文章信息
- 刘海艳, 杨丽洁, 丁艳锋, 李刚华, 王绍华, 刘正辉, 唐设, 刘仁梅, 蒋卫红
- LIU Haiyan, YANG Lijie, DING Yanfeng, LI Ganghua, WANG Shaohua, LIU Zhenghui, TANG She, LIU Renmei, JIANG Weihong
- NO对水稻孕穗期干旱胁迫下叶片光合及产量的影响
- Effects of nitric oxide on photosynthesis and yield of rice under drought stress at booting stage
- 南京农业大学学报, 2017, 40(2): 195-202
- Journal of Nanjing Agricultural University, 2017, 40(2): 195-202.
- http://dx.doi.org/10.7685/jnau.201605012
-
文章历史
- 收稿日期: 2016-05-10
2. 金湖县农业局, 江苏 淮安 223000
2. Jinhu County Agriculture Bureau, Huai'an 223000, China
干旱缺水是制约中国农业稳定发展和粮食安全的主要因素[1]。长江下游是中国主要的水稻种植区域之一, 8月盛夏持续性高温干旱频发。此时正是水稻拔节孕穗期, 也是水稻对水分胁迫最敏感的时期[2]。孕穗期干旱往往造成穗粒数减少、空秕粒率上升、干物质积累和产量下降[3]。有研究显示, 干旱下非气孔限制引起的光合速率下降, 是导致黑小麦和苋菜的产量显著降低的主要原因[4]。
叶绿素荧光动力学参数作为一种快速、无损伤探测和量化光合机构, 尤其是PSⅡ活性受伤害的诊断方式, 可提供光合机构的结构和功能相关的信息。干旱胁迫引起叶片气孔关闭, 胞间CO2浓度减少, 或引起一些非气孔限制因素的变化, 例如RuBP羧化酶的活性降低, 从而减少光合作用CO2同化[5], 以及光系统Ⅱ的结构和捕光复合体的损伤[6]等。干旱胁迫引起叶绿体中激发能过剩, 造成活性氧 (ROS) 的积累[7], 使细胞内氧化还原平衡遭到破坏, 引起氧化胁迫, 最终导致膜脂过氧化, 酶失活、DNA损伤[8]以及光合机构的氧化伤害[9]。已有相关研究通过外源喷施调节剂缓解逆境对植物光合系统造成的伤害。如外源ABA缓解干旱胁迫下水稻叶片光合速率的下降, 增加抗氧化酶活性, 最终减少产量损失[10]。Si以及脯氨酸可有效缓解干旱对光合机构的损害[11-12]。
一氧化氮 (NO), 既能介导动物细胞众多的生理功能如参与血管调节、神经传递、细胞分裂、程序性细胞死亡等过程[13], 也是植物抗逆反应中的一种重要信号物质。黄瓜、大豆、小麦、烟草在干旱时, 会促进NO的产生[14-17]。外源NO也可以通过增加SOD和CAT的活性缓解氧化伤害, 促进蛋白合成, 提高PEG模拟干旱下小麦幼苗光合速率[18], 以及增强小麦籽粒灌浆期的抗旱性以及保护PSⅡ从而提高复水后PSⅡ的恢复能力[19]。
为探究外源NO是否对孕穗期干旱的水稻叶片光合系统有保护作用以及能否加快复水后PSⅡ的恢复效果, 本研究以‘早玉香粳’为试验材料, 通过人工设置干旱胁迫和外源NO供体SNP叶面喷施预处理, 研究NO对水稻干旱胁迫下光系统Ⅱ的热耗散、细胞内ROS平衡、复水后光合特性的影响, 以及最终产量的变化探究外源NO对水稻孕穗期干旱叶片光合机构的影响。
1 材料与方法 1.1 试验时间和地点试验于2014年在南京农业大学水稻栽培实验室试验基地丹阳宝林农场 (31°54′N, 119°28′E) 进行, 采用遮雨大棚挡雨。
