2. 南京农业大学资源与环境科学学院环境工程系, 南京 210095
2. Department of Environmental Engineering, College of Resources and Environmental Sciences, Nanjing Agricultural University, Nanjing 210095
垃圾渗滤液是一种含有大量病原微生物和有毒有害物质的高浓度有机废水,对环境危害较大(董军等,2007;沈东升等,2003;刘婷等,2010).目前国内垃圾渗滤液主要采用生化加膜过滤来处理,但渗滤液中大量生物难降解的有机污染物以及高浓度氨氮对生物处理系统有较大的冲击和抑制作用,且膜过滤后的浓缩液仍是处理难点.因此,在生化处理前通常采用物化法,如混凝沉淀(Zheng et al.,2009)、活性炭吸附(Kurniawan and Lo,2009)、化学氧化(Wang et al.,2006;Cossu et al.,1998)等对垃圾渗滤液进行预处理.
高级氧化技术(Advanced oxidation technologies,AOTs)作为水处理领域的重要技术手段,已得到人们的广泛关注.大量研究证实,AOTs的机理在于具有强氧化性的羟基自由基(·OH)的产生(Zepp et al.,1992;Konstantinou et al.,2001;Okitsu et al.,2005).大部分AOTs将强氧化剂(如O3,O2或H2O2等)与催化剂(如过渡金属,铁或半导体粉末等)联合使用(Zhang et al.,2005).近几十年来,光催化氧化技术作为一种极具应用前景的水处理方法,成为环境领域的研究热点.典型的光催化氧化技术包括O3/UV,H2O2/UV,O3/H2O2/UV,H2O2/Fe2+,TiO2/UV,ZnO/UV等(Huang et al.,1993).研究表明,TiO2具有合适的能带结构、价格低廉、光催化性能好等优点,成为人们的研究热点(Nakamura et al.,2003;Uddin et al.,2007).除·OH外,过硫酸盐在热解、光解、辐射以及过渡金属活化等条件下也可产生具有强氧化性的·SO42-(Waldemer et al.,2007;Hori et al.,2007;Das et al.,1999),两种自由基最终将有机污染物降解为无机产物.Lopeza(2004)、Rivas(2004)和Zhang(2005)的研究表明,利用H2O2与Fe2+发生的Fenton反应可产生大量·OH,有效降低垃圾渗滤液中的有机污染物.若将传统Fenton反应置于紫外光照射条件下,紫外光的催化作用促使反应体系产生更多的·OH,污染物去除效果更佳(De-Morais and Zamora,2005;Kim and Vogelpohl,1998;Lau et al.,2002).尽管光催化均相Fenton反应可高效去除难降解有机污染物,但均相体系对pH要求较高,且反应后生成的含铁沉积物造成二次污染(Kim and Vogelpohl,1998;Pignatello et al.,2006),同时在一定程度上影响出水色度.因此,光催化异相Fenton反应逐渐受到人们的关注,其中黄铵铁矾、β-FeOOH为研究热点(Xu et al.,2013;Wei and Nan,2011).
虽然光催化氧化技术在环境领域的相关研究已有了大量报道,但以往的研究均为单一催化剂对污染物的去除效果,并未系统对比不同催化剂的处理效果.本文选取3种常规催化剂(TiO2、K2S2O8和Fenton试剂),同时引入新型环境材料——施氏矿物(Schwertmannite),比较了均相和异相光催化的效果差异,旨在选取垃圾渗滤液最佳光催化剂或催化剂组合,为今后光催化技术在垃圾渗滤液处理中的应用提供了相关理论依据.
2 材料与方法(Materials and methods) 2.1 供试材料渗滤液取自南京市轿子山生活垃圾填埋场,样品采用Avanti J-E型高速冷冻离心机(10000 r·min-1,10 min)去除悬浮性颗粒,并及时测定基本指标(见表 1),然后保存于4 ℃冰箱中待用(APHA et al.,1992).
| 表 1 供试垃圾渗滤液基本性质 Table 1 Prelimanary properties of the selected refuse leachate |
本试验探究了6种催化剂光化学降解垃圾渗滤液的最佳装载量,并对最佳装载条件下污染物降解效果进行对比研究.6种光催化剂分别为TiO2、K2S2O8、K2S2O8/Fe2+、施氏矿物、施氏矿物/H2O2、Fe2+/ H2O2.
具体试验方法:取若干有效容积约50 mL的石英反应管,分别放入一枚磁力搅拌子,并按试验梯度添加相应的光催化剂(固态),然后加入预先调好pH=2.5的垃圾渗滤液25 mL,根据试验需要选择性添加H2O2,最后将石英反应管放置于XPA-Ⅱ型光化学反应器中,开启磁力搅拌,在500 W汞灯照射下,25 ℃恒温反应2.5 h.待反应结束后,用滤纸过滤并收集滤液测定色度,用10 mol·L-1 NaOH或1 mol·L-1 H2SO4将滤液pH调至7.0左右测定氨氮,调至初始值8.5后测定TOC和COD.每个试验设置3个重复.
各催化剂装载量设置梯度如下:①TiO2:0、2、4、8、12、16、20 g·L-1;②K2S2O8:0、2、4、8、12、16、20 g·L-1;③K2S2O8/Fe2+:0、2、4、8、12、16、20 g·L-1 K2S2O8,其中M(K2S2O8)/ M(Fe2+)=1.5;④施氏矿物:0、2、4、8、12、16、20 g·L-1;⑤施氏矿物/H2O2:0、2、4、8、12、16、20 g·L-1 施氏矿物,其中m(施氏矿物)/V(H2O2)= 1 g·mL-1;⑥Fe2+/ H2O2:0、0.005、0.01、0.025、0.05、0.10、0.20 mol·L-1 Fe2+,其中M(H2O2)/ M(Fe2+)=1.5.上述Fe2+均以FeSO4·7H2O形式投加.其中,试验设置⑥中,FeSO4·7H2O的装载量按本试验化学合成施氏矿物(Fe8O8(OH)4.98(SO4)1.92)中Fe元素的质量百分比进行换算,确定FeSO4·7H2O装载量约为31.44 g·L-1.为考察紫外光对渗滤液中污染物的降解能力,各试验中均设置不添加任何催化剂的空白试验.
