Relationship between immunoinflammatory indicators derived from complete blood count and severity of Mycoplasma pneumoniae pneumonia in children of different ages
-
摘要:
目的 探讨9种全血细胞计数来源的免疫炎症指标与不同年龄段儿童肺炎支原体肺炎(MPP)重症化的关系。 方法 选择2023年7月1日至2024年12月31日在海军军医大学第一附属医院儿科病房住院治疗的2 132例MPP患儿为研究对象,分为重症MPP(SMPP)组和非重症MPP(NSMPP)组,按照年龄、性别1∶1匹配后根据年龄将患儿分为1~6岁、>6~16岁2个亚组进行分析,收集并比较各组的基本资料、实验室检查结果及全血细胞计数来源的免疫炎症指标。采用单因素和多因素Cox比例风险回归模型分析SMPP的影响因素,利用ROC曲线评估差异有统计学意义的指标对SMPP的预测价值。 结果 SMPP患儿220例,占MPP的10.3%。1~6岁患儿中,与NSMPP组相比,SMPP组患儿的住院时间长,血小板(PLT)计数、血小板与淋巴细胞比值(PLR)、中性粒细胞与淋巴细胞比值、衍生的中性粒细胞与淋巴细胞比值和系统性免疫炎症指数高(均P<0.05),其中PLR是患儿进展至SMPP的独立危险因素(OR=1.010,95%CI 1.003~1.018,P=0.007),PLR预测SMPP的AUC为0.635(95%CI 0.560~0.711,P<0.001),最佳截断值为125.04,此时对应的灵敏度为57.7%、特异度为70.2%;以最佳截断值为界将所有患儿分为低PLR组和高PLR组,高PLR组的重症率高于低PLR组[65.9%(60/91)vs 37.6%(44/117),P<0.001],以四分位数为界将所有患儿分为Q1~Q4组,Q4组的重症率(71.2%,37/52)高于Q1~Q3组(均P<0.05)。>6~16岁患儿中,与NSMPP组相比,SMPP组患儿的PLT和PLR高(均P<0.05),但都不是患儿进展至SMPP的独立危险因素(均P>0.05);以最佳截断值(137.03)为界将所有患儿分为低PLR组和高PLR组,高PLR组的重症率高于低PLR组[57.0%(77/135)vs 40.2%(39/97),P=0.011],以四分位数为界将所有患儿分为Q1~Q4组,Q4组的重症率(65.5%,38/58)高于Q1~Q3组(均P<0.05)。 结论 全血细胞计数来源的免疫炎症指标,尤其是PLR,对于预测不同年龄段MPP患儿的重症化具有一定的应用价值。 Abstract:Objective To investigate the relationship between 9 immunoinflammatory indicators derived from complete blood count and the severity of Mycoplasma pneumoniae pneumonia (MPP) in children of different ages. Methods Totally 2 132 children with MPP who were hospitalized in the Department of Pediatrics of The First Affiliated Hospital of Naval Medical University from Jul. 1, 2023, to Dec. 31, 2024 were enrolled, and were assigned to severe MPP (SMPP) or non-severe MPP (NSMPP) groups. According to age and gender 1∶1 matching, the children were assigned to 2 subgroups according to age (1-6 years old and >6-16 years old). The basic data, laboratory examination and immunoinflammatory indicators from complete blood count of each group were collected and compared. The influencing factors of SMPP were analyzed by univariate and multivariate Cox proportional hazards regression models. Receiver operating characteristic curves were used to analyze the predictive value of indicators that showed statistically significant differences for SMPP. Results There were 220 patients with SMPP, accounting for 10.3% of MPP. In children aged 1-6 years, compared with the NSMPP group, the SMPP group had a longer hospital stay, higher platelet (PLT) count, platelet-to-lymphocyte ratio (PLR), neutrophil-to-lymphocyte ratio, derived neutrophil-to-lymphocyte ratio and systemic immune-inflammation index (all P<0.05). PLR was an independent risk factor for SMPP (odds ratio=1.010, 95% confidence interval [CI]1.003-1.018, P=0.007). The area under curve predicted by PLR for SMPP was 0.635 (95%CI 0.560-0.711, P<0.001), the best cut-off value was 125.04, and the corresponding sensitivity and specificity were 57.7% and 70.2%, respectively. All the children were assigned to low PLR group or high PLR group using the best cut-off value as the boundary, and the severe disease rate in the high PLR group was significantly higher than that in the low PLR group (65.9%[60/91] vs 37.6%[44/117], P<0.001). All the children were assigned to Q1-Q4 groups by quartile, and the severe disease rate of the Q4 group (71.2%, 37/52) was significantly higher than that of the Q1-Q3 group (all P<0.05). In children aged >6-16 years, compared with the NSMPP group, the PLT and PLR in the SMPP group were higher (both P<0.05), but neither was an independent risk factor. All the children were assigned to low PLR group or high PLR group using the best cut-off value (137.03) as the boundary, and the severe disease rate in the high PLR group was significantly higher than that in the low PLR group (57.0%[77/135] vs 40.2%[39/97], P=0.011). All the children were assigned to Q1-Q4 groups by quartile, and the severe disease rate of the Q4 group (65.5%, 38/58) was significantly higher than that of the Q1-Q3 group (all P<0.05). Conclusion The immunoinflammatory indicators derived from complete blood count, especially PLR, have certain application value in predicting the severity of MPP children in different ages. -
