2. 贵州医科大学附属医院, 贵阳 550004;
3. 湖北医药学院附属人民医院 药学部,湖北 十堰 442000
2. The Affiliated Hospital of Guizhou Medical University, Guiyang 550004, China;
3. Department of Pharmacy, Renmin Hospital, Hubei University of Medicine, Shiyan 442000, Hubei Province, China
烟草花叶病毒 (TMV) 的防治一直是烟草优质生产栽培中的重大难题,寻找具有新型高效的抗TMV药剂是有效抑制植物病毒所造成危害的重要途径[1]。硫脲类化合物具有杀菌、消炎、抗病毒、抗HIV、抗肿瘤等广泛的生物活性[2-4],其分子设计、合成方法和生物活性研究一直是药物研究的热点,特别是因为硫脲分子中官能团硫脲键易于与底物通过氢键结合而发生拟生物酶与底物的作用[5],有利于探索手性硫脲与底物作用的构效关系、作用靶标及作用机制,并且由于其具有药害小、特异性强的优点,在农药领域中引起化学家的高度关注[4]。α-氨基酸具有多官能团和典型的手性中心,是合成手性药物的理想构建单元,其作为重要天然物质,与环境和生物体相容性好。氨基酸类药物具有杀菌、抗病毒、抗肿瘤等重要的生物活性[6-7]。将含氨基酸骨架的寡肽片段引入到药物分子中,往往可对药物的生物活性有积极作用[8-10]。α-氨基膦酸酯是天然含膦氨基酸酯类似物,它与膦酰化氨基酸、多肽的水解中间体在结构上具有相似性,同样具有广泛的生物活性[11-13]。近10年来,已有大量研究报道α-氨基膦酸酯类化合物具有较好的抗TMV活性[14-16]。
本课题组在前期工作中,采用活性基团拼接原理,设计并合成了一系列含亮氨酸、苯丙氨酸、甘氨酸等单个氨基酸的拟肽手性膦酸酯硫脲化合物[9-10, 17-19]。在此基础上,本研究开展结构改造和构型优化,在磷酸酯硫脲分子中引入含2个氨基酸的寡肽链 (丙氨酸、脯氨酸、苯丙氨酸等) 骨架,设计合成新型寡肽类手性膦酸酯硫脲化合物,通过IR、(1H、13C、19F、31P)NMR及元素分析对其结构进行表征,并测试其抗TMV活性。目标化合物的合成路线见图式1。
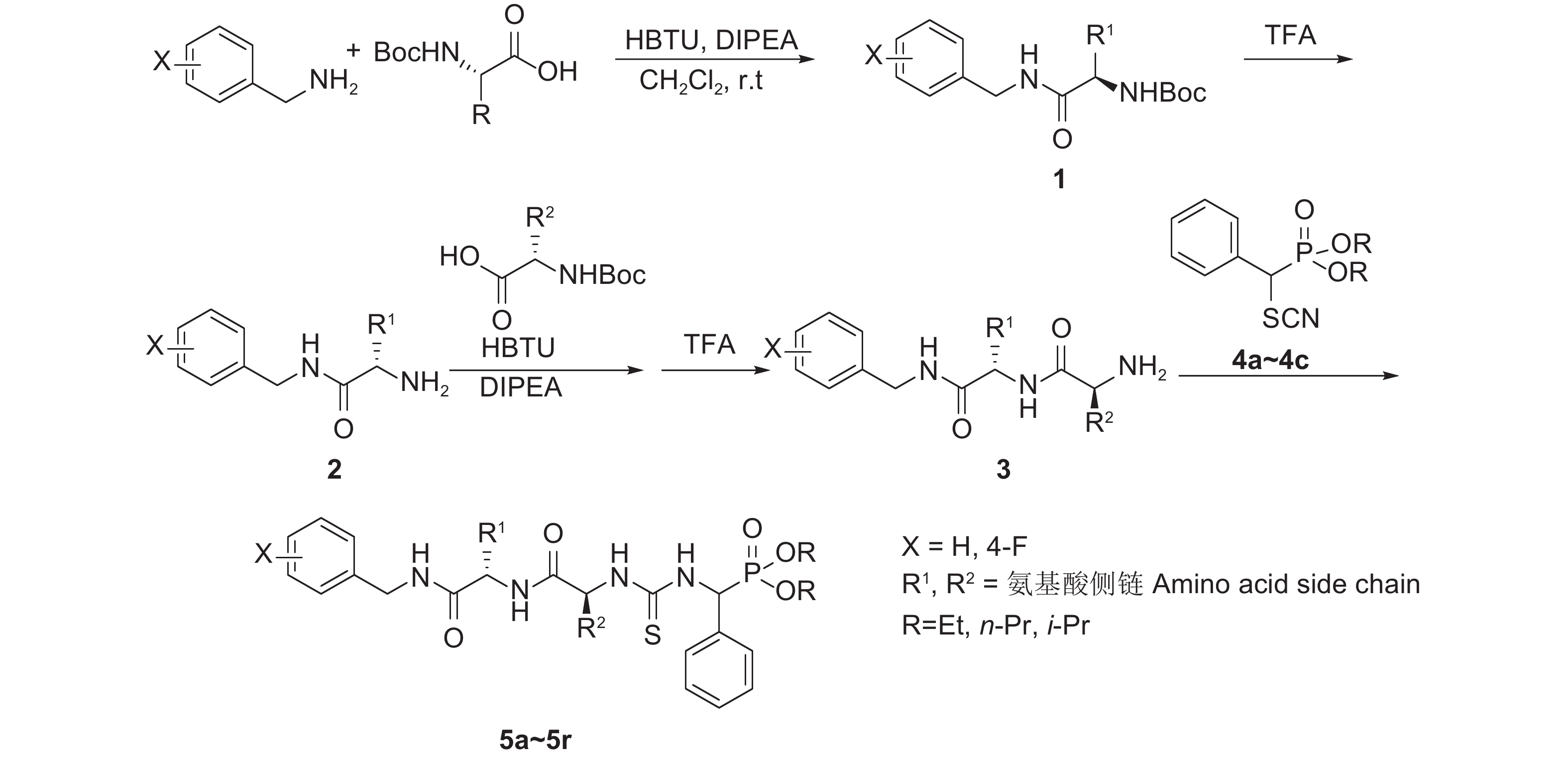
|
图式1 目标化合物5a~5r的合成路线 Scheme1 Synthetic route of target compounds 5a-5r |
1 实验部分 1.1 仪器与试剂
X-4型显微熔点测定仪 (上海易测仪器设备有限公司);Shimadzu IR Prestige-21型傅立叶红外光谱测定仪 (日本岛津公司)(KBr压片法);JEOL-ECX400型400 MHz核磁共振仪 (日本电子株式会社)(TMS为内标,CDCl3或DMSO-d6为溶剂);Elementar Vario Ⅲ型元素分析仪 (德国元素分析系统公司)。所用试剂均为分析纯。
心叶烟Nicotiana glutinosa L.种子,购于中国农业科学院烟草研究所。烟草花叶病毒 (tobacco mosaic virus,TMV),由中国科学院武汉病毒研究所提供。