1.2 试验方法以上海市农业生物基因中心提供的常规粳稻‘早玉香粳’为试验材料, 5月28号播种, 6月22号移栽。供试土壤为黄壤土, 含总氮1.11 g·kg-1、总磷0.48 g·kg-1、总钾1.96 g·kg-1, 土壤田间持水量为29.64%。移栽前土壤粉碎、过筛、风干, 混匀后装入桶中, 桶高35 cm, 桶内径34 cm, 每桶装干土18 kg。移栽时选取长势一致的小苗移栽到桶内, 每桶3穴, 每穴两苗。幼苗移栽后定量浇水并定期称质量, 一直标记叶龄培养至倒2叶心叶抽出时。水稻倒2叶抽出, 剑叶与倒2叶的叶枕距为-10~-11 cm时 (花粉母细胞减数分裂期) 开始断水, 胁迫至穗分化结束前2~3 d (减数分裂末期), 共计10 d。土壤水分控制为田间持水量的 (75±5)%, 外源喷施NO供体SNP, 试验设4个处理:对照为正常水分管理+喷施清水 (用WT表示), 正常水分管理+喷施100 μmol·L-1硝普钠 (SNP)(W100), 干旱胁迫组为干旱胁迫+喷施清水 (DS), 干旱胁迫+喷施100 μmol·L-1 SNP (D100)。每个处理3次重复、15桶, 共60桶。SNP为Sigma公司分装进口, 避光保存, 喷施浓度为100 μmol·L-1, 喷施标准是每株定时喷10 s。
在喷施SNP后1、4、7、10 d取样, 均取心叶下3片完全展开叶片, 剑叶完全抽出后取上3叶。液氮速冻30 s保存于-40 ℃冰箱, 用于各生理指标测定。第10天取样之后解除干旱胁迫, 复水, 并在复水后第3天即处理13 d测定光合及荧光参数。
1.3 测定项目与方法 1.3.1 产量成熟期每处理留3盆测产, 考察每穴穗数、穗粒数、结实率、千粒质量, 计算单盆产量。各处理均3次重复。
1.3.2 光合作用参数与叶绿素荧光动力学参数在处理后1、4、7、10 d及复水后第3天的晴天06:00—11:00和15:00—17:00, 用美国LICOR生产的LI-6400便携式光合作用测定仪测定叶绿素荧光动力学参数和光合作用参数。测定内容包括叶片净光合速率 (Pn)、气孔导度 (Gs)、蒸腾速率 (Tr)、胞间CO2浓度 (Ci)、最小荧光 (Fo)、PSⅡ最大光化学效率 (Fv/Fm)、光化学淬灭 (qP)、非光化学淬灭 (NPQ), 每个处理3个重复, 每个重复测2株水稻叶片。
1.3.3 POD、SOD、CAT活性与MDA含量测定SOD、POD酶活性测定参考李合生[20]的方法, CAT采用高锰酸钾滴定法[21]测定, MDA含量采用TCA-TBA比色法[22]测定。
1.3.4 H2O2含量与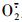
过氧化氢含量测定采用南京建成生物公司提供的试剂盒方法, 羟胺法测定超氧阴离子的产生速率[23]。
1.3.5 数据处理与分析试验结果以测定3次重复的平均值表示, 采用Microsoft Excel 2007进行整理作图, 用SPSS 19.0进行方差分析。
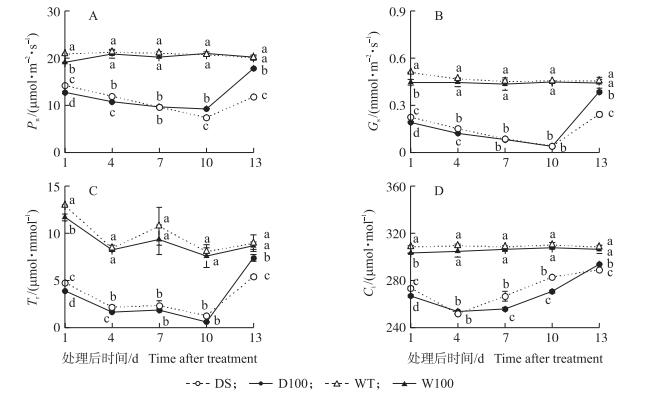
2 结果与分析 2.1 NO对水稻孕穗期干旱胁迫下叶片光合作用的影响干旱胁迫显著降低了水稻叶片净光合速率 (Pn)。处理10 d后, DS处理较WT下降了64.3%(图 1-A), 而外源NO处理10 d后D100较WT的Pn仅下降了55.4%, 显著高于DS处理; 复水后第3天即处理13 d时, D100和DS处理的Pn较WT分别恢复了93.2%和59.7%。正常水分条件下, W100处理对Pn并无显著影响。
|
图 1 NO对水稻孕穗期干旱胁迫下叶片净光合速率 (Pn)(A)、气孔导度 (Gs)(B)、蒸腾速率 (Tr)(C) 和胞间CO2浓度 (Ci)(D) 的影响 Figure 1 Effect of NO on net CO2 assimilation rate (Pn)(A), stomatal conductance (Gs)(B), transpiration rate (Tr)(C) and intercellular CO2 concentration (Ci)(D) in rice leaves under drought stress at booting stage 1) DS:干旱胁迫+叶面喷施清水; D100:干旱胁迫+叶面喷施100 μmol·L-1 SNP (硝普钠); WT:正常水分管理+叶面喷施清水; W100:正常水分管理+叶面喷施100 μmol·L-1 SNP; 2) 不同小写字母表示在5%水平上差异显著。 