


2. 盐城市第一人民医院肿瘤科,江苏 盐城 224005
 ,
ZHOU Yi-chan1,
ZHU Guo-qin1,
LI Jian-ping2,
JIAO Zheng1,
LI Xiao-lin1,
SHAO Yun1,
SUN Wei-hao1
,
ZHOU Yi-chan1,
ZHU Guo-qin1,
LI Jian-ping2,
JIAO Zheng1,
LI Xiao-lin1,
SHAO Yun1,
SUN Wei-hao1

2. Dept of Oncology, the First People′s Hospital of Yancheng City, Yancheng Jiangsu 224005, China
胃癌是影响我国人民生命健康最严重的恶性肿瘤之一,是癌症死亡的主要原因。根治性手术是目前治疗胃癌的最有效方法,但手术不能完全避免局部复发和远处转移的可能,防止肿瘤复发和转移的辅助治疗越来越受到国内外学者的重视。熊果酸(ursolic acid,UA)是广泛存在于白花蛇舌草、女贞子、乌梅和夏枯草等天然植物中的一种五环三萜类化合物,具有抑制肿瘤细胞增殖、诱导细胞凋亡、抗血管生成、抗促癌、抗突变和调节氧化应激等作用,近年来已成为肿瘤化学预防研究的热点[1-3]。临床前研究显示,UA在转基因肿瘤动物模型和人癌裸鼠移植模型中的抑瘤作用明显[3-4],具有良好的临床应用前景。我们的前期研究发现,UA促进胃癌细胞内活性氧(reactive oxygen species,ROS)生成,诱导单磷酸腺苷激活的蛋白激酶(AMPactivated protein kinase,AMPK)磷酸化,抑制信号转导与转录活化因子3(signal transducer and activator of transcription 3,STAT3)磷酸化和环氧化酶-2(cycloxygenase-2,COX-2)表达[5-6]。UA通过下调COX-2表达而抑制胃癌细胞增殖、诱导凋亡[6-7]。然而,UA抑制胃癌细胞COX-2表达的信号转导通路尚不清楚。
本研究选择2株不同分化程度但COX-2高表达的SGC-7901和MKN-45胃癌细胞株作为研究对象,观察ROS/AMPK/STAT3信号转导通路在UA影响SGC-7901和MKN-45胃癌细胞株COX-2表达中的作用,进一步探讨UA抑制胃癌细胞COX-2表达的具体机制。
1 材料与方法 1.1 药物和试剂UA、AMPK激活剂5氨基咪唑4甲酰胺核苷酸(AICAR)、二甲基亚砜(DMSO)、碘化丙啶(PI)、苯甲磺酰氟(phenylmethanesulfonyl fluoride,PMSF) 和抑肽酶为美国SigmaAldrich公司产品;AMPK抑制剂compound C和STAT3抑制剂WP1066为德国Merck公司产品;RPMI 1640培养液和胎牛血清为美国Gibco BRL公司产品;蛋白质定量BCA试剂盒为美国Pierce公司产品;抗氧化剂N乙酰L半胱氨酸(NAC)为江苏碧云天生物技术公司产品;兔抗人AMPK和磷酸化AMPK(p-AMPK)、乙酰辅酶A羧化酶(ACC)和磷酸化ACC(p-ACC)、STAT3和磷酸化STAT3(p-STAT3)、COX-2和β-ac-tin单克隆抗体为美国Cell Signaling Technology公司产品;辣根过氧化物酶标记的山羊抗兔IgG抗体为美国Bioworld Technology公司产品。PVDF膜为美国Millipore公司产品;ECL发光试剂盒为英国Amersham公司产品;其它试剂为国产分析纯级。
1.2 细胞和培养人胃腺癌中分化细胞株SGC-7901购自中国科学院上海生科院细胞资源中心,人胃腺癌低分化细胞株MKN-45购自南京凯基生物科技发展有限公司。SGC-7901和MKN-45细胞常规传代培养于含10%胎牛血清、100 kU·L-1青霉素和100 mg·L-1链霉素的RPMI 1640培养液中,37℃、5% CO2及饱和湿度的二氧化碳培养箱中培养生长。隔天换液,3 d传代1次。
1.3 药物配制UA、AICAR、compound C和WP1066先以DMSO溶解,NAC用超纯水溶解,而后均以RPMI 1640培养液稀释至所需浓度,DMSO在培养液中的浓度不超过0.1%,0.22 μm的微孔滤膜过滤除菌后4℃保存备用。