对照药剂:2%宁南霉素水剂 (四川省金珠生态农业科技有限公司)。供试药剂均用灭菌水配成质量浓度为500 μg/mL的药液。
1.2 化合物合成 1.2.1 化合物1~3的合成将0.5 mmol手性N-Boc-L-氨基酸和0.5 mmol缩合剂O-苯并三氮唑-四甲基脲六氟膦酸酯 (HBTU) 溶于干燥二氯甲烷中,加入1 mmol无水二异丙基乙胺 (DIPEA) 和0.5 mmol取代苄胺,室温下搅拌,体系从非均相逐渐成为均相。反应3~4 h后将混合液转入分液漏斗中,加入1 mol/L的盐酸洗涤,有机相经1 mol/L 盐酸洗涤,再用无水硫酸钠干燥后过滤,减压脱溶剂得到化合物N-Boc-L-氨基酸酰胺 (1);将化合物1用干燥二氯甲烷重新溶解,一次性注入5.2 mL 三氟乙酸 (TFA) 于反应液面下,在室温下搅拌反应2~4 h (脱保护)。向反应体系中加入冷的饱和碳酸钠溶液 (5 ℃, 30 mL),静置分层,水层用二氯甲烷萃取,有机相经无水硫酸钠干燥、过滤、减压脱溶剂后得到无色至浅黄色油状氨基酸酰胺类化合物 (2)。不经处理将化合物2作为氨基部分,与另一个手性N-Boc-L-氨基酸在与上述相同条件下进行缩合,经TFA脱保护得到油状氨基酸酰胺二肽粗产物,经薄层层析 (V(二氯甲烷) : V (甲醇) = 40:1) 分离纯化,得到白色固体手性二肽氨基酸酰胺衍生物 (3)。
1.2.2 化合物4a~4c的合成化合物4a~4c的合成路线见图式2,按文献[18-19] 方法合成,其理化数据与文献报道相符。
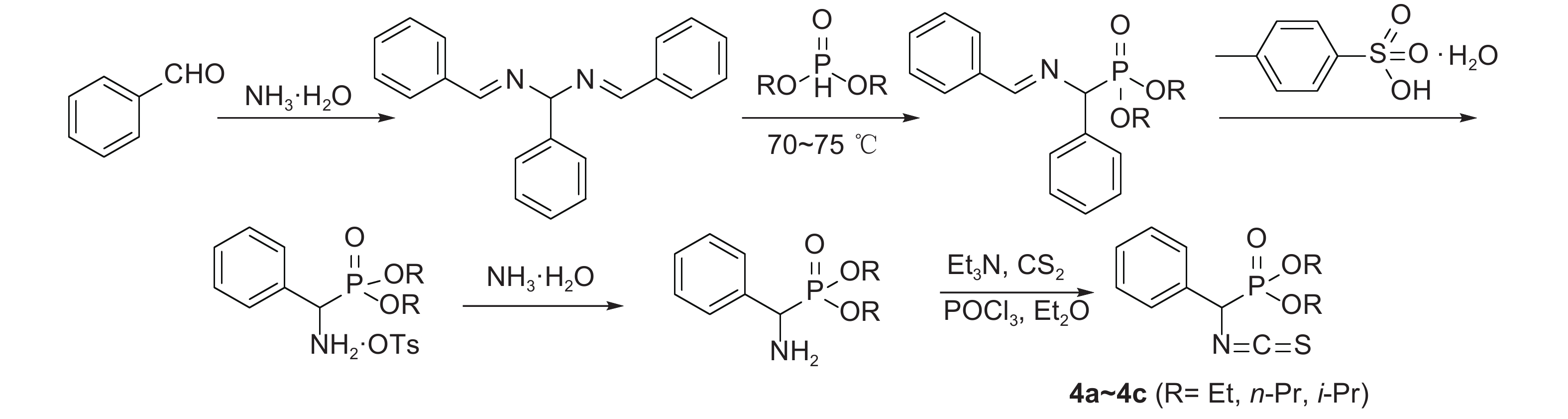
|
图式2 中间体4a~4c的合成路线 Scheme2 Synthetic route of intermediates 4a-4c |
1.2.3 目标化合物5a~5r的合成
参考文献[10]的方法合成。以5 mL无水二氯甲烷为溶剂,在1 mmol化合物3与1 mL三乙胺组成的混合液中加入1.2 mmol化合物4a~4c,在室温下搅拌反应0.5~1 h,减压脱溶剂后经柱层析 (洗脱剂为V (二氯甲烷) : V (甲醇) = 60:1) 分离,得到目标产物寡肽手性膦酸酯硫脲衍生物5a~5r。
1.3 化合物的抗TMV活性测试 1.3.1 病毒提纯采用Gooding等[20]报道的方法。选取接种TMV侵染3周以上的心叶烟上部叶片,在磷酸缓冲液 (0.01 mol/L,pH 7.2) 中匀浆,经双层纱布过滤,滤液在1 000 g下离心,沉淀物经2次聚乙二醇处理后再在10 000 g下离心,沉淀物用磷酸缓冲液 (0.01 mol/L,pH 7.2) 悬浮,即得到TMV的粗提液体。整个试验在4 ℃下进行。用紫外分光光度计测定其在260 nm波长下的吸光度值,根据公式 (1) 计算病毒的质量浓度。
| ${\text{病毒质量浓度}}\!/\!\left( {{{mg}}\!/\!{{mL}}} \right) \!=\frac{\left( {{A_{260}} \!\times \!{\text{稀释倍数}}} \right)}{E_{1\;{ cm}}^{0.1 \text{\%} \;260\;{ nm}}}$ | (1) |
其中,
