d的PC-WT品系叶状体的梢部、中部和基部的圆盘体再继续培养10 d后的生长情况;10~12,13~15,16~18.分别为日龄40、45和50 d的PC-Y1品系叶状体的梢部、中部和基部的圆盘体再继续培养10 d后的生长情况。图中标尺为2 cm 1-3,4-6,7-9. The discs from apical,middle,basal of the gametophytic blades of 40-day-old,45-day-old and 50-day-old in PC-WT were cultured for another ten days; 10-12,13-15,16-18. The discs from apical,middle,basal of the gametophytic blades of 40-day-old,45-day-old and 50-day-old in PC-Y1 were cultured for another ten days. Scale bar is 2 cm |
PC-Y1品系的各部位圆盘体,不论原藻体的日龄多少,圆盘体的直径均迅速增加,边缘光滑,至培养实验结束,也无单孢子放散。日龄40 d 的叶状体圆盘体其颜色较红,培养10 d也没有发生明显的褪色现象,生长很快(图版Ⅰ,10-12);但日龄45和50 d 的叶状体圆盘体在培养初期颜色较红,后期则颜色明显变浅,出现成熟(图版Ⅰ,13-18)。
2.2 圆盘体放散单孢子的显微观察通过显微观察发现不同日龄、不同部位的PC-WT品系的叶状体圆盘体放散单孢子的时间和数量存在较大的差异。日龄40 d的圆盘体被培养至第2天,梢部的圆盘体已开始放散单孢子,随着培养天数的增加,单孢子的放散程度越来越厉害;至第8天,梢部圆盘体的边缘基本上只剩下圆盘体的细胞壁骨架,中部和基部的圆盘体的边缘也出现单孢子大量放散后剩下的细胞壁骨架(图版Ⅱ,1-3)。
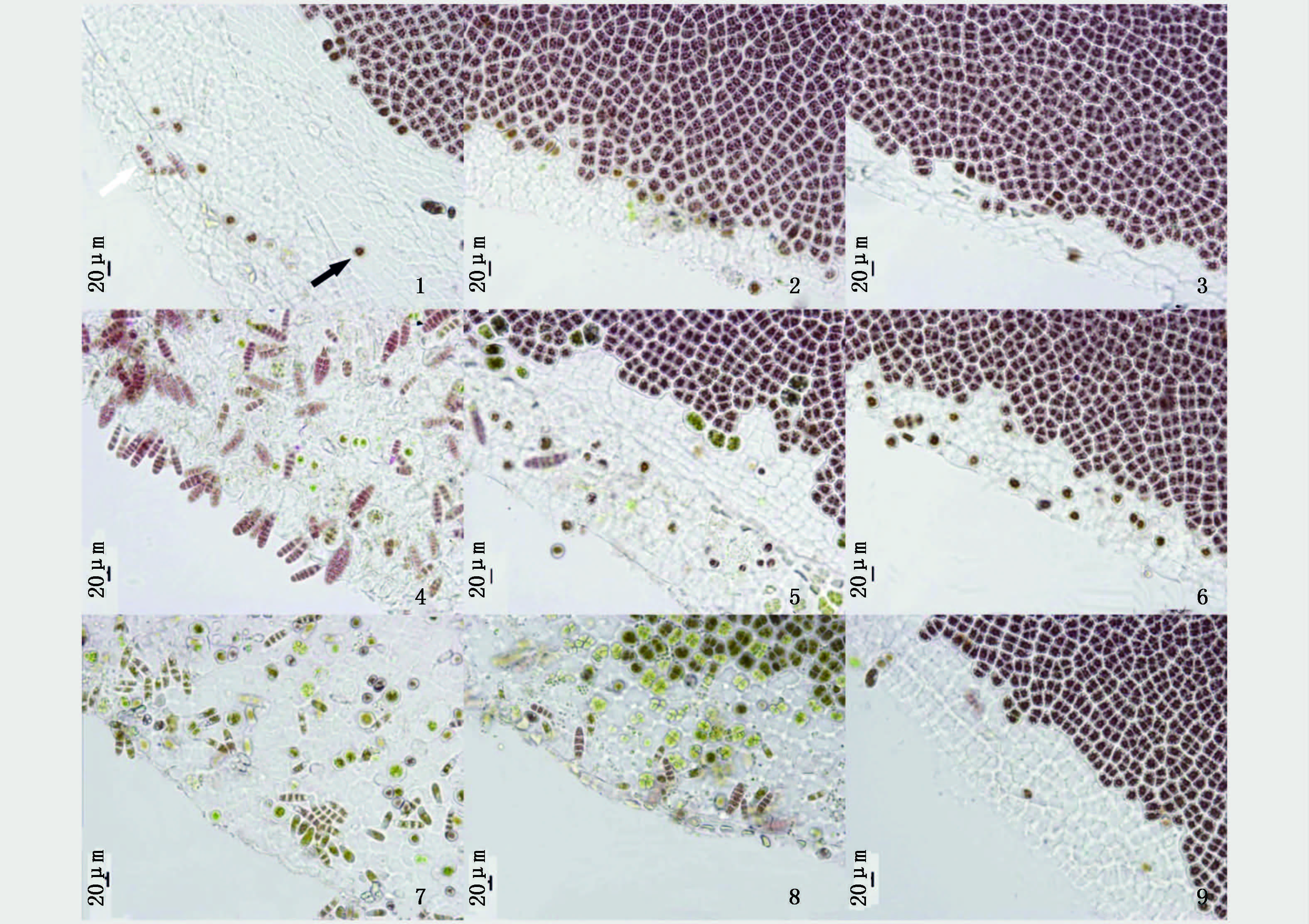
|
图 图版Ⅱ Pyropia chauhanii的野生型品系(PC-WT)不同日龄和不同部位的叶状体圆盘体的显微照片
Fig. 图版Ⅱ Micrograph of discs from the different ages and different parts of the gametophytic blades of wild-type strain (PC-WT) in Pyropia chauhanii
1~3分别为日龄40 d的PC-WT品系叶状体的梢部、中部和基部的圆盘体;4~6分别为日龄45 d的PC-WT品系叶状体的梢部、中部和基部的圆盘体;7~9分别为日龄50 d的PC-WT品系叶状体的梢部、中部和基部的圆盘体;1~6,8、9均为再继续培养8 d的圆盘体;7为再继续培养4 d的圆盘体。黑色箭头为单孢子,白色箭头为叶状体,图中标尺为20 μm 1-3. Discs from apical,middle,basal of the gametophytic blades which were 40-day-old of PC-WT; 4-6. Discs from apical,middle,basal of the gametophytic blades which were 45-day-old of PC-WT; 7-9. Discs from apical,middle,basal of the gametophytic blades which were 50-day-old of PC-WT; 1-6,8,9. The discs were cultivated for 8 days; 7. The discs were cultivated for 4 days. Monospore( |