Keywords:
- Mycoplasma pneumoniae /
- pneumonia /
- children /
- complete blood count /
- immunoinflammatory index
-
肺炎支原体肺炎(Mycoplasma pneumoniae pneumonia,MPP)是由肺炎支原体(Mycoplasma pneumoniae,MP)感染引起的肺部感染性疾病,以学龄前儿童及青少年多见,占18岁以下儿童社区获得性肺炎(community-acquired pneumonia,CAP)的10%~40%[1]。欧洲临床微生物学和传染病学会MP监测研究组的报告显示,2023年下半年中国儿童肺炎发病率的增加主要归因于MPP[2]。MP感染后引发肺炎的发病机制除了病原体对呼吸道黏膜上皮细胞的直接损伤外,免疫炎症反应引起患儿体内免疫应答失调对MPP的发生也具有重要影响,机体炎症因子水平紊乱可诱发炎症瀑布反应,导致患儿发生多器官功能障碍和全身炎症反应综合征等严重并发症[3]。
近年来,基于全血细胞计数的免疫炎症指标因能反映全身炎症水平,且具有数据易获取、检测费用低等优势,受到广泛关注,包括血小板与淋巴细胞比值(platelet-to-lymphocyte ratio,PLR)[4]、中性粒细胞与淋巴细胞比值(neutrophil-to-lymphocyte ratio,NLR)[4-7]、衍生的中性粒细胞与淋巴细胞比值(derived neutrophil-to-lymphocyte ratio,dNLR)[6]和系统性免疫炎症指数(systemic immune-inflammation index,SII)[8]等。本研究通过回顾MPP患儿的临床资料及实验室检查结果,探讨9种全血细胞计数来源的免疫炎症指标与MPP重症化之间的关系,以期为早期评估并干预MPP提供思路。
1 资料和方法
1.1 研究对象
本研究为单中心回顾性研究,以2023年7月1日至2024年12月31日于海军军医大学第一附属医院儿科病房住院的MPP患儿为研究对象。纳入标准:(1)年龄1~16岁,性别不限;(2)符合《儿童肺炎支原体肺炎诊断与治疗循证指南(2023)》[9]中MPP的诊断标准,即需结合病史、临床症状、体征及影像学表现,并满足以下任一条件——恢复期较急性期血清MP抗体滴度呈4倍及以上增高,或MP培养阳性,或MP-DNA/RNA检测阳性;(3)首次就诊时病程≤3 d(72 h)。排除标准:(1)甲型流感病毒、乙型流感病毒或新型冠状病毒等其他病原体检测阳性;(2)既往罹患川崎病、免疫缺陷综合征等影响免疫系统的疾病;(3)血常规检查前曾使用糖皮质激素或抗生素者;(4)临床资料不完整。
依据《儿童社区获得性肺炎管理指南(2024修订)》[10]中关于重症肺炎的标准,将患儿分为重症MPP(severe MPP,SMPP)组和非重症MPP(non-severe MPP,NSMPP)组。由于不同年龄段儿童的血象存在差异,白细胞分类中的中性粒细胞比例和淋巴细胞比例会有所变化,4~6岁以后血象特征逐渐与成人相似[11],因此本研究以6岁为界进行分层讨论。本研究获得海军军医大学第一附属医院伦理委员会审批(批准号:CHEC2024-321)。
1.2 研究方法
1.2.1 仪器与试剂
由经验丰富的儿科护士于入院次日清晨采集患儿空腹时的肘正中静脉血2 mL,利用日本希森美康株式会社生产的XN-9000全自动血液分析仪检测血常规,包括白细胞(white blood cell,WBC)计数、中性粒细胞(neutrophil,NEU)计数、淋巴细胞(lymphocyte,LYM)计数、单核细胞(monocyte,MON)计数、血小板(platelet,PLT)计数、红细胞(red blood cell,RBC)计数等。利用美国Beckman Coulter公司生产的AU5800型全自动生化分析仪,采用免疫比浊法测定CRP水平,正常参考值为0~10 mg/L,试剂、校准品均由美国Beckman Coulter公司提供;采用磁微粒化学发光法测定降钙素原(procalcitonin,PCT),正常参考值为0~0.05 ng/mL。采用MP核酸检测试剂盒(武汉中帜生物科技股份有限公司,双扩增法)检测MP-DNA,荧光值>1为阳性、荧光值≤1为阴性。采用MP抗体检测试剂盒(日本富士瑞必欧株式会社,被动凝集法)进行MP血清学检测,抗体滴度≥1∶160为阳性、抗体滴度<1∶160为阴性。
1.2.2 资料收集
由2名经过统一培训的儿科医生从海军军医大学第一附属医院电子病历系统中调阅并收集数据。本研究中性别为分类变量,男=“1”、女=“0”;年龄、住院天数、WBC、NEU、LYM、MON、PLT、RBC、CRP、PCT为连续变量,以原始数值记入。
1.2.3 免疫炎症指标计算
根据文献方法计算指标:SII=PLT×NEU/LYM[8, 12];系统炎症反应指数(system inflammation response index,SIRI)=NEU×MON/LYM[12];全身炎症综合指数(aggregate index of systemic inflammation,AISI)=PLT×NEU×MON/LYM[13];PLR=PLT/LYM[4, 12];NLR=NEU/LYM[4-7, 12];dNLR=NEU/(WBC-NEU)[6];单核细胞与淋巴细胞比值(monocyte-to-lymphocyte ratio,MLR)=MON/LYM[12];中性粒细胞与血小板比值(neutrophil-to-platelet ratio,NPR)=NEU/PLT×100[7];中性粒细胞与淋巴细胞和血小板比值(neutrophil-to-lymphocyte and platelet ratio,NLPR)=NEU×100/(LYM×PLT)[14]。
1.3 统计学处理
应用SPSS 26.0软件及GraphPad Prism 10.0软件进行统计学分析。计量资料行正态性与方差齐性检验,呈正态分布的计量资料以x±s表示,组间比较采用独立样本t检验;呈偏态分布的计量资料以M(Q1,Q3)表示,组间比较采用Mann-Whitney U检验。计数资料以例数和百分数表示,组间比较采用χ2检验。按1∶1比例匹配SMPP与NSMPP患儿后采用单因素及多因素Cox比例风险回归模型分析各组差异有统计学意义的指标与SMPP的相关性;绘制ROC曲线探讨各组差异有统计学意义的指标对SMPP的预测价值。检验水准(α)为0.05。
2 结果
2.1 一般情况
2023年7月1日至2024年12月31日累计收治MPP患儿2 132例,其中SMPP患儿220例(10.3%)、NSMPP患儿1 912例(89.7%)。SMPP患儿中,男96例(43.6%)、女124例(56.4%),年龄1~16岁,平均(6.64±2.95)岁,其中1~6岁者104例(47.3%)、>6~16岁者116例(52.7%)。按1∶1比例匹配同期年龄、性别与SMPP患儿无统计学差异的NSMPP患儿作为对照组。
2.2 年龄1~6岁患儿组
2.2.1 SMPP和NSMPP患儿临床资料比较
与NSMPP组相比,SMPP组患儿的住院时间更长,PLT、NLR、PLR、dNLR和SII更高(均P<0.05);两组间全血细胞计数来源的免疫炎症指标MLR、NLPR、NPR、SIRI、AISI差异均无统计学意义(均P>0.05)。见表 1。
