2. 广西田园生化股份有限公司 南宁 530000
2. Guangxi TianyuanBiochemical Co., Ltd, Nanning 530000, China
喜树碱(camptothecin,结构式见图式1) 是从中国珙桐科乔木喜树中分离出来的一种喹啉类生物碱,因其具有抗癌、抗病毒、杀虫和抑菌等多种生物活性而倍受国内外研究者关注[1-2]。
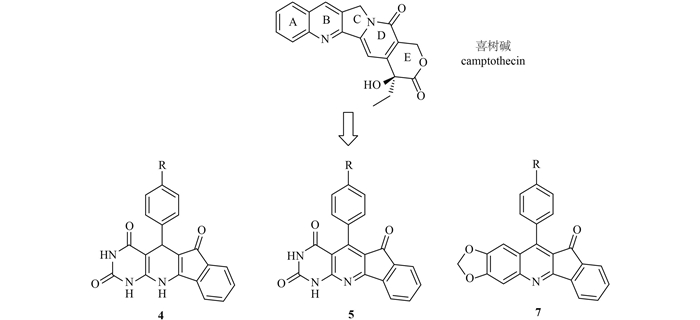
|
图式1 设计新的喜树碱类药性分子骨架 Scheme1 Design of camptothecin-inspired heterocycle scaffolds |
近年来,本课题组采用化学合成与活性筛选相结合的手段,通过对喜树碱A、B、C、D和E环进行衍生合成与结构修饰,系统分析了喜树碱母体骨架不同位点经结构修饰后的化合物与杀虫活性之间的关系,探讨了不同取代基的电性、体积及取代基位置对杀虫活性的影响,获得了喜树碱衍生物的杀虫构效关系线索[3-7]。为进一步改变因喜树碱母体结构的局限性,用传统的结构修饰方法只能实现对其母体骨架结构位点的有限衍生合成,而无法实现对结构多样性类喜树碱结构的系统调查与结构优化研究等问题,笔者以获得的构效关系线索为依据,通过计算机辅助设计,从原子水平和分子力场角度进一步探讨了喜树碱类药性分子与DNA-Top I的相互作用模式[8],并应用三组分反应合成策略构建了一系列结构新颖、易于合成的喜树碱类药性分子(化合物4、5和7),其合成路线分别见图式2和图式3。初步评价了该类化合物对朱砂叶螨的毒杀活性。
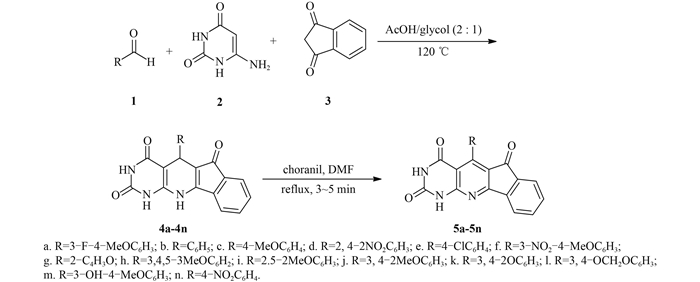
|
图式2 目标化合物4a~4n和5a~5n的合成路线 Scheme2 Synthetic routes of target compounds 4a-4n and 5a-5n |
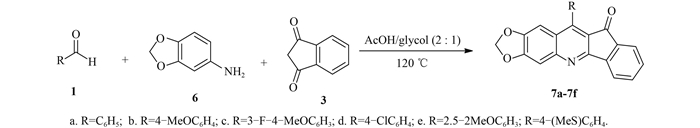
|
图式3 目标化合物7a~7f的合成路线 Scheme3 Synthetic routes of target compounds 7a-7f |
1 实验部分 1.1 仪器与试剂
X-4数字显微熔点测定仪(北京泰克仪器有限公司,温度未经校正);Bruker AM-400核磁共振波谱仪(德国Bruker公司,以氘代DMSO为溶剂,以四甲基硅烷为内标);Bruker Esquire 6 000液相色谱-质谱联用仪(德国Bruker公司)。
0.054~0.075 mm粗孔型柱层析硅胶,购自青岛海洋化工有限公司;自制薄层层析硅胶板,规格为2.5 cm × 7.5 cm,GF254硅胶购自青岛海洋化工有限公司;羧甲基纤维素钠,购自国药集团化学试剂有限公司。所有的无水溶剂均按标准方法预处理。喜树碱(CPT) 购自四川省江源天然产物有限公司;其他试剂均为市售分析纯。
1.2 化合物的合成 1.2.1 化合物4a~4n的合成参考文献[9]方法。在50 mL圆底烧瓶中,加入4 mL冰醋酸和2 mL无水乙醇后,再分别加入芳香醛(1,0.8 mmol)、6-氨基脲嘧啶(2,0.76 mmol) 和1, 3-茚满二酮(3,0.8 mmol),加热至120 ℃,持续搅拌,反应2 h,直至不再有固体产物析出时停止反应。冷却至室温,过滤,固体依次用无水乙醇(10 mL × 3) 和乙醚(3 mL × 3) 洗涤,所得粗产物通过硅胶柱层析[洗脱剂为V (三氯甲烷):V (甲醇)=50:1]纯化,得到目标化合物4a~4n。
5-(3-氟-4-甲氧基苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4a):产率75%,m. p. 203~205 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.92 (s, 1H, NH-H), 10.30 (s, 1H, NH-H), 10.17 (s, 1H, NH-H), 7.50~7.28 (m, 4H, Ar-H), 7.07~6.96 (m, 3H, Ar-H), 4.64 (s, 1H, dihydropyridine-H), 3.76 (s, 3H, Ar-OCH3); MS-ESI, m/z: 414.1 [M + Na]+.
