文章信息
- 刘丽, 季辉, 彭麟, 阮祥春, 吉利伟, 江善祥. 2015.
- LIU Li, JI Hui, PENG Lin, RUAN Xiangchun, JI Liwei, JIANG Shanxiang. 2015.
- 鸡肝原代细胞药物代谢模型的建立与优化
- The establishment and optimization of the drug metabolic model of adult chicken primary hepatocytes
- 南京农业大学学报, 38(1): 127-133
- Journal of Nanjing Agricultural University, 38(1): 127-133.
- http://dx.doi.org/10.7685/j.issn.1000-2030.2015.01.019
-
文章历史
- 收稿日期:2014-04-14
原代肝细胞由于包含了几乎肝脏所有的药物代谢酶及相应辅助因子,其代谢功能与体内肝细胞相似,因此外源性化合物的毒性筛选、危险性评价及肝损伤机制及治疗的研究,均广泛应用离体肝细胞模型[1, 2]。目前以大鼠、小鼠、猴、猪、狗、兔、鸭、鱼等作为肝源的原代培养肝细胞已广泛用于科学研究中[1, 3, 4, 5, 6]。对于成年鸡原代肝细胞的分离培养,国外已有相关报道,但培养的时间较短[7, 8, 9, 10]。而成年鸡原代肝细胞体外模型有助于研究药物的代谢途径和产物,研究参与代谢反应的酶,预测和解释药物间相互作用,评价药物对肝微粒体中细胞色素P450的诱导作用和探讨诱导机制,研究药物的细胞毒性等[11, 12]。而细胞色素P450是催化机体Ⅰ相代谢的重要酶系,它的活性直接关系着药物的清除率、效力、持续时间及药物间相互作用等[13, 14]。
本试验利用鸡肝原代细胞分离培养来建立药物代谢模型,并对鸡肝原代细胞分离培养进行优化,通过测定鸡肝细胞中与药物代谢紧密相关的3种CYP450酶亚型mRNA和蛋白的表达水平,来进一步完善和优化药物代谢模型。
1 材料与方法 1.1 试验动物
8~12周龄雄性黄羽鸡(体质量1.5~2.0 kg)购于扬州翔龙家禽发展有限公司。
1.2 主要试剂和器材
DMEM培养基、1640培养基、William′s E培养基和Leibovitz′s L-15(L-15)培养基购于Gibco公司,牛胰岛素、台盼蓝、Ⅳ型胶原酶购于Sigma公司,胎牛血清购于杭州四季青生物工程材料有限公司,青霉素、链霉素(双抗)为Invitrogen产品,HEPES sodium salt和EGTA购于Amresco公司,RNAiso plus购于TaKaRa,乳酸脱氢酶测定试剂盒购于南京建成生物工程研究所,RIPA裂解液购于长沙艾佳生物技术有限公司,其他试剂均为国产分析纯。6孔、96孔细胞培养板为Costar公司产品;倒置相差显微镜(IX70型),为日本Olympus公司产品。主要试剂配制:灌流液a:33 mmol · L-1 HEPES、127.8 mmol · L-1 NaCl、3.15 mmol · L-1 KCl,0.7 mmol · L-1 Na2HPO4 · 12H2O、0.6 mmol · L-1 EGTA,pH 7.6;灌流液b:33 mmol · L-1 HEPES、127.8 mmol · L-1 NaCl、3.15 mmol · L-1 KCl、0.7 mmol · L-1 Na2HPO4 · 12H2O、3 mmol · L-1 CaCl2;灌流液c:含0.05 g · L-1 Ⅳ型胶原酶的灌流液b。
1.3 鸡肝脏的胶原酶灌注
参照Seglen[15]二步灌流法并加以改良。操作如下:手术前鸡只禁食3 h,翅下静脉注射肝素钠1 500 IU · kg-1抗凝,并在腹腔注射50 mg · kg-1戊巴比妥钠麻醉动物,将鸡麻醉后用0.1%新洁尔灭浸泡消毒。将鸡带入无菌室,仰面固定,用酒精棉擦拭腹部皮肤。打开腹腔,先结扎入右肝的右肝门静脉的主要属支肠系膜总静脉、胰十二指肠静脉和腺胃脾静脉,再结扎入左肝门静脉的胃腹静脉、胃左静脉和腺胃后静脉,最后结扎后腔静脉和髂总静脉,取出肝脏,用含20 g · L-1双抗的生理盐水冲洗浸泡肝脏,然后在肝门静脉插管后分两步进行灌注。