参考文献[21]方法进行。选取长势一致的心叶烟,用毛笔轻轻在左半叶涂施药剂 (500 μg/mL),右半叶涂施灭菌水作对照,待叶片干后 (12 h) 接种TMV病毒。接种时先打顶,然后在需接种的叶片表面均匀喷洒1薄层400目金刚砂,用毛笔蘸取用磷酸盐缓冲液稀释过的病毒汁液 (6 × 10–3 mg/mL),在叶面 (全叶) 沿其支脉方向轻轻擦1~2次。接种后用流水 (或洗瓶) 冲洗叶片。随后在光照培养箱中保湿 (温度23 ℃ ± 1 ℃,湿度60%,照度1 × 104 lx) 培养,3~4 d后观察并记录产生枯斑的数目。每药剂处理设3株心叶烟,每株3~4片叶。每药剂重复3次。
1.3.3 药剂对TMV的活体钝化作用参考文献[21]方法进行。将药剂 (500 μg/mL) 与等体积的TMV病毒汁液混合钝化30 min,摩擦接种心叶烟左半叶,作为试验组;将灭菌水与病毒汁液混合后接种右半叶,作为对照组。随后在光照培养箱中保湿 (温度23 ℃ ± 1 ℃,湿度60%,照度1 × 104 lx) 培养,3~4 d后观察并记录产生枯斑的数目。每药剂处理设3株心叶烟,每株3~4片叶。每药剂重复3次 。
1.3.4 药剂对TMV侵染的活体治疗作用参考文献[21]方法进行。选取长势一致的心叶烟,先用毛笔蘸取TMV病毒汁液,全叶接种病毒,接种后用清水冲洗。待叶片干后,在左半叶涂施药剂,右半叶涂施对应剂量的溶剂作对照。随后在光照培养箱中保湿 (温度23 ℃ ± 1 ℃,湿度60%,照度1 × 104 lx) 培养,3~4 d后观察并记录产生枯斑的数目。每药剂处理设3株,每株3~4片叶。每药剂重复3次。
1.3.5 结果调查与分析待右边空白对照的半叶上呈现明显枯斑时 (约3~4 d),分别记录每片叶的左、右半叶的枯斑数,按 (2) 式分别计算供试化合物对烟草花叶病毒的治疗、保护和钝化作用效果 (以枯斑抑制率表示)。
| $Y/\text{\%} = \frac{\left( {C - A} \right)}{C} \times 100$ | (2) |
其中:Y为化合物对烟草花叶病毒枯斑的抑制率;C为对照组 (右半叶) 枯斑数,单位:个;A为试验组 (左半叶) 枯斑数,单位:个。左右半叶的平均枯斑数均取各组3次重复的平均数。
2 结果与讨论 2.1 目标化合物的结构表征目标化合物结构经红外光谱 (IR)、( 1H、13C、31P、19F) NMR及元素分析确证和表征,其数据如下:
O,O′-二乙基 (3-(苄胺基-甘氨酰-L-苯甘氨酰)-α-硫脲基) 苯甲膦酸酯 (5a):白色固体,产率91.6%,m. p. 179~180 ℃,
O,O′-二丙基 (3-(苄胺基-甘氨酰-L-苯甘氨酰)-α-硫脲基) 苯甲膦酸酯 (5b):白色固体,产率81.5%,m. p. 155~157 ℃,
O,O′-二异丙基 (3-(苄胺基-甘氨酰-L-苯甘氨酰)-α-硫脲基) 苯甲膦酸酯 (5c):白色固体,产率78.6%,m. p. 203~204 ℃,
O,O′-二乙基 (3-(对氟苄胺基-甘氨酰-L-苯甘氨酰)-α-硫脲基) 苯甲膦酸酯 (5d):无色晶体,产率93.3%,m. p. 74~75 ℃,
