文章信息
- 卢利霞, 杨丹丹
- LU Lixia, YANG Dandan
- 慢性子宫内膜炎患者血清MCP-1水平及其与子宫内膜息肉、炎症反应的相关性
- Serum MCP-1 level and its correlation with endometrial polyps and inflammatory response in patients with chronic endometritis
- 中国医科大学学报, 2026, 55(3): 251-258
- Journal of China Medical University, 2026, 55(3): 251-258
-
文章历史
- 收稿日期:2025-04-03
- 网络出版时间:2026-03-24 13:04:09
慢性子宫内膜炎(chronic endometritis,CE)作为子宫内膜容受性损伤的重要诱因,其病理核心在于浆细胞异常浸润导致的持续性黏膜炎症[1-2]。这种微环境改变通过干扰胚胎着床,显著增加不良妊娠结局风险。CE在不孕症群体中的检出率为2.8%~56.8%,反复种植失败女性中为14%~67.5%,复发性妊娠丢失女性中为9.3%~67.6%[3]。然而,CE通常无症状,或偶见轻微非特异性症状,如异常子宫出血、白带异常、骨盆疼痛、性交疼痛等,临床医生对CE难以识别及干预[4]。子宫内膜息肉(endometrial polyp,EP)是由腺体、间质和血管组成的异常增生物,其发生可能与炎症、激素紊乱、免疫失衡等因素有关[5]。研究[6]表明,慢性炎症是全身多种黏膜组织息肉发生的致病因素,提示CE可能通过持续性的炎性刺激导致EP的形成。单核细胞趋化蛋白-1(monocyte chemoattractant protein-1,MCP-1)是重要的炎性刺激因子之一,由炎症相关细胞和特定组织或器官的上皮细胞产生并释放,吸引免疫细胞定向迁移至炎症部位,从而放大局部炎症反应[7]。既往研究[8-10]发现MCP-1与慢性炎症密切相关,然而,MCP-1与CE和EP的关系鲜有报道。基于此,本研究旨在探讨MCP-1在CE合并EP患者血清中的水平及其与炎症反应的关系,以期为临床提供参考。
1 材料与方法 1.1 研究对象选取2021年6月至2024年6月我院妇科收治的CE患者157例作为研究对象,年龄25~80岁,平均(46.38±9.13)岁。根据是否存在EP分为EP组(n = 75)和非EP组(n = 82)。纳入标准:(1)子宫内膜活检确诊为CE;(2)EP组患者在宫腔镜息肉切除术后病理确诊为EP;(3)临床资料完整。排除标准:(1)合并其他器质性病变;(2)合并恶性肿瘤;(3)存在子宫或生殖道畸形;(4)存在其他宫腔病变。本研究经我院伦理委员会批准(HBZY2022-KY-040-01)。
1.2 基线资料收集收集患者基线资料,包括年龄、体重指数、糖尿病、高血压、口服避孕药史、吸烟史、饮酒史、CE病程及严重程度、月经周期规律性、是否绝经、孕产次数、流产次数;盆腔疼痛、子宫异常出血、白带异常等。
1.3 实验室指标检测采集患者清晨空腹血5 mL,2 000 r/min离心10 min,分离血清,置于-70 ℃冰箱保存待检。应用酶联免疫吸附试验(enzyme-linked immunosorbent assay,ELISA)测定MCP-1、白细胞介素-6(interleukin-6,IL-6)、肿瘤坏死因子α(tumor necrosis factor α,TNF-α)、白细胞介素-1β(interleukin-1β,IL-1β)、C反应蛋白(C-reactive protein,CRP)水平,ELISA试剂盒均购自上海酶联生物技术有限公司。同时采集患者清晨空腹血4 mL,应用化学发光免疫分析法(AU5800全自动生化仪,美国贝克曼公司)检测患者血清卵泡刺激素(follicle-stimulating hormone,FSH)、雌二醇(estradiol,E2)、黄体生成素(luteinizing hormone,LH)水平。
1.4 统计学分析采用SPSS 23.0软件进行统计分析,计量资料以x±s表示,2组比较采用t检验;计数资料以率(%)表示,组间比较采用χ2检验。构建分层回归模型分析临床指标与MCP-1的关系,通过多元线性回归分析并构建校正模型,分析MCP-1水平与血清性激素及炎症指标的关系;应用非条件logistic回归、RCS分析MCP-1水平与EP的关联及剂量-反应关系;采用广义多因子降维法(generalized multifactor dimensionality reduction,GMDR)模型分析炎症指标对EP的交互作用。P < 0.05为差异有统计学意义。
2 结果 2.1 MCP-1水平与CE患者临床特征的相关性不同临床特征(包括流产次数、白带异常、异常子宫出血、骨盆疼痛以及是否合并EP)患者的MCP-1水平比较,差异有统计学意义(P < 0.05),见表 1。
| Item | n | MCP-1(ng/mL) | t | P |