表 1 年龄1~6岁SMPP和NSMPP患儿的临床资料比较Table 1 Comparison of clinical data between children aged 1 to 6 years old with SMPP and NSMPPIndex SMPP N=104 NSMPP N=104 Statistic P value Age/year, M (Q1, Q3) 4.00 (3.00, 5.75) 4.00 (3.00, 5.75) Z=0.000 1.000 Gender, n (%) χ2=0.000 1.000 Male 40 (38.5) 40 (38.5) Female 64 (61.5) 64 (61.5) Length of stay/d, M (Q1, Q3) 6.00 (5.00, 7.00) 5.00 (4.00, 6.00) Z=-5.614 <0.001 CRP/(mg·L-1), M (Q1, Q3) 9.36 (4.35, 22.33) 10.12 (4.27, 21.05) Z=-0.202 0.840 PCT/(ng·mL-1), M (Q1, Q3) 0.11 (0.07, 0.20) 0.11 (0.06, 0.21) Z=-0.505 0.614 WBC/(L-1, ×109), M (Q1, Q3) 7.48 (5.46, 10.04) 7.61 (5.91, 9.74) Z=-0.196 0.845 NEU/(L-1, ×109), M (Q1, Q3) 4.06 (2.77, 6.17) 3.99 (2.55, 5.47) Z=-0.564 0.572 LYM/(L-1, ×109), M (Q1, Q3) 2.39 (1.73, 3.03) 2.59 (2.03, 3.50) Z=-1.585 0.113 MON/(L-1, ×109), M (Q1, Q3) 0.59 (0.40, 0.88) 0.63 (0.49, 0.83) Z=-0.701 0.484 PLT/(L-1, ×109), M (Q1, Q3) 311.00 (252.25, 379.75) 282.50 (240.75, 330.50) Z=-2.699 0.007 RBC/(L-1, ×1012), x±s 4.55±0.39 4.59±0.38 t=-0.877 0.382 NLR, M (Q1, Q3) 1.80 (1.05, 2.91) 1.42 (0.87, 2.40) Z=-2.100 0.036 MLR, M (Q1, Q3) 0.26 (0.18, 0.32) 0.25 (0.17, 0.30) Z=-0.889 0.374 PLR, M (Q1, Q3) 135.59 (94.23, 176.87) 109.61 (86.42, 139.82) Z=-3.372 0.001 dNLR, M (Q1, Q3) 1.34 (0.80, 2.20) 1.09 (0.64, 1.73) Z=-2.275 0.023 NLPR, M (Q1, Q3) 0.54 (0.32, 0.98) 0.48 (0.30, 0.83) Z=-1.047 0.295 NPR, M (Q1, Q3) 1.24 (0.83, 1.84) 1.33 (0.93, 1.85) Z=-0.441 0.659 SII, M (Q1, Q3) 574.33 (322.51, 838.58) 419.44 (231.25, 628.57) Z=-2.870 0.004 SIRI, M (Q1, Q3) 0.92 (0.55, 1.70) 0.95 (0.47, 1.65) Z=-0.727 0.467 AISI, M (Q1, Q3) 307.93 (151.60, 594.11) 256.75 (118.85, 474.91) Z=-1.554 0.120 SMPP: Severe Mycoplasma pneumoniae pneumonia; NSMPP: Non-severe Mycoplasma pneumoniae pneumonia; CRP: C reactive protein; PCT: Procalcitonin; WBC: White blood cell; NEU: Neutrophil; LYM: Lymphocyte; MON: Monocyte; PLT: Platelet; RBC: Red blood cell; NLR: Neutrophil-to-lymphocyte ratio; MLR: Monocyte-to-lymphocyte ratio; PLR: Platelet-to-lymphocyte ratio; dNLR: Derived neutrophil-to-lymphocyte ratio; NLPR: Neutrophil-to-lymphocyte and platelet ratio; NPR: Neutrophil-to-platelet ratio; SII: Systemic immune-inflammation index; SIRI: System inflammation response index; AISI: Aggregate index of systemic inflammation. 2.2.2 MPP进展至SMPP的影响因素
对表 1中差异有统计学意义的指标进行单因素Cox回归分析,5项指标(PLT、NLR、PLR、dNLR、SII)均P<0.1,进一步行多因素Cox回归分析,以MPP分型为因变量(SMPP赋值为1、NSMPP赋值为0),以PLT、NLR、PLR、dNLR、SII为自变量,结果显示PLR是1~6岁MPP患儿进展至SMPP的独立危险因素(OR=1.010,95%CI 1.003~1.018,P=0.007)。见表 2。
表 2 年龄1~6岁MPP患儿进展至SMPP的Cox回归分析Table 2 Cox regression analysis of MPP children aged 1 to 6 years old who progressed to SMPPVariable Univariate analysis Multivariate analysis b SE Wald OR (95%CI) P value b SE Wald OR (95%CI) P value PLT 0.005 0.002 8.238 1.005 (1.002, 1.009) 0.004 NLR 0.189 0.108 3.071 1.209 (0.978, 1.494) 0.080 PLR 0.011 0.003 11.081 1.011 (1.004, 1.017) 0.001 0.010 0.004 7.182 1.010 (1.003, 1.018) 0.007 dNLR 0.273 0.148 3.370 1.313 (0.982, 1.757) 0.066 SII 0.001 0.000 7.592 1.001 (1.000, 1.002) 0.006 MPP: Mycoplasma pneumoniae pneumonia; SMPP: Severe Mycoplasma pneumoniae pneumonia; PLT: Platelet; NLR: Neutrophil-to-lymphocyte ratio; PLR: Platelet-to-lymphocyte ratio; dNLR: Derived neutrophil-to-lymphocyte ratio; SII: Systemic immune-inflammation index; b: Regression coefficient; SE: Standard error; OR: Odds ratio; 95%CI: 95% confidence interval. 2.2.3 预测SMPP的ROC曲线
ROC曲线分析结果(图 1)显示,PLR预测SMPP的AUC为0.635(95%CI 0.560~0.711,P<0.001),最佳截断值为125.04,此时对应的灵敏度为57.7%、特异度为70.2%。
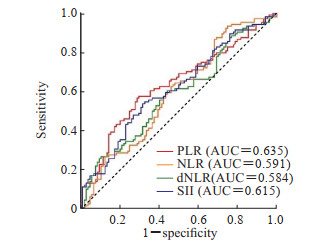 图 1 全血细胞计数来源的免疫炎症指标预测SMPP的性能分析Fig. 1 Performance analysis of immunoinflammatory indicators derived from complete blood count in predicting SMPPSMPP: Severe Mycoplasma pneumoniae pneumonia; PLR: Platelet-to-lymphocyte ratio; NLR: Neutrophil-to-lymphocyte ratio; dNLR: Derived neutrophil-to-lymphocyte ratio; SII: Systemic immune-inflammation index; AUC: Area under curve.
