文章信息
- 于雷, 饶开晴, 董传豪, 妥丽荣, 徐亚欧, 冯卫东, 蒋忠荣, 李平. 2015.
- YU Lei, RAO Kaiqing, DONG Chuanhao, TUO Lirong, XU Ya'ou, FENG Weidong, JIANG Zhongrong, LI Ping. 2015.
- 藏鸡和肉鸡种蛋皮质酮沉积与胚胎皮质酮代谢酶表达的差异分析
- Comparison analysis of corticosterone deposition in fertilized eggs and embryonic expression of corticosterone metabolic enzymes between Tibetan chickens and broiler chickens
- 南京农业大学学报, 38(2): 339-344
- Journal of Nanjing Agricultural University, 38(2): 339-344.
- http://dx.doi.org/10.7685/j.issn.1000-2030.2015.02.024
-
文章历史
- 收稿日期:2014-11-14
2. 甘孜州畜牧业科学研究所, 四川 康定 626000
2. Ganzi Prefectural Livestock Research Institute, Kangding 626000, China
在哺乳动物中,糖皮质激素(glucocorticoids,GC)可参与母体与胎儿之间的信息传递,且在调节动物的生长发育、子代的行为、生存等方面发挥重要作用。鸡蛋内的GC可改变胚胎的营养和内分泌环境,程序化影响机体的生长发育。目前,关于蛋内激素沉积的研究大多集中在野生鸟类上,对家禽的研究甚少。藏鸡是原始饲养状态保存下来的珍贵高原地方鸡种,与其他鸡种相比,有良好的高海拔、高寒、低氧适应性等优势。关于皮质酮在藏鸡蛋内沉积的研究还未见报道。
动物体内的GC水平受一系列GC代谢酶的调控[1]。已有研究表明,GC代谢酶11β-羟基类固醇脱氢酶1(11β-hydroxysteroid dehydrogenase 1,11β-HSD1)、11β-羟基类固醇脱氢酶2(11β-hydroxysteroid dehydrogenase 2,11β-HSD2)和20-羟基类固醇脱氢酶(20-hydroxysteroid dehydrogenase,20-HSD)在禽类的多数组织中表达[2, 3],其中11β-HSD1可激活GC,而11β-HSD2和20-HSD催化GC(皮质醇和皮质酮)迅速失活。哺乳动物发育初期,GC主要是通过胎盘到达胎儿体内。禽类虽然没有像哺乳动物的胎盘结构,却有类似于哺乳动物胎盘功能的结构——卵黄膜和尿囊膜。鸡胚在发育期间,胚胎生长所需的大量成分包括GC等物质也主要是通过卵黄膜和尿囊膜到达鸡胚的各个组织。因此,对禽类而言,卵黄膜和尿囊膜是母体和子代之间物质交换和信息传递的中介。目前,关于GC代谢酶相关基因在禽类卵黄膜、尿囊膜上的研究甚少, 11β-HSD1/2和20 -HSD在藏鸡和肉鸡卵黄膜和尿囊膜上表达比较的研究尚未见报道。
本试验以高原品种藏鸡和低地常氧生长的肉鸡为研究对象,比较了藏鸡与肉鸡种蛋内的皮质酮沉积水平,以及胚胎期卵黄膜和尿囊膜糖皮质激素代谢酶相关基因的表达情况,以分析品种间表型差异与种蛋内皮质酮激素水平和卵黄膜、尿囊膜基因表达之间的关系,为揭示品种间优良性状形成的机制和藏鸡的抗逆特性研究提供依据。 1 材料与方法 1.1 样品采集
藏鸡种蛋购于四川省甘孜州贡嘎山海螺沟畜牧业有限责任公司,肉鸡种蛋购于四川省温江开发区正大种鸡场。分别选取质量相近的两品种种蛋,其中藏鸡种蛋(55.73±0.38)g,肉鸡种蛋(65.19±0.72)g,差异极显著。各选取10枚用于测定蛋黄和蛋清内的皮质酮含量。将收集的其他藏、肉鸡种蛋分别在低氧(甘孜州,海拔2 000 m)和常氧(成都平原,海拔570.50 m)环境下进行孵化。不同时期两地孵化的温、湿度保持一致,入孵24 h为1胚龄。14胚龄时,藏鸡和肉鸡各选取10枚发育正常的鸡胚,采集卵黄膜和尿囊膜用于提取组织总RNA。 1.2 主要仪器和试剂
ABI 7300实时荧光定量PCR仪(美国,ABI公司);凝胶成像系统Gel Doc2000、Model 680型酶标仪(美国,Bio-Rad公司);Cary50紫外可见分光光度计(美国,Varian公司);ETC-811 PCR仪(中国,北京东胜创新生物科技有限公司);KFP352科裕微电脑全自动孵化机(中国,德州市科裕孵化设备有限公司)。
皮质酮放射免疫测定(EIA)试剂盒购自美国Cayman Chemical公司;Sep-Pak Vac 6cc(1 g)C18 Cartridges购于美国Waters Corporation;SYBR Premix Ex TaqTM、RNAiso Reagent、dNTP Mixture及随机引物均购自日本TaKaRa公司;反转录酶(M-MLV)购自Promega公司。 1.3 引物设计及合成