| Age | 1.741 | 0.242 | ||
| < 35 years | 68 | 34.50±6.24 | ||
| ≥35 years | 89 | 35.61±5.57 | ||
| Body mass index | 0.663 | 0.508 | ||
| < 22 kg/m2 | 63 | 35.05±6.06 | ||
| ≥22 kg/m2 | 94 | 35.64±5.03 | ||
| Number of miscarriages | 2.026 | 0.045 | ||
| < 2 | 140 | 34.61±5.56 | ||
| ≥2 | 17 | 37.54±6.22 | ||
| Number of births | 0.717 | 0.475 | ||
| < 2 | 142 | 35.16±5.66 | ||
| ≥2 | 15 | 36.27±6.15 | ||
| Pregnancy times | 1.462 | 0.146 | ||
| < 2 | 122 | 34.98±6.27 | ||
| ≥2 | 35 | 36.67±5.08 | ||
| CE course | 1.406 | 0.162 | ||
| < 3 years | 85 | 34.76±6.19 | ||
| ≥3 years | 72 | 36.21±6.72 | ||
| Leucorrhea anomaly | 2.101 | 0.037 | ||
| Yes | 60 | 37.53±7.06 | ||
| No | 97 | 35.51±4.97 | ||
| Menstrual regularity | 1.867 | 0.064 | ||
| Yes | 108 | 34.06±6.65 | ||
| No | 49 | 36.12±5.82 | ||
| Menopause | 1.788 | 0.076 | ||
| Yes | 72 | 34.18±5.71 | ||
| No | 85 | 35.92±6.37 | ||
| Abnormal uterine bleeding | 4.794 | < 0.001 | ||
| Yes | 43 | 39.40±5.41 | ||
| No | 114 | 34.48±5.85 | ||
| Pelvic pain | 3.835 | < 0.001 | ||
| Yes | 95 | 38.24±7.22 | ||
| No | 62 | 33.97±6.15 | ||
| EP | 4.220 | < 0.001 | ||
| Yes | 75 | 38.31±6.10 | ||
| No | 82 | 34.25±5.95 |
2.2 不同临床特征与MCP-1水平的分层回归分析
逐层加入变量,结果显示,异常子宫出血、流产次数、盆腔疼痛、白带异常和EP均是MCP-1水平升高的独立影响因素,其中EP和盆腔疼痛的效应量尤为突出,见表 2。
| Model | Non-standardized coefficient | β | t | P | |
| β | SE | ||||
| Stratification 1 | |||||
| Constant | 7.329 | 0.375 | - | 19.543 | < 0.001 |
| Abnormal uterine bleeding | 0.687 | 0.290 | 0.156 | 2.369 | 0.020 |
| Stratification 2 | |||||
| Constant | 8.146 | 0.429 | - | 18.975 | < 0.001 |
| Abnormal uterine bleeding | 0.588 | 0.217 | 0.062 | 2.715 | 0.008 |
| Number of miscarriages | 0.752 | 0.364 | 0.093 | 2.066 | 0.041 |
| Stratification 3 | |||||
| Constant | 9.759 | 0.551 | - | 17.700 | < 0.001 |
| Abnormal uterine bleeding | 0.638 | 0.207 | 0.061 | 3.073 | 0.003 |
| Number of miscarriages | 0.597 | 0.261 | 0.097 | 2.283 | 0.025 |
| Pelvic pain | 0.861 | 0.362 | 0.130 | 2.376 | 0.019 |
| Stratification 4 | |||||
| Constant | 9.448 | 0.583 | - | 16.217 | < 0.001 |
| Abnormal uterine bleeding | 0.758 | 0.229 | 0.030 | 3.303 | 0.001 |