图 1 全血细胞计数来源的免疫炎症指标预测SMPP的性能分析Fig. 1 Performance analysis of immunoinflammatory indicators derived from complete blood count in predicting SMPPSMPP: Severe Mycoplasma pneumoniae pneumonia; PLR: Platelet-to-lymphocyte ratio; NLR: Neutrophil-to-lymphocyte ratio; dNLR: Derived neutrophil-to-lymphocyte ratio; SII: Systemic immune-inflammation index; AUC: Area under curve.2.3 年龄>6~16岁患儿组
2.3.1 SMPP和NSMMP患儿临床资料比较
与NSMPP组相比,SMPP组患儿的PLT和PLR更高(均P<0.05),两组间其他全血细胞计数来源的免疫炎症指标NLR、MLR、dNLR、NLPR、NPR、SII、SIRI、AISI差异均无统计学意义(均P>0.05)。见表 3。
表 3 年龄>6~16岁SMPP和NSMPP患儿的临床资料比较Table 3 Comparison of clinical data between children aged >6-16 years old with SMPP or NSMPPIndex SMPP N=116 NSMPP N=116 Statistic P value Age/year, M (Q1, Q3) 8.50 (7.00, 10.00) 8.50 (7.00, 10.00) Z=0.000 1.000 Gender, n (%) χ2=0.000 1.000 Male 56 (48.3) 56 (48.3) Female 60 (51.7) 60 (51.7) Length of stay/d, M (Q1, Q3) 6.0 (5.0, 6.0) 5.0 (5.0, 6.0) Z=-1.147 0.251 CRP/(mg·L-1), M (Q1, Q3) 17.01 (6.73, 32.58) 16.26 (6.54, 27.72) Z=-0.702 0.482 PCT/(ng·mL-1), M (Q1, Q3) 0.09 (0.07, 0.15) 0.10 (0.06, 0.15) Z=-0.264 0.792 WBC/(L-1, ×109), M (Q1, Q3) 6.34 (5.08, 8.36) 6.47 (5.19, 8.73) Z=-0.425 0.671 NEU/(L-1, ×109), M (Q1, Q3) 3.87 (2.71, 5.50) 3.79 (2.73, 5.53) Z=-0.240 0.811 LYM/(L-1, ×109), M (Q1, Q3) 1.93 (1.43, 2.59) 1.94 (1.52, 2.43) Z=-0.506 0.613 MON/(L-1, ×109), M (Q1, Q3) 0.52 (0.37, 0.72) 0.59 (0.39, 0.73) Z=-1.054 0.292 PLT/(L-1, ×109), M (Q1, Q3) 306.50 (244.00, 350.25) 274.50 (229.00, 321.50) Z=-2.178 0.029 RBC/(L-1, ×1012), M (Q1, Q3) 4.67 (4.44, 4.88) 4.58 (4.37, 4.76) Z=-1.682 0.093 NLR, M (Q1, Q3) 2.11 (1.40, 3.08) 1.96 (1.41, 2.95) Z=-0.789 0.430 MLR, M (Q1, Q3) 0.27 (0.20, 0.34) 0.26 (0.21, 0.39) Z=-0.968 0.333 PLR, M (Q1, Q3) 158.51 (120.81, 222.62) 137.33 (111.19, 180.90) Z=-2.060 0.039 dNLR, M (Q1, Q3) 1.54 (1.14, 2.37) 1.46 (1.02, 2.12) Z=-1.192 0.233 NLPR, M (Q1, Q3) 0.74 (0.49, 1.05) 0.77 (0.46, 1.09) Z=-0.224 0.823 NPR, M (Q1, Q3) 1.34 (0.97, 1.74) 1.50 (0.99, 1.94) Z=-0.887 0.375 SII, M (Q1, Q3) 615.04 (389.76, 1 026.00) 511.31 (338.20, 787.82) Z=-1.476 0.140 SIRI, M (Q1, Q3) 0.99 (0.56, 1.61) 1.07 (0.65, 1.88) Z=-0.754 0.451 AISI, M (Q1, Q3) 277.42 (162.89, 512.39) 298.09 (165.14, 602.54) Z=-0.138 0.890 SMPP: Severe Mycoplasma pneumoniae pneumonia; NSMPP: Non-severe Mycoplasma pneumoniae pneumonia; CRP: C reactive protein; PCT: Procalcitonin; WBC: White blood cell; NEU: Neutrophil; LYM: Lymphocyte; MON: Monocyte; PLT: Platelet; RBC: Red blood cell; NLR: Neutrophil-to-lymphocyte ratio; MLR: Monocyte-to-lymphocyte ratio; PLR: Platelet-to-lymphocyte ratio; dNLR: Derived neutrophil-to-lymphocyte ratio; NLPR: Neutrophil-to-lymphocyte and platelet ratio; NPR: Neutrophil-to-platelet ratio; SII: Systemic immune-inflammation index; SIRI: System inflammation response index; AISI: Aggregate index of systemic inflammation. 2.3.2 MPP进展至SMPP的影响因素
对表 3中差异有统计学意义的指标进行单因素Cox回归分析,结果表明,虽然SMPP组的PLT和PLR均较NSMPP组升高,但不是年龄>6~16岁MPP患儿进展至SMPP的独立危险因素(均P>0.05)。见表 4。
表 4 年龄>6~16岁MPP患儿进展至SMPP的单因素Cox回归分析Table 4 Univariate Cox regression analysis of MPP children aged >6-16 years old who progressed to SMPPVariable b SE Wald OR (95%CI) P value PLT 0.003 0.002 3.382 1.003 (1.000, 1.006) 0.066 PLR 0.002 0.002 1.567 1.002 (0.999, 1.005) 0.211 MPP: Mycoplasma pneumoniae pneumonia; SMPP: Severe Mycoplasma pneumoniae pneumonia; PLT: Platelet; PLR: Platelet-to-lymphocyte ratio; b: Regression coefficient; SE: Standard error; OR: Odds ratio; 95%CI: 95% confidence interval. 2.4 不同PLR与重症率的关系
在208例1~6岁MPP患儿中,以最佳截断值为界将所有患儿分为低PLR组和高PLR组,高PLR组的重症率高于低PLR组(65.9% vs 37.6%,P<0.001);以四分位数为界将所有患儿分为Q1~Q4组,Q4组的重症率(71.2%)高于Q1~Q3组(均P<0.05)。在232例>6~16岁MPP患儿中,以最佳截断值为界将所有患儿分为低PLR组和高PLR组,高PLR组的重症率高于低PLR组(57.0% vs 40.2%,P=0.011);以四分位数为界将所有患儿分为Q1~Q4组,Q4组的重症率(65.5%)高于Q1~Q3组(均P<0.05)。见表 5。