β-actin和目的基因根据GenBank中鸡的相关cDNA序列设计,由苏州金唯智生物科技有限公司合成。引物序列见表 1。
目的基因 Target genes | GenBank登录号 GenBank accession No. | PCR产物长度/bp PCR products size | 引物对序列(5′→3′) Primer pairs sequences |
| β-actin | NM205518 | 300 | F/R:TGCGTGACATCAAGGAGAAG/TGCCAGGGTACATTGTGGTA |
| 11β-HSD1 | XM_41798.2 | 229 | F/R:GGTGGTGAAAGAGGCTGAGAAC/GGAGGCGACTTTACCTGAAACAG |
| 11β-HSD2 | XM_003209680.1 | 229 | F/R:GGTGGTGAAAGAGGCTGAGAACA/GGAGGCGACTTTACCTGAAACAG |
| 20-HSD | NM_001030795.1 | 220 | F/R:CATCCTGAGAAGATAATGTCCAACG/TGCTTTGCAGATCATCAATATCCAG |
蛋黄和蛋清内皮质酮激素的抽提步骤[4, 5]如下:每个种蛋取0.15 g蛋黄(或蛋清),加入600 μL蒸馏水稀释,涡旋30 s后-20 ℃冻存过夜。次日,添加甲醇3 mL,震荡30 min,-20 ℃冻存过夜。第3天,4 ℃,10 000 r · min-1离心10 min。取上清液,加到Sep-Pak C18柱上洗脱收集样品。吸取1 mL洗脱液在氮气流下吹干,再在500 μL EIA测定缓冲液中重悬。按照皮质酮放射免疫测定(EIA)试剂盒说明书测定蛋内皮质酮浓度。试剂盒的灵敏度30 pg · mL-1,批内变异系数5%。皮质酮EIA抗血清与皮质酮的交叉反应概率100%,与其他类固醇激素的交叉反应概率小于0.01%。 1.5 总RNA提取和cDNA合成
卵黄膜和尿囊膜总RNA用RNAiso Reagent提取,用紫外分光光度计测定总RNA浓度和纯度(A260/A280=1.7~2.0),用琼脂糖-甲醛变性凝胶电泳鉴定总RNA质量。若总RNA质量可靠,则用反转录体系对总RNA进行反转录(RT)生成cDNA。cDNA合成体系包括:2 μg总RNA、随机引物(21 μmol · L-1)1 μL、dNTP mix(10 mmol · L-1)2 μL,加无RNase水至10 μL,70 ℃变性5 min,立即放冰上冷却;再加RNA酶抑制剂(40 U · μL-1)0.5 μL,M-MLV 0.5 μL,5×RT Buffer 5 μL,用无RNase水补齐至25 μL,37 ℃反应60 min,95 ℃灭活1 min。 1.6 荧光定量RT-PCR检测基因表达
糖皮质激素代谢酶相关基因( 11β-HSD、20 -HSD)的mRNA表达情况采用Real-Time PCR进行检测。反应体系(10 μL)参照试剂盒说明书,包括Premix Ex Taq(2×)5 μL,上、下游引物(10 μmol · L-1)各0.4 μL,ROX Reference Dye(50×)0.2 μL,cDNA模板1 μL,ddH2O 3 μL。反应程序:95 ℃预变性30 s,95 ℃变性5 s,55 ℃退火30 s,72 ℃延伸30 s,共40个循环;95 ℃ 15 s,60 ℃ 30 s,95 ℃ 15 s作熔解曲线。每个样品3孔重复。选择β-actin作为内参。用2-ΔΔCT方法分析基因相对表达差异量。用待测样品RT产物混合样对反应条件进行优化。 1.7 数据分析
采用SPSS 18.0软件对试验数据进行处理,数据均用x±SE表示,差异显著性检验采用独立样本t测验法。
2 结果与分析
2.1 藏鸡和肉鸡种蛋蛋黄、蛋清内的皮质酮含量比较
如表 2所示:藏鸡种蛋蛋黄和蛋清内的皮质酮含量均低于肉鸡种蛋,且藏鸡蛋蛋黄内皮质酮含量显著低于肉鸡蛋黄内含量(P < 0.01),但两品种种蛋蛋清内皮质酮含量没有显著差异(P>0.05)。蛋黄内皮质酮含量藏鸡比肉鸡低32.6%,蛋清内皮质酮含量藏鸡比肉鸡低9.7%。
| ng · g-1 | ||
| 指标Item | 藏鸡种蛋Tibetan chicken fertilized eggs | 肉鸡种蛋Broiler chicken fertilized eggs |
| 蛋黄内皮质酮含量Corticosterone content of yolk | 1.923±0.087** | 2.853±0.211 |
| 蛋清内皮质酮含量Corticosterone content of albumen | 0.279±0.020 | 0.309±0.018 |
注:* *表示同行数据相比差异极显著(P < 0.01)。* * means significant difference in the same line at 0.01 level.