| Number of miscarriages | 0.959 | 0.399 | 0.095 | 2.403 | 0.018 |
| Pelvic pain | 0.885 | 0.280 | 0.073 | 3.158 | 0.002 |
| Leucorrhea anomaly | 0.836 | 0.407 | 0.198 | 2.053 | 0.043 |
| Stratification 5 | |||||
| Constant | 8.807 | 0.792 | - | 11.124 | < 0.001 |
| Abnormal uterine bleeding | 0.963 | 0.201 | 0.183 | 4.780 | < 0.001 |
| Number of miscarriages | 0.633 | 0.253 | 0.238 | 2.507 | 0.015 |
| Pelvic pain | 0.948 | 0.201 | 0.177 | 4.723 | < 0.001 |
| Leucorrhea anomaly | 0.605 | 0.275 | 0.252 | 2.202 | 0.032 |
| EP | 0.830 | 0.167 | 0.132 | 4.968 | < 0.001 |
2.3 2组患者一般资料比较
2组患者的体重指数、初潮时间等一般资料比较,差异均无统计学意义(P > 0.05),年龄、月经规律、绝经、异常子宫出血、骨盆疼痛、CE病程、流产次数比较,差异均有统计学意义(P < 0.05)。见表 3。
| Item | EP group(n = 75) | Non-EP group(n = 82) | χ2/t | P |
| Age(year) | 48.05±9.58 | 43.59±8.27 | 3.130 | 0.002 |
| BMI(kg/m2) | 23.19±4.77 | 22.49±5.14 | 0.882 | 0.379 |
| Time of menarche(year) | 13.94±2.03 | 14.13±1.96 | 0.596 | 0.552 |
| Menstrual cycle(d) | 28.61±2.67 | 29.07±2.33 | 1.152 | 0.251 |
| Endometrial thickness [n(%)] | 1.331 | 0.514 | ||
| 4-9 mm | 56(74.67) | 66(80.49) | ||
| > 9-14 mm | 11(14.67) | 10(12.20) | ||
| > 14 mm | 8(10.66) | 6(7.31) | ||
| Hypertension [n(%)] | 3.495 | 0.062 | ||
| Yes | 20(26.67) | 12(14.63) | ||
| No | 55(73.33) | 70(85.37) | ||
| Diabetes [n(%)] | 2.373 | 0.123 | ||
| Yes | 10(13.33) | 5(6.10) | ||
| No | 65(86.67) | 77(93.90) | ||
| Smoking history [n(%)] | 1.366 | 0.242 | ||
| Yes | 6(8.00) | 3(3.66) | ||
| No | 69(92.00) | 79(96.34) | ||
| Drinking history [n(%)] | 0.043 | 0.836 | ||
| Yes | 11(14.67) | 13(15.85) | ||
| No | 64(85.33) | 69(84.15) | ||
| Oral contraceptive [n(%)] | 1.352 | 0.245 | ||
| Yes | 3(4.00) | 7(8.54) | ||
| No | 72(96.00) | 75(91.46) | ||
| Menstrual regularity [n(%)] | 5.168 | 0.023 | ||
| Yes | 45(60.00) | 63(76.83) | ||
| No | 30(40.00) | 19(23.17) | ||
| Menopause [n(%)] | 5.947 | 0.015 | ||
| Yes | 42(56.00) | 30(36.59) | ||
| No | 33(44.00) | 52(63.41) | ||
| Abnormal uterine bleeding [n(%)] | 14.041 | < 0.001 | ||
| Yes | 31(41.33) | 12(14.63) | ||
| No | 44(58.67) | 70(85.37) | ||
| Pelvic pain [n(%)] | 12.044 | 0.001 | ||
| Yes | 56(74.67) | 39(47.56) | ||
| No | 19(25.33) | 43(52.44) | ||