第一步,消化前灌流:用100 mL玻璃注射器抽取37~38 ℃灌流液a冲洗肝脏,待肝脏稍鼓胀时用针头在后腔静脉上扎洞,直至肝脏的颜色变为土黄色,流出的液体清亮为止(约用400 mL)。然后,用37~38 ℃灌流液b冲洗以去除残留的EGTA。第二步,消化灌流:将肝脏转移到无菌的250 mL烧杯中,把烧杯浸泡在37~38 ℃恒温水浴锅中,以20 mL · min-1速率灌入37 ℃预热的灌流液c。
1.4 肝细胞分离纯化及计数
在超净工作台将胶原酶消化好的肝脏转移到无菌的烧杯中,用含双抗的Hank′s液冲洗干净,放入平皿中。用小止血钳轻轻撕开肝被膜,用含双抗的Hank′s液洗脱肝细胞,并用移液枪反复吹打分散细胞,然后用100目和200目的不锈钢筛将细胞悬液过滤到烧杯中。静置沉淀细胞10 min,从液面吸弃部分细胞上悬液,剩余的约100 mL细胞悬液分装到2个50 mL塑料离心管中,500 r · min-1离心3 min后弃上清液,每个离心管用20 mL含双抗的Hank′s重悬细胞,500 r · min-1离心3 min后弃上清液,每个离心管用15 mL不同的贴壁培养基重悬细胞。贴壁培养基为加入体积分数为10%的胎牛血清、1%双抗(100 U · mL-1青霉素和100 μg · mL-1链霉素)、0.5 mg · L-1牛胰岛素的基础培养液,基础培养液分别选用William′s E、L-15、1640或DMEM培养基。将2个离心管的细胞合在一起,反复吹打,分散细胞。取0.5 mL细胞悬液用培养基稀释10倍后,取细胞悬液0.5 mL与0.5 mL 4 g · L-1台盼蓝溶液混匀,1 min后在血细胞计数板上进行细胞计数,计算出获得的肝细胞产量和肝细胞实时存活率。
1.5 鸡肝细胞原代基础培养液的筛选和形态学的观察
为了考察不同基础培养液对鸡肝细胞原代培养的影响,用上述试验中不同基础培养液配制的贴壁培养液,将获得的肝细胞稀释成5×105 mL-1,接种于96孔细胞培养板,每孔接种200 μL。William′s E、DMEM和1640培养液置于37 ℃的CO2培养箱培养,L-15培养液置于37 ℃恒温箱培养。4 h后吸弃旧培养液,用Hank′s液轻轻洗细胞2次,更换细胞培养液,每孔200 μL。接着培养20 h后更换培养液,以后每24 h换1次培养液。连续培养8 d,观察记录细胞培养的全过程。
1.6 MTT法绘制生长曲线及LDH活性的测定
将获得的肝细胞稀释成5×105 mL-1细胞悬液,接种于96孔细胞培养板,每孔200 μL。置37 ℃恒温箱培养,4 h后吸弃旧培养液,用Hank′s液轻轻洗细胞2次,更换培养液,每孔200 μL。继续培养20 h后更换培养液。以后每24 h换1次培养液。每天换液时用MTT法检测[16]细胞生长情况,绘制生长曲线。
将获得的肝细胞稀释成5×105 mL-1,分别接种3 mL于3个细胞培养皿中,每天更换并收集培养液,离心除去浮游细胞及碎片,将培养上清液置于-20 ℃的冰箱保存。连续取样8 d,按照说明书用试剂盒检测LDH酶活性。
1.7 细胞内3种主要代谢酶CYP 1A4、CYP1A5和CYP3A37 mRNA表达水平的测定
将获得的肝细胞稀释成5×105 · mL-1,分别接种3 mL于4个细胞培养皿中,分别在0、2、4、6和8 d时吸弃旧培养液,用Hank′s液轻轻洗细胞2次,加0.5 mL Trizol Reagent吹打,使细胞裂解完全,采用Trizol试剂法提取肝细胞的总RNA,逆转录为cDNA,并以此为模板进行PCR扩增,采用10 g · L-1琼脂糖凝胶电泳测定CYP1A 4、CYP1A5、CYP3A37 与β-actin的相对吸光值并进行半定量分析。CYP 1A4 的引物序列(PCR产物长度为105 bp)为:5′-AGGACGGAGGCTGACAAG-3′/5′-CAGGATGGTGGTGAGGAAGA-3′。CYP 1A5 的引物序列(PCR产物长度为201 bp)为:5′-TCACCATCCCGCACAGCA-3′/5′-AAGTCATCACCTTCGCATC-3′。CYP 3A37 的引物序列(PCR产物长度为145 bp)为:5′-CGAATCCCAGAAATCAGA-3′/5′-AGCCAGGTAACCAAGTGT-3′。β-actin目的基因引物序列(PCR产物长度为300 bp)为:5′-TGCGTGACATCAAGGAGAAG-3′/5′-TGCCAGGGTACATTGTGGTA-3′。
1.8 细胞内3种主要代谢酶CYP1A4、CYP1A5和CYP3A37的蛋白表达水平测定