日龄45 d的圆盘体被培养至第4天,梢部的圆盘体释放很多的单孢子,同时形成果胞和精子囊;中部的圆盘体也放散单孢子和形成性细胞。至第8天,梢部的圆盘体上已经长出许多单孢子小苗,中部和基部的圆盘体留下较多的细胞壁骨架(图版Ⅱ,4-6)。梢部和中部的圆盘体由于出现很多的性细胞,单孢子放散量相对较少,基部的圆盘体未形成性细胞,单孢子放散量较大。
日龄50 d的叶状体梢部的圆盘体再培养2 d就形成了大量的性细胞,至第4天,已经看不到营养细胞(图版Ⅱ,7),第6天之后,圆盘体已全部解体。中部圆盘体被培养至第8天,形成了大量的性细胞,单孢子放散较少(图版Ⅱ,8)。基部圆盘体没有形成性细胞,放散了大量的单孢子,边缘只留下了细胞壁骨架(图版Ⅱ,9)。
日龄40、45和50 d的PC-Y1叶状体的圆盘体在培养过程中,不论是梢部、中部和基部,其圆盘体均无单孢子放散。日龄40 d的叶状体梢、中、基部的圆盘体均为营养细胞,颜色较鲜艳(图版Ⅲ,1-3)。日龄45 d的叶状体圆盘体,第4天时细胞虽然均为营养细胞,但颜色开始变暗,第8天时出现了大量的果胞(图版Ⅲ,4-6)。日龄50 d的叶状体其梢部、中部和基部的圆盘体颜色没有较大差异,但均较暗淡,在培养初期就呈现出由营养细胞向性细胞发育的趋势(图版Ⅲ,7-9)。

|
图 图版Ⅲ Pyropia chauhanii的诱变品系(PC-Y1)不同日龄和不同部位的叶状体圆盘体的显微照片
Fig. 图版Ⅲ Micrograph of discs from the different ages and different parts of the gametophytic blades ofinduced strain (PC-Y1) in Pyropia chauhanii
~3分别为日龄40 d的PC-Y1品系叶状体的梢部、中部和基部的圆盘体; 4~6分别为日龄45 d的PC-Y1品系叶状体的梢部、中部和基部的圆盘体; 7~9分别为日龄50 d的PC-Y1品系叶状体的梢部、中部和基部的圆盘体;1~9均为再继续培养8 d的圆盘体。图中标尺为20 μm 1-3. Discs from apical,middle,basal of the gametophytic blades which were 40-day-old of PC-Y1; 4-6. Discs from apical,middle,basal of the gametophytic blades which were 45-day-old of PC-Y1; 7-9. Discs from apical,middle,basal of the gametophytic blades which were 50-day-old of PC-Y1; 1-9. The discs were cultivated for 8 days. Scale bar is 20 μm |
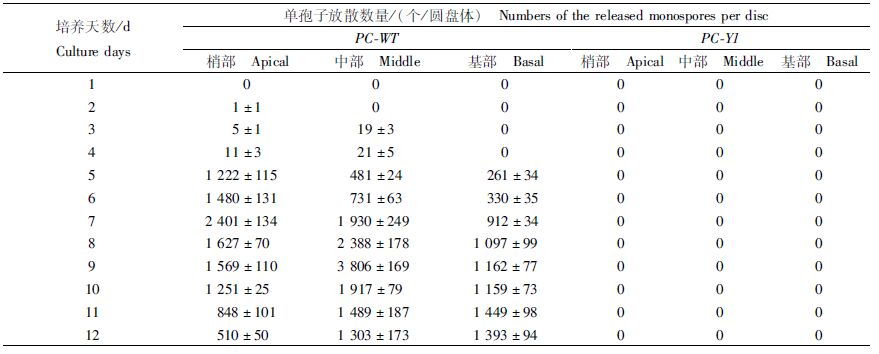
如表 1所示,日龄40 d的PC-WT叶状体梢部、中部和基部的圆盘体分别在第2天、第3天和第5天开始放散单孢子,梢部圆盘体在第7天出现单孢子放散高峰,中部圆盘体则在第9天出现放散高峰。梢部和中部圆盘体的单孢子日放散量呈先升后降的趋势,基部则呈上升趋势。日龄40 d的PC-Y1叶状体的圆盘体均没有放散单孢子。
|
表 1 日龄40 d的Pyropia chauhanii 的PC-WT和PC-Y1品系叶状体圆盘体再培养12 d的单孢子放散量 Tab.1 Numbers of the released monospores of the 40-day-old gametophytic blades discs of PC-WT and PC-Y1 |
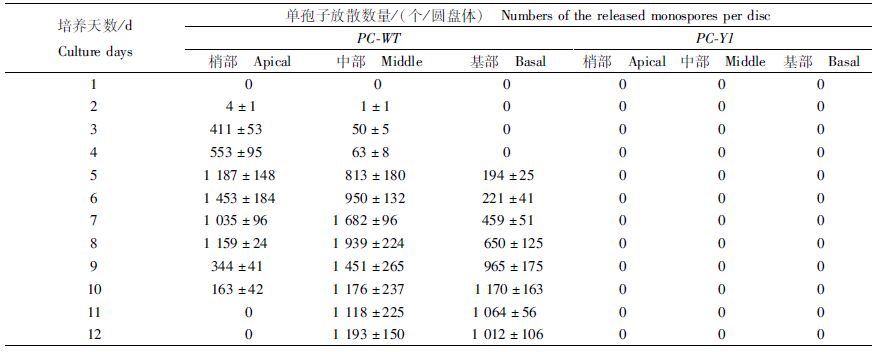
如表 2所示,日龄45 d的PC-WT叶状体梢部和中部的圆盘体均在第2天开始释放单孢子,基部则在第5天开始放散,为194个/圆盘体;梢部和中部的圆盘体分别在第6天和第8天出现单孢子放散高峰,分别为1 453个/圆盘体和1 939个/圆盘体;培养后期,基部圆盘体的日放散量维持在约1 000个/圆盘体。日龄45 d的PC-Y1叶状体的圆盘体在培养过程中仍没有单孢子释放。