1) DS:Drought stress and water sprayed on leaves; D100:Drought stress and 100 μmol·L-1 SNP (sodium nitroprusside) sprayed on leaves; WT:Irrigation and water sprayed on leaves; W100:Irrigation and 100 μmol·L-1 SNP sprayed on leaves. 2) Different letters are significantly different at 5% probability level. The same as below. |
气孔导度 (Gs) 是对干旱胁迫最敏感的光合作用参数之一, 干旱胁迫10 d后, DS处理的Gs降低92.7%, 外源NO处理D100的Gs降低87.02%;复水后第3天 (处理后13 d) D100处理基本恢复至正常水平 (图 1-B), 而DS处理仅恢复到正常水平的53.3%。正常水分条件下, 外源喷施NO的W100处理的Gs在处理后第1天明显下降, 之后逐渐回升, 与WT基本无差异。
Tr变化表现出与Gs相似的规律, 干旱条件下Tr显著低于正常水分处理, 外源NO处理D100和W100的Tr在短期内均有所下降。复水后第3天D100处理恢复较快 (图 1-C), 显著高于DS处理。
干旱条件下胞间CO2浓度 (Ci) 显著低于正常水分条件下, 并随着干旱的持续呈先下降后上升的趋势, D100处理减缓了干旱4 d后胞间CO2浓度的上升。复水后胞间CO2浓度均呈上升趋势 (图 1-D)。
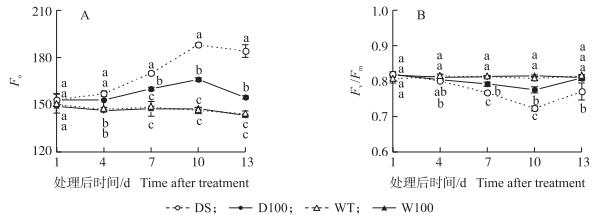
2.2 NO对水稻孕穗期干旱胁迫下叶片Fo和Fv/Fm的影响干旱条件下Fo随着干旱的持续明显增加, 干旱后10 d, DS较WT增加了28.8%, 差异达显著水平; 而D100处理明显缓解了干旱条件下Fo的增加, 处理后7 d与DS处理间差异达到显著水平 (图 2-A)。干旱降低了Fv/Fm, DS在处理后4 d与WT处理差异达显著水平, 之后差异逐渐加大, 干旱10 d, DS较WT下降了9.9%, 复水后有所回升, 但仍显著低于WT处理; 而D100处理缓解了干旱条件下Fv/Fm的降低 (图 2-B), 复水后恢复较快, 第3天就能恢复到正常水平。
|
图 2 NO对水稻孕穗期干旱胁迫下叶片最小荧光 (Fo)(A) 和最大光化学效率 (Fv/Fm)(B) 的影响 Figure 2 Effect of NO on minimal fluorescence level (Fo)(A) and maximum photochemical efficiency ofPSⅡ(Fv/Fm)(B) in rice leaves under drought stress at booting stage |
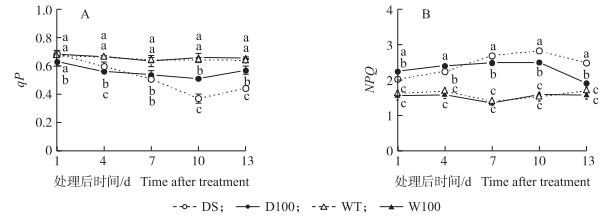
干旱条件下的qP低于正常水分处理, 随着干旱时间的加长, 差异逐渐加大, 处理4 d后就达到显著水平, 10 d后DS较WT下降了42.5%。SNP处理显著缓解了干旱条件下qP的下降, D100较WT仅下降了20.5%, 但NO对正常水分下的qP并无显著影响 (图 3-A)。干旱处理显著增加了NPQ, 干旱10 d时DS处理比WT处理上升了86.7%, 而NO处理在干旱前期进一步增加了干旱条件下的NPQ, 对正常水分条件下的NPQ无显著影响 (图 3-B)。复水后第3天, D100处理的qP和NPQ恢复较快。
|