2.3 测定方法分别使用精密pH计(FE20型,METTLER TOLEDO,Switzerland)和电导率仪(FE30型,METTLER TOLEDO,Switzerland)测定pH和电导率;色度采用稀释法测定(奚旦立等,2004);COD采用重铬酸钾-分光光度法测定(周俊等,2009);使用总有机碳/总氮分析仪(multi N/C 3100型,Analytik Jena,Germany)测定TOC含量;氨氮、菌落总数以及总大肠菌群和粪大肠菌群分别采用滴定法、平皿计数法和多管发酵法测定(国家环保总局,2006).
3 结果与讨论(Results and discussion) 3.1 TiO2光催化处理垃圾渗滤液的效果TiO2多相光催化能有效降解多种对环境有害的污染物,使有害物质矿化为CO2、H2O及其它无机小分子物质(沈伟韧等,1998),贾陈忠等(2013)研究发现,TiO2/UV光催化氧化对垃圾渗滤液具有较好的降解效果.TiO2光催化反应过程主要包括以下几个方面(张金龙,2004;张立德和牟季美,2001):TiO2粒子被光照后,若所受能量大于或等于禁带宽度,价带上的电子便被光子激发,进而发生跃迁到达导带,此时价带上产生空穴,TiO2内部产生电子-空穴对(e--h+);其中,e-具有强还原性,可诱发还原反应,h+则具有强氧化性,可氧化TiO2表面的H2O和OH-,生成另一种强氧化剂羟基自由基(·OH),该自由基可将TiO2表面的有机物氧化分解,亦可进入液相中,将液相中的有机物质完全氧化为CO2.
由图 1可以看出,随着TiO2加入量的增加,COD和TOC去除率显著上升,在TiO2加入量为16 g·L-1时达到最大值,分别为25.6%和26.9%,此时色度由40倍降至5倍.继续增加TiO2的量,色度基本保持不变,COD和TOC去除率呈下降趋势.可见,TiO2的添加量应适宜,若添加量过少,则TiO2被紫外光激发后产生的电子-空穴对(e--h+)不足,有效光能无法完全转化为化学能,使得污染物去除效果不佳;若添加过量,过多的TiO2将导致反应体系透光度下降,造成光散射,进而降低了紫外光的有效利用率(Carlson et al.,2015;杨运平等,2006).
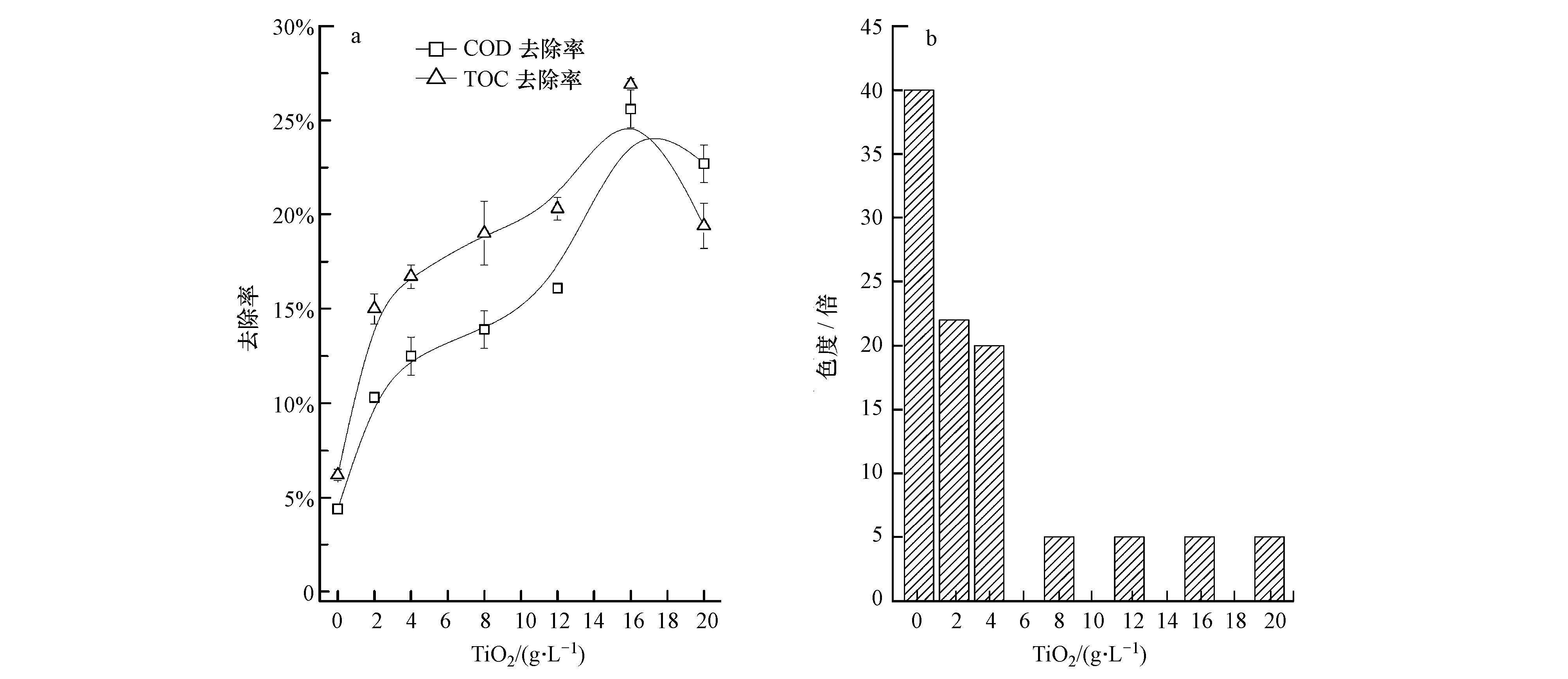 |
| 图 1 光催化条件下TiO2添加量对垃圾渗滤液各指标的影响 Fig. 1 Impact of TiO2 on COD and TOC removal efficiency(a)and chromaticity(b)of refuse leachate |
过硫酸盐在水中电离产生S2O82-,其标准氧化还原电位(E0 =+ 2.01 V)(Liang et al.,2003),高于MnO4-(E0= +1.68 V)和H2O2(E0= +1.70 V),与O3(E0 = +2.07 V)相近.过硫酸盐分子中所含的过氧基(—O—O—)具有较强的氧化性,但在常温常态下反应速度较慢,对有机物的氧化效果不明显,但在紫外光的激发下,过硫酸盐活化分解为·SO-4(House,1962),该自由基的氧化还原电位E0=+2.6 V,与·OH(E0=+2.8 V)相近(Liang et al.,2003),理论上可矿化大部分有机物.