将获得的肝细胞稀释成5×105 mL-1,分别接种3 mL于4个细胞培养皿中,分别在0、2、4、6和8 d,用Hank′s液轻轻洗细胞2次,用细胞刷将细胞轻轻刮下,置于1.5 mL离心管中,4 000 r · min-1离心5 min,弃去Hank′s液。参照RIPA蛋白裂解液说明书的处理方法提取总蛋白并参照BCA蛋白定量试剂盒说明书对上述获得的总蛋白进行蛋白定量。用加样缓冲液分别配制成12%的分离胶和5%的浓缩胶,每孔加50 μg样品,进行电泳。电泳条件为浓缩胶80 V 30 min,分离胶100 V 2.5 h。电泳结束后将蛋白转移到聚偏二氟乙烯(PVDF)膜上。
2 结果与分析 2.1 肝脏的离体灌流
将肝脏完整取出后,肝门静脉插管前肝脏血液充盈,灌注a、b液后,血液被冲洗干净,肝脏呈现土黄色,但灌注完胶原酶后,整个肝脏非常柔软,肝组织与肝包膜分离。撕开肝包膜,肝组织就松散流出。图 1为灌流前后的肝脏图。
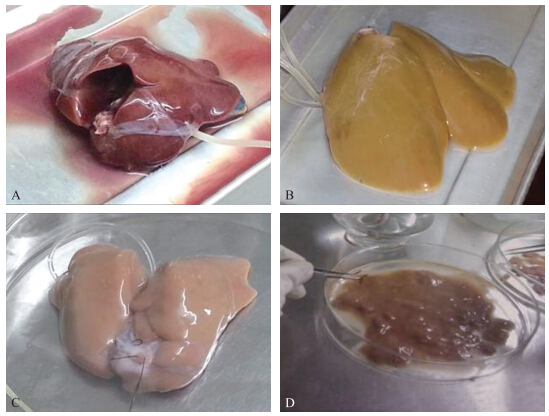 | 图 1 灌流前后的肝脏图Fig. 1 Morphology of chicken liver before and after perfusionA.肝门静脉插管灌流开始;B.冲洗液a、b灌流结束;C.灌注胶原酶结束;D.灌流结束,撕去肝包膜。 A.Intubate from hepatic portal vein before perfusion;B.After perfusate a and b were perfused;C.After collagenase was perfused;D.Liver capsule was teared after perfusion. |
通过细胞计数,计算出从每个肝脏获得的肝细胞总量为(9.2±0.2)×108个,以台盼蓝拒染试验得到肝细胞实时存活率为(93±1)%。
2.2 细胞培养条件筛选和形态学观察
借助细胞形态学[17]判断肝细胞纯度和质量,在倒置显微镜下,刚分离得到的肝细胞呈圆形,边缘较光滑,直径约为牛、鼠肝细胞的1/2,胞浆内尤其是靠近细胞膜的地方无明显气泡,贴壁后呈上皮细胞样生长,多角形,胞浆内容物均匀丰富,细胞核清晰透亮,少数有双核,细胞生长状态良好。在不同的培养基中培养后,发现William′s E培养基贴壁最好,DMEM和L-15次之,1640最差,在长期培养过程中DMEM培养基中细胞稳定周期长,可长期培养2~3周,而且与William′s E相比,DMEM更经济,最终选择用DMEM培养肝原代细胞。图 2为肝细胞在DMEM培养基中分别培养8 h和2、3、5、12、21 d时,倒置显微镜下的细胞形态。由图 2可见:培养8 h后,肝细胞依然呈圆形,边缘光滑。培养2 d后,肝细胞增殖旺盛,可见较多双核细胞。新生的肝细胞排列在岛状周围,外周伸有锯齿状突起。培养5 d后,绝大多数岛屿状肝细胞互相汇合成片,细胞排列紧密,细胞间界限清晰,形成胆小管样结构。培养12 d时,肝细胞逐渐出现老化现象,表现为胞体萎缩拱起,胞膜与核膜破损,胞间界限模糊,胞质出现颗粒化。培养至21 d时,肝上皮细胞间出现非肝上皮细胞,呈长条形,并伸有细长的突起生长,并最终完全取代肝上皮细胞。
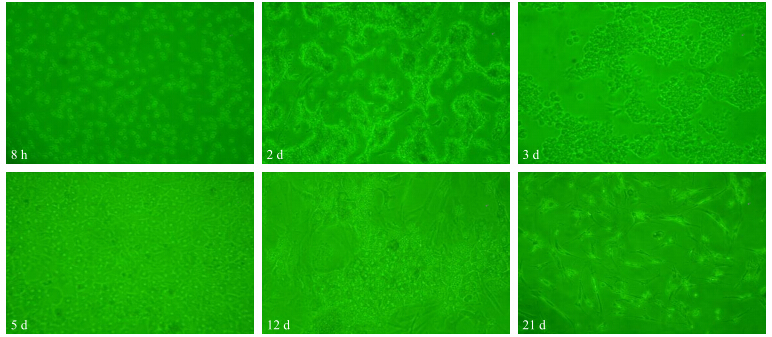 | 图 2 DMEM培养基中不同培养阶段鸡肝细胞的形态Fig. 2 Morphology of chicken hepatocytes in different culture period in DMEM medium |
通过MTT法得到8 d内细胞的生长曲线如图 3。在DMEM培养基培养条件下,鸡肝细胞体外培养8 d内的吸光值显示:原代培养2~4 d,肝细胞数量稍微降低;培养4~6 d,肝细胞数量下降的比较多;之后的2 d,继续下降。
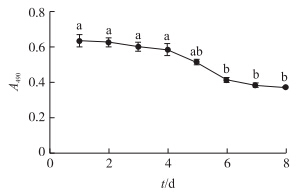 | 图 3 鸡肝原代细胞在DMEM培养基中的生长曲线Fig. 3 Growth state of primary culture chicken hepatocytes (MTT assay)cultured in DMEM medium不同小写字母表示差异显著。The different small letters mean significant difference at 0.05 level. |
由图 4可以看出:培养8 h,上清液中LDH的活性最大,随着时间的增加,LDH的活性开始下降;培养3~8 d,LDH的活性在低水平保持稳定。