不同基因均以藏鸡基因表达量做对照分析鸡胚卵黄膜皮质酮代谢酶相关基因,结果(图 1)显示:14胚龄藏鸡与肉鸡鸡胚卵黄膜 11β-HSD1 mRNA表达水平差异不显著(P>0.05);藏鸡 11β-HSD2、20 -HSD mRNA表达量显著或极显著低于肉鸡(P < 0.05或P < 0.01)。
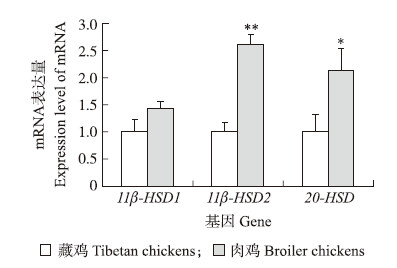 |
图 1 4胚龄藏鸡和肉鸡卵黄膜 11β-HSD1、11β-HSD2、20 -HSD mRNA表达的品种差异 Fig. 1 Breed differences of chicken embryonic 11β-HSD1,11β-HSD2 and 20 -HSD mRNA expression in yolk sac of Tibetan and broiler chickens on embryonic day of 14
*和* *分别表示同一基因不同品种相比差异显著(P < 0.05)或极显著(P < 0.01)。 * and * * indicate significant difference(P < 0.05)or extremely signifi-cant difference(P < 0.01)between breeds for the same gene.The same as follows. |
以藏鸡基因表达量做对照分析鸡胚尿囊膜皮质酮代谢酶相关基因,结果(图 2)显示:14胚龄藏鸡尿囊膜 11β-HSD1 mRNA水平显著显著高于肉鸡(P < 0.05);藏鸡尿囊膜 11β-HSD2、20 -HSD mRNA表达量极显著高于肉鸡(P < 0.01)。
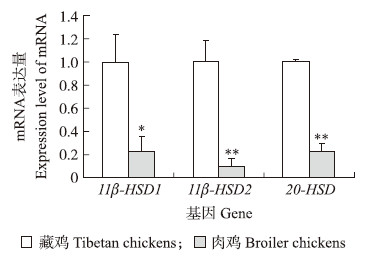 | 图 2 14胚龄藏鸡和肉鸡尿囊膜 11β-HSD1、11β-HSD2和20 -HSD mRNA表达的品种差异 Fig. 2 Breed differences of chicken embryonic 11β-HSD1,11β-HSD2 and 20 -HSD mRNA expression in chorioallantoic membrene(CAM)of Tibetan and broiler chickens on embryonic day of 14 |
以藏鸡卵黄膜 20 -HSD mRNA表达量做对照,计算两品种不同组织皮质酮代谢酶基因的表达。由表 3可见:除了藏鸡卵黄膜 20 -HSD mRNA表达水平显著低于其尿囊膜内水平(P < 0.01),无论藏鸡还是肉鸡,其卵黄膜内的皮质酮代谢酶基因表达量均显著高于尿囊膜内水平(P < 0.01)。
| 基因Genes |
|
|
| 11β-HSD1 | 9.449±2.143 | 0.064±0.016** | 13.591±1.278 | 0.015±0.009** |
| 11β-HSD2 | 6.816±1.227 | 0.183±0.033** | 17.727±1.328 | 0.018±0.012** |
| 20-HSD | 1.000±0.334 | 3.095±0.082** | 2.149±0.392 | 0.703±0.214** |
注: * *表示同一品种不同组织间差异极显著(P<0.01)。* * means extremely significant difference(P<0.01)between tissues for the same breed.
糖皮质激素包括皮质酮和皮质醇,在禽类存在的主要是皮质酮。目前,已在许多野生鸟类如金丝雀[6]、黄腿海鸥[7]、日本鹌鹑[8]、欧洲椋鸟[9]的蛋内检测到皮质酮的存在,在鸡蛋[10]内也证实含有皮质酮激素。本试验中,我们检测到藏、肉鸡种蛋蛋黄内的皮质酮含量有极显著的种间差异,但蛋清内的含量差异不显著。这与Navara等[11]的报道一致,其研究显示白来航和海兰褐鸡蛋黄内的皮质酮含量差异显著,而蛋清内含量差异不显著。他们认为可能由于蛋清沉积时间较短,蛋清内皮质酮浓度只是一个短期的累积,所以导致蛋清内的含量没有显著差异。
另外,本试验藏鸡蛋质量显著低于肉鸡蛋质量。据文献报道,鸡蛋大小不会对种间蛋黄、蛋清内的皮质酮浓度产生显著影响[2],蛋内皮质酮含量只是反映了不同品种间血液内皮质酮含量的差异,或在蛋形成过程,皮质酮在鸡蛋内沉积率的不同[2]。此外,本试验中无论藏鸡还是肉鸡,其蛋黄内的皮质酮含量都高于蛋清内含量,这是由于类固醇激素主要沉积在蛋黄内。蛋形成期间,蛋黄的积累是发生在产蛋前7~12 d[12],而蛋清的积累是在产蛋前大约6 h[13]。鉴于此,蛋黄内皮质酮浓度高于蛋清内浓度具有合理性。蛋内皮质酮沉积水平受饲养条件[14, 15]、基因型[16]、母体营养[17]等因素影响。长期限制应激能显著增加海鸟蛋内的皮质酮沉积水平[18]。肉种鸡的饲养方式不同于藏鸡的散养,本试验种蛋内皮质酮沉积水平的品种间差异,是否与肉种鸡在繁育期间的限饲或拥挤的饲养条件等应激有关还有待进一步研究。因为藏鸡长期生活在高原,已适应高海拔和低氧的环境。