表 5 不同年龄段MPP患儿PLR与重症率的关系Table 5 Relationship between PLR and severe rate in MPP children of different agesSubgroup 1-6 years >6-16 years Section N Severe case, n (%) χ2 value P value Section N Severe case, n (%) χ2 value P value Quartile 13.929 0.003 6.791 0.079 Q1 ≤90.33 52 21 (40.4)* ≤114.94 58 27 (46.6)* Q2 >90.33-119.59 52 19 (36.5)* >114.94-143.70 58 24 (41.4)* Q3 >119.59-159.73 52 27 (51.9)* >143.70-194.42 58 27 (46.6)* Q4 >159.73 52 37 (71.2) >194.42 58 38 (65.5) Cut-off value 16.430 <0.001 6.396 0.011 Low PLR ≤125.04 117 44 (37.6) ≤137.03 97 39 (40.2) High PLR >125.04 91 60 (65.9) >137.03 135 77 (57.0) *P<0.05 vs Q4 subgroup. MPP: Mycoplasma pneumoniae pneumonia; PLR: Platelet-to-lymphocyte ratio. 3 讨论
CAP是儿童常见的感染性疾病。由于儿童的气管、支气管短且狭窄,纤毛运动功能和清除病原体能力较差,以及免疫系统尚未发育成熟,抵抗力较弱,一旦感染CAP,容易发展至重症,严重威胁患儿的生命安全。在发展中国家,CAP已成为导致儿童死亡的主要原因之一[15]。MP是我国儿童需住院治疗CAP的常见病原体之一,在流行期间占CAP的20%~40%,部分地区高达50%[1, 16],感染后多数病情轻,但部分患儿会进展为SMPP。研究表明,SMPP患儿会出现更明显的炎症反应,可能会发生严重的肺内和肺外并发症,如坏死性肺炎、弥散性血管内凝血、急性呼吸窘迫综合征等,还可能遗留严重的后遗症,如肺纤维化、感染后闭塞性细支气管炎等[17-18]。因此,早期识别并及时规范治疗对改善SMPP患儿的预后极为重要。SMPP的发病机制尚未完全明确,但多数研究认为其与过度免疫炎症相关[3, 19]。免疫功能与炎症反应相互影响,后者通常是机体免疫应答过程中的重要环节,因此评估MPP患儿的机体炎症免疫状态,有助于指导临床治疗。
血常规是所有具备辅助检查条件的医疗机构都能开展的实验室检测,具有检测速度快、价格低廉的特点,是CAP患儿的常规检测,易于获得结果。基于血常规全血细胞计数的指标不仅能反映机体炎症程度和免疫状态,还可用来评估各类急慢性疾病的预后,如脓毒症[7, 14]、急性缺血性脑卒中[20]等。PLT是感染急性期的炎症反应物,可通过释放趋化因子和细胞因子等炎症介质加剧炎症反应[21]。同时,PLT含有识别病原体的模式识别受体,因此PLT亦在抗感染过程中发挥重要的辅助作用[22]。本研究结果显示,SMPP可影响常见炎症相关标志物的水平,不论患儿处于哪一个年龄段,SMPP组的PLT均高于NSMPP组,进一步表明SMPP中存在过度的炎症反应。既往研究报道SMPP患者的PLT高于NSMPP患者[23],本研究结果与之相符。
LYM是机体细胞免疫的重要组成部分,在适应性免疫应答中发挥重要作用。LYM主要功能包括识别病原体及其毒性产物。当MP侵入机体引发炎症反应时,LYM的数量和功能状态可能因病原体感染或机体整体健康状况而发生改变,可作为反映机体免疫状态的重要指标[22]。有研究表明儿童感染MP后,重症患儿LYM数量下降更显著,免疫功能受损更严重,可能与过度炎症反应诱导的LYM凋亡有关[24]。本研究中虽然SMPP与NSMPP患儿的LYM差异无统计学意义,但基于PLT和LYM计算的PLR在SMPP患儿中明显升高,PLR升高提示血小板活化程度增强,与感染、血液系统异常和免疫疾病有关。
NEU作为非特异性免疫系统抵御感染的第一道细胞防线,在抗MP免疫应答中起着关键作用[25]。MP感染宿主后,会诱导宿主外周血、肺组织中的NEU异常增多[26],而过度升高的NEU可导致肺炎加重[27]。基于NEU和LYM计算的NLR是一种成本低、易测量的生物标志物,可反映炎症细胞的活化状态。Li等[5]研究表明,高NLR患者更易发生坏死性肺炎和难治性MPP,并需要重症监护,总发热持续时间和住院时间更长,住院费用更高。SII作为一种新型炎症生物标志物,结合了PLT、NEU和LYM,可反映机体的炎症和免疫状态。Wang等[8]的研究显示,SII可能是预测儿童MPP严重程度的有效指标,其灵敏度和特异度均优于NLR、PLR和SIRI。本研究也显示,1~6岁SMPP患儿的NLR、dNLR和SII均明显高于NSMPP患儿。
为了评估基于全血细胞计数来源的免疫炎症指标预测SMPP的价值,本研究对SMPP和NSMPP患儿中差异有统计学意义的指标进行了单因素和多因素Cox回归分析及绘制了ROC曲线。在1~6岁患儿中,单因素Cox回归分析结果显示PLT(OR=1.005,95%CI 1.002~1.009,P<0.05)、PLR(OR=1.011,95%CI 1.004~1.017,P<0.05)和SII(OR=1.001,95%CI 1.000~1.002,P<0.05)是MPP重症化的影响因素,多因素Cox回归分析结果显示PLR是患儿进展至SMPP的独立危险因素(OR=1.010,95%CI 1.003~1.018,P=0.007),PLR预测SMPP的ROC曲线AUC为0.635(95%CI 0.560~0.711,P<0.001)。而在>6~16岁儿童中,PLT与PLR都不是患儿进展至SMPP的独立危险因素(均P>0.05)。
虽然探讨全血细胞计数来源的免疫炎症指标与MPP相关性的研究较多,但全面探讨PLR、NLR、dNLR等9项指标与MPP重症化关系的研究较少。Shao等[12]研究了NLR、PLR、SII、SIRI和MLR对儿童MPP的预测价值,发现MPP患儿这5项指标均明显高于健康儿童。进一步的logistic回归分析结果显示这5项指标均是MPP的危险因素、MLR是MPP的独立危险因素,但是该研究未进一步评估这些指标与MPP严重程度的相关性,也未将dNLR、NLPR、NPR、AISI纳入研究。Zhuo等[28]回顾性分析了来自3家医疗机构的不限年龄的598例NSMPP和230例SMPP患者淋巴细胞与单核细胞比值、NLR、SII和PLR指标,发现SMPP组NLR、PLR和SII均明显高于NSMPP组,与本研究1~6岁患儿组的研究结果相符,但该研究未进一步行单因素和多因素回归分析,也未研究dNLR、NLPR、NPR、SIRI和AISI与MPP重症化的关系。
与既往研究相比,本研究充分考虑了不同年龄段儿童的血象特点,在排除性别与年龄的影响后,较为全面的探讨了基于全血细胞计数来源的免疫炎症指标对儿童SMPP的预测价值,且这些指标均易于获取、成本低廉。然而,本研究也存在一定的局限性:(1)本研究为单中心回顾性研究,样本例数少且病例选择可能存在偏倚;(2)本研究结果未行多中心验证或引入外部测试集;(3)收治的SMPP患儿整体病情相对较轻,未包括坏死性肺炎或需机械通气的危重病例,因此导致某些在其他研究中有显著差异的指标(如SIRI、AISI)在本研究中未呈现明显差异。基于以上不足,未来我们将扩大合作范围,开展长期的多中心和更大样本的前瞻性研究,以进一步验证全血细胞计数来源的免疫炎症指标在SMPP预测中的价值。
-
图 1 全血细胞计数来源的免疫炎症指标预测SMPP的性能分析
Fig. 1 Performance analysis of immunoinflammatory indicators derived from complete blood count in predicting SMPP
SMPP: Severe Mycoplasma pneumoniae pneumonia; PLR: Platelet-to-lymphocyte ratio; NLR: Neutrophil-to-lymphocyte ratio; dNLR: Derived neutrophil-to-lymphocyte ratio; SII: Systemic immune-inflammation index; AUC: Area under curve.