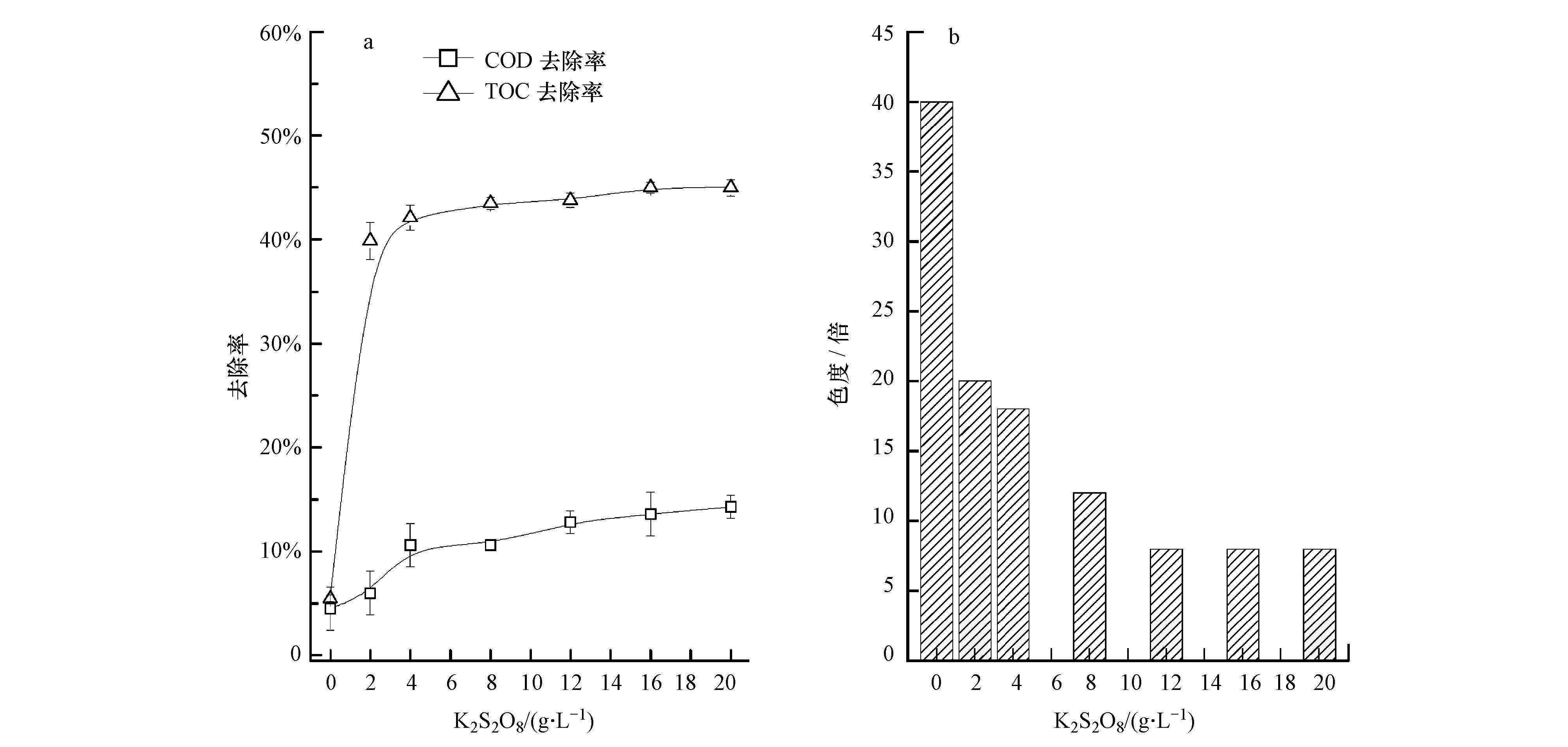 |
| 图 2 光催化条件下K2S2O8添加量对垃圾渗滤液各指标的影响 Fig. 2 Impact of K2S2O8 on COD and TOC removal efficiency(a)and chromaticity(b)of refuse leachate |
图 2为K2S2O8添加量对垃圾渗滤液中污染物去除效果的影响.随着K2S2O8添加量的增加,有机污染物的去除率呈先上升后逐渐平稳的趋势.在K2S2O8添加量为4 g·L-1时,COD和TOC去除率分别为10.6%和42.1%,色度为18倍;当K2S2O8的添加量达到20 g·L-1时,COD和TOC去除率分别为14.3%和45.0%,此时色度为8倍.由此可知,在一定范围内增加K2S2O8的加入量可较高程度上提高光催化降解污染物的效率,但是过多的K2S2O8水解产生的S2O82-氧化还原电位远低于·SO-4,大量的S2O2-8会与有机污染物争夺·SO-4,导致污染物去除率无法显著提高.由此可知,K2S2O8光催化可在一定程度上去除有机污染物,降低渗滤液色度.
3.3 K2S2O8/Fe2+光催化处理垃圾渗滤液的效果目前关于活化K2S2O8的研究多为添加金属离子、紫外光照和加热等.例如,Oh等(2009)研究发现,升高温度可提高过硫酸盐对聚乙烯醇的降解率,在20 ℃条件下反应120 min,聚乙烯醇降解率仅为30%,而在60 ℃和80 ℃条件下,聚乙烯醇分别在30 min和10 min时被完全氧化.由本文3.2节可知,K2S2O8在紫外光照条件下对垃圾渗滤液中污染物的去除效果并不明显,因此,本试验联合Fe2+和紫外光照共同活化K2S2O8的方式进行比较.
由图 3a可知,在K2S2O8光催化体系中添加Fe2+(M(K2S2O8)/(Fe2+)=1.5)后,COD、TOC去除率较不添加Fe2+时有所提高,在m(K2S2O8)= 20 g·L-1分别由14.3%和45.0%提高到17.9%和64.1%.但是由于Fe2+的介入,出水色度受Fe3+影响,由初始的40倍增加至450倍(图 3b).