1.4 实验分组将传代后处于对数生长期的细胞分为对照(control)组、UA(30 μmol·L-1)组、抗氧化剂(NAC,5 mmol·L-1)组、NAC+UA组、AMPK激活剂(AICAR,0.5 mmol·L-1)组、AICAR+UA组、AMPK抑制剂(compound C,2.5 μmol·L-1)组、compound C+UA组、STAT3抑制剂(WP1066,5 μmol·L-1)组、WP1066+UA组。NAC+UA组NAC预处理30 min后UA再干预培养24 h;AICAR+UA组AICAR预处理2 h后UA再干预培养24 h;compound C+UA组compound C预处理1 h后UA再干预培养24 h;WP1066+UA组WP1066预处理1 h后UA再干预培养24 h。
1.5 细胞蛋白提取和Western blot检测各组干预培养后的细胞用预冷的PBS洗涤3次,以100 μL细胞裂解液(PBS内含:Nonidet P-40 1%,脱氧胆酸钠5 g·L-1,SDS 1 g·L-1,PMSF 0.1 g·L-1和抑肽酶10 mg·L-1)4℃处理60 min。细胞裂解物经11 000×g 4℃离心10 min后取上清,BCA试剂盒测定其蛋白浓度。常规进行SDSPAGE电泳后转印至PVDF膜,室温封闭2 h,分别加入一抗(APMK抗体、p-AMPK抗体、ACC抗体、p-ACC抗体、STAT3抗体、p-STAT3抗体、COX-2抗体和β-ac-tin抗体),4℃孵育过夜,辣根过氧化物酶标记的山羊抗兔IgG抗体为第二抗体,4℃孵育2 h,ECL发光,使用ImageJ(National Institute of Health,Bethesda,MD)图像分析软件对蛋白电泳带的灰度进行半定量分析。
1.6 统计学处理实验结果以x±s表示,所有数据均应用SPSS 17.0统计软件包进行分析,多组间均数的比较采用单因素方差分析(ANOVA),组间两两比较用LSD法检验。
2 结果 2.1 UA和抗氧化剂NAC对胃癌细胞AMPK、STAT3磷酸化和COX-2表达的影响Western blot检查结果显示,抗氧化剂NAC有效抑制UA诱导的AMPK磷酸化,ACC是AMPK的下游直接效应靶蛋白,p-ACC被认为是观察AMPK被激活的最佳指标[8](Fig 1),UA抑制STAT3磷酸化和COX-2表达的作用被NAC所逆转(Fig 2)。
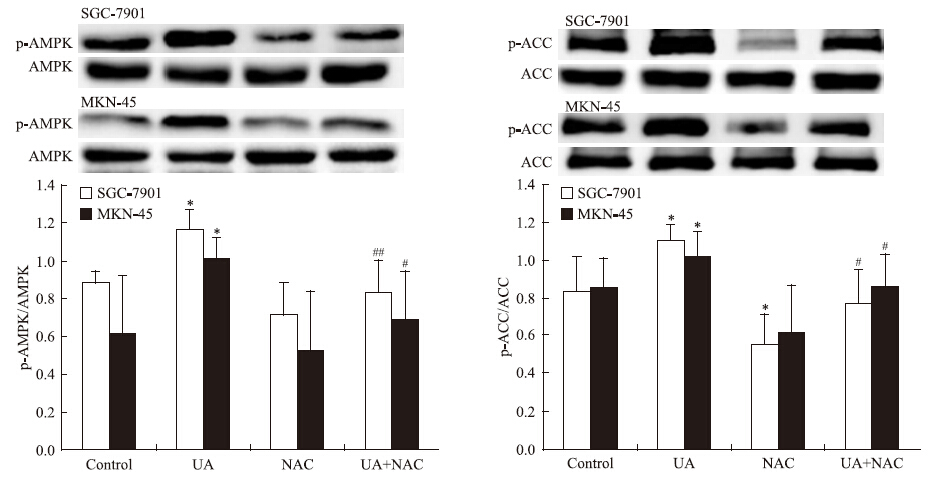
|
| Fig 1 Effects of NAC on ursolic acid(UA)-induced AMPK and ACC phosphorylation in gastric cancer cells The top panels show a representative immunoblot of six separate experiments undertaken. The histograms at the bottom represent the relative expression of phosphoAMPK(p-AMPK) or phosphoACC(p-ACC) compared with total AMPK or total ACC respectively. All data represent the x±s of six independent experiments.*P<0.05 vs control.#P<0.05,##P<0.01 vs UA treatment without NAC pre-incubation. |
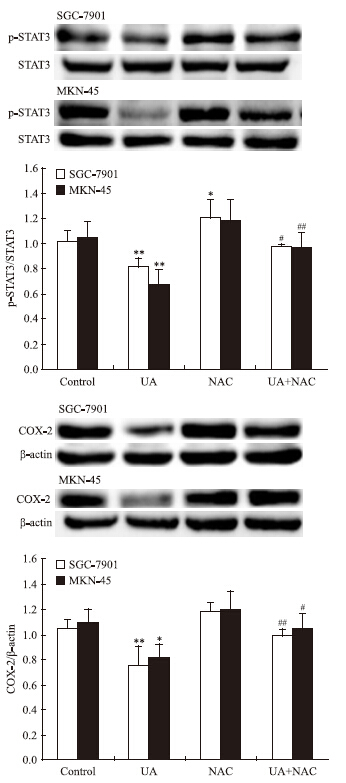
|
| Fig 2 Effects of NAC on UAinhibited STAT3 phosphorylation and COX-2 expression in gastric cancer cells The top panels show a representative immunoblot of six separate experiments undertaken. The histograms at the bottom represent the relative expression of phosphoSTAT3(p-STAT3) or COX-2 compared with total STAT3 or β-ac-tin respectively. All data represent the x±s of six independent experiments.*P<0.05,**P<0.01 vs control.#P<0.05,##P<0.01 vs UA treatment without NAC pre-incubation. |
Western blot检查结果显示,AMPK激活剂AICAR单用或联合UA均可诱导SGC-7901和MKN-45细胞AMPK磷酸化(Fig 3),抑制STAT3磷酸化和COX-2表达,联合作用大于单药作用(Fig 4)。
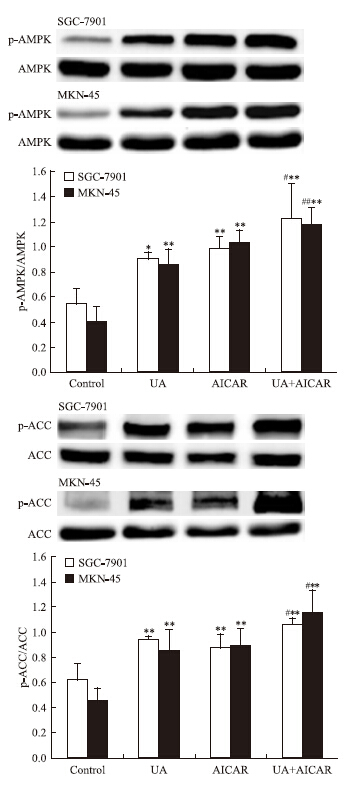
|
| Fig 3 Effects of AMPK activator AICAR(5-amino-4-imidazolecarboxamide ribosid-1-b-Dribofuranoside) on UA -induced AMPK and ACC phosphorylation in gastric cancer cells The top panels show a representative immunoblot of six separate experiments undertaken. The histograms at the bottom represent the relative expression of phosphoAMPK(p-AMPK) or phosphoACC(p-ACC) compared with total AMPK or total ACC respectively. All data represent the x±s of six independent experiments.*P<0.05,**P<0.01 vs control.#P<0.05,##P<0.01 vs UA treatment without AICAR pre-incubation. |
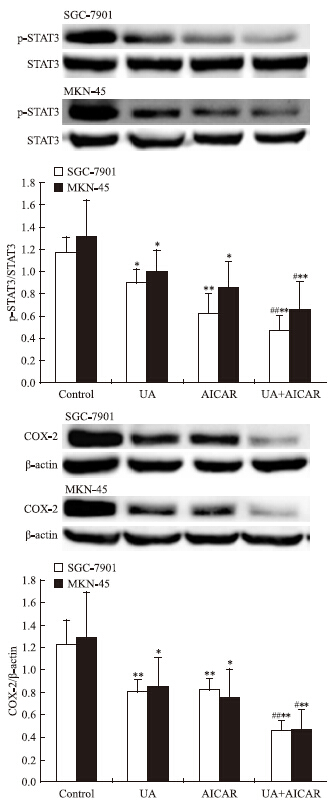
|
| Fig 4 Effects of AMPK activator AICAR on UAinhibited STAT3 phosphorylation and COX-2 expression in gastric cancer cells The top panels show a representative immunoblot of six separate experiments undertaken. The histograms at the bottom represent the relative expression of phosphoSTAT3(p-STAT3) or COX-2 compared with total STAT3 or β-ac-tin respectively. All data represent the x±s of six independent experiments.*P<0.05,**P<0.01 vs control.#P<0.05,##P<0.01 vs UA treatment without AICAR pre-incubation |
AMPK抑制剂compound C有效抑制UA诱导的AMPK磷酸化(Fig 5),UA抑制STAT3磷酸化和COX-2表达的作用被compound C所逆转(Fig 6)。
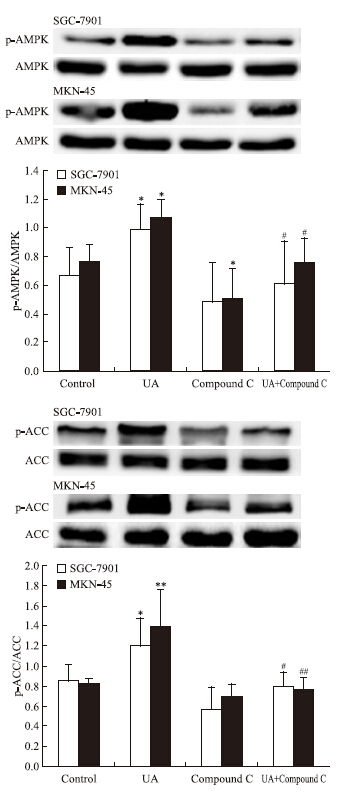
|
| Fig 5 Effects of AMPK inhibitor compound C on UA-induced AMPK and ACC phosphorylation in gastric cancer cells The top panels show a representative immunoblot of six separate experiments undertaken. The histograms at the bottom represent the relative expression of phosphoAMPK(p-AMPK) or phosphoACC(p-ACC) compared with total AMPK or total ACC respectively. All data represent the x±s of six independent experiments.*P<0.05,**P<0.01 vs control.#P<0.05,##P<0.01 vs UA treatment without compound C pre-incubation. |
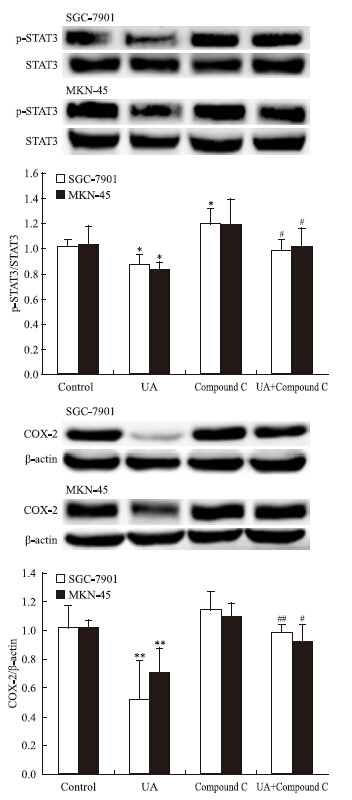
|
| Fig 6 Effects of AMPK inhibitor Compound C on UAinhibited STAT3 phosphorylation and COX-2 expression in gastric cancer cells he top panels show a representative immunoblot of six separate experiments undertaken. The histograms at the bottom represent the relative expression of phosphoSTAT3(p-STAT3) or COX-2 compared with total STAT3 or β-ac-tin respectively. All data represent the x±s of six independent experiments.*P<0.05,**P<0.01 vs control.#P<0.05,##P<0.01 vs UA treatment without Compound C pre-incubation. |
STAT3抑制剂WP1066对UA诱导的AMPK磷酸化无明显影响(Fig 7),WP1066单用或联合UA均可抑制SGC-7901和MKN-45细胞STAT3磷酸化和COX-2表达,且联合作用大于单药作用(Fig 8)。
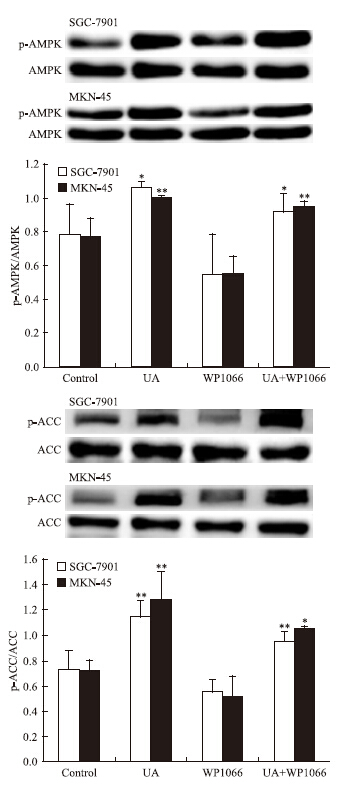
|
| Fig 7 Effects of STAT3 inhibitor WP1066 on UA-induced AMPK and ACC phosphorylation in gastric cancer cells The top panels show a representative immunoblot of six separate experiments undertaken. The histograms at the bottom represent the relative expression of phosphoAMPK(p-AMPK) or phosphoACC(p-ACC) compared with total AMPK or total ACC respectively. All data represent the x±s of six independent experiments.*P<0.05,**P<0.01 vs control. |
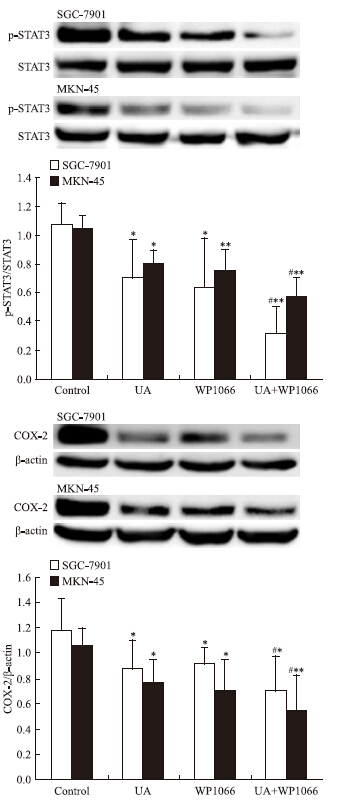
|
| Fig 8 Effects of STAT3 inhibitor WP1066 on UAinhibited STAT3 phosphorylation and COX-2 expression in gastric cancer cells The top panels show a representative immunoblot of six separate experiments undertaken. The histograms at the bottom represent the relative expression of phosphoSTAT3(p-STAT3) or COX-2 compared with total STAT3 or β-ac-tin respectively. All data represent the x±s of six independent experiments.*P<0.05,**P<0.01 vs control.#P<0.05 vs UA treatment without WP1066 pre-incubation. |