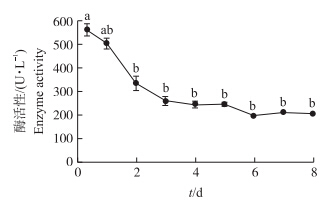 | 图 4 鸡肝原代细胞培养8 d内LDH的活性变化Fig. 4 Change of LDH activities of chicken primary culture hepatocytes in 8 days |
如图 5所示:原代培养肝细胞内的3种主要代谢酶CYP 1A4、CYP1A5和CYP3A37 mRNA的相对表达水平在前2 d稳定,第4天时稍微降低,在第6、8天都有所提高。但CYP 1A5 mRNA在第8天的相对表达量比第6天低。
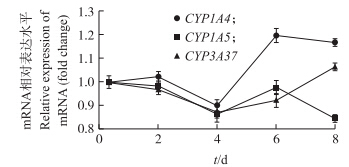 | 图 5 不同培养时间原代肝细胞内CYP 1A4、CYP1A5 和CYP3A37 mRNA的相对表达量Fig. 5 The relative expression level of CYP 1A4,CYP1A5,CYP3A37 mRNA in cell by different culture time |
3种主要代谢酶CYP1A4、CYP1A5和CYP3A37在原代培养肝细胞内主要表达为CYP1A和CYP3A 2种蛋白。如图 6所示:CYP1A蛋白表达量在前2 d稳定,第4天时稍微升高,但在第6、8天时降低。CYP3A蛋白表达量8 d内保持稳定。
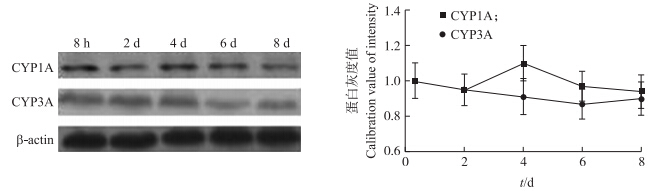 | 图 6 不同培养时间原代肝细胞内CYP1A、CYP3A蛋白的表达Fig. 6 The expression of CYP1A,CYP3A protein in cell by different culture time |
肝细胞分离方法有机械法和酶消化法,通过机械法[18]和简单的肝剪切酶消化法[19]获得的肝细胞的活力都不高,而且细胞膜损伤严重并且不易修复;胶原酶灌注消化法是目前最理想也是最常用的分离肝细胞的方法,此方法获得的肝细胞活力强,完整性好,细胞产量高,培养一段时间内还能保持肝细胞在体内的生理功能,是研究肝细胞功能的理想工具。
本研究通过肝脏离体循环灌流分离肝细胞,短时间细胞自由沉淀、低速离心来纯化肝实质细胞,获得很好的纯化效果,肝细胞纯度和存活率达90%以上。
不同类型的培养液因其成分和功能有较大的差别,对肝细胞的培养有明显的影响[20]。刘开永等[21]和陈黎龙等[22]选用William′s E培养液做基础培养基,培养成年鸡的肝原代细胞效果良好。但本研究中发现DMEM培养条件下细胞生长状态好,细胞稳定周期长,可培养2~3周,而且DMEM更经济。
鸡肝细胞原代培养过程中检测细胞活性常用的方法是台盼蓝拒染试验及MTT试验。通过MTT法绘制该试验生长曲线发现,在DMEM培养条件下,鸡肝细胞体外原代培养8 d细胞数量大且稳定,随后有所降低。此外,通过多种酶活性水平监测细胞的存活情况。LDH是一种胞内酶[23],存在于肝细胞内,当细胞膜破损时,LDH会从细胞内漏出到细胞外环境中,通过测定细胞培养基上清液中的LDH活性,可以间接了解到细胞膜的受损程度。本研究对不同培养时间段的培养基上清液中LDH活性检测,表明在胶原酶灌注消化鸡肝脏和分离肝细胞过程中,细胞膜受到了一定程度的损伤;培养2 d后,肝细胞逐渐适应了新环境并自我修复;培养3 d后培养基上清液中LDH水平在较低水平持续,说明此时肝细胞膜完整,细胞状态好,适合用于有关肝细胞代谢、中毒、基因表达的研究。
因为细胞色素P450是催化机体Ⅰ相代谢的重要酶系,在药物的生物转化中起着决定性作用,其中CYP1A和CYP3A在药物代谢过程中起重要作用,因此本研究考察了鸡肝原代细胞培养过程中CYP 1A4、CYP1A5、CYP3A37 mRNA和蛋白表达规律,为鸡肝原代细胞用于药物体外代谢提供理论基础和依据。文献[24, 25]报道,小鼠连续以50 mg · kg-1剂量腹腔注射苯巴比妥钠7 d对CYP450产生诱导作用,是转录后调节,不影响mRNA的表达。本试验是单次腹腔注射50 mg · kg-1戊巴比妥钠麻醉动物,但在鸡肝细胞原代培养过程中,3种代谢酶CYP1A4、CYP1A5、CYP3A37在细胞内mRNA的相对表达量和蛋白定量均没有显著性变化。表明本试验操作离体灌流单次腹腔注射50 mg · kg-1戊巴比妥钠未显著影响3种主要代谢酶的表达。