5-苯基-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4b):产率88%,m.p. 186~188 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.84 (s, 1H, NH-H), 10.27 (s, 1H, NH-H), 10.11 (s, 1H, NH-H), 7.01~7.16 (m, 4H, Ar-H), 7.26~7.44 (m, 5H, Ar-H), 4.67 (s, 1H, dihydropyridine-H); MS-ESI, m/z: 366.3[M + Na]+.
5-(4-甲氧基苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4c):产率75%,m. p.188~190 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.88 (s, 1H, NH-H), 10.29 (s, 1H, NH-H), 10.12 (s, 1H, NH-H), 7.49~7.26 (m, 4H, Ar-H), 7.17~7.12 (m, 2H, Ar-H), 6.80~6.76 (m, 2H, Ar-H), 4.62 (s, 1H, dihydropyridine-H), 3.68 (s, 3H, Ar-OCH3); MS-ESI, m/z: 396.3 [M + Na]+.
5-(2, 4-二硝基苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4d):产率62%,m. p.198~200 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.90 (s, 1H, NH-H), 10.49 (s, 1H, NH-H), 10.29 (s, 1H, NH-H), 7.49~7.34 (m, 4H, Ar-H), 7.14~7.06 (m, 3H, Ar-H), 4.68 (s, 1H, dihydropyridine-H); MS-ESI, m/z: 456.2 [M + Na]+.
5-(4-氯苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4e):产率70%,m. p.173~176 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.93 (s, 1H, NH-H), 10.32 (s, 1H, NH-H), 10.19 (s, 1H, NH-H), 7.50~7.31 (m, 4H, Ar-H), 7.08~7.07 (m, 4H, Ar-H), 4.68 (s, 1H, dihydropyridine-H); MS-ESI, m/z: 400.1 [M + Na]+.
5-(3-硝基-4-甲氧基苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4f):产率66%,m. p.180~182 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.95 (s, 1H, NH-H), 10.36 (s, 1H, NH-H), 10.24 (s, 1H, NH-H), 7.71~7.22 (m, 7H, Ar-H), 4.73 (s, 1H, dihydropyridine-H), 3.86 (s, 3H, Ar-OCH3); MS-ESI, m/z: 441.1 [M + Na]+.