ROS是氧在机体代谢过程中产生的中间产物及其衍生物,参与细胞增殖、分化、转化和凋亡以及细胞内重要信号途径的转导[9],近年来研究表明,抗肿瘤药物诱导细胞凋亡与其引起肿瘤细胞内ROS水平增加密切相关[10-12]。AMPK是细胞能量代谢的主要调节器[13],激活AMPK可诱导胃癌和胰腺癌等肿瘤细胞发生凋亡[14-15],UA和白藜芦醇(resveratrol)的抗肿瘤作用与ROS依赖的AMPK活化有关[5, 16-17]。
AMPK除参与细胞内的能量代谢调节外还调控一些基因的转录和表达[18],本研究结果显示,UA诱导AMPK磷酸化,抑制STAT3磷酸化和COX-2表达;抗氧化剂NAC能逆转UA对STAT3磷酸化和COX-2表达的抑制作用,提示ROS是UA抑制STAT3活性和COX-2表达的重要介质[5]。AMPK激活剂AICAR是AMP类似物,明显抑制STAT3磷酸化和COX-2表达,UA与AICAR联合使用对STAT3磷酸化和COX-2表达的抑制作用和单用UA组相比差异有显著性,提示UA联合AICAR后AMPK得到进一步的激活。而AMPK抑制剂compound C则逆转UA对STAT3磷酸化和COX-2表达的抑制作用,进一步证明UA通过AMPK信号通路抑制胃癌细胞STAT3活化和COX-2表达。
我们前期研究发现,JAK2/STAT3和PI3K/Akt信号途径能诱导胃癌细胞COX-2的表达[19],而UA能阻断STAT3通路抑制多发性骨髓瘤细胞增殖[20]。STAT3在各种类型的人胃癌细胞株和胃癌组织中都有较高的活性,JAK/STAT信号转导途径可能在胃癌的发生、发展中起重要的作用[21]。因此,我们推测UA通过ROS/AMPK/STAT3通路下调COX-2表达。当然,由于细胞内信号转导机制十分复杂,UA抑制胃癌细胞COX-2表达尚不排除有其它通路的存在,也不排除不同通路之间存在关联(如crosstalk等),要全面了解其机制需大量而深入的研究。
| [1] | Kalani K, Yadav D K, Khan F, et al. Pharmacophore, QSAR, and ADME based semisynthesis and in vitro evaluation of ursolic acid analogs for anticancer activity[J]. J Mol Model, 2012, 18 (7) : 3389-413 doi:10.1007/s00894-011-1327-6. |
| [2] | Zhao C, Yin S, Dong Y, et al. Autophagy-dependent EIF2AK3 activation compromises ursolic acid-induced apoptosis through upregulation of MCL1 in MCF-7 human breast cancer cells[J]. Autophagy, 2013, 9 (2) : 196-207 doi:10.4161/auto.22805. |
| [3] | Shanmugam M K, Manu K A, Ong T H, et al. Inhibition of CXCR4/CXCL12 signaling axis by ursolic acid leads to suppression of metastasis in transgenic adenocarcinoma of mouse prostate model[J]. Int J Cancer, 2011, 129 (7) : 1552-63 doi:10.1002/ijc.v129.7. |
| [4] | Prasad S, Yadav V R, Sung B, et al. Ursolic acid inhibits growth and metastasis of human colorectal cancer in an orthotopic nude mouse model by targeting multiple cell signaling pathways: chemosensitization with capecitabine[J]. Clin Cancer Res, 2012, 18 (18) : 4942-53 doi:10.1158/1078-0432.CCR-11-2805. |
| [5] | 庞姗姗, 朱悦, 周逸婵, 等. 熊果酸通过ROS/AMPK/STAT3信号通路抑制胃癌细胞COX-2表达[J]. 江苏医药, 2015, 41 (12) : 1368-70 . Pang S S, Zhu Y, Zhou Y C, et al. Ursolic acid inhibiting COX-2 expression in gastric cancer cell through ROS/AMPK/STAT3 signal pathway[J]. Jiangsu Med J, 2015, 41 (12) : 1368-70 . |
| [6] | 唐丹, 李剑萍, 郑锡凤, 等. 熊果酸通过STAT3通路调控胃癌细胞增殖和凋亡[J]. 中国药理学通报, 2012, 28 (2) : 179-84 . Tang D, Li J P, Zheng X F, et al. Regulation of ursolic acid on proliferation and apoptosis of gastric cancer cells via STAT3 signaling pathway[J]. Chin Pharmacol Bull, 2012, 28 (2) : 179-84 . |
| [7] | Zhang H, Li X, Ding J, et al. Delivery of ursolic acid(UA) in polymeric nanoparticles effectively promotes the apoptosis of gastric cancer cells through enhanced inhibition of cyclooxygenase 2(COX-2)[J]. Int J Pharm, 2013, 441 (1-2) : 261-8 doi:10.1016/j.ijpharm.2012.11.034. |
| [8] | Shaw R J. Glucose metabolism and cancer[J]. Curr Opin Cell Biol, 2006, 18 (6) : 598-608 doi:10.1016/j.ceb.2006.10.005. |
| [9] | Ray P D, Huang B W, Tsuji Y. Reactive oxygen species(ROS) homeostasis and redox regulation in cellular signaling[J]. Cell Signal, 2012, 24 (5) : 981-90 doi:10.1016/j.cellsig.2012.01.008. |
| [10] | Yi B, Liu D, He M, et al. Role of the ROS/AMPK signaling pathway in tetramethylpyrazine-induced apoptosis in gastric cancer cells[J]. Oncol Lett, 2013, 6 (2) : 583-9 . |
| [11] | Li X, Zheng D, Lu X, et al. Enhanced cytotoxicity and activation of ROS-dependent c-Jun NH2-terminal kinase and caspase-3 by low doses of tetrandrine-loaded nanoparticles in Lovo cells-A possible Trojan strategy against cancer[J]. Eur J Pharm Biopharm, 2010, 75 (3) : 334-40 doi:10.1016/j.ejpb.2010.04.016. |
| [12] | Li X, Lu X, Xu H, et al. Paclitaxel/tetrandrine coloaded nanoparticles effectively promote the apoptosis of gastric cancer cells based on "oxidation therapy"[J]. Mol Pharm, 2012, 9 (2) : 222-9 doi:10.1021/mp2002736. |
| [13] | Hardie D G. AMPK: positive and negative regulation, and its role in whole-body energy homeostasis[J]. Curr Opin Cell Biol, 2015, 33 : 1-7 . |
| [14] | Saitoh M, Nagai K, Nakagawa K, et al. Adenosine induces apoptosis in the human gastric cancer cells via an intrinsic pathway relevant to activation of AMP-activated protein kinase[J]. Biochem Pharmacol, 2004, 67 (10) : 2005-11 doi:10.1016/j.bcp.2004.01.020. |
| [15] | Wang B, Wang X B, Chen L Y, et al. Belinostat-induced apoptosis and growth inhibition in pancreatic cancer cells involve activation of TAK1-AMPK signaling axis[J]. Biochem Biophys Res Commun, 2013, 437 (1) : 1-6 doi:10.1016/j.bbrc.2013.05.090. |
| [16] | Zheng Q Y, Jin F S, Yao C, et al. Ursolic acid-induced AMP-activated protein kinase(AMPK) activation contributes to growth inhibition and apoptosis in human bladder cancer T24 cells[J]. Biochem Biophys Res Commun, 2012, 419 (4) : 741-7 doi:10.1016/j.bbrc.2012.02.093. |
| [17] | Yuan Y, Xue X, Guo R B, et al. Resveratrol enhances the antitumor effects of temozolomide in glioblastoma via ROS-dependent AMPK-TSC-mTOR signaling pathway[J]. CNS Neurosci Ther, 2012, 18 (7) : 536-46 doi:10.1111/j.1755-5949.2012.00319.x. |
| [18] | McGee S L, Hargreaves M. AMPK and transcriptional regulation[J]. Front Biosci, 2008, 13 : 3022-33 doi:10.2741/2907. |
| [19] | Xu W, Chen G S, Shao Y, et al. Gastrin acting on the cholecystokinin2 receptor induces cyclooxygenase-2 expression through JAK2/STAT3/PI3K/Akt pathway in human gastric cancer cells[J]. Cancer Lett, 2013, 332 (1) : 11-8 doi:10.1016/j.canlet.2012.12.030. |
| [20] | Pathak A K, Bhutani M, Nair A S, et al. Ursolic acid inhibits STAT3 activation pathway leading to suppression of proliferation and chemosensitization of human multiple myeloma cells[J]. Mol Cancer Res, 2007, 5 (9) : 943-55 doi:10.1158/1541-7786.MCR-06-0348. |
| [21] | Huang W, Yu L F, Zhong J, et al. Stat3 is involved in angiotensin II-induced expression of MMP2 in gastric cancer cells[J]. Dig Dis Sci, 2009, 54 (10) : 2056-62 doi:10.1007/s10620-008-0617-z. |