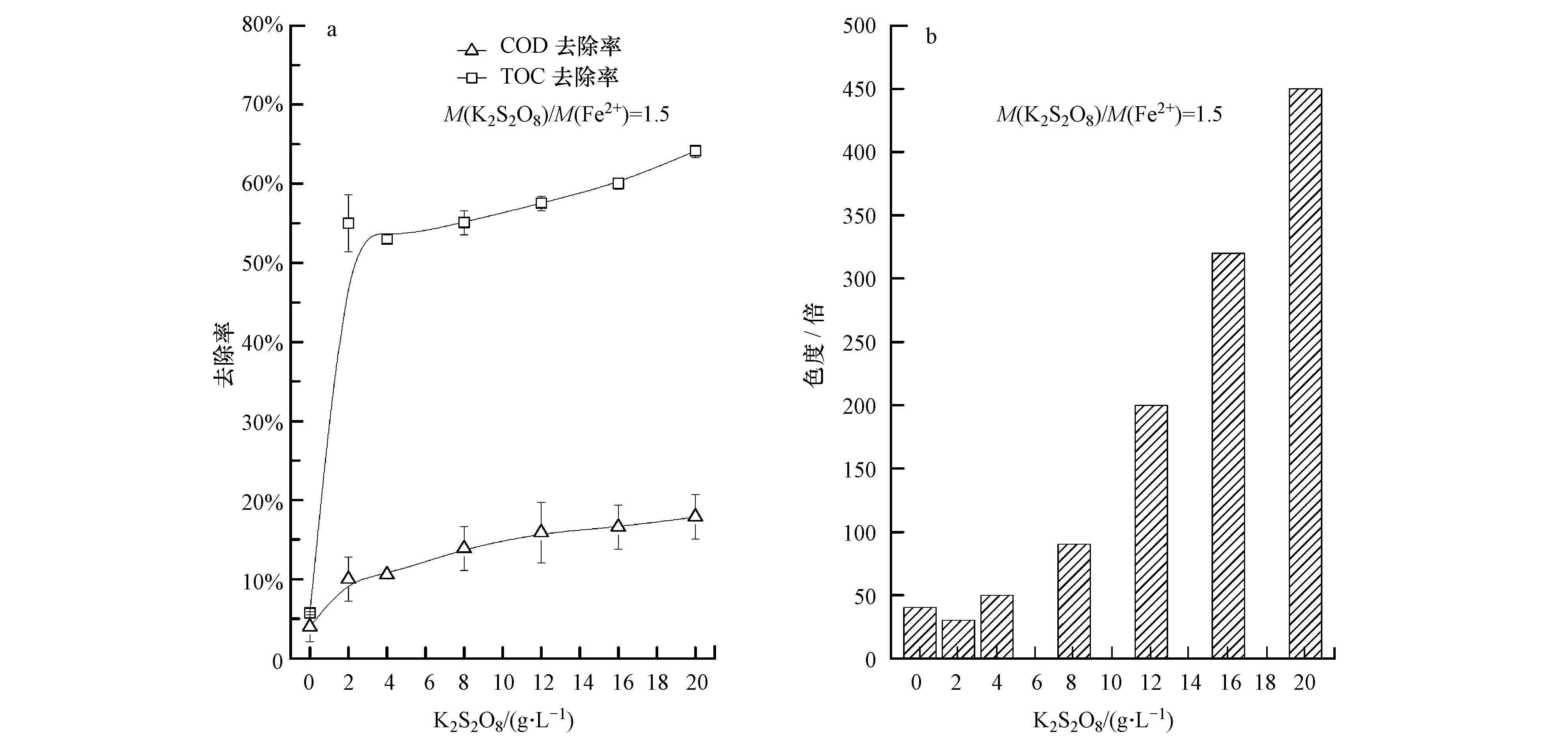 |
| 图 3 光催化条件下K2S2O8/Fe2+添加量对垃圾渗滤液各指标的影响 Fig. 3 Impact of K2S2O8/ Fe2+ on COD and TOC removal efficiency(a)and chromaticity(b)of refuse leachate |
Fenton反应可高效去除COD,但Fenton反应后产生的大量含铁污泥难以沉降去除(Kim and Vogelpohl,1998;Pignatello et al.,2006).有研究表明,传统Fenton可通过紫外光照进行改进,进而减少含铁污泥的产生(Kavitha and Palanivelu,2004;Brillas et al.,1998).另有大量研究证明,光催化Fenton反应对污染物的去除效率高于传统Fenton反应(Bali et al.,2003;Ghaly et al.,2001;Sarasa et al.,2006;Kim and Huh,1997;Kim and Vogelpohl,1998).
本试验将500 W汞灯与传统Fenton反应相结合,对垃圾渗滤液进行光催化Fenton处理.由图 4a可知,当M(H2O2)/M(Fe2+)=1.5时,随着Fe2+浓度的增加,COD与TOC去除率呈逐渐上升的趋势,在M(Fe2+)=0.2 mol·L-1时达到最大,分别为46.5%和78.1%;出水色度与本文3.3节的结果相似,均受Fe2+影响,在COD与TOC去除率达到最大时,色度达到175倍(图 4b),远高于渗滤液原始色度(40倍).