|
表 2 日龄45 d的Pyropia chauhanii的PC-WT和PC-Y1品系叶状体圆盘体再培养12 d的单孢子放散量 Tab.2 Numbers of the released monospores of the 45-day-old gametophytic blades discs of PC-WT and PC-Y1 strains in Pyropia chauhanii after being cultured for another 12 days |
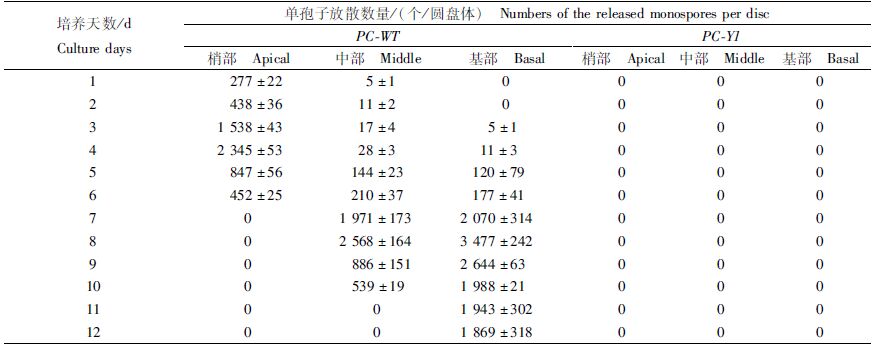
如表 3所示,日龄50 d的PC-WT叶状体梢部和中部的圆盘体在第1天就开始放散单孢子,基部的圆盘体在第3天也开始放散单孢子;至第6天,梢部的圆盘体就已全部放散;第8天,中部和基部的圆盘体的单孢子日放散量最多,分别为2 568个/圆盘体和3 477个/圆盘体。日龄50 d的PC-Y1叶状体的梢、中和基部的圆盘体均没有放散单孢子。
|
表 3 日龄50 d的Pyropia chauhanii的PC-WT和PC-Y1品系叶状体圆盘体再培养12 d的单孢子放散量 Tab.3 Numbers of the released monospores of the 50-day-old gametophytic blades discs of PC-WT and PC-Y1 strains in Pyropia chauhanii after being cultured for another 12 days |
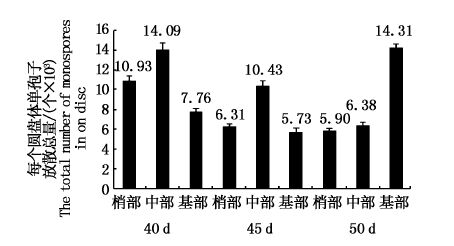
统计12 d的圆盘体单孢子放散总量,发现PC-WT的叶状体不同部位间具有一定的差异(图 1)。日龄40和45 d的圆盘体其单孢子放散总量次序为:中部>梢部>基部;日龄50 d的叶状体,其梢部和中部的圆盘体单孢子放散总量差异不明显,均比日龄40和45 d相同部位圆盘体的单孢子放散总量少,而基部圆盘体的放散总量最多,为1.43万个/圆盘体。
|
图 1 Pyropia chauhanii的PC-WT品系不同日龄和不同部位的叶状体圆盘体再培养12 d后的单孢子放散总量
Fig. 1 Numbers of monospores from discs of the different ages and different parts of gametophytic blades of PC-WT strain in Pyropia chauhanii after being cultured for another 12 days
|
紫菜属的生活史包括微型的丝状体和大型的叶状体两个阶段[13],绝大部分的紫菜种在丝状体成熟后,由放散的壳孢子长成叶状体,其中条斑紫菜和甘紫菜等[14-16]的叶状体长到一定日龄后,可放散单孢子,后者再萌发成叶状体。在生产上,紫菜叶状体放散的单孢子不仅能增加栽培网帘上的附苗量,还能利用单孢子进行二次采苗和栽培[17]。本文结果证实,P. chauhanii的野生型品系(PC-WT)叶状体也能放散单孢子,但它的诱变品系(PC-Y1)不放散单孢子,推测可能是基因发生变异所致,因为本实验室对该品系已经进行了连续数代的培养,其子代均未出现单孢子。YAN[18]等发现条斑紫菜的野生品系只能放散少量的单孢子,但经过人工诱变的红色和绿色突变体可以放散大量的单孢子,以致叶状体长不大,这证明与单孢子放散相关的基因发生了改变,并且这种改变是可以遗传的,本文的结果与他们的观察结果基本一致,即与紫菜单孢子放散相关的基因是可以通过人工诱变而改变的,但紫菜放散单孢子的详细分子生物学机制还有待研究。