5-(2-呋喃基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4g):产率63%,m. p.179~181 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.96 (s, 1H, NH-H), 10.40 (s, 1H, NH-H), 10.29 (s, 1H, NH-H), 7.51~7.32 (m, 5H, Ar-H, furan-H), 6.29 (s, 1H, furan-H), 6.09 (s, 1H, furan-H), 4.80 (s, 1H, dihydropyridine-H); MS-ESI, m/z: 356.1 [M + Na]+.
5-(3, 4, 5-三甲氧基苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4h):产率92%,m. p.186~187 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.92 (s, 1H, NH-H), 10.32 (s, 1H, NH-H), 10.13 (s, 1H, NH-H), 7.49~7.29 (m, 4H, Ar-H), 6.53 (s, 2H, Ar-H), 4.67 (s, 1H, dihydropyridine-H), 3.70 (s, 6H, Ar-OCH3), 3.59 (s, 3H, Ar-OCH3); MS-ESI, m/z: 456.1 [M + Na]+.
5-(2, 5-二甲氧基苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4i):产率90%,m. p.201~203 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.79 (s, 1H, NH-H), 10.28 (s, 1H, NH-H), 10.09 (s, 1H, NH-H), 7.48~7.22 (m, 4H, Ar-H), 6.84~6.67 (m, 3H, Ar-H), 4.83 (s, 1H, dihydropyridine-H), 3.68 (s, 3H, Ar-OCH3), 3.65 (s, 3H, Ar-OCH3); MS-ESI, m/z: 426.5 [M + Na]+.
5-(3, 4-二甲基苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4j):产率78%,m. p.184~186 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.86 (s, 1H, NH-H), 10.30 (bs, 1H, NH-H), 10.10 (bs, 1H, NH-H), 7.46~7.23 (m, 4H, Ar-H), 6.97~6.90 (m, 3H, Ar-H), 4.56 (s, 1H, dihydropyridine-H), 2.11 (s, 3H,Ar-CH3), 2.10 (s, 3H, Ar-CH3); MS-ESI, m/z: 394.3 [M + Na]+.
5-(3, 4-二氯苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4k):产率77%,m. p.187~190 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.91 (s, 1H, NH-H), 10.39 (bs, 1H, NH-H), 10.28 (bs, 1H, NH-H), 7.51~7.44 (m, 4H, Ar-H), 7.39~7.24 (m, 3H, Ar-H), 5.09 (s, 1H, dihydropyridine-H); MS-ESI, m/z: 434.1 [M + Na]+.
5-(3, 4-亚甲基二氧苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4l):产率91%,m. p.187~190 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.92 (s, 1H, NH-H), 10.30 (bs, 1H, NH-H), 10.14 (bs, 1H, NH-H), 7.79~7.29 (m, 4H, Ar-H), 6.81~6.68 (m, 3H, Ar-H), 5.93 (s, 2H, -OCH2O-), 4.61 (s, 1H, dihydropyridine-H); MS-ESI, m/z: 410.1 [M + Na]+.
5-(3-羟基-4-甲氧基苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4m):产率80%,m. p.182~186 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 10.91 (s, 1H, NH-H), 10.32 (bs, 1H, NH-H), 10.12 (bs, 1H, NH-H), 8.80 (s, 1H, Ar-OH), 7.49~7.27 (m, 4H, Ar-H), 6.76~6.61 (m, 3H, Ar-H), 4.53 (s, 1H, dihydropyridine-H), 3.68 (s, 3H, Ar-OCH3); MS-ESI, m/z: 412.1 [M + Na]+.
5-(4-硝基苯基)-5, 11-二氢-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(4n):产率65%,m. p.189~191 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 11.44 (s, 1H, NH-H), 10.61 (s, 1H, NH-H), 10.42 (s, 1H, NH-H), 8.28~7.77 (m, 4H, Ar-H), 7.67~7.34 (m, 4H, Ar-H), 5.38 (s, 1H, dihydropyridine-H); MS-ESI, m/z: 411.3 [M + Na]+.