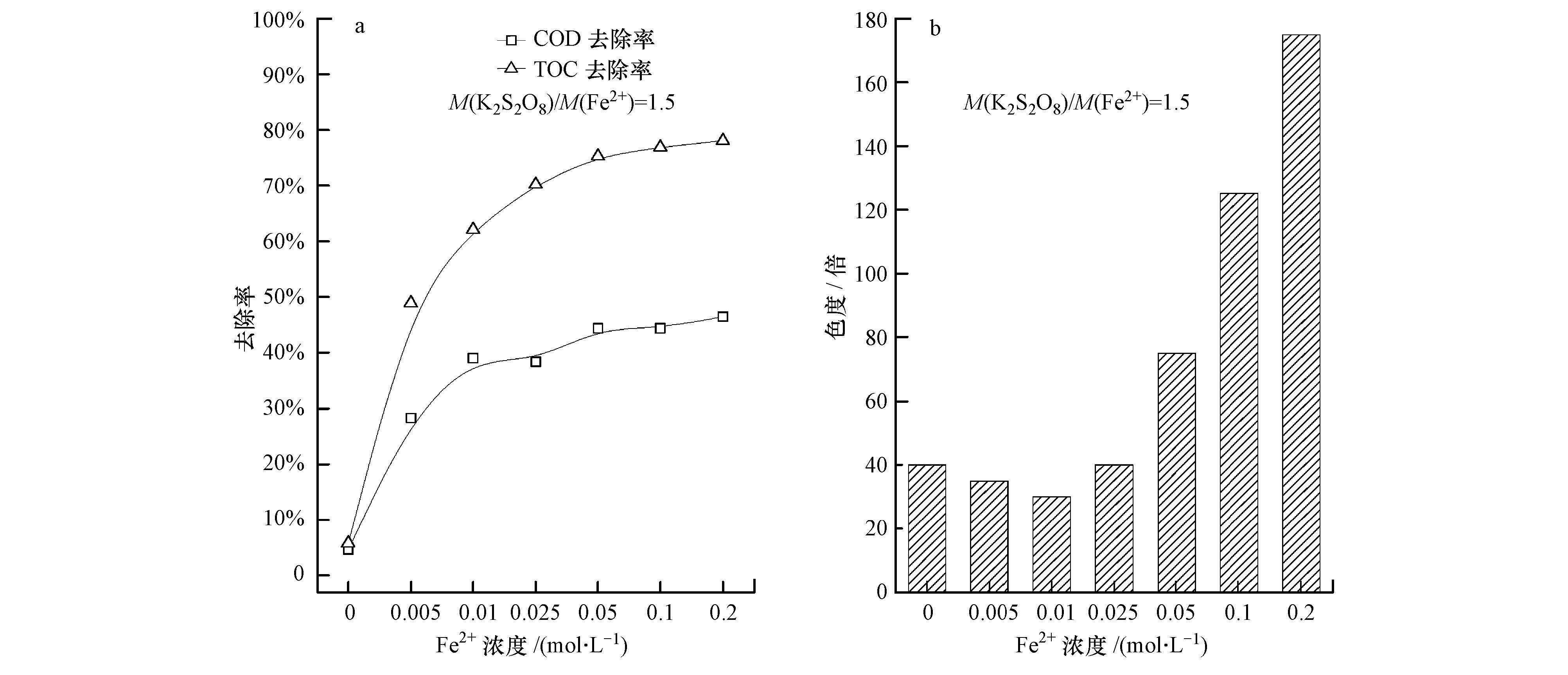 |
| 图 4 光催化条件下H2O2/Fe2+添加量对垃圾渗滤液各指标的影响 Fig. 4 Impact of H2O2/Fe2+ on COD and TOC removal efficiency(a)and chromaticity(b)of refuse leachate |
施氏矿物是Bigham等在酸性矿山废水中的沉积物以及污泥生物沥浸环境中发现的一种次生羟基硫酸铁矿物(Bigham et al.,1990;Liao et al.,2009).由于施氏矿物具有较大的比表面积,可吸附污染水体中的重金属离子(Jönsson et al.,2005;Liao et al.,2011;李浙英等,2011),亦可吸附渗滤液中的有机污染物;且施氏矿物结构中含有大量羟基,在紫外光照射下,存在产生·OH降解有机污染物的可能性.
图 5为施氏矿物添加量对渗滤液中污染物去除效果的影响.由图 5a易知,COD和TOC去除率随施氏矿物添加量的增加而逐渐提高,在m(施氏矿物)=20 g·L-1时,分别达到28.6%和52.9%;受Fe3+影响,色度去除效果低于ZnO、TiO2、K2S2O8光催化的去除效果,最低出水色度为15倍(图 5b).
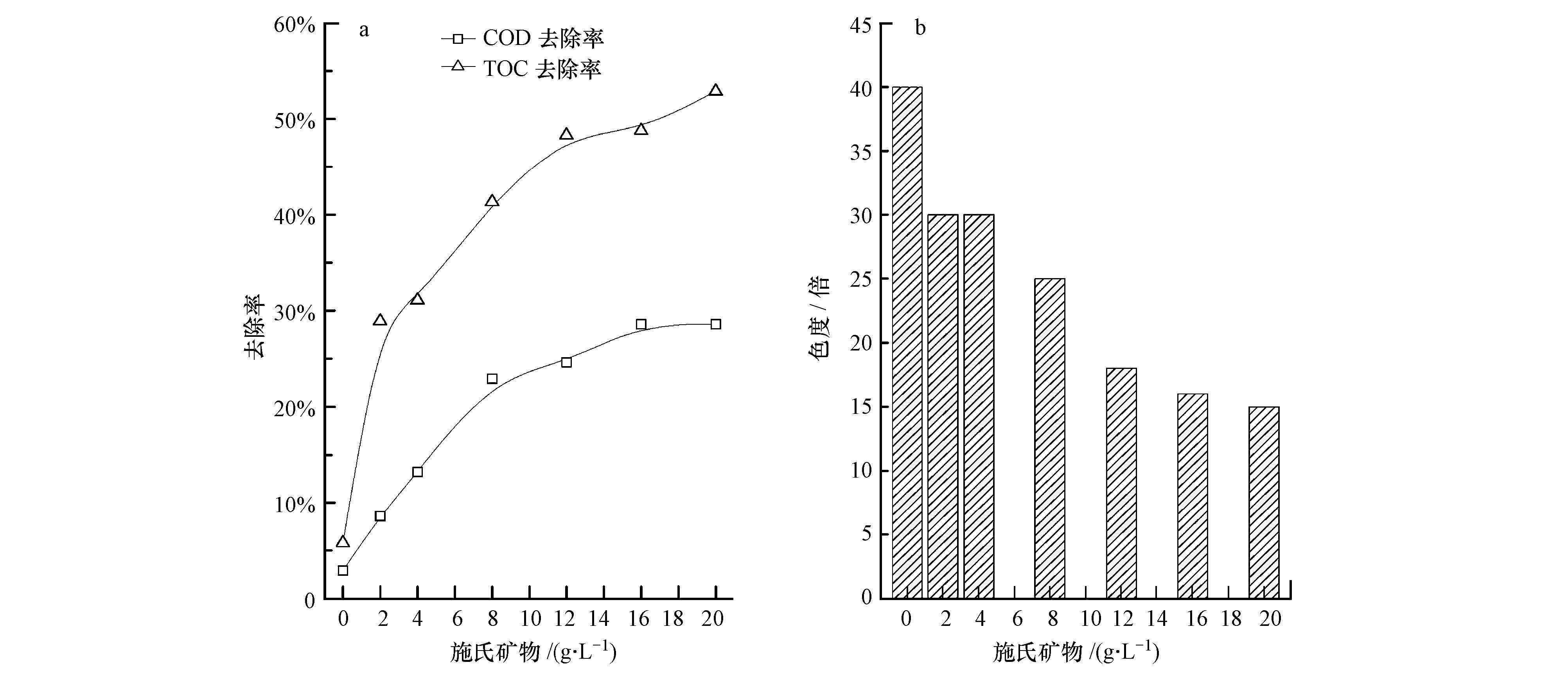 |
| 图 5 光催化条件下施氏矿物添加量对垃圾渗滤液各指标的影响 Fig. 5 Impact of schwertmannite on COD and TOC removal efficiency(a)and chromaticity(b)of refuse leachate |
由本文3.4节试验结果可知,传统Fenton反应在紫外光照条件下可有效去除垃圾渗滤液中的有污染物,但出水色度远高于初始色度,且反应后产生的Fe(OH)3絮状物难以沉淀去除,造成二次污染.另有研究表明,传统光Fenton反应为均相反应,Fe2+对反应体系的pH值要求较高,只在狭窄的pH范围内(2.5~3.0)具有高的氧化效率(Gumy et al.,2005;Huang and Huang,2008).以上两个缺点制约着均相光Fenton反应的实际应用.若将Fe3+固定在固相中,不仅避免了含铁污泥的产生,还在保证了高氧化效率的同时拓宽了光Fenton反应的pH范围.因此本试验利用化学合成的施氏矿物,在紫外光和H2O2共存的条件下,采用异相光Fenton法处理垃圾渗滤液.