图 3 NO对水稻孕穗期干旱胁迫下叶片光化学淬灭 (qP)(A) 和非光化学淬灭 (NPQ)(B) 的影响 Figure 3 Effect of NO on photochemical fluorescence quenching efficiency (qP)(A) and non-photochemicalquenching (NPQ)(B) in rice leaves under drought stress at booting stage |
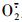
从图 4可见:干旱条件下H2O2含量和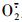
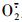
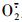
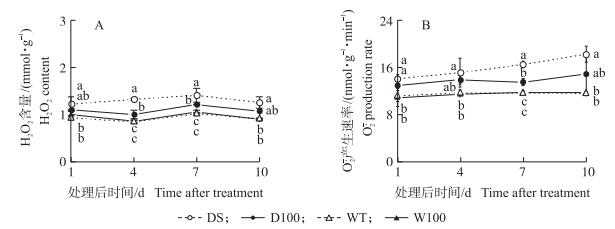
|
图 4 NO对水稻孕穗期干旱胁迫下叶片H2O2含量 (A) 和  |
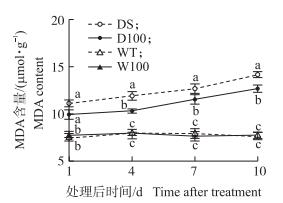
图 5显示:干旱胁迫增加了MDA的积累, 并随着干旱胁迫时间的延长呈增加趋势, 干旱10 d后DS较WT增加了85.56%, 而D100处理较DS显著降低了MDA的含量。正常水分条件下, 外源NO对MDA含量影响不显著。
|
图 5 NO对水稻孕穗期干旱胁迫下叶片MDA含量的影响 Figure 5 Effect of NO on MDA content in rice leafunder drought stress at booting stage |
干旱胁迫增加了POD、CAT、SOD的活性, 但影响程度不同 (图 6)。干旱条件下, POD活性随着干旱时间的持续急剧增加; CAT活性则呈现先增加后下降的趋势; SOD活性随时间的变化较小。在干旱10 d时, POD、CAT和SOD活性DS处理较WT处理分别增加了107.89%、8.09%、16.46%。NO处理后, 进一步增加了干旱条件下POD、CAT、SOD的活性, 但对正常水分条件下的POD、CAT、SOD活性并无显著影响。
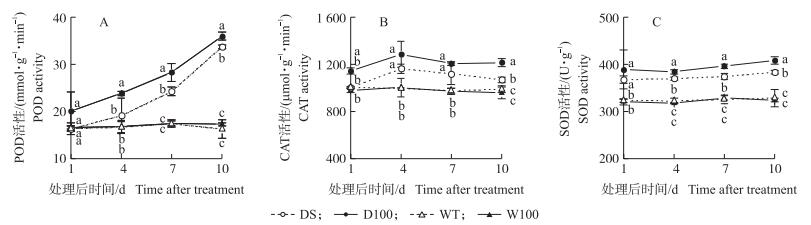
|
图 6 NO对水稻孕穗期干旱胁迫下叶片POD (A)、CAT (B) 和SOD (C) 活性的影响 Figure 6 Effect of NO on POD (A), CAT (B) and SOD (C) activities in rice leavesunder drought stress at booting stage |
与WT相比, 孕穗期DS处理使得水稻的每盆产量降低了36.5%(图 7), 而NO处理D100每盆产量较DS显著提高了21.0%, 显著降低了孕穗期干旱胁迫对水稻产量造成的损失。正常水分处理下, NO处理 (W100) 对水稻产量并没有显著影响。
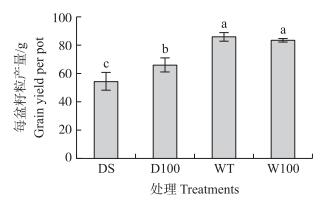
|
图 7 NO对水稻孕穗期干旱胁迫下产量的影响 Figure 7 Effect of NO on grain yield of rice underdrought stress at booting stage |
干旱通过限制气孔开度, 降低胞间CO2浓度 (Ci)[24], 或通过改变光合代谢[25]影响光合作用。胞间CO2浓度可用来区分光合速率下降的主导因素[26]。本试验中, 干旱处理前期水稻叶片净光合速率Pn显著下降, Gs、Tr、Ci与Pn趋势相一致, 说明前期Pn的降低主要由于气孔开度限制, 而在干旱处理后期Ci呈上升趋势, 因此干旱后期水稻Pn的下降是以非气孔开度限制因素为主导作用, 这可能是由于RuBP酶活性降低, PSⅡ结构的改变或PSⅡ受到损伤等引起的。叶绿素荧光动力学参数可提供植物在逆境下PSⅡ活性以及光合代谢变化的相关信息[27]。本研究结果显示, 干旱下Fo显著上升, Fv/Fm显著下降, 表明光合机构以及细胞膜系统受到了损伤, 光化学效率降低, PSⅡ反应中心部分关闭, 导致过剩光能的积累和PSⅡ反应中心失活, 这与已有相关研究结果一致[28-29]。本研究中, 外源NO在干旱前期由于进一步降低了气孔导度, 引起Pn低于未喷施NO的处理; 正常水分条件下, 外源NO也能降低气孔导度, 这与Farooq等[30]的研究结果一致。随着干旱的加重, 外源喷施NO缓解Pn下降, 胞间CO2浓度上升转折点也晚于干旱未喷施NO的处理, 同时缓解Fo的增加以及Fv/Fm的降低, 说明NO延缓了干旱胁迫对水稻叶片光合机构的伤害, 这可能是由于NO通过改善叶黄素循环的方式提高了过剩光能的热耗散[31]。
在干旱条件下, 植物通过捕光复合体上叶黄素循环[32]和黄体素循环[33]的热耗散方式, 消耗过剩的光能, 起到了一种光保护作用, 而这一机制与光化学反应具有竞争作用, 可导致光合作用的变化。非荧光淬灭NPQ反映热耗散能力, 光化学淬灭qP反映植物光合活性高低。本研究中, 干旱条件下NPQ显著增加, qP显著下降, 说明干旱条件下水稻叶片PSⅡ热耗散增加, 而在干旱后期NPQ的显著上升, 可能由于重度胁迫下光合机构严重损伤导致。外源喷施NO, 在干旱前期进一步增加了NPQ, 减少了qP, 说明NO提高了水稻干旱下叶片PSⅡ过剩激发能的热耗散能力增加, 这是对光合机构的一种保护机制[34], 延缓了干旱胁迫对光合机构的损伤。外源NO降低大豆叶片的qP, 增加NPQ[35], 这与本研究结果一致。当植物受到干旱胁迫时, 光抑制和光呼吸引起ROS的产生[36], 过量的ROS会产生氧化胁迫, 引起膜脂过氧化, 造成细胞膜损伤以及酶失活。而抗氧化酶系统可有效清除ROS的积累, 在不同作物中具有不同的表现, 有些酶活性上升, 而有些酶活性被抑制, 这可能由于植物体内不同的ROS水平对应不同的响应[37]。丙二醛 (MDA) 含量是衡量植物脂质过氧化的重要指标[38]。本研究结果显示, H2O2含量和
植物在受到中度胁迫后恢复很快, 受到严重干旱胁迫时, 只恢复最大光合速率的40%~60%, 且随着时间延长持续恢复[39]。Kirschbaum[40]认为重度干旱胁迫后, 光合机构的恢复主要分两个步骤:第一步气孔张开; 第二步光合蛋白的从头合成。本研究结果显示, 复水后第3天, 气孔导度上升, 叶绿素荧光动力学参数也都有所恢复, Pn达到了最大光合速率的56%;外源SNP处理, 复水后第3天恢复了最大光合速率的81%, 气孔导度以及叶绿素荧光动力学参数的恢复程度也显著高于未喷施NO的处理, 但对正常水分条件下叶绿素荧光动力学参数无显著影响。由此认为, 可能由于外源NO减轻对细胞膜以及PSⅡ的伤害, 使得复水后光合作用恢复较快。有研究显示, 小麦复水后psbA转录水平迅速上升, 促进D1蛋白合成[19], D1蛋白是PSⅡ中重要的功能蛋白, 参与光化学反应[41]。本研究中PSⅡ在复水后恢复较快, 是否与D1蛋白合成关键基因psbA有关, 其机制有待进一步研究。
本研究中, 干旱处理下产量显著降低, 而外源NO处理缓解了产量的降低, 在长期干旱下, 外源NO处理的叶片光合速率显著高于未处理NO的, 由此认为, 产量的减少与光合作用下降相关, 而叶绿素荧光动力学参数也可用来评估作物生产力, 这与Jedmowski等[42]的研究一致。已有相关研究显示, 对于作物的抗旱能力, 复水后光合作用的恢复是抵抗干旱胁迫以及减少产量损失的主要参考指标[43]。本研究中, 外源NO处理下水稻叶片复水后光合作用恢复较快, 产量也显著高于干旱下未喷施NO的。
孕穗期干旱显著降低了水稻叶片光合速率, 增加了细胞内ROS含量, 并对光系统Ⅱ造成了一定的损伤。外源喷施NO显著缓解了干旱下PSⅡ光化学反应的失调、ROS的积累以及细胞膜的损伤, 并促进复水后光合作用的恢复, 从而减少了产量的损失。
| [1] |
王春乙, 娄秀荣, 王建林. 中国农业气象灾害对作物产量的影响[J].
自然灾害学报, 2007, 16(5): 37–43.
Wang C Y, Lou X R, Wang J L. Influence of agricultural meteorological disasters on output of crop in China[J]. Journal of Natural Disasters, 2007, 16(5): 37–43. (in Chinese) |
| [2] |
李成业, 熊昌明, 魏仙君. 中国水稻抗旱研究进展[J].
作物研究, 2006(5): 426–429.
Li C Y, Xiong C M, Wei X J. Progress of China rice drought research[J]. Crop Research, 2006(5): 426–429. (in Chinese) |
| [3] |
张玉屏, 朱德峰, 林贤青, 等. 不同灌溉方式对水稻需水量和生长的影响[J].
灌溉排水学报, 2007, 26(2): 83–85.
Zhang Y P, Zhu D F, Lin X Q, et al. Effects of different irrigation methods on water demand and growth of rice[J]. Journal of Irrigation and Drainage, 2007, 26(2): 83–85. (in Chinese) |
| [4] | Hura T, Hura K, Grzesiak M, et al. Effect of long-term drought stress on leaf gas exchange and fluorescence parameters in C3 and C4 plants[J]. Acta Physiologiae Plantarum, 2007, 29(2): 103–113. DOI: 10.1007/s11738-006-0013-2 |
| [5] | Carmo-Silva A E, Keys A J, Andralojc P J, et al. Rubisco activities, properties, and regulation in three different C4 grasses under drought[J]. Journal of Experimental Botany, 2010, 61(9): 2355–2366. DOI: 10.1093/jxb/erq071 |
| [6] | Hura T, Hura K, Grzesiak M, et al. Effect of long-term drought stress on leaf gas exchange and fluorescence parameters in C3 and C4 plants[J]. Acta Physiologiae Plantarum, 2007, 29(2): 103–113. DOI: 10.1007/s11738-006-0013-2 |
| [7] | Loggini B, Scartazza A, Brugnoli E, et al. Antioxidative defense system, pigment composition, and photosynthetic efficiency in two wheat cultivars subjected to drought[J]. Plant Physiology, 1999, 119(3): 1091–1099. DOI: 10.1104/pp.119.3.1091 |
| [8] | Moldovan L, Moldovan N I. Oxygen free radicals and redox biology of organelles[J]. Histochemistry and Cell Biology, 2004, 122(4): 395–412. DOI: 10.1007/s00418-004-0676-y |
| [9] | Monneveux P, Pastenes C, Reynolds M P. Limitations to photosynthesis under light and heat stress in three high-yielding wheat genotypes[J]. Journal of Plant Physiology, 2003, 160(6): 657–666. DOI: 10.1078/0176-1617-00772 |
| [10] |
郭贵华, 刘海艳, 李刚华, 等. ABA缓解水稻孕穗期干旱胁迫生理特性的分析[J].
中国农业科学, 2014, 47(22): 4380–4391.
Guo G H, Liu H Y, Li G H, et al. The analysis of physiological characteristics about ABA alleviating rice booting stage drought stress[J]. Scientia Agricultura Sinica, 2014, 47(22): 4380–4391. (in Chinese) |
| [11] | Chen W, Yao X, Cai K, et al. Silicon alleviates drought stress of rice plants by improving plant water status, photosynthesis and mineral nutrient absorption[J]. Biological Trace Element Research, 2011, 142(1): 67–76. DOI: 10.1007/s12011-010-8742-x |
| [12] | Farooq M, Wahid A, Lee D J. Exogenously applied polyamines increase drought tolerance of rice by improving leaf water status, photosynthesis and membrane properties[J]. Acta Physiologiae Plantarum, 2009, 31(5): 937–945. DOI: 10.1007/s11738-009-0307-2 |
| [13] | Wink D A, Mitchell J B. Chemical biology of nitric oxide:insights into regulatory, cytotoxic, and cytoprotective mechanisms of nitric oxide[J]. Free Radical Biology and Medicine, 1998, 25(4/5): 434–456. |
| [14] | Arasimowicz-Jelonek M, Floryszak-Wieczorek J, Kubi As' J. Interaction between polyamine and nitric oxide signaling in adaptive responses to drought in cucumber[J]. Journal of Plant Growth Regulation, 2009, 28(2): 177–186. DOI: 10.1007/s00344-009-9086-7 |
| [15] | Leshem Y Y, Haramaty E. The characterization and contrasting effects of the nitric oxide free radical in vegetative stress and senescence of Pisum sativum Linn.Foliage[J]. Journal of Plant Physiology, 1996, 148(3): 258–263. |
| [16] | Gould K S, Lamotte O, Klinguer A, et al. Nitric oxide production in tobacco leaf cells:a generalized stress response?[J]. Plant, Cell and Environment, 2003, 26(11): 1851–1862. DOI: 10.1046/j.1365-3040.2003.01101.x |
| [17] | Kolbert Z, Bartha B, Erdei L. Generation of nitric oxide in roots of Pisum sativum, Triticuma estivum and Petroselinum crispum plants under osmotic and drought stress[J]. Acta Biol Szegediensis, 2005, 49: 13–16. |
| [18] | Tan J, Zhao H, Hong J, et al. Effects of exogenous nitric oxide on photosynthesis, antioxidant capacity and proline accumulation in wheat seedlings subjected to osmotic stress[J]. World J Agric Sci, 2008, 4(3): 307–313. |
| [19] | Wang Y, Suo B, Zhao T, et al. Effect of nitric oxide treatment on antioxidant responses and psbA gene expression in two wheat cultivars during grain filling stage under drought stress and rewatering[J]. Acta Physiologiae Plantarum, 2011, 33(5): 1923–1932. DOI: 10.1007/s11738-011-0740-x |
| [20] |
李合生.
植物生理生化原理与技术[M]. 北京: 高等教育出版社, 2000: 184-185.
Li H S. Plant Physiology and Biochemistry Test Principles and Techniques[M]. Beijing: Higher Education Press, 2000: 184-185. (in Chinese) |
| [21] |
李长宁, SrivastavaM K, 农倩, 等. 水分胁迫下外源ABA提高甘蔗抗旱性的作用机制[J].
作物学报, 2010, 36(5): 863–870.
Li C N, Srivastava M K, Nong Q, et al. Mechanism of tolerance to drought in sugarcane plant enhanced by foliage dressing of abscisic acid under water stress[J]. Acta Agronomica Sinica, 2010, 36(5): 863–870. DOI: 10.3724/SP.J.1006.2010.00863 (in Chinese) |
| [22] |
姚满生, 杨小环, 郭平毅. 脱落酸与水分胁迫下棉花幼苗水分关系及保护酶活性的影响[J].
棉花学报, 2005, 17(3): 141–145.
Yao M S, Yang X H, Guo P Y. Effects of abscisic acid on water relationship and defensive enzymes activities in cotton seedling under water stress[J]. Cotton Science, 2005, 17(3): 141–145. (in Chinese) |
| [23] |
陈银萍, 蘧苗苗, 苏向南, 等. 外源一氧化氮对镉胁迫下紫花苜蓿幼苗活性氧代谢和镉积累的影响[J].
农业环境科学学报, 2015, 34(12): 2261–2271.
Chen Y P, Qu M M, Su X N, et al. Effect of exogenous nitric oxide on active oxygen metabolism and cadmium accumulation in alfalfa seedlings under cadmium stress[J]. Journal of Agro-Environment Science, 2015, 34(12): 2261–2271. (in Chinese) |
| [24] | Flexas J, Bota J, Loreto F, et al. Diffusive and metabolic limitations to photosynthesis under drought and salinity in C3 plants[J]. Plant Biology, 2004, 6(3): 269–279. DOI: 10.1055/s-2004-820867 |
| [25] | Lawlor D W, Cornic G. Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants[J]. Plant, Cell and Environment, 2002, 25(2): 275–294. DOI: 10.1046/j.0016-8025.2001.00814.x |
| [26] |
徐俊增, 彭世彰, 魏征, 等. 节水灌溉水稻叶片胞间CO2浓度及气孔与非气孔限制[J].
农业工程学报, 2010, 26(7): 76–80.
Xu J Z, Peng S Z, Wei Z, et al. Intercellular CO2 concentration and stomatal or non-stomatal limitation of rice under water saving irrigation[J]. Transactions of the CSAE, 2010, 26(7): 76–80. (in Chinese) |
| [27] | Schnabel G, Strittmatter G, Noga G. Changes in photosynthetic electron transport in potato cultivars with different field resistance after infection with Phytophthora infestans[J]. Journal of Phytopathology, 1998, 146(5): 205–210. |
| [28] | Bartošková H, Komenda J, Nauš J. Functional changes of photosystemⅡ in the moss Rhizomnium punctatum (Hedw.) induced by different rates of dark desiccation[J]. Journal of Plant Physiology, 1999, 154(5): 597–604. |
| [29] | Pastenes C, Horton P. Resistance of photosynthesis to high temperature in two bean varieties (Phaseolus vulgaris L.)[J]. Photosynthesis Research, 1999, 62(2): 197–203. |
| [30] | Farooq M, Basra S M A, Wahid A, et al. Exogenously applied nitric oxide enhances the drought tolerance in fine grain aromatic rice (Oryza sativa L.)[J]. Journal of Agronomy and Crop Science, 2009, 195(4): 254–261. DOI: 10.1111/jac.2009.195.issue-4 |
| [31] | Song L, Yue L, Zhao H, et al. Protection effect of nitric oxide on photosynthesis in rice under heat stress[J]. Acta Physiologiae Plantarum, 2013, 35(12): 3323–3333. DOI: 10.1007/s11738-013-1365-z |
| [32] | Demmig-Adams B, Adams W W. The role of xanthophyll cycle carotenoids in the protection of photosynthesis[J]. Trends in Plant Science, 1996, 1(1): 21–26. DOI: 10.1016/S1360-1385(96)80019-7 |
| [33] | García-Plazaola J I, Hernández A, Olano J M, et al. The operation of the lutein epoxide cycle correlates with energy dissipation[J]. Functional Plant Biology, 2003, 30(3): 319–324. DOI: 10.1071/FP02224 |
| [34] | Yang W, Sun Y, Chen S, et al. The effect of exogenously applied nitric oxide on photosynthesis and antioxidant activity in heat stressed chrysanthemum[J]. Biologia Plantarum, 2011, 55(4): 737–740. DOI: 10.1007/s10535-011-0178-4 |
| [35] | Wodala B, Örd g A, Horváth F. The cost and risk of using sodium nitroprusside as a NO donor in chlorophyll fluorescence experiments[J]. Journal of Plant Physiology, 2010, 167(13): 1109–1111. DOI: 10.1016/j.jplph.2010.03.013 |
| [36] | Noctor G, Veljovic-Jovanovic S, Driscoll S, et al. Drought and oxidative load in the leaves of C3 plants:a predominant role for photorespiration?[J]. Annals of Botany, 2002, 89(7): 841–850. DOI: 10.1093/aob/mcf096 |
| [37] | Cruz de Carvalho M H. Drought stress and reactive oxygen species:production, scavenging and signaling[J]. Plant Signaling and Behavior, 2008, 3(3): 156–165. DOI: 10.4161/psb.3.3.5536 |
| [38] | Smirnoff N. The role of active oxygen in the response of plants to water deficit and desiccation[J]. New Phytologist, 1993(1): 27–58. |
| [39] | Kirschbaum M U F. Water stress in Eucalyptus pauciflora:comparison of effects on stomatal conductance with effects on the mesophyll capacity for photosynthesis, and investigation of a possible involvement of photoinhibition[J]. Planta, 1987, 171(4): 466–473. DOI: 10.1007/BF00392293 |
| [40] | Kirschbaum M U F. Recovery of photosynthesis from water stress in Eucalyptus pauciflora:a process in two stages[J]. Plant, Cell and Environment, 1988, 11(8): 685–694. DOI: 10.1111/pce.1988.11.issue-8 |
| [41] | Liu W J, Yuan S, Zhang N H, et al. Effect of water stress on photosystem 2 in two wheat cultivars[J]. Biologia Plantarum, 2006, 50(4): 597–602. DOI: 10.1007/s10535-006-0094-1 |
| [42] | Jedmowski C, Ashoub A, Momtaz O, et al. Impact of drought, heat, and their combination on chlorophyll fluorescence and yield of wild barley (Hordeum spontaneum)[J]. Journal of Botany, 2015, 2015(6): 1–9. |
| [43] | Chaves M M, Flexas J, Pinheiro C. Photosynthesis under drought and salt stress:regulation mechanisms from whole plant to cell[J]. Annals of Botany, 2009, 103(4): 551–560. |