| CE course [n(%)] | 7.613 | 0.006 | ||
| < 3 years | 32(42.67) | 53(64.63) | ||
| ≥3 years | 43(57.33) | 29(35.37) | ||
| Pregnancy times [n(%)] | 3.282 | 0.070 | ||
| < 2 | 63(84.00) | 59(71.95) | ||
| ≥2 | 12(16.00) | 23(28.05) | ||
| Number of births [n(%)] | 1.385 | 0.239 | ||
| < 2 | 70(93.33) | 72(87.80) | ||
| ≥2 | 5(6.67) | 10(12.20) | ||
| Number of miscarriages [n(%)] | 4.490 | 0.034 | ||
| < 2 | 71(94.67) | 69(84.15) | ||
| ≥2 | 4(5.33) | 13(15.85) |
2.4 2组患者血清性激素比较
EP组患者FSH(7.19 U/L±0.98 U/L)、LH(3.15 U/L±0.93 U/L)和E2(168.52 pmol/L±65.12 pmol/L)水平均高于无EP组患者(6.63 U/L±1.13 U/L、2.64 U/L±0.86 U/L和130.24 pmol/L±53.29 pmol/L),差异有统计学意义(P < 0.05)。
2.5 2组患者炎症指标比较EP组患者TNF-α、IL-1β、IL-6和CRP水平均高于无EP组患者,差异有统计学意义(P < 0.05)。见表 4。
| Group | n | TNF-α(ng/L) | IL-1β(ng/L) | IL-6(ng/L) | CRP(mg/L) |
| EP group | 75 | 20.35±3.36 | 31.97±5.31 | 125.82±8.63 | 15.70±4.13 |
| Non-EP group | 82 | 17.88±3.24 | 21.26±3.63 | 109.21±8.55 | 12.25±3.14 |
| t | 4.688 | 14.86 | 12.105 | 5.922 | |
| P | < 0.001 | < 0.001 | < 0.001 | < 0.001 |
2.6 CE患者EP的多因素logistic回归分析
将单因素分析中具有显著差异的指标纳入多因素logistic回归分析,结果显示,MCP-1、FSH、LH、E2、TNF-α、IL-1β、IL-6是CE患者合并EP的独立危险因素(P < 0.05)。见表 5。
| Project | β | SE | Wald χ2 | OR | 95%CI | P |
| Menopause | 0.381 | 0.209 | 3.266 | 1.463 | 0.971-2.204 | 0.071 |
| Abnormal uterine bleeding | 0.432 | 0.228 | 3.810 | 1.545 | 1.005-2.371 | 0.054 |
| Pelvic pain | 0.276 | 0.174 | 2.715 | 1.321 | 0.953-1.852 | 0.088 |
| CE course | 0.143 | 0.087 | 2.471 | 1.159 | 0.968-1.377 | 0.117 |
| MCP-1 | 1.120 | 0.243 | 30.279 | 3.521 | 2.173-5.246 | < 0.001 |
| FSH | 0.339 | 0.163 | 4.414 | 1.398 | 1.025-1.933 | 0.042 |
| LH | 0.417 | 0.192 | 4.678 | 1.520 | 1.036-2.204 | 0.036 |
| E2 | 0.851 | 0.138 | 36.731 | 2.342 | 1.765-3.100 | < 0.001 |
| TNF-α | 0.622 | 0.169 | 13.293 | 1.857 | 1.330-2.600 | 0.001 |
| IL-1β | 1.152 | 0.183 | 34.030 | 2.859 | 2.015-4.072 | < 0.001 |
| IL-6 | 1.182 | 0.231 | 26.297 | 3.251 | 2.072-5.110 | < 0.001 |
| CRP | 0.160 | 0.101 | 2.562 | 1.173 | 0.964-1.435 | 0.103 |
2.7 MCP-1水平与血清性激素及炎症指标的相关性分析
多元线性回归分析结果显示,E2和TNF-α、IL-1β、IL-6、CRP在未调整模型和调整模型中,均与MCP-1水平呈显著正相关(P < 0.05),见表 6。
| Project | MCP-1 | |||
| β | OR | 95%CI | P | |
| Unadjusted model | ||||
| FSH | 0.496 | 1.642 | 1.433-1.776 | 0.079 |
| LH | 0.341 | 1.406 | 1.145-1.532 | 0.111 |
| E2 | 0.553 | 1.738 | 1.423-2.123 | < 0.001 |
| TNF-α | 0.889 | 2.433 | 1.666-3.551 | < 0.001 |
| IL-1β | 2.651 | 14.168 | 10.518-19.085 | < 0.001 |
| IL-6 | 0.622 | 1.863 | 1.472-2.357 | < 0.001 |
| CRP | 1.193 | 3.297 | 2.545-4.270 | < 0.001 |
| Model 1 | ||||
| FSH | 0.203 | 1.225 | 1.001-1.357 | 0.425 |
| LH | 0.107 | 1.113 | 0.904-1.323 | 0.427 |
| E2 | 0.243 | 1.275 | 1.067-1.524 | 0.015 |
| TNF-α | 0.355 | 1.426 | 1.082-1.881 | 0.024 |
| IL-1β | 1.941 | 6.966 | 5.441-8.917 | < 0.001 |
| IL-6 | 0.338 | 1.402 | 1.196-1.643 | < 0.001 |
| CRP | 0.883 | 2.418 | 1.802-3.245 | < 0.001 |
| Model 2 | ||||
| FSH | 0.329 | 1.391 | 1.145-1.586 | 0.282 |
| LH | 0.178 | 1.195 | 0.897-1.469 | 0.147 |
| E2 | 0.301 | 1.351 | 1.171-1.559 | < 0.001 |
| TNF-α | 0.569 | 1.766 | 1.332-2.343 | < 0.001 |
| IL-1β | 2.296 | 9.934 | 7.625-12.944 | < 0.001 |
| IL-6 | 0.361 | 1.435 | 1.175-1.752 | 0.001 |
| CRP | 0.785 | 2.192 | 1.628-2.953 | < 0.001 |
| Model 1,adjust BMI,menarche time,menstrual cycle,endometrial thickness,hypertension,diabetes,smoking history,drinking history,oral contraceptives,pregnancy times and parity;Model 2,based on Model 1,adjust age,menstrual regularity,menopause,abnormal uterine bleeding,pelvic pain,CE course and abortion times. | ||||
2.8 MCP-1水平与CE患者合并EP的剂量-反应分析
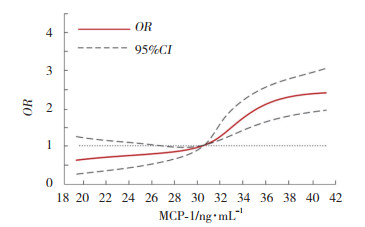
应用限制性立方样条模型分析MCP-1水平与CE患者合并EP的剂量-反应关系。结果显示,MCP-1水平与CE患者合并EP的关联强度存在非线性剂量-反应关系(非线性检验P < 0.001),呈显著正相关,尤其当MCP-1 > 30.30 ng/mL时,随着MCP-1水平升高,CE患者EP患病风险显著上升。见图 1。
|
| 图 1 基于限制性立方样条模型分析MCP-1水平与CE患者EP发生的关系 Fig.1 Analysis of the relationship between MCP-1 levels and EP in patients with CE based on the restricted cubic spline model |
2.9 MCP-1与炎症指标对CE患者EP的GMDR交互作用分析
以MCP-1与TNF-α、IL-1β、IL-6、CRP水平的中位数值作为界值,将各项高水平赋值为1,低水平赋值为0,采用GMDR模型进行交互作用分析,模型结果显示,在CE合并EP患者中,最佳交互作用模型为MCP-1、TNF-α、IL-1β、IL-6高水平之间的交互作用(P < 0.05)。见表 7。
| Model | Test balance accuracy | Replacement test(P) | Cross-validation consistency |
| A1 | 0.532 | 7(0.109) | 6/10 |
| A2/A4 | 0.564 | 6(0.055) | 7/10 |
| A1/A2/A4 | 0.582 | 7(0.023) | 7/10 |
| A1/A2/A3/A4* | 0.611 | 10(0.001) | 9/10 |
| A1/A2/A3/A4/A5 | 0.602 | 9(0.012) | 10/10 |
| A1,A2,A3,A4 and A5 respectively represent high expression of MCP-1,TNF-α,IL-1β,IL-6 and CRP. *,best model. | |||
3 讨论
尽管CE是良性病变,仍能引发不良妊娠结局、降低生育能力[11]。有研究[12]显示,在CE患者宫腔中通常能观察到仅由子宫内膜黏膜的小血管化突起构成的微息肉,微息肉的形成机制可能涉及大量炎症细胞的局部积聚,进而刺激子宫内膜组织过度增殖并诱发异常血管生成,最终形成EP。本研究157例CE患者中有75例伴有EP,发生率为47.77%。研究[13]发现,MCP-1是C-C趋化因子家族的一员,通过与其受体C-C趋化因子受体2(cc chemokine receptor 2,CCR2)结合激活核因子κB(nuclear factor-kappa B,NF-ĸB)等关键的炎症信号通路,在调控细胞迁移的同时,促进白细胞的募集,在感染期间发挥保护性免疫反应的核心作用,MCP-1由子宫内膜、腹膜间皮、巨噬细胞、单核细胞、内皮细胞和成纤维细胞分泌,分泌后可导致单核细胞、巨噬细胞等免疫细胞浸润,并可能导致免疫平衡向Th2极化[14]。
本研究发现在不同临床特征(包括流产次数、白带异常、异常子宫出血、骨盆疼痛以及是否合并EP)的患者中,MCP-1水平均存在统计学差异,且EP患者的MCP-1水平显著高于非EP患者,限制性立方样条模型分析显示,当MCP-1水平高于30.30 ng/mL时,EP的患病风险显著增加。研究[8]表明,病原体相关分子模式、损伤相关分子模式(damage-associa-ted molecular pattern,DAMP)在发挥抗感染作用的同时,也可能导致宿主细胞损伤,当宿主细胞识别这些分子模式后,可激活NF-κB和TNF-α的过度表达,活化的NF-κB进入细胞核内,进而上调MCP-1、TNF-α、IL-6、IL-1β等多种促炎细胞因子的表达,这可能是EP发生发展的重要机制。本研究结果显示,EP组患者的TNF-α、IL-1β、IL-6、CRP水平均显著高于非EP组患者,且与MCP-1水平呈显著正相关,提示促炎细胞因子可能通过上调MCP-1的表达参与EP的发生发展。GMDR交互作用分析结果显示,MCP-1、TNF-α、IL-1β、IL-6与CE患者发生EP之间存在显著的交互作用,且协同作用比单一指标的作用更强,可能是因为MCP-1作为炎症反应的中游分子,通过结合其受体CCR2,吸引单核细胞和巨噬细胞迁移至炎症部位,这些被募集的巨噬细胞进一步释放更多的炎性细胞因子,形成持续的促炎细胞因子正反馈网络。
本研究结果显示,EP组患者的FSH、LH和E2水平显著高于非EP组患者,且E2与MCP-1水平呈显著正相关。雌激素可以通过快速激活细胞膜表面丝裂原活化蛋白激酶等信号通路对其他转录因子进行调控和激活,其他转录因子可激活下游靶基因的转录,参与细胞的生理过程[15-16]。
综上所述,在CE患者中,MCP-1水平与EP的发生密切相关,并受到性激素E2及炎症指标TNF-α、IL-6、IL-1β、CRP的正向调控。MCP-1可能在CE合并EP的发生发展中发挥关键作用。
| [1] |
SINGH N, SETHI A. Endometritis-diagnosis, treatment and its impact on fertility-a scoping review[J]. JBRA Assist Reprod, 2022, 26(3): 538-546. DOI:10.5935/1518-0557.20220015 |
| [2] |
乌恩岳苏, 杨晓燕. 蒙药苏格木勒-7通过抑制TLR4/NF-κB通路减轻子宫内膜炎大鼠炎症反应[J]. 中国医科大学学报, 2024, 53(9): 845-852. DOI:10.12007/j.issn.0258-4646.2024.09.013 |
| [3] |
林明媚, 李蓉. 慢性子宫内膜炎对女性生育结局影响的研究进展[J]. 中华医学杂志, 2024, 104(20): 1893-1897. DOI:10.3760/cma.j.cn112137-20230922-00522 |
| [4] |
KITAYA K, YASUO T, YAMAGUCHI T. Bridging the diagnostic gap between histopathologic and hysteroscopic chronic endometritis with deep learning models[J]. Medicina (Kaunas), 2024, 60(6): 972. DOI:10.3390/medicina60060972 |
| [5] |
田文艳, 张慧英, 仝佳丽, 等. 子宫内膜息肉诊治中国专家共识(2022年版)[J]. 中国实用妇科与产科杂志, 2022, 38(8): 809-813. DOI:10.19538/j.fk2022080112 |
| [6] |
林苏云, 胡曙昉, 温莉虹, 等. 子宫内膜息肉患者慢性子宫内膜炎发生情况及相关性分析[J]. 中国计划生育学杂志, 2023, 31(12): 2937-2941. DOI:10.3969/j.issn.1004-8189.2023.12.031 |
| [7] |
CHEN BJ, FRANGOGIANNIS NG. Chemokines in myocardial infarction[J]. J Cardiovasc Transl Res, 2021, 14(1): 35-52. DOI:10.1007/s12265-020-10006-7 |
| [8] |
CHEN YY, LIU SY, WU LL, et al. Epigenetic regulation of chemo-kine (CC-motif) ligand 2 in inflammatory diseases[J]. Cell Prolif, 2023, 56(7): e13428. DOI:10.1111/cpr.13428 |
| [9] |
RUDNICKA E, SUCHTA K, GRYMOWICZ M, et al. Chronic low grade inflammation in pathogenesis of PCOS[J]. Int J Mol Sci, 2021, 22(7): 3789. DOI:10.3390/ijms22073789 |
| [10] |
王宏锋, 段雪娇, 马曙铮, 等. 血清ENA-78、HE4、MCP-1、IL-6联合检测在子宫内膜异位症中的表达水平及临床意义[J]. 中国妇产科临床杂志, 2022, 23(5): 513-514. DOI:10.13390/j.issn.1672-1861.2022.05.023 |
| [11] |
郭丽萍, 刘娟, 李思佳. 慢性子宫内膜炎患者子宫内膜息肉发生情况及其与IVF助孕结局的关系研究[J]. 中国性科学, 2024, 33(4): 57-61. DOI:10.3969/j.issn.1672-1993.2024.04.015 |
| [12] |
TSONIS O, GKROZOU F, DIMITRIOU E, et al. Hysteroscopic features suggestive of chronic endometritis: a systematic review[J]. Hum Fertil (Camb), 2023, 26(6): 1530-1543. DOI:10.1080/14647273.2023.2265155 |
| [13] |
YOSHIMURA T, LI CN, WANG YZ, et al. The chemokine monocyte chemoattractant protein-1/CCL2 is a promoter of breast cancer metastasis[J]. Cell Mol Immunol, 2023, 20(7): 714-738. DOI:10.1038/s41423-023-01013-0 |
| [14] |
GEORGAKIS MK, BERNHAGEN J, HEITMAN LH, et al. Targeting the CCL2-CCR2 axis for atheroprotection[J]. Eur Heart J, 2022, 43(19): 1799-1808. DOI:10.1093/eurheartj/ehac094 |
| [15] |
HEIDARI S, KOLAHDOUZ-MOHAMMADI R, KHODAVERDI S, et al. Expression levels of MCP-1, HGF, and IGF-1 in endometriotic patients compared with non-endometriotic controls[J]. BMC Wo-mens Health, 2021, 21(1): 422. DOI:10.1186/s12905-021-01560-6 |
| [16] |
NASIRI-ANSARI N, SPILIOTI E, KYROU I, et al. Estrogen receptor subtypes elicit a distinct gene expression profile of endothelial-derived factors implicated in atherosclerotic plaque vulnerability[J]. Int J Mol Sci, 2022, 23(18): 10960. DOI:10.3390/ijms231810960 |
 2026, Vol. 55
2026, Vol. 55