污染物去除效果随施氏矿物添加量的变化如图 6所示.当m(施氏矿物)=20 g·L-1时,COD和TOC去除率分别达到41.6%和76.0%(图 6a),与传统光Fenton反应的去除效果相当(图 4a),但异相光Fenton处理后,垃圾渗滤液的出水色度可达到0(图 6b),且没有含铁污泥产生.根据Xu等(2013)分析认为,其机理在于有机污染物被吸附在施氏矿物表面,在紫外光照射下,施氏矿物与H2O2发生系列光化学反应,该过程产生的大量羟基自由基(·OH)可直接将难降解污染物氧化为简单物质,且在一定范围内提高施氏矿物的装载量可增加·OH的产生量而去除污染物.
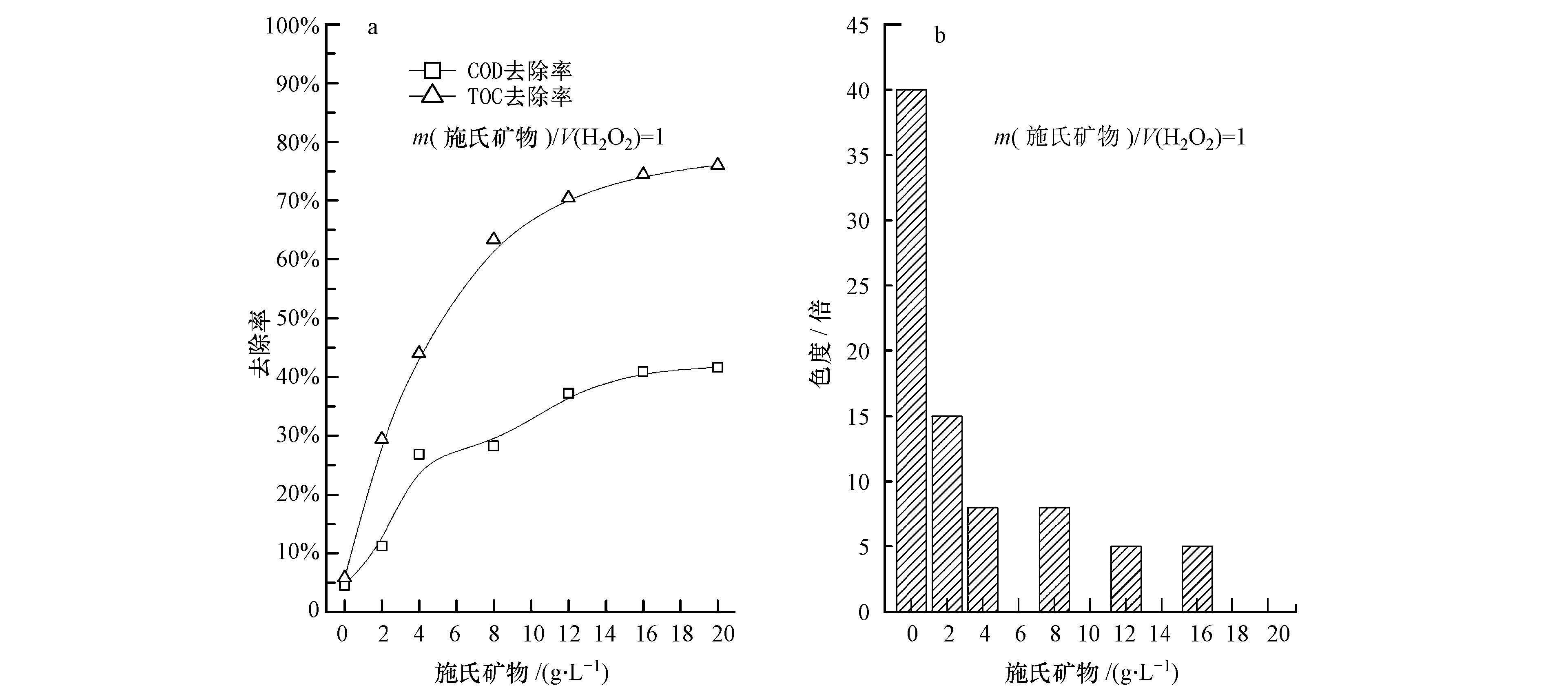 |
| 图 6 光催化条件下施氏矿物/H2O2添加量对垃圾渗滤液各指标的影响 Fig. 6 Impact of schwertmannite/ H2O2on COD and TOC removal efficiency(a)and chromaticity(b)of refuse leachate |
1) 采用TiO2、K2S2O8、K2S2O8/Fe2+、施氏矿物、施氏矿物/H2O2、Fe2+/ H2O2 作为催化剂来光化学降解垃圾渗滤液,COD去除率分别为25.5%、14.3%、17.9%、28.6%、41.6%、46.5%,TOC去除率分别为26.9%、45.0%、64.1%、52.9%、76.0%、78.1%,色度分别由40倍变为5倍、8倍、450倍、15倍、0、175倍.