O,O′-二丙基 (3-(对氟苄胺基-甘氨酰-L-苯甘氨酰)-α-硫脲基) 苯甲膦酸酯 (5e):白色固体,产率75.5%,m. p. 133~134 ℃,
O,O′-二异丙基 (3-(对氟苄胺基-甘氨酰-L-苯甘氨酰)-α-硫脲基) 苯甲膦酸酯 (5f):白色固体,产率77.8%,m. p. 176~177 ℃,
O,O′-二乙基 (3-(苄胺基-甘氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5g):白色固体,产率66.8%,m. p. 167~168 ℃,
O,O′-二丙基 (3-(苄胺基-甘氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5h):白色固体,产率71.4%,m. p. 148~149 ℃,
O,O′-二异丙基 (3-(苄胺基-甘氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5i):无色晶体,产率70.4%,m. p. 72~73 ℃,
O,O′-二乙基 (3-(对氟苄胺基-甘氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5j):白色固体,产率98.7%,m. p. 75~76 ℃,
O,O′-二丙基 (3-(对氟苄胺基-甘氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5k):白色固体,产率94.3%,m. p. 95~96 ℃,
O,O′-二异丙基 (3-(对氟苄胺基-甘氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5l):白色固体,产率95.0%,m. p. 62~63 ℃,
O,O′-二乙基 (3-(苄胺基-L-脯氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5m):白色固体,产率92.6%,m. p. 103~104 ℃,
O,O′-二丙基 (3-(苄胺基-L-脯氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5n):白色固体,产率90.2%,m. p. 117~118 ℃,
O,O′-二异丙基 (3-(苄胺基-L-脯氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5o):白色固体,产率81.8%,m. p. 89~90 ℃,
O,O′-二乙基 (3-(对氟苄胺基-L-脯氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5p):白色固体,产率93.5%,m. p. 97~98 ℃,
O,O′-二丙基 (3-(对氟苄胺基-L-脯氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5q):白色固体,产率53.9%,m. p. 107~108 ℃,
O,O′-二异丙基 (3-(对氟苄胺基-L-脯氨酰-L-苯丙氨酰)-α-硫脲基) 苯甲膦酸酯 (5r):白色固体,产率82.4%,m. p. 83~84 ℃,
从IR数据可以看出,目标化合物酰胺基和硫脲基N-H伸缩振动吸收峰在3 000~3 300 cm–1处附近;在2 930~2 980 cm–1处为甲基不对称吸收峰;在1 511~1 533 cm–1附近有苯环C=C骨架振动;硫脲基的C=S伸缩振动峰出现在1 349 cm–1附近;在1 222 cm–1和999 cm–1附近分别出现P=O双键和P-O-C键较强的伸缩振动吸收峰;同时芳基、烃基的C-H伸缩振动吸收峰都很明显。
在 1H NMR谱中,化学位移值δ 7.48~7.06为苯环上Ar-H;由于氮原子和磷原子的电负性作用,与之相连的次甲基上的氢化学位移向低场移动,出现在δ 6.79附近;δ 4.68~4.46处为多重峰,是酰胺基团中与氮相连芳基上的氢;膦酸酯酯键上的亚甲基和次甲基受到P的耦合裂分,出现在δ 4.33附近;δ 1.40~0.88处为多重峰,为甲基氢。
2.2 目标化合物的合成 2.2.1 反应溶剂对目标化合物收率的影响以目标化合物5a的合成为例,考察了不同溶剂对其产率的影响。在50 mL三颈瓶中加入中间体3a (X = H,R1 = H,R2 = Ph)(1 mmol)、5 mL溶剂和1 mL三乙胺,室温条件下搅拌反应5 min再加入中间体异硫氰酸酯4a (1.2 mmol),以TLC (展开剂V (二氯甲烷) : V (甲醇) = 20:1) 监测反应,1 h反应完毕。反应液直接减压浓缩蒸除溶剂,得浅黄色液体,经柱层析分离 (V (二氯甲烷) : V (甲醇) = 60:1) 得5a。不同溶剂对目标化合物收率的影响见表1。
|
|
表 1 不同溶剂对合成目标化合物5a收率的影响 Table 1 Effect of different solvents on the synthesis of the target compound 5a |
通过表1可看出,采用不同溶剂合成目标化合物5a,其收率相差较大。其中以二氯甲烷为溶剂产物收率最高,达95%;而以极性较小的环己烷为溶剂,产物收率仅为35%。这可能是由于不同极性的溶剂对手性二肽胺的溶解性、极化程度等的不同,从而导致底物中游离氨基与异硫氰基的结合速率不同。
2.2.2 中间体α-异硫氰基苯甲膦酸酯 (4) 的合成方法分别采用氯甲酸乙酯、硫酸铜和三氯氧磷[4]3种试剂合成中间体4,并比较了3种方法的产物收率。以无水乙醚为溶剂,向氨基膦酸酯中加入二硫化碳和三乙胺,冰浴下搅拌反应2 h后分别滴加氯乙酸乙酯 (硫酸铜或三氯氧磷) 的无水乙醚混合液,再在冰浴下搅拌反应2 h[18-19]。反应过程见图式3。结果显示,三氯氧磷法的收率 (68%) 比氯甲酸乙酯 (51%) 和硫酸铜法的收率 (18%) 高,此法合成异硫氰酸酯的副产物较少,后处理简便,易于提纯。因此,最终选用三氯氧磷法制备中间体α-异硫氰基苯甲膦酸酯 (4)。
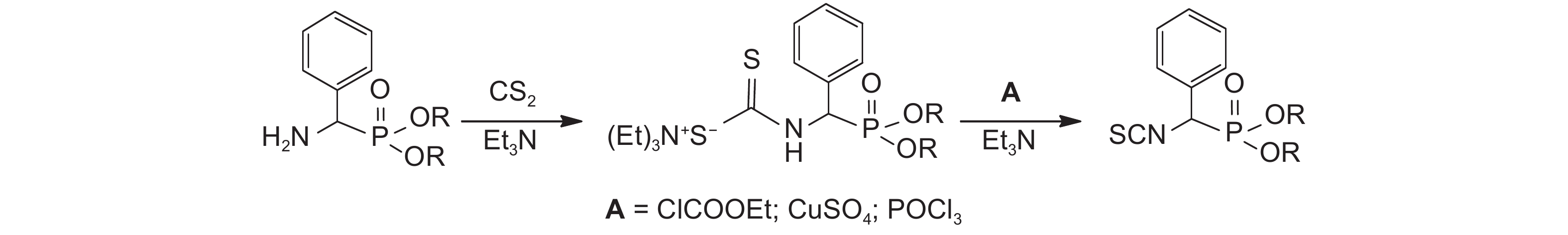
|
图式3 中间体4的合成路线 Scheme3 Synthesis of intermediate 4 |
2.3 目标化合物抗TMV活性
由表2数据可以看出:在500 μg/mL时供试化合物对TMV具有中等至较好的治疗活性,部分化合物对TMV活体治疗、保护和钝化作用接近对照药剂宁南霉素。其中,化合物5i、5j和5p对TMV治疗作用效果 (枯斑抑制率) 分别为52.6%、55.7%和56.1%,其保护作用效果 (枯斑抑制率) 分别为57.8%、52.4%和50.6%,钝化作用效果 (枯斑抑制率) 分别为83.9%、80.7%和72.3%。从结构上看,该类化合物中,当X = p-F,R = Et或i-Pr,R1、R2 = 芳环或杂环 (脯氨酸侧链) 时,化合物抗TMV的活性显著提高。可见,在该类寡肽膦酸酯硫脲中,引入不同种类氨基酸以及改变氨基酸连接次序,将明显影响其目标产物的生物活性。
3 结论采用活性基团拼接原理,以手性α-氨基酸、取代苄胺、取代苯甲醛和亚膦酸二烷基酯等为原料,将氟原子、氨基酸膦酸酯和氨基酸引入到硫脲结构中,合成了18个新型寡肽手性膦酸酯硫脲衍生物。对目标化合物抗TMV活性测试表明:该类化合物具有较好的抗TMV活性,尤其是化合物5j (X = p-F,R=Et,R1 = H,R2 = L-Bn) 和5p (X = p-F,R = Et,R1 = 脯氨酸侧链,R2 = L-Bn) 抗TMV的治疗作用效果较高,具有进一步深入研究价值。
|
|
表 2 目标化合物的抗TMV活性 Table 2 Anti-TMV activity of the target compound |
| [1] |
SONG B A, YANG S, JIN L H, et al. Environment-friendly antiviral agents for plants[M]. Berlin: Springer Press, 2010: 1-10.
|
| [2] |
FUJII N, MALLARI J P, HANSELL E J, et al. Discovery of potent thiosemicarbazone inhibitors of rhodesain and cruzain[J]. Bioorg Med Chem Lett, 2005, 15(1): 121-123. DOI:10.1016/j.bmcl.2004.10.023 |
| [3] |
EASMON J, PURSTINGER G, HEINISCH G, et al. Synthesis, cytotoxicity, and antitumor activity of copper(II) and iron(II) complexes of (4)N-azabicyclo[3.2.2]nonane thiosemicarbazones derived from acyl diazines[J]. J Med Chem, 2001, 44(13): 2164-2171. DOI:10.1021/jm000979z |
| [4] |
CHEN M H, CHEN Z, SONG B A, et al. Synthesis and antiviral activities of chiral thiourea derivatives containing an α-aminophosphonate moiety
[J]. J Agric Food Chem, 2009, 56(4): 1383-1388. |