| [1] | Battle T,Stacey G. Cell culture models for hepatotoxicology[J]. Cell Biology and Toxicology,2001,17(4/5):287-299 |
| [2] | de la Rosa L C,Schoemaker M H,Vrenken T E,et al. Superoxide anions and hydrogen peroxide induce hepatocyte death by different mechanisms:involvement of JNK and ERK MAP kinases[J]. J Hepatol,2006,44(5):918-929 |
| [3] | van de Kerkhove M P,Germans M R,Deurholt T,et al. Evidence for Galalpha(1-3)Gal expression on primary porcine hepatocytes:implications for bioartificial liver systems[J]. J Hepatol,2005,42(4):541-547 |
| [4] | Yao Y Q,Zhang D F,Tang N,et al. Replication of hepatitis B virus in primary duck hepatocytes transfected with linear viral DNA[J]. World Journal of Gastroenterology,2005,11(32):5019-5021 |
| [5] | Rose K A,Kostrubsky V,Sahi J. Hepatobiliary disposition in primary cultures of dog and monkey hepatocytes[J]. Molecular Pharmaceutics,2006,3(3):266-274 |
| [6] | Tuschl G,Lauer B,Mueller S. Primary hepatocytes as a model to analyze species-specific toxicity and drug metabolism[J]. Expert Opinion on Drug Metabolism & Toxicology,2008,4(7):855-870 |
| [7] | Lee S H,Lee M Y,Lee J H,et al. A potential mechanism for short time exposure to hypoxia-induced DNA synthesis in primary cultured chicken hepatocytes:correlation between Ca2+/PKC/MAPKs and PI3K/Akt/mTOR[J]. Journal of Cellular Biochemistry,2008,104(5):1598-1611 |
| [8] | Ferioli A,Harvey C,de Matteis F. Drug-induced accumulation of uroporphyrin in chicken hepatocyte cultures. Structural requirements for the effect and role of exogenous iron[J]. Biochem J,1984,224(3):769-777 |
| [9] | Jacobs J M,Nichols C,Marek D,et al. Effect of arsenite on the induction of CYP1A4 and CYP1A5 in cultured chick embryo hepatocytes[J]. Toxicol Appl Pharmacol,2000,168(3):177-182 |
| [10] | Kennedy S W,Jones S P,Bastien L J. Efficient analysis of cytochrome p4501A catalytic activity,porphyrins,and total proteins in chicken-embryo hepatocyte cultures with a fluorescence plate reader[J]. Analytical Biochemistry,1995,226(2):362-370 |
| [11] | Hakala K S,Link M,Szotakova B,et al. Characterization of metabolites of sibutramine in primary cultures of rat hepatocytes by liquid chromatography-ion trap mass spectrometry[J]. Analytical and Bioanalytical Chemistry,2009,393(4):1327-1336 |
| [12] | Rodriguez-Fuentes N,Lopez-Rosas I,Roman-Cisneros G,et al. Biotin deficiency affects both synthesis and degradation of pyruvate carboxylase in rat primary hepatocyte cultures[J]. Mol Genet Metab,2007,92(3):222-228 |
| [13] | Ma Y,Sachdeva K,Liu J,et al. Clofibrate and perfluorodecanoate both upregulate the expression of the pregnane X receptor but oppositely affect its ligand-dependent induction on cytochrome P450 3A23[J]. Biochemical Pharmacology,2005,69(9):1363-1371 |
| [14] | Gelboin H V,Krausz K. Monoclonal antibodies and multifunctional cytochrome P450:drug metabolism as paradigm[J]. The Journal of Clinical Pharmacology,2006,46(3):353-372 |
| [15] | Seglen P O. Preparation of isolated rat liver cells[J]. Methods Cell Biology,1976,13:29-83 |
| [16] | 郝新保,张利朝,殷缨,等. MTT比色法测定细胞生长曲线[J]. 第四军医大学学报,1997,18(4):95-96 [Hao X B,Zhang L C,Yin Y,et al. MTT assay determined growth curve[J]. Journal of the Fourth Military Medical University,1997,18(4):95-96(in Chinese with English abstract)] |
| [17] | Puviani A C,Ottolenghi C,Tassinari B,et al. An update on high-yield hepatocyte isolation methods and on the potential clinical use of isolated liver cells[J]. Comparative Biochemistry and Physiology A:Molecular and Integrative Physiology,1998,121(2):99-109 |
| [18] | 江青艳,傅伟龙,高淑静,等. 鸡离体肝细胞几种分离方法的比较[J]. 华南农业大学学报,2001,22(1):66-69 [Jiang Q Y,Fu W L,Gao S J,et al. Comparison of several methods for isolating chicken hepatocytes[J]. Journal of Huanan Agricultural University,2001,22(1):66-69(in Chinese with English abstract)] |
| [19] | Howard R B,Christensen A K,Gibbs F A,et al. The enzymatic preparation of isolated intact parenchymal cells from rat liver[J]. The Journal of Cell Biology,1967,35(3):675-684 |
| [20] | Hamilton G A,Westmoreland C,George E. Effects of medium composition on the morphology and function of rat hepatocytes cultured as spheroids and monolayers[J]. In Vitro Cellular and Developmental Biology Animal,2001,37(10):656-667 |
| [21] | 刘开永,黄显会,高海,等. 成年鸡原代肝细胞的原位分离和长期培养[J]. 中国兽医学报,2009,29(9):1193-1196 [Liu K Y,Huang X H,Gao H,et al. In situ isolation and primary long-term culture of adult chicken hepatocytes[J]. Journal of Chinese Veterinary Science,2009,29(9):1193-1196(in Chinese with English abstract)] |
| [22] | 陈黎龙,江青艳,朱晓彤,等. 成年鸡肝细胞的分离与原代[J]. 江西农业大学学报,2008,30(3):385-389 [Chen L L,Jiang Q Y,Zhu X T,et al. Separation and primary culture of adult chicken hepatocytes[J]. Journal of Jiangxi Agricultural University,2008,30(3):385-389(in Chinese with English abstract)] |
| [23] | 杨辉,杨丹凤,张华山,等. 4种典型纳米材料对小鼠胚胎成纤维细胞毒性的初步研究[J]. 生态毒理学报,2007,2(4):427-434 [Yang H,Yang D F,Zhang H S,et al. Study on cytotoxicity of four typical nanomaterials in mouse embryo fibroblasts[J]. Asian Journal of Ecotoxicology,2007,2(4):427-434(in Chinese with English abstract)] |
| [24] | 程建峰,滕树保,陈东鸿,等. 甘草酸和苯巴比妥钠对小鼠肝P450 3A酶的影响[J]. 第四军医大学学报,2000,21(8):968-970 [Cheng J F,Teng S B,Chen D H,et al. Effect of glycyrrhizin and phenobarbital pretreat-ment on hepatic P450 3A enzyme in mice[J]. Journal of the Fourth Military Medical University,2000,21(8):968-970(in Chinese with English abstract)] |
| [25] | 李平,程建峰,陈东鸿,等. 地塞米松、苯巴比妥钠和利多卡因对昆明种小鼠肝P450 3A酶的影响[J]. 第四军医大学学报,2001,22(1):26-28 [Li P,Cheng J F,Chen D H,et al. Effect of dexamethasone,phenobarbital and lido-caine on hepatic P450 3A enzyme in Kunming mice[J]. Journal of the Fourth Military Medical University,2001,22(1):26-28(in Chinese with English abstract)] |