2) 施氏矿物/H2O2/UV与Fe2+/H2O2/UV对COD和TOC的去除效果相当,但后者产生大量难以沉降去除的Fe(OH)3絮状物,且处理后渗滤液的色度明显增加.相对而言,施氏矿物/H2O2作为催化剂更适用于垃圾渗滤液光化学处理.
| [1] | APHA,AWWA, WEF. 2012. Standard Method for the Examination of Water and Wastewater[M]. Amer Public Health Assn, Washington, DC, USA |
| [2] | Bali U, Çatalkaya E Ç, Şengül F. 2003. Photochemical degradation and mineralization of phenol: a comparative study[J]. Journal of Environmental Science and Health, Part A , 38 (10) : 2259–2275. DOI:10.1081/ESE-120023373 |
| [3] | Brillas E, Mur E, Sauleda R, et al. 1998. Aniline mineralization by AOP's: anodic oxidation, photocatalysis, electro-Fenton and photoelectro-Fenton processes[J]. Applied Catalysis B: Environmental , 16 (1) : 31–42. DOI:10.1016/S0926-3373(97)00059-3 |
| [4] | Bigham J M, Schwertmann U, Carlson L. 1990. A poorly crystallized oxyhydroxysulfate of iron formed by bacterial oxidation of Fe (II) in acid mine waters[J]. Geoehirn Cosmochim Aeta , 54 (10) : 2743–2758. DOI:10.1016/0016-7037(90)90009-A |
| [5] | Cossu R, Polcaro A M, Lavagnolo M C, et al. 1998. Electrochemical treatment of landfill leachate: Oxidation at Ti/PbO2 and Ti/SnO2 anodes[J]. Environmental Science Technology , 32 (22) : 3570–3573. DOI:10.1021/es971094o |
| [6] | Das S, Kamat P V, Padmaja S, et al. 1999. Free radical induced oxidation of the azo dye Acid Yellow 9[J]. Journal of the Chemical Society, Perkin Transactions 2 , 999 (6) : 1219–1223. |
| [7] | De-Morais J L, Zamora P P. 2005. Use of advanced oxidation processes to improve the biodegradability of mature landfill leachates[J]. Journal of Hazardous Materials , 123 (1/3) : 181–186. |
| [8] | 董军, 赵勇胜, 张伟红, 等.2007. 渗滤液中有机物在不同氧化还原带中的降解机理与效率研究[J]. 环境科学 , 2007, 28 (9) : 2041–2045. |
| [9] | Ghaly M Y, Härtel G, Mayer R, et al. 2001. Photochemical oxidation of p-chlorophenol by UV/H2O2 and photo-Fenton process. A comparative study[J]. Waste Manage , 21 : 41–47. DOI:10.1016/S0956-053X(00)00070-2 |
| [10] | Gumy D, Fernández-Ibáñez P, Malato S, et al. 2005. Supported Fe/C and Fe/Nafion/C catalysts for the photo-Fenton degradation of Orange II under solar irradiation[J]. Catalysis Today , 101 (3/4) : 375–382. |
| [11] | 国家环保总局. 2006. 水和废水监测分析方法[M]. 第4版. 北京: 中国环境科学出版社: 211 -213. |
| [12] | House D A. 1962. Kinetics and mechanism of oxidations by peroxydisulfate[J]. Chemical Reviews , 62 (3) : 185–203. DOI:10.1021/cr60217a001 |
| [13] | Huang C P, Dong C, Tang Z. 1993. Advanced chemical oxidation: Its present role and potential future in hazardous waste treatment[J]. Waste Manage , 13 (5/7) : 361–377. |
| [14] | Huang C P, Huang Y H. 2008. Comparison of catalytic decomposition of hydrogen peroxide and catalytic degradation of phenol by immobilized iron oxides[J]. Applied Catalysis A: General , 346 (1/2) : 140–148. |
| [15] | Hori H, Yamamoto A, Koike K, et al. 2007. Persulfate-induced photochemical decomposition of a fluorotelomer unsaturated carboxylic acid in water[J]. Water Research , 41 (13) : 2962–2968. DOI:10.1016/j.watres.2007.02.033 |
| [16] | J nsson J, Persson P, Sj berg S, et al. 2005. Schwertmannite precipitated from acid mine drainage: phase transformation, sulfate release and surface properties[J]. APPIGeoehem , 20 : 179–191. |
| [17] | 贾陈忠, 刘松, 张彩香, 等.2013. 光催化氧化降解垃圾渗滤液中溶解性有机物[J]. 环境工程学报 , 2013, 7 (2) : 451–456. |
| [18] | Kim Y K, Huh I R. 1997. Enhancing biological treatability of landfill leachate by chemical oxidation[J]. Environmental Engineering Science , 14 (1) : 73–79. DOI:10.1089/ees.1997.14.73 |
| [19] | Kurniawan T A, Lo W H. 2009. Removal of refractory compounds from stabilized landfill leachate using an integrated H2O2 oxidation and granular activated carbon (GAC) adsorption treatment[J]. Water Resource , 43 (16) : 4079–4091. |
| [20] | Kavitha V, Palanivelu K. 2004. The role of ferrous ion in Fenton and photo-Fenton processes for the degradation of phenol[J]. Chemosphere , 55 (9) : 1235–1243. DOI:10.1016/j.chemosphere.2003.12.022 |
| [21] | Konstantinou I K, Sakellarides T M, Sakkas V A, et al. 2001. Photocatalytic degradation of selected s-Triazine herbicides and organophosphorus insecticides over aqueous TiO2 suspensions[J]. Environmental Science &Technology , 35 (2) : 398–405. |
| [22] | Kim S M, Vogelpohl A. 1998. Degradation of Organic Pollutants by the Photo-Fenton-Process[J]. Chemical Engineering &Technology , 21 (2) : 187–191. |
| [23] | Liang C J, Bruell C J, Marley M C, et al. 2003. Thermally activated persulfate oxidation of trichloroethylene (TCE) and 1,1,1-trichloroethane (TCA) in aqueous systems and soil slurries[J]. Soil and Sediment Contamination , 12 (2) : 207–228. DOI:10.1080/713610970 |
| [24] | Liao Y H, Zhou L X, Bai S Y, et al. 2009. Occurrence of biogenic schwertmannite in sludge bioleaching environments and its adverse effect on solubilization of sludge-borne metals[J]. Applied Geochemistry , 24 : 1739–1746. DOI:10.1016/j.apgeochem.2009.05.003 |