| [5] |
GOODYER C L M, CHINJE E C, JAFFAR M, et al. Synthesis of N-benzyl-and N-phenyl-2-amino-4,5-dihydrothiazoles and thioureas and evaluation as modulators of the isoforms of nitric oxide synthase
[J]. Bioorg Med Chem, 2003, 11(19): 4189-4206. DOI:10.1016/S0968-0896(03)00451-6 |
| [6] |
BARRETT G C. Chemistry and biochemistry of the amino acids[M]. London: Chapman and Hall, 1985: 297-338.
|
| [7] |
SEITZ T, WOLLWEBER D. Substituted valinamide derivatives: DE 4026966[P]. 1992-03-04.
|
| [8] |
SZEKERK M. Cyclic phosphoramide mustard (NSC-69945) derivatives of amino acids and peptides[J]. Cancer Treat Rep, 1976, 60(4): 347-354. |
| [9] |
杨家强, 谷晴, 胡月维, 等. 超声辐射下O,O′-二烷基-α-取代苯基-α-苯磺酰氧基甲基膦酸酯的合成与生物活性
[J]. 有机化学, 2013, 33(10): 2226-2231. YANG J Q, GU Q, HU Y W, et al. Synthesis of O,O′-dialkyl-α-phenyl-α-phenylsulfonyloxy-methylphosphonate under ultrasound and their antitumor activity [J]. Chinese J Org Chem, 2013, 33(10): 2226-2231. |
| [10] |
LIU J Z, SONG B A, BHADURY P S, et al. Synthesis and bioactivities of α-aminophosphonate derivatives containing benzothiazole and thiourea moieties
[J]. Phosphorus Sulfur Silicon Relat Elem, 2012, 187(1): 61-70. DOI:10.1080/10426507.2011.575422 |
| [11] |
BOOJAMRA C G, LIN K Y, MACKMAN R L, et al. Preparation of amino acid-containing nucleotides as prodrugs and antiviral agents: WO 2006015261[P]. 2006-02-09.
