Supported by the National Natural Science Foundation of China under Grant No. 41574076, the Young Core Teacher Scheme of Henan Province under Grant No. 2014GGJS-108, and the Fundamental and Cutting-edge Technology Research Program of Henan Province under Grant No. 152300410218
Supported by the National Natural Science Foundation of China under Grant No. 41574076, the Young Core Teacher Scheme of Henan Province under Grant No. 2014GGJS-108, and the Fundamental and Cutting-edge Technology Research Program of Henan Province under Grant No. 152300410218
† Corresponding author. E-mail:
We compare the direct free energy interpolation (DFEI) method and the quasi-harmonic approximation (QHA) in calculating of the equation of states and thermodynamic properties of prototype Al. The Gibbs free energy of Al is calculated using the DFEI method based on the high-temperature phonon density of states reduced from classical molecular dynamics simulations. Then, we reproduce the thermal expansion coefficients, the specific heat, the isothermal bulk modulus of Al accurately. By comparing the results from the DFEI method and the QHA, we find that the DFEI method is indeed more accurate in calculating anharmonic properties than the QHA.
Like platinum and tungsten, aluminum is another frequently used pressure standard material in high-pressure experiments, such as the shock wave experiment. The pressure-volume-temperature equation of states (EOS) provides key information for it as a pressure standard.[1] Al is a typical sp-bonded simple metal and crystallized in face-centered-cubic structure under ambient conditions. With the simple electronic and lattice structures, it is often taken as a typical prototype for theoretical calculations.[2–3] For the theoretical calculation of free energy, the accurate anharmonic effects are very hard to be taken into account in the theoretical EOS in previous methods.
Usually, the quasi-harmonic approximation (QHA) was used to calculate the temperature effects and it only takes account into the volume dependence of phonon frequencies of lattice vibrations. However, in real materials the phonon frequencies depend largely on temperature, especially in the highly anharmonic solids or at the temperature close to melting. This is because that the QHA omits part of anharmonicity and is only accurate when temperature below Debye temperature.[4–6] In order to overcome these difficulties of QHA, several anharmonic free energy calculation techniques have been developed, such as the thermodynamic integration technique[7–8] and the self-consistent ab initio lattice dynamics (SCAILD) method.[9–10]
Molecular dynamics (MD) simulations include anharmonicity to any order beyond Debye temperature,[11–12] especially applicable to high-temperature free energy calculations. Thus it can correct the free energy of the QHA considerably in the free energy calculations. In this paper, we apply the MD simulations to extract anharmonic free energy of Al and then construct more accurate EOS beyond the QHA.
The rest of the paper is organized as follows. In Sec.
The free energy of Al was first calculated by the quasi-harmonic approximation and then corrected by taking into account the phonon-phonon interactions. The quasi-harmonic approximation (QHA) describes volume-dependent thermal effects based on lattice vibration models known as phonon, neglecting the phonon-phonon interactions. In the framework of the QHA, the Helmholtz free energy of a crystal system is,
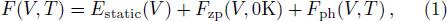
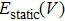
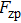
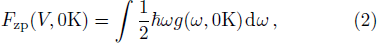
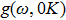
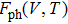
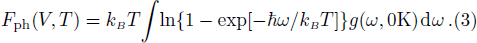
In the harmonic approximation, the PDOS 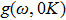
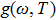
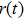
According to the Wiener–Khintchine theorem,[17–18] the autocorrelation of position function 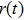
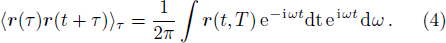
The PDOS at T is,
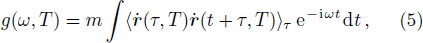
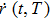
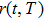
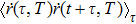
The Helmholtz free energy of a crystal system is then rewritten as,
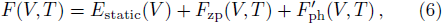
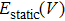
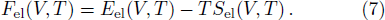
In Eq. (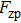
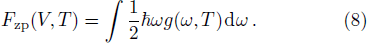
The term 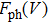
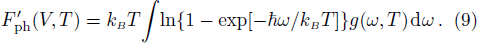
After obtaining the finite number of temperature dependent PDOS (TD-PDOS) data, we use a much strait-forward and simple method named the direct free energy interpolation (DFEI) method, to construct full anharmonic effects at any temperature.
The DFEI method extracts anharmonic free energy at any temperature based on the limited several TD-PDOS data from MD simulations. The free energies 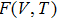
We perform classical MD simulations to simulate Al at different pressure and temperature. Then, we gather the trajectories of all the atoms in crystal to calculate phonon density of states. The embedded-atom-method (EAM)[19–20] potential developed by Mishin et al.[21–22] was used to describe the interatomic interactions of Al atoms. For a metal system containing N atoms, the total potential energy 
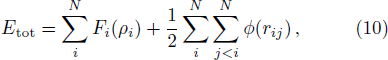
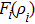
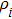
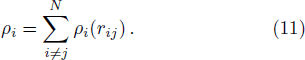
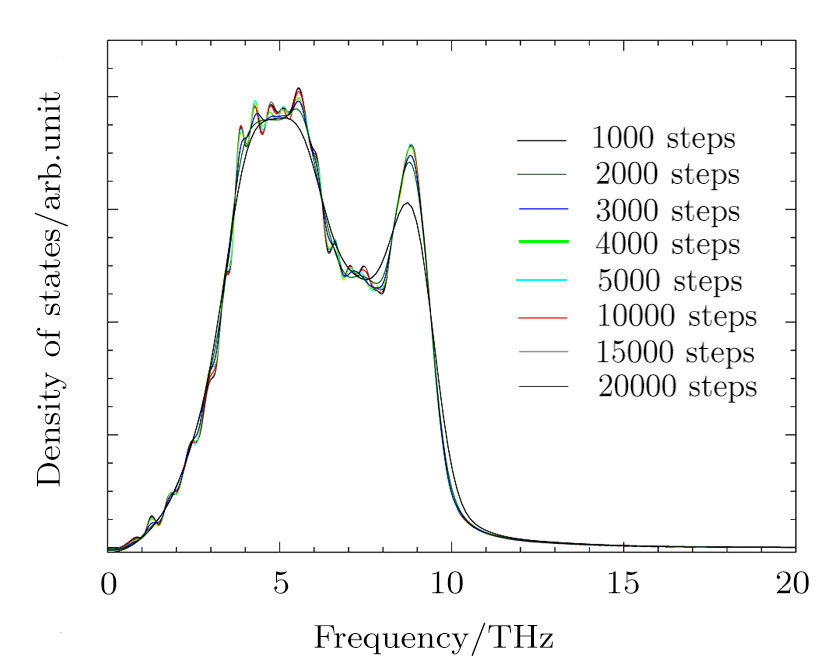
The MD simulations were conducted with the large-scale atomic/molecular massively parallel simulator (LAMMPS)[23–24] package. The simulation box constructed from the multiplication 14×14×14 of the face-centered-cubic (fcc) conventional unit cell including 10976 atoms. The simulations were conducted for all the supercells with different volumes in the canonical ensemble (NVT). The periodic boundary condition was used for all the atoms in the simulation box. The time step was 1 fs and the total number of time steps were 10000. From Fig.
The phonon dispersion curve of Al were calculated using the direct method from molecular dynamics simulations.[25] The calculated phonon dispersion curve of Al are compared well with experimental data at 298 K[26] in Fig. 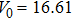
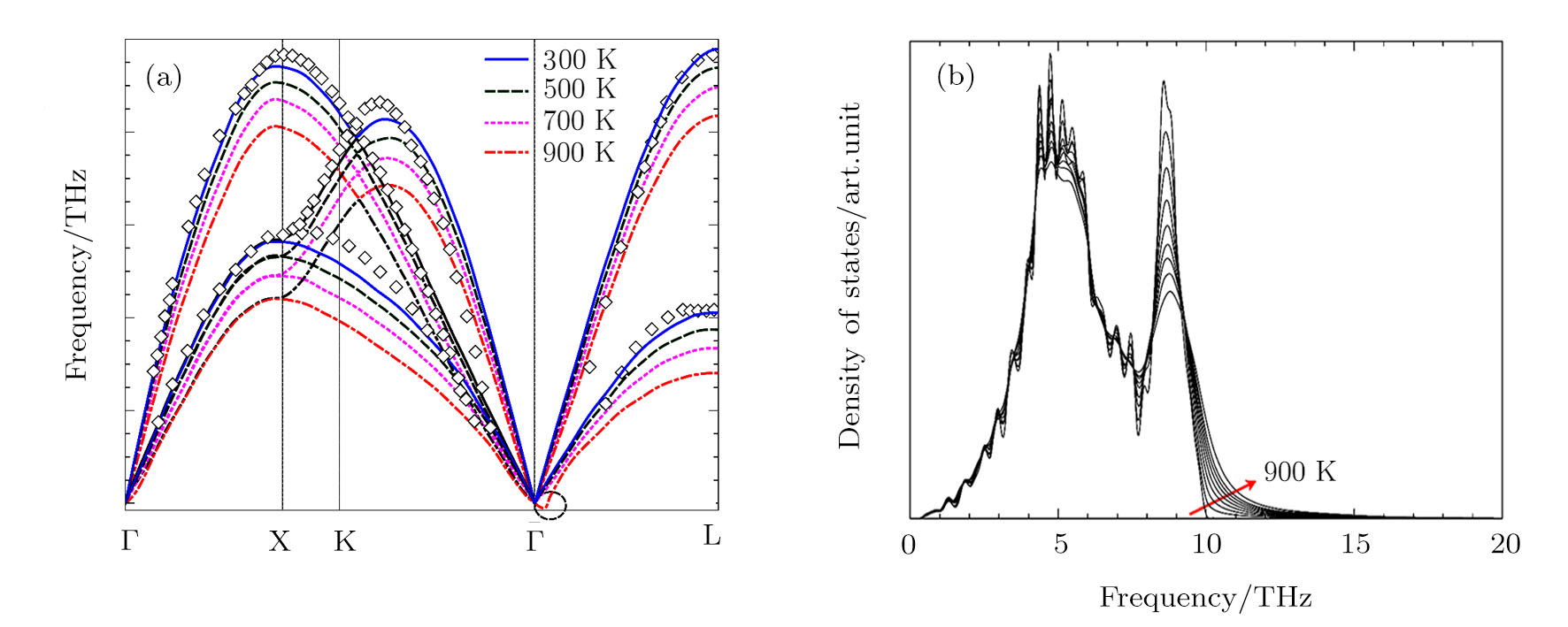 | Fig. 2 (a) The comparison of the calculated 0 GPa phonon dispersion curves of Al with experimental data at 298 K.[26] The circle indicates the negative frequencies at 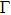 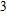 |
The volumes of supercells used for PDOS calculations had volumes varying from 1.08 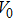
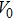
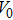
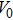
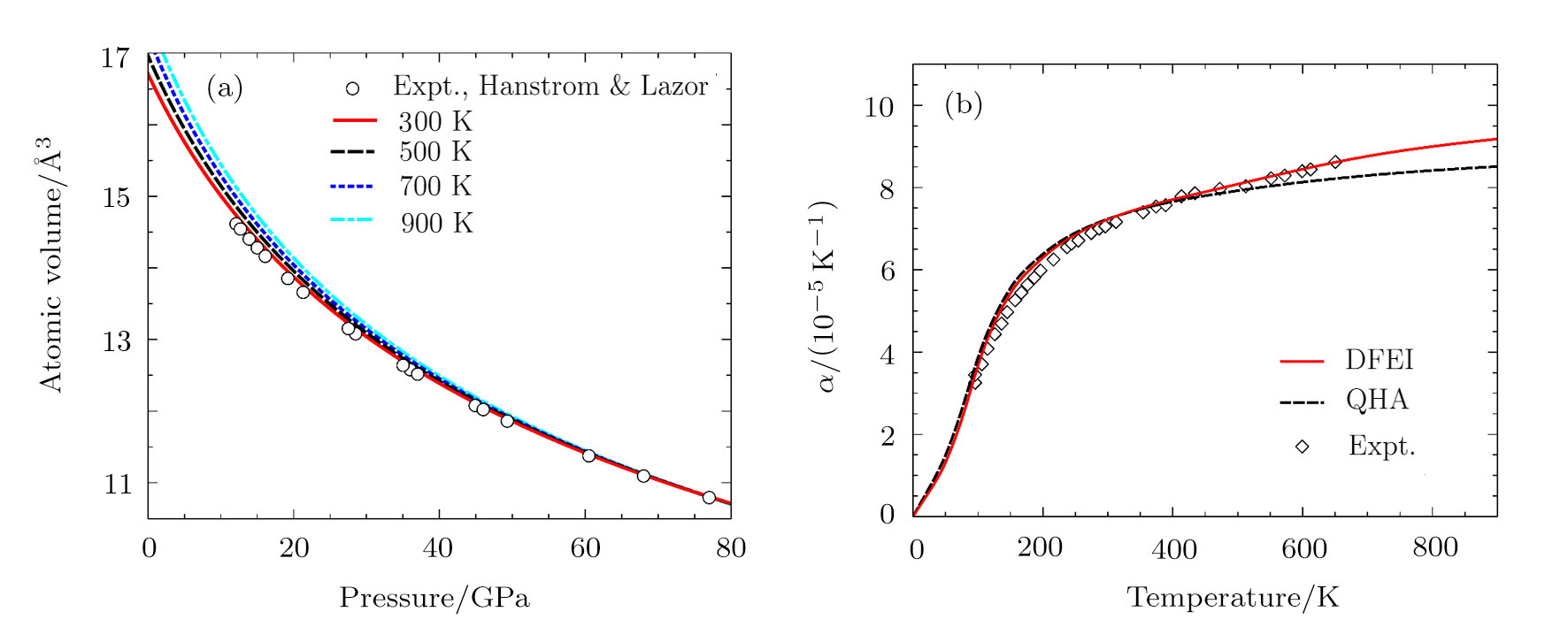
We calculate the Helmholtz free energy from the PDOS according to Eq. (
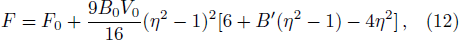
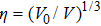
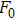
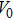
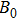
The accuracy of the calculated thermal properties was checked by the thermal expansion coefficient defined by,
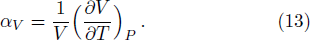
The calculated thermal expansion coefficients as a function of temperature are shown in Fig.
The specific heat at constant volume was calculated by
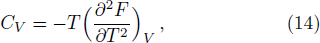
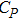
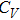
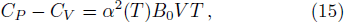
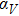
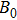
Figure 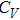
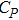
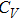
 | Fig. 4 The specific heat of Al at constant volume and at constant pressure compared with the experimental data.[35] |
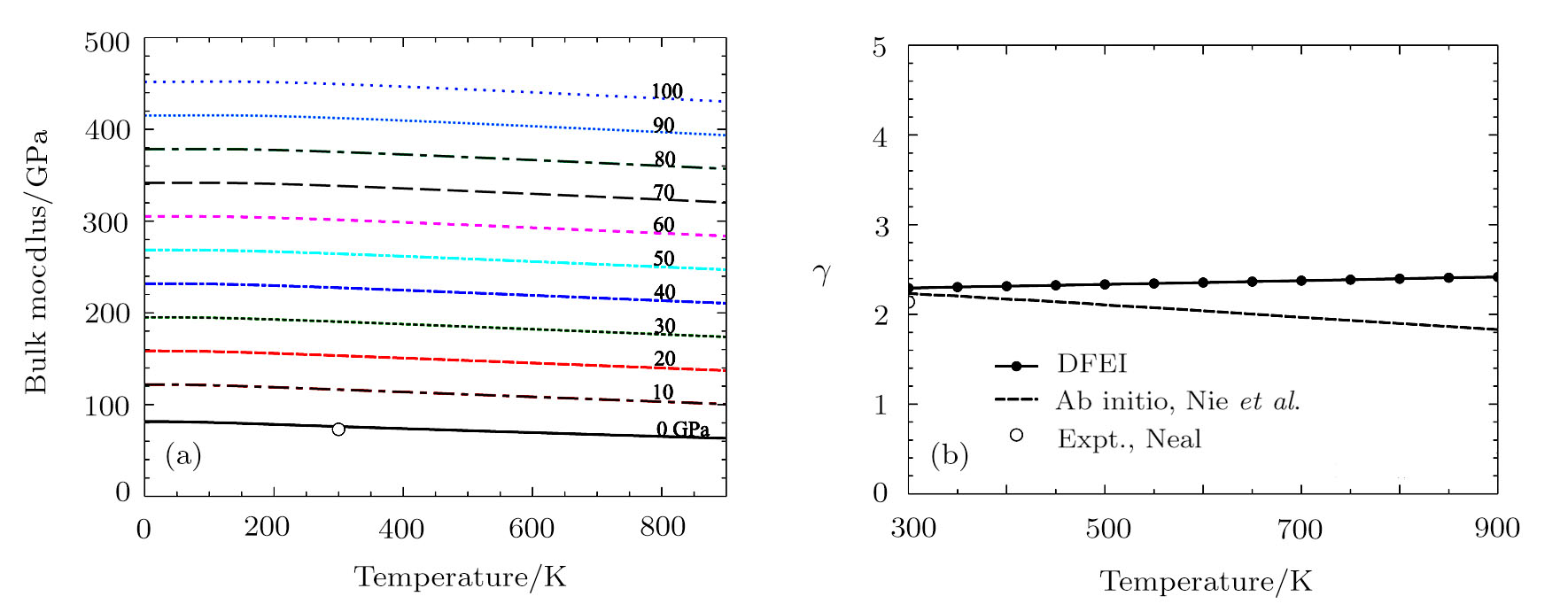
The thermodynamic Grüneisen parameter was achieved by
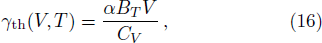
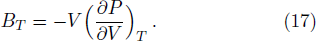
The calculated isothermal bulk modulus versus temperature curves at different pressures are shown in Fig.
In conclusion, we calculate and correct the thermodynamic properties of Al using the direct calculation method of full free energy of lattice vibrations, i.e., the DFEI method which makes accurate anharmonic corrections for the thermodynamic properties of materials beyond the QHA. The calculated thermal expansion coefficients of Al using DFEI method agree better with experiment than the QHA results. Furthermore, the calculated isotherms, the constant volume and constant pressure heat capacities, the bulk moduli using DFEI are all in good agreement with experimental data. This indicates that the DFEI method is indeed an efficient and accurate method to determining the thermodynamic properties of materials from the high temperature phonon density of states.
| [1] | |
| [2] | |
| [3] | |
| [4] | |
| [5] | |
| [6] | |
| [7] | |
| [8] | |
| [9] | |
| [10] | |
| [11] | |
| [12] | |
| [13] | |
| [14] | |
| [15] | |
| [16] | |
| [17] | |
| [18] | |
| [19] | |
| [20] | |
| [21] | |
| [22] | |
| [23] | |
| [24] | |
| [25] | |
| [26] | |
| [27] | |
| [28] | |
| [29] | |
| [30] | |
| [31] | |
| [32] | |
| [33] | |
| [34] | |
| [35] | |
| [36] | |
| [37] | |
| [38] |