表 1 年龄1~6岁SMPP和NSMPP患儿的临床资料比较
Table 1 Comparison of clinical data between children aged 1 to 6 years old with SMPP and NSMPP
Index SMPP N=104 NSMPP N=104 Statistic P value Age/year, M (Q1, Q3) 4.00 (3.00, 5.75) 4.00 (3.00, 5.75) Z=0.000 1.000 Gender, n (%) χ2=0.000 1.000 Male 40 (38.5) 40 (38.5) Female 64 (61.5) 64 (61.5) Length of stay/d, M (Q1, Q3) 6.00 (5.00, 7.00) 5.00 (4.00, 6.00) Z=-5.614 <0.001 CRP/(mg·L-1), M (Q1, Q3) 9.36 (4.35, 22.33) 10.12 (4.27, 21.05) Z=-0.202 0.840 PCT/(ng·mL-1), M (Q1, Q3) 0.11 (0.07, 0.20) 0.11 (0.06, 0.21) Z=-0.505 0.614 WBC/(L-1, ×109), M (Q1, Q3) 7.48 (5.46, 10.04) 7.61 (5.91, 9.74) Z=-0.196 0.845 NEU/(L-1, ×109), M (Q1, Q3) 4.06 (2.77, 6.17) 3.99 (2.55, 5.47) Z=-0.564 0.572 LYM/(L-1, ×109), M (Q1, Q3) 2.39 (1.73, 3.03) 2.59 (2.03, 3.50) Z=-1.585 0.113 MON/(L-1, ×109), M (Q1, Q3) 0.59 (0.40, 0.88) 0.63 (0.49, 0.83) Z=-0.701 0.484 PLT/(L-1, ×109), M (Q1, Q3) 311.00 (252.25, 379.75) 282.50 (240.75, 330.50) Z=-2.699 0.007 RBC/(L-1, ×1012), x±s 4.55±0.39 4.59±0.38 t=-0.877 0.382 NLR, M (Q1, Q3) 1.80 (1.05, 2.91) 1.42 (0.87, 2.40) Z=-2.100 0.036 MLR, M (Q1, Q3) 0.26 (0.18, 0.32) 0.25 (0.17, 0.30) Z=-0.889 0.374 PLR, M (Q1, Q3) 135.59 (94.23, 176.87) 109.61 (86.42, 139.82) Z=-3.372 0.001 dNLR, M (Q1, Q3) 1.34 (0.80, 2.20) 1.09 (0.64, 1.73) Z=-2.275 0.023 NLPR, M (Q1, Q3) 0.54 (0.32, 0.98) 0.48 (0.30, 0.83) Z=-1.047 0.295 NPR, M (Q1, Q3) 1.24 (0.83, 1.84) 1.33 (0.93, 1.85) Z=-0.441 0.659 SII, M (Q1, Q3) 574.33 (322.51, 838.58) 419.44 (231.25, 628.57) Z=-2.870 0.004 SIRI, M (Q1, Q3) 0.92 (0.55, 1.70) 0.95 (0.47, 1.65) Z=-0.727 0.467 AISI, M (Q1, Q3) 307.93 (151.60, 594.11) 256.75 (118.85, 474.91) Z=-1.554 0.120 SMPP: Severe Mycoplasma pneumoniae pneumonia; NSMPP: Non-severe Mycoplasma pneumoniae pneumonia; CRP: C reactive protein; PCT: Procalcitonin; WBC: White blood cell; NEU: Neutrophil; LYM: Lymphocyte; MON: Monocyte; PLT: Platelet; RBC: Red blood cell; NLR: Neutrophil-to-lymphocyte ratio; MLR: Monocyte-to-lymphocyte ratio; PLR: Platelet-to-lymphocyte ratio; dNLR: Derived neutrophil-to-lymphocyte ratio; NLPR: Neutrophil-to-lymphocyte and platelet ratio; NPR: Neutrophil-to-platelet ratio; SII: Systemic immune-inflammation index; SIRI: System inflammation response index; AISI: Aggregate index of systemic inflammation. 表 2 年龄1~6岁MPP患儿进展至SMPP的Cox回归分析
Table 2 Cox regression analysis of MPP children aged 1 to 6 years old who progressed to SMPP
Variable Univariate analysis Multivariate analysis b SE Wald OR (95%CI) P value b SE Wald OR (95%CI) P value PLT 0.005 0.002 8.238 1.005 (1.002, 1.009) 0.004 NLR 0.189 0.108 3.071 1.209 (0.978, 1.494) 0.080 PLR 0.011 0.003 11.081 1.011 (1.004, 1.017) 0.001 0.010 0.004 7.182 1.010 (1.003, 1.018) 0.007 dNLR 0.273 0.148 3.370 1.313 (0.982, 1.757) 0.066 SII 0.001 0.000 7.592 1.001 (1.000, 1.002) 0.006 MPP: Mycoplasma pneumoniae pneumonia; SMPP: Severe Mycoplasma pneumoniae pneumonia; PLT: Platelet; NLR: Neutrophil-to-lymphocyte ratio; PLR: Platelet-to-lymphocyte ratio; dNLR: Derived neutrophil-to-lymphocyte ratio; SII: Systemic immune-inflammation index; b: Regression coefficient; SE: Standard error; OR: Odds ratio; 95%CI: 95% confidence interval. 表 3 年龄>6~16岁SMPP和NSMPP患儿的临床资料比较
Table 3 Comparison of clinical data between children aged >6-16 years old with SMPP or NSMPP
Index SMPP N=116 NSMPP N=116 Statistic P value Age/year, M (Q1, Q3) 8.50 (7.00, 10.00) 8.50 (7.00, 10.00) Z=0.000 1.000 Gender, n (%) χ2=0.000 1.000 Male 56 (48.3) 56 (48.3) Female 60 (51.7) 60 (51.7) Length of stay/d, M (Q1, Q3) 6.0 (5.0, 6.0) 5.0 (5.0, 6.0) Z=-1.147 0.251 CRP/(mg·L-1), M (Q1, Q3) 17.01 (6.73, 32.58) 16.26 (6.54, 27.72) Z=-0.702 0.482 PCT/(ng·mL-1), M (Q1, Q3) 0.09 (0.07, 0.15) 0.10 (0.06, 0.15) Z=-0.264 0.792 WBC/(L-1, ×109), M (Q1, Q3) 6.34 (5.08, 8.36) 6.47 (5.19, 8.73) Z=-0.425 0.671 NEU/(L-1, ×109), M (Q1, Q3) 3.87 (2.71, 5.50) 3.79 (2.73, 5.53) Z=-0.240 0.811 LYM/(L-1, ×109), M (Q1, Q3) 1.93 (1.43, 2.59) 1.94 (1.52, 2.43) Z=-0.506 0.613 MON/(L-1, ×109), M (Q1, Q3) 0.52 (0.37, 0.72) 0.59 (0.39, 0.73) Z=-1.054 0.292 PLT/(L-1, ×109), M (Q1, Q3) 306.50 (244.00, 350.25) 274.50 (229.00, 321.50) Z=-2.178 0.029 RBC/(L-1, ×1012), M (Q1, Q3) 4.67 (4.44, 4.88) 4.58 (4.37, 4.76) Z=-1.682 0.093 NLR, M (Q1, Q3) 2.11 (1.40, 3.08) 1.96 (1.41, 2.95) Z=-0.789 0.430 MLR, M (Q1, Q3) 0.27 (0.20, 0.34) 0.26 (0.21, 0.39) Z=-0.968 0.333 PLR, M (Q1, Q3) 158.51 (120.81, 222.62) 