鸡胚发育阶段,GC是胎儿组织分化、成熟以及中枢神经系统发育所必需的激素,但过多接触GC则会影响胎儿的发育,使子代出现低出生体质量、生长缓慢、免疫力低下等现象[19, 20, 21]。哺乳动物中,胎盘 11β-HSD2 是阻止母体GC到达胎儿的屏障,研究显示胎盘 11β-HSD2 基因的表达水平与胎儿出生体质量成正相关[22],且胎盘GC屏障功能减弱即 11β-HSD2 基因的表达水平下降是导致胎儿生长受限(fetal growth restriction,FCR)发生的原因之一[23]。在禽类, 11β-HSD2和20 -HSD是保护胎儿免受母体高水平皮质酮影响的重要屏障。本试验中,肉鸡种蛋蛋黄内的皮质酮含量较高,且皮质酮代谢酶基因主要是在卵黄膜内表达,可见肉鸡鸡胚卵黄膜内 11β-HSD2和20 -HSD mRNA的高表达量主要是为了保护胚胎免受过多的母源性GC对胚胎发育的损害。引人注意的是,藏鸡鸡胚皮质酮代谢酶基因在尿囊膜上的表达量均显著高于肉鸡。在采样过程我们发现,与肉鸡相比,藏鸡尿囊膜的血管异常丰富,而在哺乳动物的相关研究表明,GC可通过影响胎盘上血管内皮生长因子的表达,抑制胎盘血管的形成[24]。藏鸡与肉鸡相比,种蛋内的皮质酮含量相对较低,这是否与藏鸡尿囊膜血管丰富相关尚需进一步验证。另外,在哺乳动物上的研究显示,GC代谢酶基因的表达具有组织特异性[25],这在一定程度也能解释两品种鸡胚皮质酮代谢酶基因在卵黄膜、尿囊膜组织表达的差异。
综上,本研究结果显示藏鸡和肉鸡种蛋内皮质酮水平及胚胎卵黄膜、尿囊膜皮质酮代谢酶基因表达水平都存在着一定差异。但关于母体内激素是如何沉积到蛋内,以及禽类“胎盘”组织——卵黄膜和尿囊膜 11β-HSDs、20 -HSD基因表达差异与两品种表现性状差异的关系仍有待进一步研究。
| [1] | Edwards C R W, Benediktsson R, Lindsay R S, et al. 11β-Hydroxysteroid dehydrogenases:key enzymes in determining tissue-specific glucocorticoid effects[J]. Steroids, 1996, 61(4):263-269 |
| [2] | Ahmed A A, Ma W, Guo F, et al. Differences in egg deposition of corticosterone and embryonic expression of corticosterone metabolic enzymes between slow and fast growing broiler chickens[J]. Comparative Biochemistry and Physiology Part A:Molecular and Integrative Physiology, 2013, 164(1):200-206 |
| [3] | 饶开晴. Leptin对鸡胚卵黄膜和尿囊膜基因表达及血管形成的影响[D]. 南京:南京农业大学, 2009 [Rao K Q. Effect of leptin on yolk sac and chorioallantoic membrane gene expression and angiogenesis in the chicken embryo[D]. Nanjing:Nanjing Agricultural University, 2009(in Chinese with English abstract)] |
| [4] | Okuliarová M, rnikov B, Rettenbacher S, et al. Yolk testosterone and corticosterone in hierarchical follicles and laid eggs of Japanese quail exposed to long-term restraint stress[J]. General and Comparative Endocrinology, 2010, 165(1):91-96 |
| [5] | Williams T D, Ames C E, Kiparissis Y, et al. Laying-sequence-specific variation yolk oestrogen levels, and relationship to plasma oestrogen in female zebra finches(Taeniopygia guttata)[J]. Proceedings of the Royal Society B:Biological Sciences, 2005, 272:173-177 |
