2. CSIR-Central Electrochemical Research Institute, Karaikudi-630003, India;
3. Defence Research Development Laboratory, Hyderabad, India
1 Introduction
MDN 250 is a 18% nickel, cobalt strengthened steel (C-type), with excellent mechanical properties, workability and heat treatment characteristics. It is weldable without preheat, in both the annealed and aged condition. The alloy is very tough, relatively soft (30/34 Rc), therefore, readily machined and formed. Typical applications for maraging include missile and rocket motor cases, landing and takeoff gear, munitions, aerospace, extrusion tooling, die casting, high performance shafting, gears and fasteners. MDN 138 is a precipitation, age hardenable stainless steel. Its principal features are high transverse toughness, good resistance to general and stress corrosion cracking, high strength that is developed by a single low temperature heat treatment and best welded in the solution annealed condition. This alloy has been used in aircraft components such as landing gear and structural sections, valves, shafts, and components in the petrochemical and nuclear industries. Many ship superstructures and liquid cargo containers are made of aluminum due to its low weight and acceptable strength. The most popular aluminium alloys for use in corrosive environments such as seawater are the 5xxx and 6xxx series alloys, which demonstrate adequate strength and excellent corrosion resistance. These series are highly suitable in various marine structures, machinery and port buildings. Alloy 6063 is a heat treatable aluminium-magnesium-silicon alloy. It provides good combination of extrudability and mechanical properties (Wan Nik et al., 2010a; Trdan and Grum, 2015; Jafarzadeh et al., 2008; Wan Nik et al., 2010b; Hosni et al., 2008). With light weight and high performance characteristics HE 20 aluminium alloy has a wide range of mechanical and corrosion resistance properties and hence acts as an important material in defence & aerospace applications and yacht construction, truck bodies, bicycle frames to screw machine parts, . In the present study the galvanic corrosion behaviour of the metal combinations HE 20/ MDN 138 and HE 20/MDN 250, with 1:1 area ratio, has been studied in natural seawater using the open well facility of CECRI’s Offshore Platform at Tuticorin for a year, which is first of its kind in the literature of Indian waters.
2 Materials and methodsThe materials (HE 20, MDN 138 & MDN 250) were supplied by Defence Research and Development Laboratory, Hyderabad. The composition of HE 20 is Cu:0.24%, Si:0.64%, Mg:0.88%, Fe:0.16%, Mn:0.17%, Cr:0.16%, Zn:0.05%, Ti: 0.01% and Al: balance. The composition of MDN 138 is Al: 1.0%, C: 0.04%, Cr: 12.5%, Mn: 0.18%, Mo: 2.0%, Ni: 7.5%, N: 0.01%, P: 0.01%, Si:0.1%, S: 0.007% and Fe: balance. The composition of MDN 250 is Al:0.07%, C: 0.03%, Cr: 0.5%, Mn: 0.10%, Mo: 4.6%, Ti: 0.3%, Ni: 17.5%, Co: 7.5%, Cu: 0.5%, P: 0.01%, Si:0.1%, S: 0.008% and Fe: balance. The coupons of HE 20, MDN 250 & MDN 138 of size 75 mm×50 mm×6 mm, were cut from the respective sheets, mechanically polished with different grits silicon carbide metallurgical paper (180, 220, 400, and 600), cleaned and degreased with acetone and weighed to an accuracy of 10–4 g, and stored in desiccators until use. Galvanic contact between the coupons HE 20 & MDN 250 and HE 20 & MDN 138 was effected by PVC sheathed 320SWG copper wires and the contact points were sealed using marine epoxy to prevent crevice corrosion. The galvanically coupled coupons HE 20 & MDN 250 and HE 20 & MDN 138 were fixed on a wooden frame with grooving and immersed in the natural seawater at a water depth 2 m below the mean low tide level using the open well facility of CECRI’s Offshore Platform at Tuticorin. Periodic monitoring of the open circuit potential of the coupons HE 20, MDN 250 & MDN 138 and the mixed potentials of the galvanic couples HE 20/MDN 250 and HE 20/MDN 138, were made daily using a high impedance voltmeter (Tektronix, Model DMM155) with an SCE. The galvanic current of the couples HE 20/MDN 250 and HE 20/MDN 138 was monitored periodically using zero impedance ammeter. Subsurface seawater was collected from the three test locations on a quarterly basis during the 12 months study period using a Hydro-Bios (Kiel) water sampler. Analyses were carried out following the standard procedures outlined by Strickland and Parsons (1972) which included general physico-chemical parameters, dissolved nutrients and major ions. Heavy metals in the water samples were extracted following APDC-MIBK pre-concentration procedure (Brooks et al., 1967) and estimated on an atomic absorption spectrometer (GBC 932 Plus).
Potentiodynamic polarization curves of HE 20, MDN 250 & MDN 138 were drawn using AUTOLAB PGSTAT30 feed potential range of-1000 mV to 1000 mV with 3 electrodes cell arrangement with HE 20/MDN 250/MDN 138 as working electrode, Ag/AgCl as reference electrode and platinum as counter electrode in natural seawater. The field studies were terminated after 330 days and the digital images of the galvanically coupled coupons HE 20 & MDN 138, and HE 20 & MDN 250, were recorded, with biomass & corrosion products and after removing biomass & corrosion products. The fouling organisms on the surfaces of the galvanically coupled coupons (HE 20 & MDN 250 and HE 20 & MDN 138) were recorded and visual observations were made on the surfaces of coupons after removal of biomass & corrosion products. The calcareous deposits on MDN 138 & MDN 250 were analyzed using XRD (X’pert PRO PAN analytical diffractometer with Syn master 793s, Netherlands). The extent of galvanic protection offered by HE 20 to MDN 138 & MDN 250 has been calculated from the gravimetric corrosion rate values of both freely corroded and galvanically coupled MDN 138, MDN 250 & HE 20.
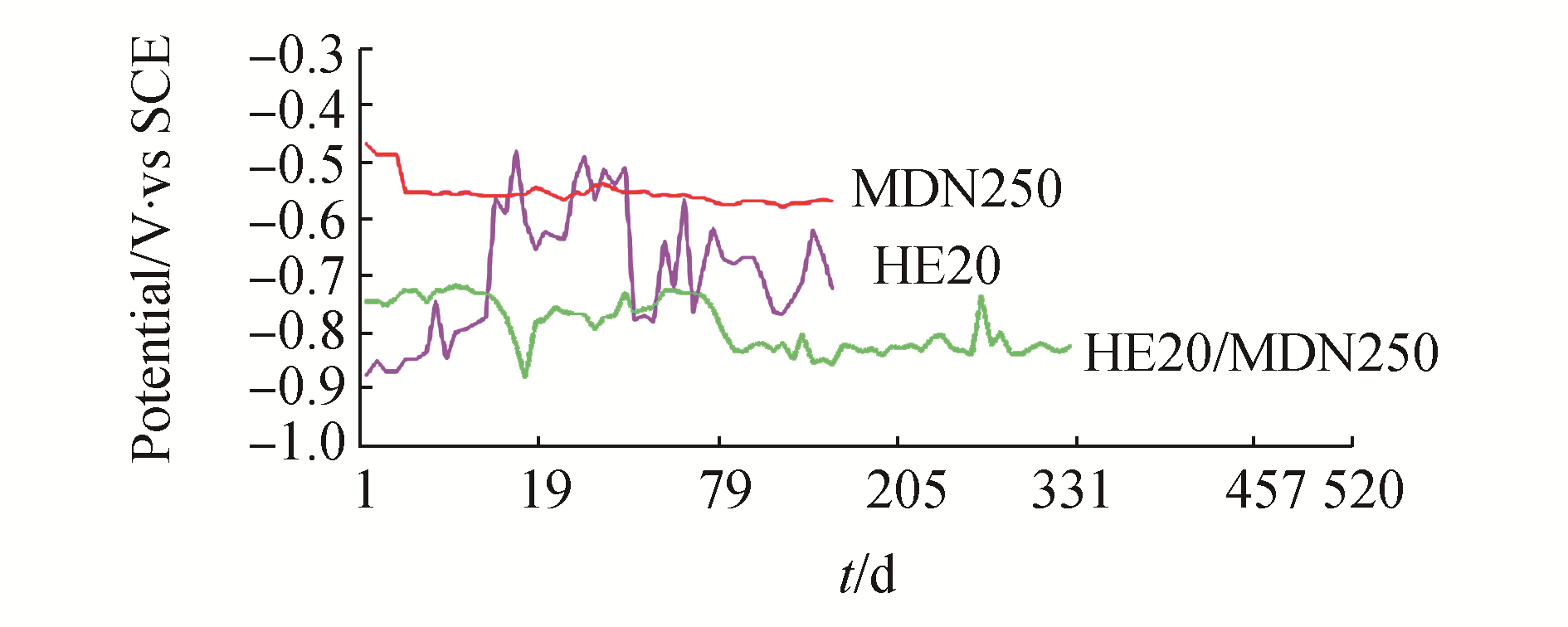
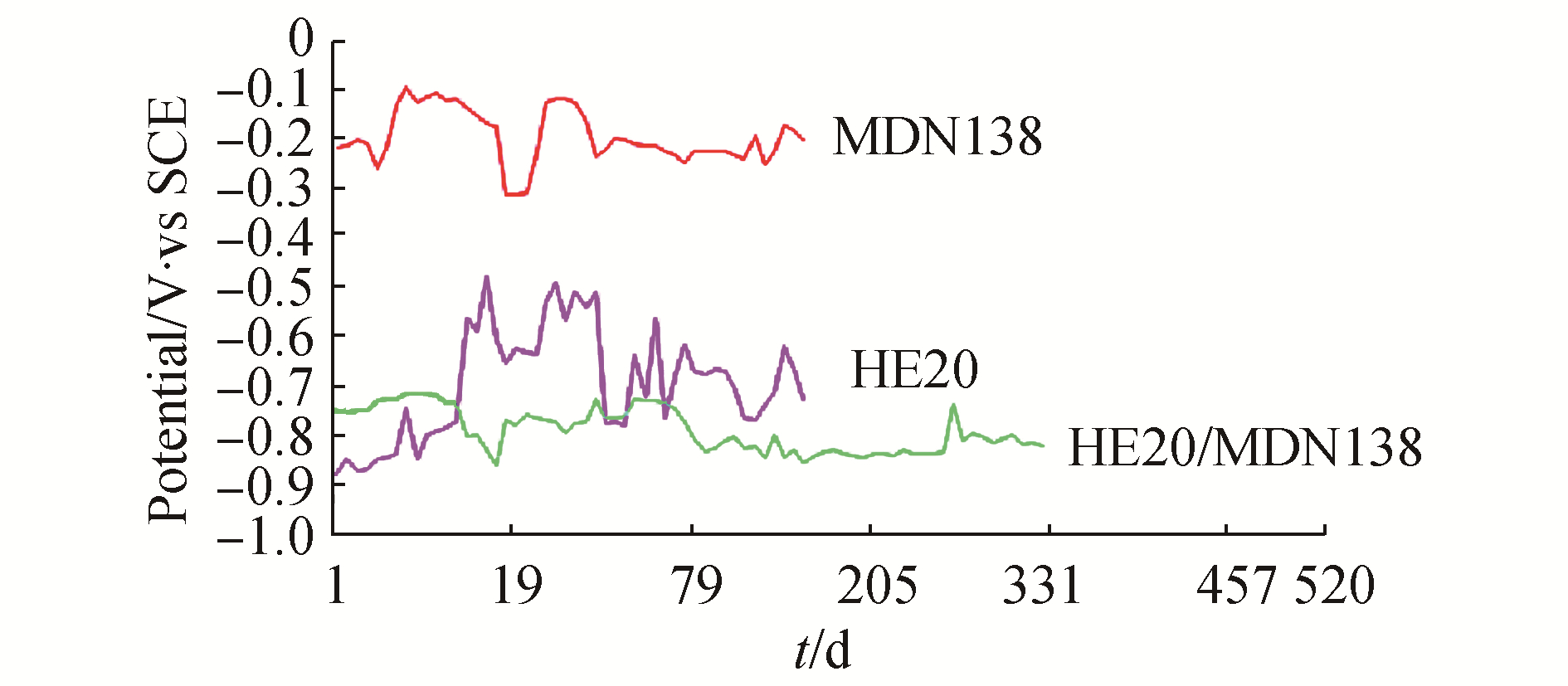
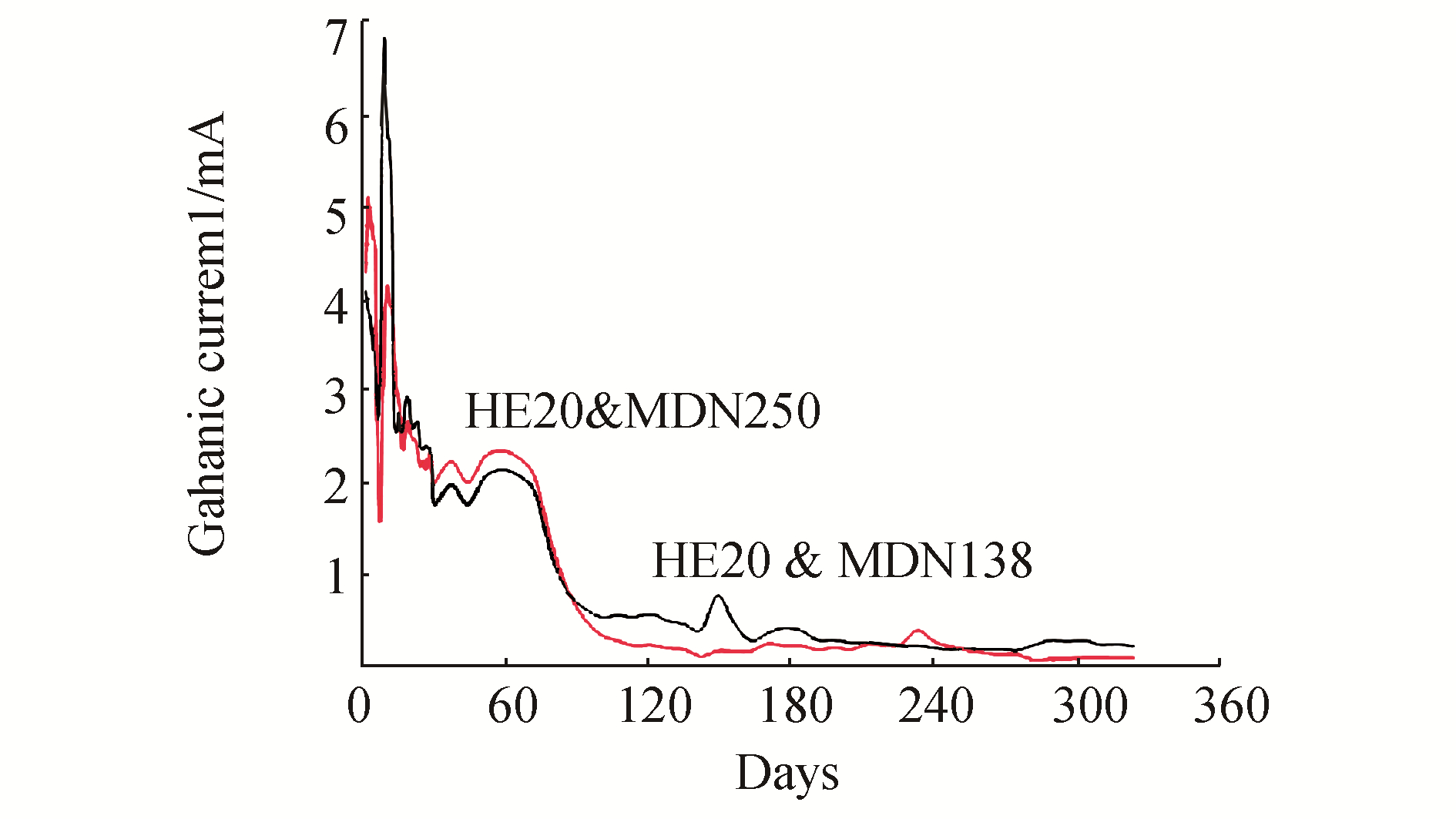
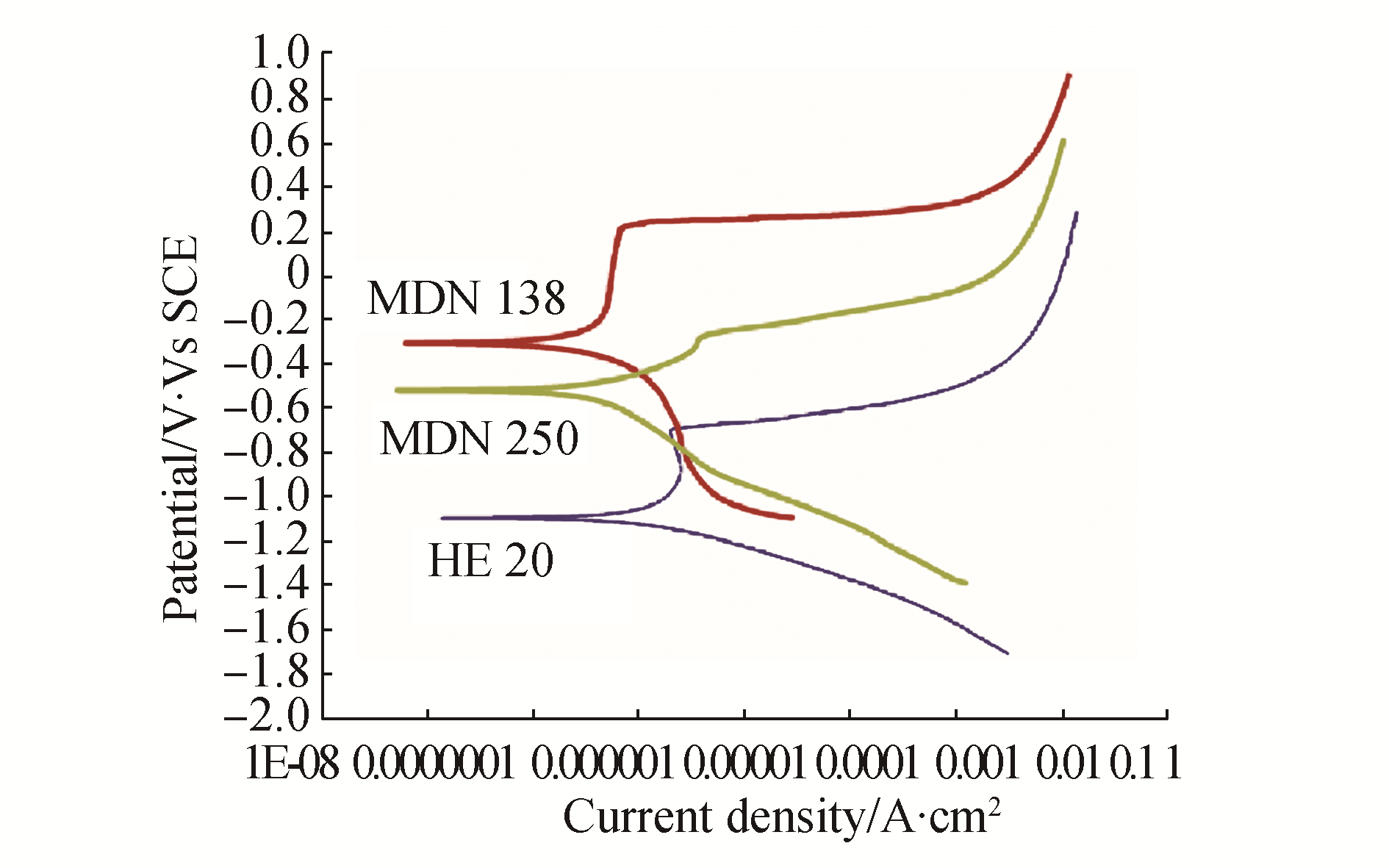
3 Results and DiscussionThe mean values of physicochemical parameters of seawater of Open sea–Tuticorin are presented in Table 1. In general the values of the water quality parameters were found to be normal, and did not show much seasonal variation during the study period. Hence the mean values of the four quarters are taken for consideration. Fig. 1 portrays the open circuit potential values of HE 20 & MDN 250 and the mixed potential values of the couple HE 20/MDN 250 and Fig. 2 portrays the open circuit potential values of HE 20 & MDN 138 and the mixed potential values of the couple HE 20/MDN 138, recorded throughout the study period. The galvanic current values of the galvanic couples HE 20/MDN 250 and HE 20/MDN 138, recorded throughout the study period are given in Fig. 3. The Potentiodynamic polarization scans of the freely corroded HE 20, MDN 250 & MDN 138 are given in Fig. 4.
| Parameters | Open sea-Tuticorin | |
| Mean | Standard deviation | |
| Salinity/‰ | 35.00 | 0.74 |
| pH | 8.1 | 0.113 389 |
| Dissolved Oxygen/(ml∙l−1) | 4.85 | 0.265 922 |
| Inorganic Phosphate/(µmol∙l−1) | 0.725 | 0.074 066 |
| Total Phosphorous/(µmol∙l−1) | 3.27 | 1.195 894 |
| Nitrite/(µmol∙l−1) | 0.017 | 0.000 983 |
| Nitrate/(µmol∙l−1) | 4.37 | 0.297 |
| Silicate/(µmol∙l−1) | 18.83 | 2.76 |
| Ammonia/(µmol∙l−1) | 2.25 | 0.134 164 |
| Calcium/(mg∙l−1) | 400 | 19.820 6 |
| Magnesium/(mg∙l−1) | 1275 | 97.247 84 |
| Copper/(µg∙l−1) | 2.7 | 0.47 |
| Cadmium/(µg∙l−1) | 1.30 | 0.217 |
| Lead/(µg∙l−1) | 14 | 0.727 8 |
| Iron/(µg∙l−1) | 54.0 | 15.928 |
| Manganese/(µg∙l−1) | 5.50 | 0.790 569 |
| Zinc/(µg∙l−1) | 1.570 | 0.594 7 |
| Mercury/(µg∙l−1) | 1.196 | 0.234 379 |
|
| Figure 1 Open circuit potential and mixed potential of HE 20 & MDN 250 |
|
| Figure 2 Open circuit potential and mixed potential of HE 20 & MDN 138 |
|
| Figure 3 Galvanic current values of the couples HE 20/MDN 138 & HE 20/MDN 250 |
|
| Figure 4 Potentiodynamic polarization curves of HE 20, MDN 250 & MDN 138 |
Figs. 5 & 6, portray the digital images of the galvanically coupled coupons HE 20 & MDN 138, with biomass & corrosion products and after removing biomass & corrosion products, respectively. The surface of the galvanically coupled HE 20 is characterized by barnacles with white corrosion products, while that of MDN 138 is characterized by oysters as primary foulers and barnacles as secondary foulers. The surface of the galvanically coupled HE 20 after removal of corrosion products and biomass is characterized by minute pits beneath passive corrosion products, whereas no characteristic pitting was observed on MDN 138.

|
| Figure 5 Surface of the galvanically coupled coupons of HE 20 (Left) & MDN 138 (Right) exposed in natural seawater for 330 days |

|
| Figure 6 Surface of the galvanically coupled coupons of HE 20 & MDN 138 after removal of biomass and corrosion products |
Figs. 7 & 8, portray the digital images of the galvanically coupled coupons HE 20 & MDN 250, with biomass & corrosion products and after removing biomass & corrosion products, respectively. The surface of the galvanically coupled HE 20 is characterized by fewer barnacles with white corrosion products, while that of MDN 250 is characterized by oysters as primary foulers and barnacles as secondary foulers. The surface of the galvanically coupled HE 20 after removal of corrosion products and biomass is characterized by minute pits beneath passive corrosion products, whereas no characteristic pitting was observed on MDN 250.

|
| Figure 7 Surface of the galvanically coupled coupons of HE 20 (Left) & MDN 250 (Right) exposed in natural seawater for 330 days |

|
| Figure 8 Surface of the galvanically coupled coupons of HE 20 & MDN 250 after removal of biomass and corrosion products |
The nature of calcareous deposits formed on the galvanically protected MDN 138 & MDN 250 as analyzed by XRD are found to be same and given in Fig. 9. The compounds were identified as CaCO3 (calcite, aragonite and vaterite), MgCO3 (magnesite), Mg(OH)2 (brucite) and MgO (brucite). With particular reference to CaCO3, more aragonite phases were recorded than calcite or vaterite.
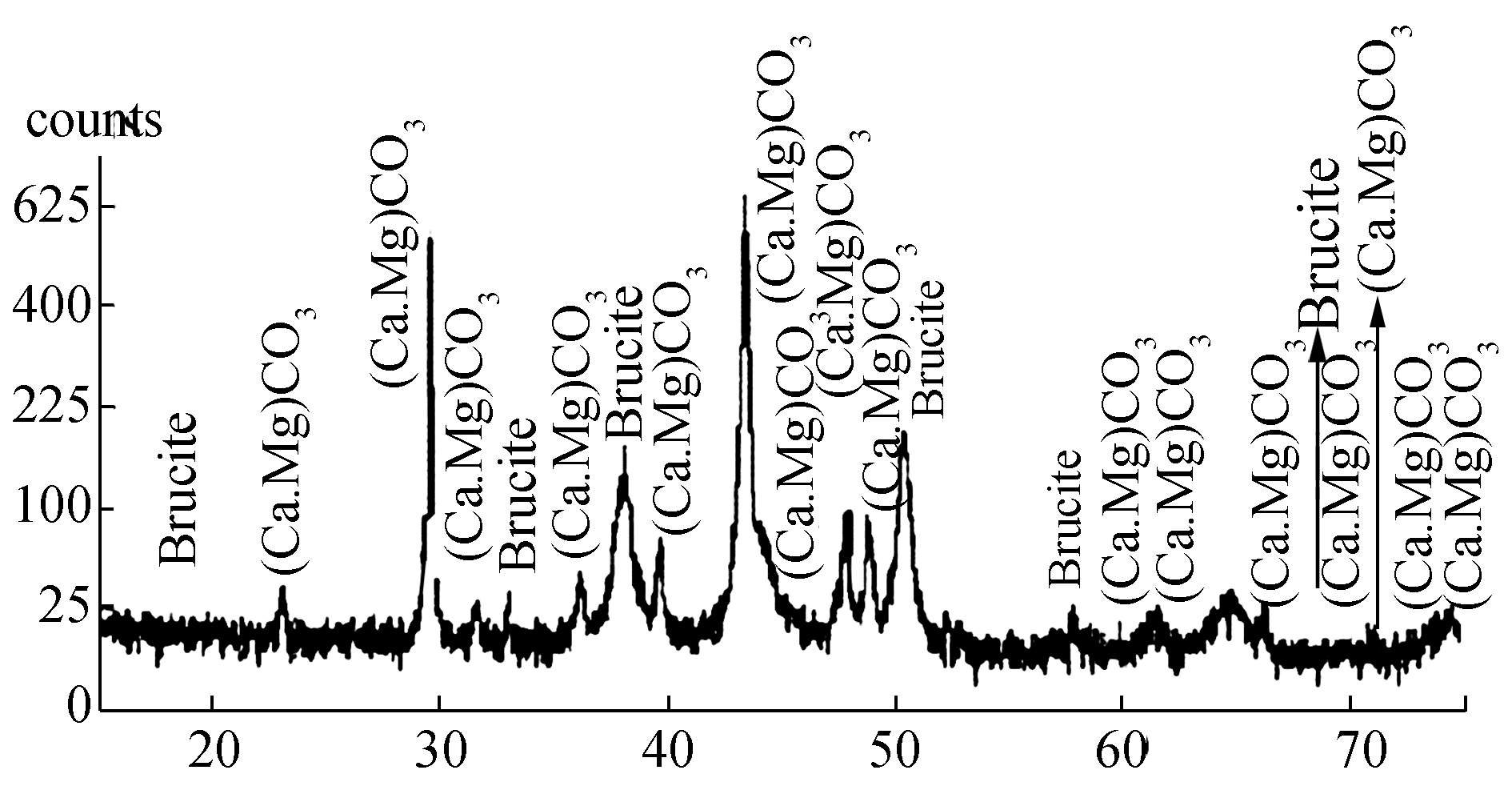
|
| Figure 9 XRD pattern of calcareous deposits formed on the surfaces of galvanically protected MDN 138 & MDN 250 |
The OCP of HE 20 from the commencement (-0.877 V) of experiment, experienced a steady uptrend up to 16th day (-0.482 V), i.e. a positive shift of 305 V has been observed. There has been a downtrend from 17th day to 21st day, from thereupon a sharp increase up to 23rd day (-0.491 V). Many zigzag were observed between 23rd day to 142nd day, and at the end of the experiment the OCP was-0.726 V. The OCP of MDN 138 from the commencement (-0.217V) of experiment had experienced many upward and downward surges during the study period, which is more pronounced up to 28th day and the fluctuation ranges between-0.312 V & -0.098 V.
The mixed potential of the couple HE 20/MDN 138 from the commencement (-0.751 V) of the experiment to till the end of the study (-0.823 V), lies in the range of-0.862 to −0.738 V. No major fluctuations in the mixed potential values are observed throughout the study period. The potential difference drive between the two metals vary between-0.45 V & -0.65 V, throughout the study period. Hence HE 20 would have offered maximum protection to MDN 138.
The galvanic current values of the couple HE 20/MDN 138 experienced a downward drift from the 1st day (4.1 mA) to the 6th day (2.65 mA), thereafter a steep increase up to 9th day (6.85 mA); and a sudden downward surge from 10th day (5.85 mA) up to 14th day (2.54 mA). The steep increase in the galvanic current values from 7th day to 9th day could be attributed to the initial current demand for effective protection of the cathode surface until the formation of an adherent thin film of calcareous deposit. Cathodic protection alters the ionic concentration at the interface, increasing the hydroxyl ion concentration (Sarlak et al.2009 and Wieng et al. 2010). The consequent pH increase reduces the solubility of calcium, magnesium and bicarbonate ions at the interface favouring the precipitation of a calcareous scale according to the following reactions:
O2+ 2H2O + 4e-→4OH-
2H2O + 2e-→2OH-+ H2
Ca2+ + CO32-→CaCO3
HCO3-+ OH-→H2O + CO32-
Mg2+ + 2OH-→Mg(OH)2
A steady and gradual decline in the galvanic current values between 16th day (2.54 mA) & 135th day (0.462 mA) has been observed. A steady state has been observed between 226th day (0.221 mA) & 324th day (0.221 mA). Thus the onset of steady state current has been postponed to the tail end of the experiment (226-324 days); which can be accounted owing to the impairment caused to the calcareous deposit film, by the combined effect of turbulence in the sea and settlement of fouling organisms on the cathodic surface (MDN 138). Despite the overwhelming evidence that bacteria and calcareous deposits coexist on cathodically protected surfaces, their interrelationships are not understood (Little and Wagner, 1993). The growth of calcareous deposits can interact in different ways with biofilms still present on the surfaces. Some bacteria can cause their dissolution. Some deposits can be disbanded from surfaces, and surface acidification can occur even under cathodic protection. Deposits lead to an increase of concentration polarization values and promote the effectiveness of cathodic protection. Porous deposits could become harmful with cathodic protection current blackout (Dexter and Lafontaine, 1989; Dickinson et al., 1996). The pattern of the curve infers that HE 20 would have offered required amount of protection to MDN 138. This has been further supported by the digital image of the surface of the coupon MDN 138 and visual observation after removal of biomass & corrosion products.
3.3.2 HE 20/MDN 250The OCP of HE 20 from the commencement (-0.877 V) of experiment, experienced a steady uptrend up to 16th day (-0.482 V), i.e. a positive shift of 305 V has been observed. There has been a downtrend from 17th day to 21st day, from thereupon a sharp increase up to 23rd day (-0.491 V). Many zigzag were observed between 23rd day to 142nd day, and at the end of the experiment the OCP was-0.726 V. The OCP of MDN 250 from the commencement (-0.468 V) of the experiment, experienced only minor fluctuations (-0.553 to-0.568V) throughout the study period. The galvanic potential of the couple HE 20/MDN 250 from the commencement (-0.746 V) to till the end (-0.826 V) of the experiment lies in the range of-0.881 to-0.838 V. No major fluctuations are observed throughout the study period. Hence HE 20 would have offered reasonable protection to MDN 250.
Applying, Cathodic Protection (CP) to metallic surfaces in seawater causes the formation of calcareous deposit on cathodic surface owing to the precipitation of calcium and magnesium salts, such as calcite, aragonite and brucite. These deposits increase the throwing power of CP, decreasing the cathodic current demand and consequently the cost of CP. Both the chemical composition and structure of the deposit greatly influence the effectiveness of CP. Many parameters affect this deposition, including flow velocity (Lee and Ambrose, 1986; Hack and Guanti, 1989), the calcium and magnesium content of seawater (Culberson, 1983), temperature (Kunjapur et al. 1985), dissolved organic matter (Edyvean, 1987) and pressure. However, interfacial pH (Culberson, 1983) is the most significant factor and any factor which affects this can modify the deposition kinetics, along with the nature and the stability of the scale.
The galvanic current values of the couple experienced a gradual and steep decrease between 2nd day (5.1 mA) and 7th day (1.584 mA), thereafter from 8th day (2.68 mA) to 10th day (4.15 mA) a positive surge; and from 12th day (3.9 mA) to 121st day (0.234 mA), a gradual & steady decline have been observed. The steady state values are observed between 282 days (0.073 mA) & 324 days (0.09 mA). Thus the onset of steady state current has been postponed to the tail end of the experiment (226-324 days); which can be accounted owing to the impairment caused to the calcareous deposit film, by the combined effect of turbulence in the sea and settlement of fouling organisms on the cathodic surface (MDN 250). The main consequence of biofilm formation on protected surfaces appears to be an increase in the current density necessary to polarize the metal to the protection potential. The presence of large numbers of cells on cathodically protected surfaces does mean that in the event that cathodic protection is intermittent, discontinuous, or discontinued, the corrosion attack due to the microorganisms will be more aggressive (Little and Wagner, 1993). The shape of the curve indicates that HE 20 would have offered reasonable protection to MDN 250. This has been further supported by the digital image of the surface of the coupon MDN 250 and visual observation after removal of biomass & corrosion products.
3.3.3 Potentiodynamic polarization scans of HE 20, MDN 138 & MDN 250Potentiodynamic polarization scans of freely corroded HE 20, MDN 138 & MDN 250 in natural seawater reveal the characteristic nature of the individual metal and the trend corroborates with open circuit potential values of the individual metal.
3.3.4 Extent of galvanic protection offered by HE 20 to MDN 138 & MDN 250The galvanic protection offered by HE 20 to MDN 138 & MDN 250 during the study period of 330 days amounts to 98% and 94%, respectively. This further reaffirms that the galvanic protection offered by HE 20 is continuous and effective, which has been evinced from the adherent nature of the calcareous deposit film comprising compounds such as CaCO3 (calcite, aragonite and vaterite), MgCO3 (magnesite), Mg(OH)2 (brucite) and MgO (brucite), despite the local disturbances by the combined effect of turbulence in the sea and settlement of fouling organisms on the cathodic surface.
4 ConclusionsThe galvanic protection offered by HE 20 to MDN 138 & MDN 250 in natural seawater amounts to 98% and 94%, respectively, revealing that the galvanic protection offered by HE 20 is continuous and effective, which has been further evinced from the adherent nature of the calcareous deposit film comprising compounds such as CaCO3 (calcite, aragonite and vaterite), MgCO3 (magnesite), Mg(OH)2 (brucite) and MgO (brucite), despite the local disturbances by the combined effect of turbulence in the sea and settlement of fouling organisms on the cathodic surface.
Acknowledgements:The authors wish to thank the Director, The CSIR-Central Electrochemical Research Institute, Karaikudi, for permission and encouragements. The authors also thank the Project Director, DRDL, Hyderabad for the financial support (Project No. SSP0709)
| Little BJ, Wagner PA, 1993. The interrelationship between marine biofouling and cathodic protection. Corrosion 1993. NACE, Houston, TX. Paper No. 525. |
| Brooks RR, Presley BJ, Kaplan IR, 1967. APDC-MIBK extraction system for the determination of trace elements in saline waters by atomic absorption. Talanta, 14, 806–816. |
| Culberson CH, 1983. Effect of seawater chemistry on the formation of calcareous deposits. Corrosion 1983. NACE, Houston, TX. Paper No. 61. |
| Dexter SC, Lafontaine JP, 1989. Effect of natural marine biofilms of galvanic corrosion. Corrosion, 54, 851–861. |
| Dickinson WH, Caccavo F, Lewandowski Z, 1996. The ennoblement of stainless steel by manganic oxide biofilms. Corrosion Science, 38, 1407–1422. DOI:10.1016/0010-938X(96)00031-5 |
| Edyvean RGJ, 1987. Interactios between microfouling and the calcareous deposit formd on cathodically protected steel in seawater. 6th Intl Cong Marine Corrosion and Fouling, Vol: Marine Biology, Athens, 469-475. |
| Hack HP, Guanti RJ, 1989. Effect of high flow on calcareous deposits and cathodic protection current density. Mat Perf., 28, 29–35. |
| Hosni E, El-Houd A, El-Shawesh F, 2008. A study on the corrosion behavior of aluminum alloys in seawater. Materials and Design, 29, 801–805. DOI:10.1016/j.matdes.2007.01.02 |
| Jafarzadeh K, ShahrabiT, Hosseini MG, 2008. EIS study on pitting corrosion of AA5083-H321 aluminum-magnesium alloy in stagnant 3.5% NaCl solution. Journal of Materials Science and Technology-Shenyang, 24(2), 215–219. |
| Kunjapur M, Hartt W, Smith S, 1985. Influence of temperature on calcareous deposition cathodically polarized steel in seawater.Corrosion 1985. NACE, Houston, Paper No. 316. |
| Lee R, Ambrose J, 1986. A hydrodynamical and chemical study of calcareous deposits. Corrosion 1986. NACE, Houston, TX. Paper No. 292. |
| Sarlak M, Shahrabi T, Zamanzade M, 2009. Investigation of calcareous deposits formation on copper and 316L stainless steel under cathodic polarization in artificial seawater. Protection of Metals and Physical Chemistry of Surfaces, 45(2), 216–222. DOI:10.1134/S2070205109020166 |
| Strickland JDH, Parsons TR, 1972. A practical handbook of seawater analysis. Ottawa: Fisheries Research Board of Canada. |
| Trdan AU, Grum J, 2015. Investigation of corrosion behaviour of aluminium alloy subjected to laser shock peening without a protective coating. Advances in Materials Science and Engineering, 2015, Article ID 705306, 1-9.DOI: 10.1155/2015/705306 |
| Wan Nik WB, Sulaiman O, Eng Giap SG, Rosliza R, 2010a. Evaluation of inhibitive action of sodium benzoate on corrosion behaviour of AA6063 in seawater. International Journal of Technology, 1, 20–28. DOI:10.14716/ijtech.v1i1.39 |
| Wan NikWB, Sulaiman O, Ffadhli A, Rosliza R, 2010b. Corrosion behaviour of aluminum alloy in seawater. Proceedings of MARTEC 2010. The International Conference on Marine Technology 2010, BUET, Dhaka, Bangladesh, 175-180. |
| Wieng SM, Osvoll H, Gartland PO, 2007. Efficient cathodic protection to stainless steel small bore tubing. Corrosion 2007. NACE. Houston, Paper No. 07078. |