137.33 (111.19, 180.90) Z=-2.060 0.039 dNLR, M (Q1, Q3) 1.54 (1.14, 2.37) 1.46 (1.02, 2.12) Z=-1.192 0.233 NLPR, M (Q1, Q3) 0.74 (0.49, 1.05) 0.77 (0.46, 1.09) Z=-0.224 0.823 NPR, M (Q1, Q3) 1.34 (0.97, 1.74) 1.50 (0.99, 1.94) Z=-0.887 0.375 SII, M (Q1, Q3) 615.04 (389.76, 1 026.00) 511.31 (338.20, 787.82) Z=-1.476 0.140 SIRI, M (Q1, Q3) 0.99 (0.56, 1.61) 1.07 (0.65, 1.88) Z=-0.754 0.451 AISI, M (Q1, Q3) 277.42 (162.89, 512.39) 298.09 (165.14, 602.54) Z=-0.138 0.890 SMPP: Severe Mycoplasma pneumoniae pneumonia; NSMPP: Non-severe Mycoplasma pneumoniae pneumonia; CRP: C reactive protein; PCT: Procalcitonin; WBC: White blood cell; NEU: Neutrophil; LYM: Lymphocyte; MON: Monocyte; PLT: Platelet; RBC: Red blood cell; NLR: Neutrophil-to-lymphocyte ratio; MLR: Monocyte-to-lymphocyte ratio; PLR: Platelet-to-lymphocyte ratio; dNLR: Derived neutrophil-to-lymphocyte ratio; NLPR: Neutrophil-to-lymphocyte and platelet ratio; NPR: Neutrophil-to-platelet ratio; SII: Systemic immune-inflammation index; SIRI: System inflammation response index; AISI: Aggregate index of systemic inflammation. 表 4 年龄>6~16岁MPP患儿进展至SMPP的单因素Cox回归分析
Table 4 Univariate Cox regression analysis of MPP children aged >6-16 years old who progressed to SMPP
Variable b SE Wald OR (95%CI) P value PLT 0.003 0.002 3.382 1.003 (1.000, 1.006) 0.066 PLR 0.002 0.002 1.567 1.002 (0.999, 1.005) 0.211 MPP: Mycoplasma pneumoniae pneumonia; SMPP: Severe Mycoplasma pneumoniae pneumonia; PLT: Platelet; PLR: Platelet-to-lymphocyte ratio; b: Regression coefficient; SE: Standard error; OR: Odds ratio; 95%CI: 95% confidence interval. 表 5 不同年龄段MPP患儿PLR与重症率的关系
Table 5 Relationship between PLR and severe rate in MPP children of different ages
Subgroup 1-6 years >6-16 years Section N Severe case, n (%) χ2 value P value Section N Severe case, n (%) χ2 value P value Quartile 13.929 0.003 6.791 0.079 Q1 ≤90.33 52 21 (40.4)* ≤114.94 58 27 (46.6)* Q2 >90.33-119.59 52 19 (36.5)* >114.94-143.70 58 24 (41.4)* Q3 >119.59-159.73 52 27 (51.9)* >143.70-194.42 58 27 (46.6)* Q4 >159.73 52 37 (71.2) >194.42 58 38 (65.5) Cut-off value 16.430 <0.001 6.396 0.011 Low PLR ≤125.04 117 44 (37.6) ≤137.03 97 39 (40.2) High PLR >125.04 91 60 (65.9) >137.03 135 77 (57.0) *P<0.05 vs Q4 subgroup. MPP: Mycoplasma pneumoniae pneumonia; PLR: Platelet-to-lymphocyte ratio. -
[1] DING G, ZHANG X, VINTURACHE A, et al. Challenges in the treatment of pediatric Mycoplasma pneumoniae pneumonia[J]. Eur J Pediatr, 2024, 183(7): 3001-3011. DOI: 10.1007/s00431-024-05519-1. [2] MEYER SAUTEUR P M, BEETON M L; European Society of Clinical Microbiology and Infectious Diseases (ESCMID) Study Group for Mycoplasma and Chlamydia Infections (ESGMAC), and the ESGMAC Mycoplasma pneumoniae Surveillance (MAPS) study group. Pneumonia outbreaks due to re-emergence of Mycoplasma pneumoniae[J]. Lancet Microbe, 2024, 5(6): e514. DOI: 10.1016/s2666-5247(23)00406-8. [3] ZHU Y, LUO Y, LI L, et al. Immune response plays a role in Mycoplasma pneumoniae pneumonia[J]. Front Immunol, 2023, 14: 1189647. DOI: 10.3389/fimmu.2023.1189647. [4] MOOSMANN J, KRUSEMARK A, DITTRICH S, et al. Age- and sex-specific pediatric reference intervals for neutrophil-to-lymphocyte ratio, lymphocyte-to-monocyte ratio, and platelet-to-lymphocyte ratio[J]. Int J Lab Hematol, 2022, 44(2): 296-301. DOI: 10.1111/ijlh.13768. [5] LI D, GU H, CHEN L, et al. Neutrophil-to-lymphocyte ratio as a predictor of poor outcomes of Mycoplasma pneumoniae pneumonia[J]. Front Immunol, 2023, 14: 1302702. DOI: 10.3389/fimmu.2023.1302702. [6] ARDESTANI S K, SALEHI M R, ATTARAN B, et al. Neutrophil to lymphocyte ratio (NLR) and derived NLR combination: a cost-effective predictor of moderate to severe COVID-19 progression[J]. Iran J Allergy Asthma Immunol, 2022, 21(3): 241-253. DOI: 10.18502/ijaai.v21i3.9798. [7] ZHANG Y, PENG W, ZHENG X. The prognostic value of the combined neutrophil-to-lymphocyte ratio (NLR) and neutrophil-to-platelet ratio (NPR) in sepsis[J]. Sci Rep, 2024, 14(1): 15075. DOI: 10.1038/s41598-024-64469-8. [8] WANG S, WAN Y, ZHANG W. The clinical value of systemic immune inflammation index (SⅡ) in predicting the severity of hospitalized children with Mycoplasma pneumoniae pneumonia: a retrospective study[J]. Int J Gen Med, 2024, 17: 935-942. DOI: 10.2147/IJGM.S451466. [9] 中华医学会儿科学分会呼吸学组, 国家呼吸系统疾病临床医学研究中心, 中华儿科杂志编辑委员会. 儿童肺炎支原体肺炎诊断与治疗循证指南(2023)[J]. 