1.2.2 化合物5a~5n的合成参考文献[9]方法合成。在50 mL圆底烧瓶中,加入6 mLN, N-二甲基甲酰胺(DMF) 和化合物4a~4n (0.8 mmol),加热搅拌至80 ℃后加入氯醌(o-chloranil) (0.8 mmol),升温至回流并持续搅拌4 min;加入1 mL水后再加热1 min,用薄层层析[TLC,展开剂为V (三氯甲烷):V (甲醇)=20:1]监测至原料反应完全。将反应液冷却至室温,搅拌10 min,有黄色固体析出。固体的纯化方法同1.2.1节。
5-(3-氟-4-甲氧基苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5a):产率88%,m. p.201~203 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.21 (s, 1H, NH-H), 11.32 (s, 1H, NH-H), 7.85-7.61 (m, 4H, Ar-H), 7.19~7.04 (m, 3H, Ar-H), 3.92 (s, 3H, Ar-OCH3); MS-ESI, m/z: 412.2 [M + Na]+.
5-苯基-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5b):产率92%,m. p.183~186 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.21 (s, 1H, NH-H), 11.28 (s, 1H, NH-H), 7.87~7.59 (m, 4H, Ar-H), 7.42~7.24 (m, 5H, Ar-H); MS-ESI, m/z: 364.4 [M + Na]+.
5-(4-甲氧基苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5c):产率85%,m. p.188~191 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.18 (s, 1H, NH-H), 11.28 (s, 1H, NH-H), 7.85~7.59 (m, 4H, Ar-H), 7.24~7.20 (m, 2H, Ar-H), 6.94~6.91 (m, 2H, Ar-H), 3.83 (s, 3H, Ar-OCH3); MS-ESI, m/z: 394.2 [M + Na]+.
5-(2, 4-二硝基苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5d):产率84%,m. p.177~179 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.21 (s, 1H, NH-H), 11.28 (s, 1H, NH-H), 7.86~7.58 (m, 4H, Ar-H), 7.40~7.34 (m, 3H, Ar-H); MS-ESI, m/z: 454.1 [M + Na]+.
5-(4-氯苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5e):产率70%,m. p.187~190 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.25 (s, 1H, NH-H), 11.34 (s, 1H, NH-H), 7.86~7.59 (m, 4H, Ar-H), 7.45~7.28 (m, 4H, Ar-H); MS-ESI, m/z: 398.2 [M + Na]+.
5-(3-硝基-4-甲氧基苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5f):产率87%,m. p.185~187 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.27 (s, 1H, NH-H), 11.39 (s, 1H, NH-H), 7.89~7.61 (m, 5H, Ar-H), 7.40~7.37 (m, 2H, Ar-H), 4.01 (s, 3H, Ar-OCH3); MS-ESI, m/z: 439.1 [M + Na]+.
5-(2-呋喃基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5g):产率82%,m. p.183~185 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.24 (s, 1H, NH-H), 11.40 (s, 1H, NH-H), 7.85~7.75 (m, 3H, Ar-H), 7.70~7.63 (m, 2H, Ar-H, furan-H), 6.85 (d, 1H, furan-H, J=4 Hz), 6.67 (s, 1H, furan-H); MS-ESI, m/z: 357.1 [M + Na]+.
5-(3, 4, 5-三甲氧基苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5h):产率93%,m. p.196~198 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.19 (s, 1H, NH-H), 11.29 (s, 1H, NH-H), 7.86~7.61 (m, 4H, Ar-H), 6.60 (s, 2H, Ar-H), 3.75 (s, 3H, Ar-OCH3), 3.71 (s, 6H, Ar-OCH3); MS-ESI, m/z: 454.1 [M + Na]+.
5-(2, 5-二甲氧基苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5i):产率90%,m. p.185~189 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.22 (s, 1H, NH-H), 11.31 (s, 1H, NH-H), 7.86~7.62 (m, 4H, Ar-H), 6.94~6.70 (m, 3H, Ar-H), 3.69 (s, 3H, Ar-OCH3), 3.57 (s, 3H, Ar-OCH3); MS-ESI, m/z: 424.4 [M + Na]+.
5-(3, 4-二甲基苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5j):产率94%,m. p.177~179 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.20 (s, 1H, NH-H), 11.27 (s, 1H, NH-H), 7.85~7.58 (m, 4H, Ar-H), 7.14~6.96 (m, 3H, Ar-H), 2.30 (s, 3H, Ar-CH3), 2.23 (s, 3H, Ar-CH3); MS-ESI, m/z: 392.2 [M + Na]+.
5-(3, 4-二氯苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5k):产率70%,m. p.188~190 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.40 (s, 1H, NH-H), 11.48 (s, 1H, NH-H), 7.89~7.78 (m, 4H, Ar-H), 7.69~7.30 (m, 3H, Ar-H); MS-ESI, m/z: 432.2 [M + Na]+.
5-(3, 4-亚甲基二氧苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5l):产率90%,m. p.181~183 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.21 (s, 1H, NH-H), 11.31 (s, 1H, NH-H), 7.85~7.61 (m, 4H, Ar-H), 6.92~6.73 (m, 3H, Ar-H), 6.09 (s, 2H, -OCH2O-); MS-ESI, m/z: 408.2 [M + Na]+.
5-(3-羟基-4-甲氧基苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5m):产率78%,m. p.182~185 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.14 (s, 1H, NH-H), 11.24 (s, 1H, NH-H), 8.87 (s, 1H, Ar-OH), 7.81~7.58 (m, 4H, Ar-H), 6.88~6.61 (m, 3H, Ar-H), 3.81 (s, 3H, Ar-OCH3); MS-ESI, m/z: 410.3 [M + Na]+.
5-(4-硝基苯基)-1H-茚并[2′, 1′:5, 6]吡啶[2, 3-d]嘧啶-2, 4, 6 (3H)-三酮(5n):产率85%,m. p.167~169 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 12.36 (s, 1H, NH-H), 11.44 (s, 1H, NH-H), 8.27 (d, 2H, Ar-H, J=8.8Hz), 7.88~7.56 (m, 6H, Ar-H), 5.38 (s, 1H, dihydropyridine-H); MS-ESI, m/z: 409.4 [M + Na]+.
1.2.3 化合物7a~7f的合成参考文献[9]方法合成。在50 mL圆底烧瓶中,加入4 mL冰醋酸和2 mL无水乙醇后,再分别加入芳香醛(1,0.8 mmol)、3, 4-亚甲二氧基苯胺(6,0.76 mmol) 和1, 3-茚满二酮(3,0.8 mmol),加热至120 ℃,持续搅拌反应2 h,直至不再有固体产物析出时止。冷却至室温,过滤,固体依次用无水乙醇(10 mL × 3) 和乙醚(3 mL × 3) 洗涤,所得粗产物通过硅胶柱层析[洗脱剂为V (三氯甲烷):V (甲醇)=50:1]纯化得到目标化合物7a~7f。
11-苯基-10H-[1, 3]间二氧杂环戊烯[4, 5-g]茚并[1, 2-b]喹啉-10-酮(7a):产率69%,m.p. 166~169 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 7.99 (d, 1H, Ar-H, J=8.0 Hz), 7.78~7.74 (m, 2H, Ar-H), 7.65~7.57 (m, 5H, Ar-H), 7.44 (s, 2H, Ar-H), 6.78 (s, 1H, Ar-H), 6.25 (s, 2H, -OCH2O-); MS-ESI, m/z: 374.1 [M + Na]+.
11-(4-甲氧基苯基)-10H-[1, 3]间二氧杂环戊烯[4, 5-g]茚并[1, 2-b]喹啉-10-酮(7b):产率30%,m.p.187~190 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 7.97 (d, 1H, Ar-H, J=8.0 Hz), 7.77~7.73 (m, 1H, Ar-H), 7.66~7.62 (m, 3H, Ar-H), 7.58~7.54 (m, 2H, Ar-H), 7.50~7.47 (m, 2H, Ar-H), 6.79 (s, 1H, Ar-H), 6.25 (s, 2H, -OCH2O-); MS-ESI, m/z: 408.2 [M + Na]+.
11-(3-氟-4-甲氧基苯基)-10H-[1, 3]间二氧杂环戊烯[4, 5-g]茚并[1, 2-b]喹啉-10-酮(7c):产率37%,m.p.197~199 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 7.94 (d, 1H, Ar-H, J=8.0 Hz), 7.77 (d, 1H, Ar-H, J=8.0 Hz), 7.65~7.60 (m, 2H, Ar-H), 7.40~7.36 (m, 2H, Ar-H), 7.13~7.09 (m, 2H, Ar-H), 7.04~7.02 (m, 1H, Ar-H), 6.85 (s, 1H, Ar-H), 6.23 (s, 2H, -OCH2O-), 3.88 (s, 3H, Ar-OCH3); MS-ESI, m/z: 404.1 [M + Na]+.
11-(4-氯苯基)-10H-[1, 3]间二氧杂环戊烯[4, 5-g]茚并[1, 2-b]喹啉-10-酮(7d):产率39%,m.p.199~202 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 7.96 (d, 1H, Ar-H, J=8.0 Hz), 7.76~7.71 (m, 1H, Ar-H), 7.63~7.51 (m, 3H, Ar-H), 7.16~7.08 (m, 2H, Ar-H), 6.84 (d, 1H, Ar-H, J=4.0 Hz), 6.69 (s, 1H, Ar-H), 6.24 (s, 2H, -OCH2O-), 3.74 (s, 3H, Ar-OCH3), 3.58 (s, 3H, Ar-OCH3); MS-ESI, m/z: 434.1 [M + Na]+.
11-(2, 5-二甲氧基苯基)-10H-[1, 3]间二氧杂环戊烯[4, 5-g]茚并[1, 2-b]喹啉-10-酮(7e):产率33%,m.p.196~198 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 7.96 (d, 1H, Ar-H, J=8.0 Hz), 7.74~7.71 (m, 1H, Ar-H), 7.62~7.52 (m, 3H, Ar-H), 7.39~7.32 (m, 2H, Ar-H), 7.22 (d, 1H, Ar-H, J=4.0 Hz), 6.88 (s, 1H, Ar-H), 6.25 (s, 2H, -OCH2O-), 3.97 (s, 3H, Ar-OCH3); MS-ESI, m/z: 422.1 [M + Na]+.
11-(4-(甲硫基) 苯基)-10H-[1, 3]间二氧杂环戊烯[4, 5-g]茚并[1, 2-b]喹啉-10-酮(7f):产率32%,m.p.189~190 ℃; 1H NMR (DMSO-d6, 400 MHz), δ: 7.97 (d, 1H, Ar-H, J=8.0 Hz), 7.77 (d, 1H, Ar-H, J=8.0 Hz), 7.67~7.62 (m, 2H, Ar-H), 7.59~7.55 (m, 2H, Ar-H), 7.40 (d, 2H, Ar-H, J=8.0 Hz), 7.34 (s, 1H, Ar-H), 6.85 (s, 1H, Ar-H), 6.25 (s, 2H, -OCH2O-), 2.56 (s, 3H, Ar-SCH3); MS-ESI, m/z: 420.1 [M + Na]+.
1.3 杀虫活性测定 1.3.1 供试昆虫朱砂叶螨Tetranychus cinnabarinus,采自甘肃省农业科学院田间黄豆苗。
1.3.2 毒力测试方法参照FAO (联合国粮农组织) 推荐的玻片浸渍法[10],并加以改进。将目标化合物用水稀释成500、250、100、50和10 mg/L系列质量浓度药液。以浸渍清水为空白对照,以喜树碱为药剂对照。挑选大小一致、体色鲜艳、行动活泼的雌成螨,将其背部粘在双面胶玻璃片一端,每片3行,每行10头。在温度(25±1) ℃、相对湿度85%左右的生化培养箱中放置4 h后,用双目镜观察,剔除死亡或不活泼个体。将带螨玻片的一端浸入溶液中,轻轻摇动5 s后取出,迅速吸干螨体及其周围多余药液。置于上述生化培养箱中,24 h后取出置于显微镜下,用柔软毛笔挑动螨足,螨足不动的记为死亡数,取3次平行实验测试值均值。采用SPSS (13.0版本) 分析计算50%致死浓度LC50。
2 结果与分析毒力测试结果(表 1) 表明:所有目标化合物对朱砂叶螨均表现出不同程度的毒杀活性,其LC50值在25.93~112.0 mg/L之间,其中化合物4l (LC50 25.93 mg/L)、7c (LC50 30.67 mg/L) 和7d (LC50 28.16 mg/L) 的杀螨活性与喜树碱相当。
|
|
表 1 目标化合物4a~4n、5a~5n和7a~7f对朱砂叶螨的毒杀活性(24 h) Table 1 Insecticidal activity of target compounds 4a-4n, 5a-5n and 7a-7f againstTeranychus reinnabarinus (24 h) |
从表 1数据还可看出:4系列化合物中4l活性较好,且产率较高;而4进一步反应生成的5系列化合物活性则不理想;虽然7系列化合物产率较低,但活性整体高于4和5。综合来看,化合物4l和7系列化合物可以作为先导结构进一步优化。
| [1] | THOMAS C J, RAHIER N J, HECHT S M. Camptothecin:current perspectives[J]. Bioorg Med Chem, 2004, 12(7): 1585–1604. doi:10.1016/j.bmc.2003.11.036 |
| [2] | LIU Y Q, LI W Q, MORRIS-NATCHKE S L, et al. Perspectives on biologically active camptothecin derivatives[J]. Med Res Rev, 2015, 35(4): 753–789. doi:10.1002/med.2015.35.issue-4 |
| [3] | YANG L, ZHANG Z J, LIU Y Q, et al. Evaluation of insecticidal activity of camptothecin analogs against Brontispa longissima[J]. Med Chem Res, 2014, 23(2): 980–986. doi:10.1007/s00044-013-0703-4 |
| [4] | LI W Q, LIU Y Q, ZHAO Y L, et al. Antifeedant activity of camptothecin and its semisynthetic derivatives against Spodoptera litura (Fabricius) (Lepidoptera:Noctuidae) larvae[J]. Curr Bioact Compd, 2012, 8(3): 291–295. doi:10.2174/157340712802762438 |
| [5] | LIU Y Q, DAI W, TIAN J, et al. Synthesis and insecticidal activities of novel spin-labeled derivatives of camptothecin[J]. Heteroatom Chem, 2011, 22(6): 687–691. doi:10.1002/hc.v22.6 |
| [6] | WU D, ZHANG S Y, LIU Y Q, et al. Synthesis, biological activities, and quantitative structure-activity relationship (QSAR) study of novel camptothecin analogues[J]. Molecules, 2015, 20(5): 8634–8653. doi:10.3390/molecules20058634 |
| [7] |
李忠榜, 陈振, 陈安良, 等. 7-(1-酰基哌嗪-4-基) 甲基喜树碱衍生物的合成及杀虫活性[J]. 农药学学报, 2015, 17(2): 136–142.
LI Z B, CHEN Z, CHEN A L, et al. Synthesis and insecticidal activity of 7-(1-carbonyl-piperazin-4-yl) methyl-camptothecin derivatives[J]. Chin J Pestic Sci, 2015, 17(2): 136–142. |
| [8] | SONG Z L, WANG M J, LI L L, et al. Design, synthesis, cytotoxic activity and molecular docking studies of new 20(S)-sulfonylamidine camptothecin derivatives[J]. Eur J Med Chem, 2016, 115: 109–120. doi:10.1016/j.ejmech.2016.02.070 |
| [9] | EVDOKIMOV N M, VAN SLAMBROUCK S, HEFFETER P, et al. Structural simplification of bioactive natural products with multicomponent synthesis. 3. Fused uracil-containing heterocycles as novel topoisomerase-targeting agents[J]. J Med Chem, 2011, 54(7): 2012–2021. doi:10.1021/jm1009428 |
| [10] | BUSVINE J R. FAO plant protection paper 21. "Recommended methods for measurement of pest resistance to pesticide"[M]. Rome: FAO, 1980: 49-54. |
 2017, Vol. 19
2017, Vol. 19