| [6] | Schwabl H. Yolk is a source of maternal testosterone for developing birds[J]. Proceedings of the National Academy of Sciences, 1993, 90(24):11446-11450 |
| [7] | Rubolini D, Romano M, Boncoraglio G, et al. Effects of elevated egg corticosterone levels on behavior, growth, and immunity of yellow-legged gull(Larus michahellis)chicks[J]. Hormones and Behavior, 2005, 47(5):592-605 |
| [8] | Hayward L S, Richardson J B, Grogan M N, et al. Sex differences in the organizational effects of corticosterone in the egg yolk of quail[J]. General and Comparative Endocrinology, 2006, 146(2):144-148 |
| [9] | Love O P, Williams T D. The adaptive value of stress-induced phenotypes:effects of maternally derived corticosterone on sex-biased investment, cost of reproduction, and maternal fitness[J]. The American Naturalist, 2008, 172(4):135-149 |
| [10] | Rettenbacher S, Möstl E, Hackl R, et al. Corticosterone in chicken eggs[J]. Annals of the New York Academy of Sciences, 2005, 1046(1):193-203 |
| [11] | Navara K J, Pinson S E. Yolk and albumen corticosterone concentrations in eggs laid by white versus brown caged laying hens[J]. Poultry Science, 2010, 89(7):1509-1513 |
| [12] | Johnson A L. Avian Physiology[M]. 4th ed. New York:Springer-Verlag, 1986:403-431 |
| [13] | Downing J A, Bryden W L. Determination of corticosterone concentrations in egg albumen:a non-invasive indicator of stress in laying hens[J]. Physiology and Behavior, 2008, 95(3):381-387 |
| [14] | Lay D C, Jr, Fulton R M, Hester P Y, et al. Hen welfare in different housing systems[J]. Poultry Science, 2011, 90(1):278-294 |
| [15] | Fraisse F, Cockrem J F. Corticosterone and fear behavior in white and brown caged laying hens[J]. British Poultry Science, 2006, 47(2):110-119 |
| [16] | Nestor K E, Anderson J W, Patterson R A. Genetics of growth and reproduction in the turkey.14. Changes in genetic parameters over thirty generations of selection for increased body weight[J]. Poultry Science, 2000, 79(4):445-452 |
| [17] | Love O P, Chin E H, Wynne-Edwards K E, et al. Stress hormones:a link between maternal condition and sex-biased reproductive investment[J]. American Naturalist, 2005, 166(6):751-766 |