梅俊学等[19]发现处于不同发育时期的叶状体在开始放散单孢子的时间上存在较大的差异,成熟的藻体仍可放散单孢子。本研究对日龄为40、45和50 d的PC-WT品系叶状体进行圆盘体培养实验,其结果也说明日龄对单孢子的放散时间早晚和数量均有较大影响,如果圆盘体所处的藻体部位相同,日龄50 d的圆盘体最早开始放散单孢子,日龄40 d的圆盘体则最晚,这说明叶状体放散单孢子的时间与叶状体日龄和成熟度是相关的,但准确的关系还需依据更多的实验结果予以阐明。如图 1所示:日龄40 d的叶状体,其单孢子放散量是中部最多、梢部次之、基部最少,日龄45 d的叶状体的单孢子放散量与此相类似,但3个部位的总放散量却明显下降。日龄50 d 的叶状体,单孢子的放散量是基部最多、中部其次、梢部最少。随着日龄的增加和成熟度的提高,梢部放散的单孢子量越来越少,暗示单孢子的放散量与藻体的成熟度是相关的。另外,还发现圆盘体生长速度越快,面积越大,放散单孢子的量也越大。随着日龄增大,梢部和中部的圆盘体生长越来越慢,性细胞出现量也越来越多,单孢子的放散量也越来越少,但基部的生长和单孢子放散量趋势与此相反。
本文结果也证实,藻体的某个部位越早成熟,它的性细胞的数量就越多,而单孢子的放散量越少。所以,叶状体圆盘体放散单孢子的时间早晚和数量与藻体发育和分化的程度可能存在某种关系,其机理有待阐明。
| [1] | SUTHERLAND J E, LINDSTROM S C, NELSON W A, et al.A new look at an ancient order: generic revision of the Bangiales (Rhodophyta)[J]. Journal of Phycology, 2011, 47(5): 1131–1151. DOI:10.1111/j.1529-8817.2011.01052.x |
| [2] | YOSHIDA T.Catalogue of species of Porphyra in the world, with special reference to the type locality and bibliography[J]. National History Research, 1997, 3: 5–18. |
| [3] | 马家海, 蔡守清. 条斑紫菜的栽培与加工[M]. 北京: 科学出版社, 1996: 1-58. MA J H, CAI S Q. Cultivation and process of Porphyra yezoensis[M]. Beijing: Science Press, 1996: 1-58. |
| [4] | 张岩岩. 两种印度产紫菜的室内培养与生活史观察[D]. 上海: 上海海洋大学, 2013: 25-28. ZHANG Y Y. Indoor cultivation and a part life-cycle of two species of Pyropia from India[D]. Shanghai: Shanghai Ocean University, 2013: 25-28. |
| [5] | 张聪, 严兴洪.Pyropia churharii新品系的分离与特性分析[J]. 水产学报, 2014, 38(9): 1457–1465. ZHANG C, YAN X H.Isolation and characterization of new strains of Pyropia churharii (Bangiales, Rhodophyta)[J]. Journal of Fisheries of China, 2014, 38(9): 1457–1465. |
| [6] | 郑宝福, 陈美琴, 费修绠.培养光强对条斑紫菜丝状体生长发育的影响[J]. 海洋与湖沼, 1980, 11(4): 362–369. ZHENG B F, CHEN M Q, FEI X G.On the effect of culture light intensity on the growth and development of the conchocelis phase of development of the conchocelis phase of Porphyra yezoensis Ueda[J]. Oceanologia et Limnologia Sinica, 1980, 11(4): 362–369. |
| [7] | 程凌江, 蒋丽金, 马金石.条斑紫菜中R-藻红蛋白的纯化及其α和β亚基的分离与发色团含量的测定[J]. 海洋与湖沼, 1990, 21(4): 337–342. CHENG L J, JIANG L J, MA J S.Purification of R-phycoerythrin in Porphyra yezoensis and separation, chromophore determination of its α and β subunits[J]. Oceanologia et Limnologia Sinica, 1990, 21(4): 337–342. |
| [8] | 陈美琴, 郑宝福, 任国忠.温度对条斑紫菜壳孢子和单孢子附着的影响[J]. 海洋湖沼通报, 1985: 66–69. CHEN M Q, ZHENG B F, REN G Z.The influence of temperature on the abherence of the conchospores and the monospores of Porphyra yezoensis Ueda[J]. Transactions of Oceanology and Limnology, 1985: 66–69. |
| [9] | 王素娟, 章景荣, 刘家驹, 等.条斑紫菜(Porphyra yezoensis Ueda)自然附苗养殖的初步研究[J]. 水产学报, 1964, 1(1/2): 85–94. WANG S J, ZHANG J R, LIU J J, et al.Preliminary study on the natural cultivation of Porphyra yezoensis Ueda[J]. Journal of Fisheries of China, 1964, 1(1/2): 85–94. |
| [10] | KATO M, ARUGA Y.Comparative studies on the growth and photosynthesis of the pigmentation mutants of Porphyra yezoensis in laboratory culture[J]. Japanese Journal of Phycology, 1984, 32: 333–347. |
| [11] | 梁志强. 坛紫菜遗传育种的初步研究[D]. 上海: 上海水产大学, 2004: 10-22. LIANG Z Q. Primary study on genetics and breeding of Porphyra haitanenis[D]. Shanghai: Shanghai Fisheries University, 2004: 10-22. |
| [12] | 王素娟, 张小平, 徐志东, 等.坛紫菜营养细胞和原生质体培养的研究Ⅰ[J]. 海洋与湖沼, 1986, 17(3): 217–221. WANG S J, ZHANG X P, XU Z D, et al.A study on the cultivation of the vegetative cells and protoplasts of P. haitanensis Ⅰ[J]. Oceanologia et Limnologia Sinica, 1986, 17(3): 217–221. |
| [13] | NIWA K.Genetic analysis of artificial green and red mutants of Porphyra yezoensis Ueda (Bangiales, Rhodophyta)[J]. Aquaculture, 2010, 308(1/2): 6–12. |
| [14] | XU L, ZHOU Y H, WANG S J, et al.Ultrastructural and molecular evidence for monospore formation in the thallus of Porphyra yezoensis[J]. Journal of Fisheries of China, 2003, 27(6): 519–527. |
| [15] | LI L, SAGA N, MIKAMI K.Phosphatidylinositol 3-kinase activity and asymmetrical accumulation of F-actin are necessary for establishment of cell polarity in the early development of monospores from the marine red alga Porphyra yezoensis[J]. Journal of Experimental Botany, 2008, 59(13): 3575–3586. DOI:10.1093/jxb/ern207 |
| [16] | NOTOYA M, KIKUCHI N, MATSUO M, et al.Culture studies of four species of Porphyra (Rhodophyta) from Japan[J]. Nippon Suisan Gakkaishi, 1993, 59(3): 431–436. DOI:10.2331/suisan.59.431 |
| [17] | 李世英, 崔广法.条斑紫菜单孢子和壳孢子幼苗生长发育的初步观察[J]. 海洋与湖沼, 1980, 11(4): 370–374. LI S Y, CUI G F.An observation on the growth and development of sporelings from concho-spores and monospores of Porphyra yezoensis[J]. Oceanologia et Limnologia Sinica, 1980, 11(4): 370–374. |
| [18] | YAN X H, FUJITA Y, ARUGA Y.High monospore-producing mutants obtained by treatment with MNNG in Porphyra yezoensis Ueda (Bangiales, Rhodophyta)[J]. Hydrobiologia, 2004, 512(1/3): 133–140. |
| [19] | 梅俊学, 费修绠, 王斌.条斑紫菜单孢子的研究[J]. 海洋与湖沼, 2001, 32(4): 402–407. MEI J X, FEI X G, WANG B.Study on monospore production of Porphyra yezoensis[J]. Oceanologia et Limnologia Sinica, 2001, 32(4): 402–407. |
2. Key Laboratory of Exploration and Utilization of Aquatic Genetic Resources, Ministry of Education, Shanghai Ocean University, Shanghai 201306, China
 2017,
Vol. 26
2017,
Vol. 26