| [25] | Liao Y H, Liang J R, Zhou L X. 2011. Adsorptive removal of As(III) by biogenic schwertmannite from simulated As-contaminated groundwater[J]. Chemosphere , 83 : 295–301. DOI:10.1016/j.chemosphere.2010.12.060 |
| [26] | Lopeza A, Paganoa M, Volpe A, et al. 2004. Fenton's pre-treatment of mature landfill leachate[J]. Chemosphere , 54 (7) : 1005–1010. DOI:10.1016/j.chemosphere.2003.09.015 |
| [27] | Lau I W C, Wang P, Chiu S S T, et al. 2002. Photoassisted fenton oxidation of refractory organics in UASB-pretreated leachate[J]. Journal of Environmental Sciences , 14 (3) : 388–392. |
| [28] | 刘婷, 陈朱蕾, 唐素琴, 等.2010. 不同填埋时间、不同季节的垃圾渗滤液生物毒性[J]. 环境科学 , 2010, 31 (2) : 541–546. |
| [29] | Lutterbeck C A, Wilde M L, Baginska E, et al. 2015. Degradation of 5-FU by means of advanced (photo)oxidation processes: UV/H2O2, UV/Fe2+/H2O2 and UV/TiO2-Comparison of transformation products, ready biodegradability and toxicity[J]. Science of The Total Environment : 527-528–232-245. |
| [30] | 李浙英, 梁剑茹, 柏双友, 等.2011. 生物与化学成因施氏矿物吸附去除水中As(III)效果的比较研究[J]. 环境科学学报 , 2011, 31 (5) : 912–918. |
| [31] | Nakamura R, Imanishi A, Murakoshi K, et al. 2003. In situ FTIR studies of primary intermediates of photocatalytic reactions on nanocrystalline TiO2 films in contact with aqueous solutions[J]. Journal of the American Chemical Society , 125 (24) : 7443–7450. DOI:10.1021/ja029503q |
| [32] | Okitsu K, Iwasaki K, Yobiko Y, et al. 2005. Sonochemical degradation of azo dyes in aqueous solution: a new heterogeneous kinetics model taking into account the local concentration of OH radicals and azo dyes[J]. Ultrason Sonochem , 12 (4) : 255–262. DOI:10.1016/j.ultsonch.2004.01.038 |
| [33] | Oh S Y, Kim H W, Park J M, et al. 2009. Oxidation of polyvinyl alcohol by persulfate activated with heat, Fe2+, and zero-valent iron[J]. Journal of Hazardous Materials , 168 (1) : 346–351. DOI:10.1016/j.jhazmat.2009.02.065 |
| [34] | Pignatello J J, Oliveros E, MacKay A. 2006. Advanced oxidation processes for organic contaminant destruction based on the Fenton reaction and related chemistry[J]. Critical Reviews in Environmental Science and Technology , 36 (1) : 1–84. DOI:10.1080/10643380500326564 |
| [35] | Rivas F J, Beltrán F, Carvalho F, et al. 2004. Stabilized leachates: sequential coagulation-flocculation+chemical oxidation process[J]. Journal of Hazardous Materials , 116 (1/2) : 95–102. |
| [36] | Sarasa J, Llabrés T, Ormad P, et al. 2006. Characterization and photo-Fenton treatment of used tires leachate[J]. Journal of Hazardous Materials , 136 (3) : 874–881. DOI:10.1016/j.jhazmat.2006.01.023 |
| [37] | 沈东升, 何若, 刘宏远. 2003. 生活垃圾填埋生物处理技术[M]. 北京: 化学工业出版社 . |
| [38] | 沈伟韧, 赵文宽, 贺飞, 等.1998. TiO2光催化反应及其在废水处理中的应用[J]. 化学进展 , 1998, 10 (4) : 349–361. |
| [39] | Uddin M M, Hasnat M A, Samed A J, et al. 2007. Influence of TiO2 and ZnO photocatalysts on adsorption and degradation behaviour of Erythrosine[J]. Dyes Pigments , 75 (1) : 207–212. DOI:10.1016/j.dyepig.2006.04.023 |
| [40] | Wei C Z, Nan Z D. 2011. Effects of experimental conditions on one-dimensional single-crystal nanostructure of β-FeOOH[J]. Materials Chemistry and Physics , 127 (1/2) : 220–226. |
| [41] | Wang F Q, Smith D W, El-Din M G. 2006. Aged raw landfill leachate: Membrane fractionation, O3 only and O3/H2O2 oxidation, and molecular size distribution analysis[J]. Water Resource , 40 (3) : 463–474. |
| [42] | Waldemer R H, Tratnyek P G, Johnson R L, et al. 2007. Oxidation of chlorinated ethenes by heat-activated persulfate: Kinetics and products[J]. Environmental Science &Technology , 41 (3) : 1010–1015. |
| [43] | Xu Z H, Liang J R, Zhou L X. 2013. Photo-Fenton-like degradation of azo dye methyl orange using synthetic ammonium and hydronium jarosite[J]. Journal of Alloys and Compounds , 546 (5) : 112–118. |
| [44] | 奚旦立, 孙欲生, 刘秀英. 2004. 环境监测[M]. 北京: 高等教育出版社 . |
| [45] | 杨运平, 唐金晶, 方芳, 等.2006. UV/TiO2/Fenton光催化氧化垃圾渗滤液的研究[J]. 中国给水排水 , 2006, 22 (7) : 34–37. |
| [46] | Zepp R G, Faust B C, Hoigne J. 1992. Hydroxyl radical formation in aqueous reactions (pH 3-8) of iron (Ⅱ) with hydrogen peroxide: the photo-Fenton reaction[J]. Environmental Science &Technology , 26 (2) : 313–319. |
| [47] | Zhang H, Choi H J, Huang C P. 2005. Optimization of Fenton process for the treatment of landfill leachate[J]. Journal of Hazardous Materials , 125 (1/3) : 166–174. |
| [48] | Zheng Z, Zhang H, He P J, et al. 2009. Co-removal of phthalic acid esters with dissolved organic matter from landfill leachate by coagulation and flocculation process[J]. Chemosphere , 75 (2) : 180–186. DOI:10.1016/j.chemosphere.2008.12.011 |
| [49] | 张金龙. 2004. 光催化[M]. 上海: 华东理工大学出版社 . |
| [50] | 张立德, 牟季美. 2001. 纳米材料和纳米结构[M]. 北京: 科学出版社 . |
| [51] | 周俊, 杨新萍, 周立祥.2009. 自配消解液分光光度法测定污水中的COD[J]. 环境工程学报 , 2009, 3 (11) : 1956–1960. |
 2016, Vol. 36
2016, Vol. 36