| [18] | Quillfeldt P, Poisbleau M, Parenteau C, et al. Measuring corticosterone in seabird egg yolk and the presence of high yolk gestagen concentrations[J]. General and Comparative Endocrinology, 2011, 173(1):11-14 |
| [19] | Nyirenda M J, Lindsay R S, Kenyon C J, et al. Glucocorticoid exposure in late gestation permanently programs rat hepatic phosphoenolpyruvate carboxykinase and glucocorticoid receptor expression and causes glucose intolerance in adult offspring[J]. Journal of Clinical Investigation, 1998, 101(10):2174-2181 |
| [20] | Reynolds L P, Borowicz P P, Caton J S, et al. Developmental programming:the concept, large animal models, and the key role of uteroplacental vascular development[J]. Journal of Animal Science, 2010, 88(13):E61-E72 |
| [21] | Bale T L. Neuroendocrine and immune influences on the CNS:it's a matter of sex[J]. Neuron, 2009, 64(1):13-16 |
| [22] | 邱玉芳, 童华, 郭锡熔, 等. 妊娠高血压综合征伴胎儿宫内生长迟缓胎盘11-β羟类固醇脱氢酶的表达[J]. 中国医师进修杂志, 2008, 31(18):1-4 [Qiu Y F, Tong H, Guo X R, et al. 11beta-hydroxysteroid dehydrogenase gene expression in placenta of pregnancy induced hypertension complicated by intrauterine growth retardation[J]. Chinese Journal of Postgraduates of Medicine, 2008, 31(18):1-4(in Chinese with English abstract)] |
| [23] | 王秀, 叶连红, 暴蕾, 等. FGR孕妇血清皮质醇浓度与胎盘11β-HSD2表达水平的关系[J]. 吉林医学, 2010, 31(31):5490-5492 [Wang X, Ye L H, Bo L, et al. The relationship between pregnancy serum cortisol concentration and placental tissue 11β-HSD2 expression level in fetal growth restriction[J]. Jilin Medical Journal, 2010, 31(31):5490-5492(in Chinese with English abstract)] |
| [24] | Hewitt D P, Mark P J, Waddell B J. Glucocorticoids prevent the normal increase in placental vascular endothelial growth factor expression and placental vascularity during late pregnancy in the rat[J]. Endocrinology, 2006, 147(12):5568-5574 |
| [25] | Speirs H J, Seckl J R, Brown R W. Ontogeny of glucocorticoid receptor and 11beta-hydroxysteroid dehydrogenase type-1 gene expression identifies potential critical periods of glucocorticoid susceptibility during development[J]. Journal of Endocrinology, 2004, 181(1):105-116 |