|
| [12] |
石德清, 陈茹玉. 含α-氨基膦酸酯的脱落酸酰胺类似物的合成与生物活性
[J]. 应用化学, 2012, 19(8): 780-782. SHI D Q, CHEN R Y. Synthesis and biological activities of abscisic amide derivatives containing α-amino phosphonate [J]. Chin J Appl Chem, 2012, 19(8): 780-782. |
| [13] |
马姣丽, 朱文娟, 李静, 等. 含噻吩并[3,2-c]吡啶α-氨基膦酸酯类衍生物的合成及生物活性的研究
[J]. 有机化学, 2013, 33(7): 1472-1477. MA J L, ZHU W J, LI J, et al. Synthesis and biological activities of α-aminophosphonates derivatives containing thieno[3,2-c] pyridine [J]. Chinese J Org Chem, 2013, 33(7): 1472-1477. |
| [14] |
HU D Y, WAN Q Q, YANG S, et al. Synthesis and antiviral activities of amide derivatives containing the α-aminophosphonate moiety
[J]. J Agric Food Chem, 2008, 56(3): 998-1001. DOI:10.1021/jf072394k |
| [15] |
LONG N, CAI X J, SONG B A, et al. Synthesis and antiviral activities of cyanoacrylate derivatives containing an α-aminophosphonate moiety
[J]. J Agric Food Chem, 2008, 56(13): 5242-5246. DOI:10.1021/jf800405m |
| [16] |
ZHANG P W, TANG C H, CHEN Z W, et al. Design, synthesis, and antiviral activity of α-aminophosphonates bearing a benzothiophene moiety
[J]. Phosphorus Sulfur Silicon Relat Elem, 2014, 189(4): 530-540. DOI:10.1080/10426507.2013.829837 |
| [17] |
王伟, 张国平, 宋宝安, 等. O,O′-二烷基-α-(取代苯并噻唑-2-基)氨基-(取代苯基甲基)膦酸酯的合成与抗烟草花叶病毒活性
[J]. 有机化学, 2007, 27(2): 279-284. WANG W, ZHANG G P, SONG B A, et al. Synthesis and anti-tobacco mosaic virus activity of O,O′-dialkyl-α-(substituted benzothiazol-2-yl)amino-(substituted phenylmethyl)phosphonate [J]. Chinese J Org Chem, 2007, 27(2): 279-284. |
| [18] |
LIU J Z, SONG B A, FAN H T, et al. Synthesis and in vitro study of pseudo-peptide thioureas containing α-aminophosphonate moiety as potential antitumor agents
[J]. Eur J Med Chem, 2010, 45(11): 5108-5112. DOI:10.1016/j.ejmech.2010.08.021 |
| [19] |
LIU J Z, YANG S, LI X Y, et al. Synthesis and antiviral bioactivity of chiral thioureas containing leucine and phosphonate moieties[J]. Molecules, 2010, 15(8): 5112-5123. DOI:10.3390/molecules15085112 |
| [20] |
GOODING JR G V, HEBERT T T. A simple technique for purification of tobacco mosaic virus in large quantities[J]. Phytopathology, 1967, 57(11): 1285. |
| [21] |
SONG B A, ZHANG H P, WANG H, et al. Synthesis and antiviral activity of novel chiral cyanoacrylate derivatives[J]. J Agric Food Chem, 2005, 53(20): 7886-7891. DOI:10.1021/jf051050w |
 2018, Vol. 20
2018, Vol. 20