中华儿科杂志, 2024, 62(12): 1137-1144. [10] 中华医学会儿科学分会呼吸学组, 中华儿科杂志编辑委员会, 中国医药教育协会儿科专业委员会. 儿童社区获得性肺炎管理指南(2024修订)[J]. 中华儿科杂志, 2024, 62(10): 920-930. [11] 时玉霞, 周霖, 雷蕾, 等. 基于血常规的儿童甲型流行性感冒多参数预测模型[J]. 海军军医大学学报, 2024, 45(11): 1371-1380. DOI: 10.16781/j.CN31-2187/R.20240372. SHI Y, ZHOU L, LEI L, et al. Multi-parameter prediction model based on blood routine in children with influenza A[J]. Acad J Naval Med Univ, 2024, 45(11): 1371-1380. DOI: 10.16781/j.CN31-2187/R.20240372. [12] SHAO L, YU B, LYU Y, et al. The clinical value of novel inflammatory biomarkers for predicting Mycoplasma pneumoniae infection in children[J]. J Clin Lab Anal, 2025, 39(3): e25150. DOI: 10.1002/jcla.25150. [13] ZINELLU A, PALIOGIANNIS P, MANGONI A A. Aggregate index of systemic inflammation (AISI), disease severity, and mortality in COVID-19: a systematic review and meta-analysis[J]. J Clin Med, 2023, 12(14): 4584. DOI: 10.3390/jcm12144584. [14] WANG H, ZHANG R, XU J, et al. Development of a prognosis prediction model for pediatric sepsis based on the NLPR[J]. J Inflamm Res, 2024, 17: 7777-7791. DOI: 10.2147/JIR.S479660. [15] WANG X, LI Y, DELORIA-KNOLL M, et al. Global burden of acute lower respiratory infection associated with human metapneumovirus in children under 5 years in 2018: a systematic review and modelling study[J]. Lancet Glob Health, 2021, 9(1): e33-e43. DOI: 10.1016/S2214-109X(20)30393-4. [16] WAITES K B, XIAO L, LIU Y, et al. Mycoplasma pneumoniae from the respiratory tract and beyond[J]. Clin Microbiol Rev, 2017, 30(3): 747-809. DOI: 10.1128/CMR.00114-16. [17] YANG S, LU S, GUO Y, et al. A comparative study of general and severe Mycoplasma pneumoniae pneumonia in children[J]. BMC Infect Dis, 2024, 24(1): 449. DOI: 10.1186/s12879-024-09340-x. [18] WANG Z, PENG Y, YANG S, et al. Risk factors for complications of Mycoplasma pneumoniae pneumonia in hospitalized children in China: a systematic review and meta-analysis[J]. BMC Pediatr, 2024, 24(1): 810. DOI: 10.1186/s12887-024-05279-9. [19] TAHMASEBI H, BABAEIZAD A, MOHAMMADLOU M, et al. Reemergence of Mycoplasma pneumoniae disease: pathogenesis and new approaches[J]. Microb Pathog, 2024, 196: 106944. DOI: 10.1016/j.micpath.2024.106944. [20] 王丽君, 张萍, 呼延梅华, 等. 外周血炎症标志物与急性缺血性脑卒中的关系[J]. 海军军医大学学报, 2024, 45(2): 174-180. DOI: 10.16781/j.CN31-2187/R.20230539. WANG L, ZHANG P, HUYAN M, et al. Relationships between peripheral blood inflammatory markers and acute ischemic stroke[J]. Acad J Naval Med Univ, 2024, 45(2): 174-180. DOI: 10.16781/j.CN31-2187/R.20230539. [21] BALTA S, DEMıRKOL S, KUCUK U. The platelet lymphocyte ratio may be useful inflammatory indicator in clinical practice[J]. Hemodial Int, 2013, 17(4): 668-669. DOI: 10.1111/hdi.12058. [22] KOSIDŁO J W, WOLSZCZAK-BIEDRZYCKA B, MATOWICKA-KARNA J, et al. Clinical significance and diagnostic utility of NLR, LMR, PLR and SⅡ in the course of COVID-19: a literature review[J]. J Inflamm Res, 2023, 16: 539-562. DOI: 10.2147/JIR.S395331. [23] LI L, REN B, GUO J, et al. Expression levels of blood platelet and C-reactive protein in patients with severely pneumonic and their predictive values for efficacy[J]. Cell Mol Biol (Noisy-le-grand), 2020, 66(2): 105-110. doi: 10.14715/cmb/2020.66.2.17 [24] TEZOL O, BOZLU G, SAGCAN F, et al. Value of neutrophil-to-lymphocyte ratio, monocyte-to-lymphocyte ratio, platelet-to-lymphocyte ratio and red blood cell distribution width in distinguishing between reactive lymphadenopathy and lymphoma in children[J]. Bratisl Lek Listy, 2020, 121(4): 287-292. DOI: 10.4149/BLL_2020_045. [25] ZHANG Z, WAN R, YUAN Q, et al. Cell damage and neutrophils promote the infection of Mycoplasma pneumoniae and inflammatory response[J]. Microb Pathog, 2022, 169: 105647. DOI: 10.1016/j.micpath.2022.105647. [26] YANG L, ZHANG Y, SHEN C, et al. Clinical features and risk factors of plastic bronchitis caused by Mycoplasma pneumoniae pneumonia in children[J]. BMC Pulm Med, 2023, 23(1): 468. DOI: 10.1186/s12890-023-02766-0. [27] WANG C, WEN J, YAN Z, et al. Suppressing neutrophil itaconate production attenuates Mycoplasma pneumoniae pneumonia[J]. PLoS Pathog, 2024, 20(11): e1012614. DOI: 10.1371/journal.ppat.1012614. [28] ZHUO L Y, HAO J W, SONG Z J, et al. Predicting the severity of Mycoplasma pneumoniae pneumonia in pediatric and adult patients: a multicenter study[J]. Sci Rep, 2024, 14(1): 22978. DOI: 10.1038/s41598-024-74251-5.

 下载:
下载:
