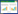文章信息
- 贾圣英, 王力, 孙志军, 王传合, 关琳, 田雪, 张胜波, 徐红梅, 蒋丽, 赵福禄, 陈转霞, 石晨
- JIA Shengying, WANG Li, SUN Zhijun, WANG Chuanhe, GUAN Lin, TIAN Xue, ZHANG Shengbo, XU Hongmei, JIANG Li, ZHAO Fulu, CHEN Zhuanxia, SHI Chen
- 外周血Hsa-circRNA0001380水平对急性前壁ST段抬高型心肌梗死后心力衰竭的预测价值
- Diagnostic value of peripheral blood Hsa-circRNA0001380 level in heart failure after acute anterior wall ST-segment elevation myocardial infarction
- 中国医科大学学报, 2026, 55(3): 234-239
- Journal of China Medical University, 2026, 55(3): 234-239
-
文章历史
- 收稿日期:2025-10-27
- 网络出版时间:2026-03-24 15:13:48
2. 中国医科大学附属盛京医院心血管内科, 沈阳 110004
2. Department of Cardiology, Shengjing Hospital of China Medical University, Shenyang 110004, China
急性心肌梗死(acute myocardial infarction,AMI)是全球意外死亡的主要原因。急性前壁ST段抬高型心肌梗死(ST-segment elevation myocardial infarction,STEMI)的病死率及并发症的发生率居高不下[1]。前壁STEMI比其他部位AMI更容易诱发左心室收缩功能障碍[2]。环状RNA(circular RNA,circRNA)是一种非编码RNA,广泛参与各种生理及病理过程[3-6]。研究[7]表明,circRNA MICR能够预测AMI后4个月的左心室收缩功能,可用于风险分层。本课题组前期研究[8]通过高通量测序数据分析证实,Mmu-circ-0001380在小鼠心肌缺血/再灌注损伤模型中表达显著上调。因此,本研究拟进一步分析其人源同源分子Hsa-circRNA0001380对急性前壁STEMI急诊经皮冠状动脉介入治疗(percutaneous coronary intervention,PCI)术后发生心力衰竭的预测价值。
1 材料与方法 1.1 研究对象选择2022年1月至12月期间大连理工大学附属中心医院(大连市中心医院)心血管内科收治的急性前壁STEMI(包括前间隔心肌梗死和广泛前壁心肌梗死)患者62例作为研究组。所有患者在发病12 h内成功接受急诊PCI。平均年龄(61.34±12.23)岁,其中男30例,女32例。排除既往有心肌梗死病史、慢性心力衰竭、先天性或瓣膜性心脏病、原发性心肌病、肺源性心脏病、心房颤动、严重肾功能不全(肌酐清除率 < 40 mL/min)、恶性肿瘤、近1个月内输血史、近2周内外伤或手术史、血液系统疾病、系统性炎症反应、自身免疫性疾病、妊娠和哺乳期的患者。根据左室射血分数(left ventricular ejection fraction,LVEF)将研究组患者再分为LVEF≥50%组和LVEF < 50%组。另选取22例同期健康体检者作为对照组,年龄和性别均与研究组匹配。本研究获得大连市中心医院伦理委员会批准(YN2022-039-30)。所有研究对象签署知情同意书。
根据Killip分级评估急性前壁STEMI心功能:Ⅰ级,无明显心功能不全;Ⅱ级,心力衰竭,肺部湿啰音范围 < 50%肺野;Ⅲ级,急性肺水肿,肺部闻及广泛湿啰音;Ⅳ级,心源性休克。
1.2 研究方法 1.2.1 收集临床资料收集研究组和对照组的一般临床资料、实验室检查结果。一般资料包括年龄、性别、既往病史、入院时生命体征。依据Killip分级(Ⅰ~Ⅳ级)评估心功能状态。实验室指标包括肝功能、肾功能、肌钙蛋白I、N末端脑钠肽前体(N-terminal pro-brain natriuretic peptide,NT-proBNP)。
1.2.2 实时定量PCR采集2组研究对象的静脉血2 mL,1 500 g离心5 min,分离血清后置于-80 ℃保存。应用TRIpure试剂提取血清RNA,反转录为cDNA。使用2×Taq PCR MasterMix(北京索莱宝科技有限公司)和ExicyclerTM 96荧光定量PCR仪(韩国Bioneer公司)进行PCR扩增。β-actin作内参照。引物序列如下:Hsa-circRNA0001380,正向5’- GCCACCCATTGATTTGA -3’,反向5’- GGTTCCACCAGTATTTCC -3’;β-actin,正向5’- GGCACCCAGCACAATGAA -3’,反向5’- TAGAAGCATTTGCGGTGG -3’。采用2-ΔΔCt法计算Hsa-circRNA0001380的相对表达量。
1.2.3 评估心室功能应用二维超声心动图评估左心室功能。采用改良Simpson法测量并记录LVEF和左心室舒张末容积(left ventricular end-diastolic volume,LVEDV)。
1.2.4 药物治疗所有急性前壁STEMI患者均于诊断明确后行急诊PCI,术前给予阿司匹林及P2Y12抑制剂(替格瑞洛或氯吡格雷)双联抗血小板治疗,常规应用他汀类药物进行调血脂及稳定斑块治疗。依据急性冠脉综合征管理指南及患者临床状况,个体化给予肾素-血管紧张素-醛固酮系统抑制剂。
1.3 统计学分析采用SPSS 25.0软件进行统计分析。正态分布的计量资料用x±s表示,非正态分布计量资料用M(P25~P75)表示。符合正态分布且方差齐的2组间比较采用独立样本t检验,不符合上述条件的两样本计量资料比较采用非参数秩和检验。计数资料用率(%)表示,组间比较采用χ2检验或Fisher确切概率法。对Hsa-circRNA0001380与心脏功能指标之间进行相关分析时,若双变量正态分布或近似正态分布则采用直线相关分析,否则采用秩相关分析。通过绘制受试者操作特征(receiver operating characteristic,ROC)曲线,计算曲线下面积(area under the curve,AUC),分析Hsa-circRNA0001380与NT-proBNP对急性前壁STEMI后心力衰竭的诊断价值。P < 0.05为差异有统计学意义。
2 结果 2.1 一般资料比较研究组患者62例,平均年龄(61.34±12.23)岁,其中男48例,女14例;对照组32例,平均年龄(61.23±11.66)岁,其中男10例,女12例。研究组中,心功能Killip分级Ⅰ、Ⅱ、Ⅲ、Ⅳ级的患者分别为31、20、7、4例;根据LVEF分类,LVEF≥50%组患者27例,均为Killip分级Ⅰ级;LVEF < 50%组患者35例,其中Killip分级Ⅰ、Ⅱ、Ⅲ、Ⅳ级的患者分别为4、20、7、4例。
2.2 各组Hsa-circRNA0001380水平比较与对照组比较,研究组患者Hsa-circRNA0001380水平显著升高,且随着Killip分级升高呈上升趋势,差异有统计学意义(P < 0.001)。此外,LVEF < 50%组急性前壁STEMI患者的Hsa-circRNA0001380表达水平显著高于LVEF≥50%组,差异有统计学意义(P < 0.001)。说明Hsa-circRNA0001380与急性前壁STEMI患者心力衰竭的严重程度有关,提示其可作为急诊PCI术后心力衰竭危险分层的潜在标志物。见表 1。
| Group | n | Hsa-circRNA0001380 | Z | P |
| Control | 22 | 0.008 5(0.007 6-0.011 7) | 4.220 | < 0.001 |
| Anterior-wall STEMI | 62 | 0.012 7(0.010 8-0.015 6) | ||
| Killip class Ⅰ | 31 | 0.010 8(0.009 3-0.011 7) | -6.073 | < 0.001 |
| Killip classes Ⅱ-Ⅳ | 31 | 0.015 3(0.013 4-0.016 6) | ||
| LVEF ≥50% | 27 | 0.010 5(0.009 2-0.011 5) | -6.316 | < 0.001 |
| LVEF < 50% | 35 | 0.014 9(0.013 4-0.016 7) | ||
| LVEF,left ventricular ejection fraction. | ||||
2.3 基于Hsa-circRNA0001380中位表达水平分析心脏功能
根据Hsa-circRNA0001380的中位表达水平(0.012 7),将研究组患者分为高表达组(血清水平≥0.012 7,n = 31)和低表达组(血清水平 < 0.012 7,n = 31)。结果显示,高表达组中Killip分级Ⅱ~Ⅳ级患者比例高于低表达组。同时,高表达组肌钙蛋白I、NT-proBNP水平显著高于低表达组。此外,入院时高表达组患者LVEDV较高,LVEF较低,见表 2。
| Item | Low-level group(Hsa-circRNA0001380 < 0.012 7) | High-level group(Hsa-circRNA0001380≥0.012 7) | P |
| Killip class Ⅱ-Ⅳ [n(%)] | 3(9.7) | 28(90.3) | < 0.001 |
| LVEDV(mm) | 49.94±4.03 | 54.58±6.27 | 0.001 |
| LVEF(%) | 55.97±5.76 | 40.81±6.94 | < 0.001 |
| TnI(ng/mL) | 3.24±1.02 | 8.68±2.32 | < 0.001 |
| NT-proBNP(pg/mL) | 794.0(302.0-1 968.1) | 2 131.2(1 520.7-3 538.2) | 0.002 |
| LVEDV,left ventricular end-diastolic volume;LVEF,left ventricular ejection fraction;TnI,troponin I;NT-proBNP,N-terminal pro-brain natriuretic peptide. | |||
2.4 相关性分析
血清中Hsa-circRNA0001380表达水平与心肌损伤标志物肌钙蛋白I、心力衰竭相关指标NT-proBNP以及心脏结构参数LVEDV均呈显著正相关(r = 0.700,P < 0.01;r = 0.644,P < 0.01;r = 0.594,P < 0.01)。表明随着Hsa-circRNA0001380水平升高,上述反映心肌损伤、心功能下降及左心室容积的指标均相应上升。相反,Hsa-circRNA0001380与LVEF呈显著负相关(r =-0.838,P < 0.01),提示其水平增高伴随左心室收缩功能下降。见图 1。因此,Hsa-circRNA0001380水平与多项心脏结构及功能指标具有显著相关性,提示其可能成为评估心脏功能的潜在生物标志物。
|
| 图 1 Hsa-circRNA0001380与心脏功能指标的相关性 Fig.1 Correlation between Hsa-circRNA0001380 and cardiac function indicators |
2.5 Hsa-circRNA0001380对急性前壁STEMI患者心力衰竭的预测价值
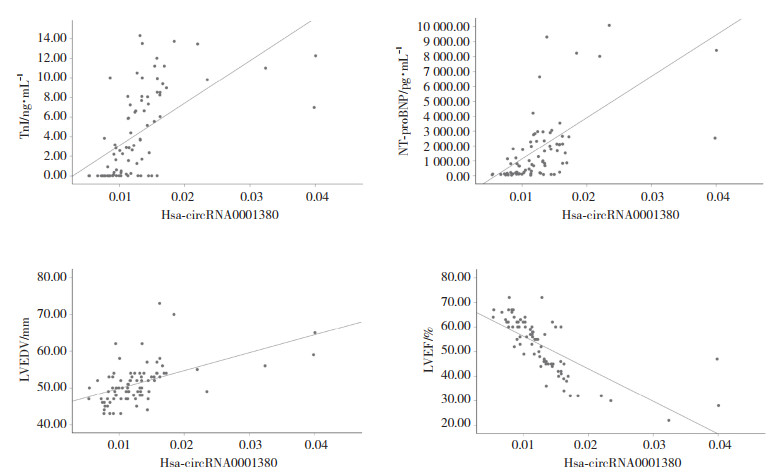
ROC曲线显示,Hsa-circRNA0001380的AUC为0.953(95%CI:0.895~1.000),> NT-proBNP [AUC为0.845(95%CI:0.751~0.940)],提示Hsa-circRNA0001380对急性前壁STEMI患者急诊PCI术后心力衰竭的发生较NT-proBNP具有更高的预测效能。Hsa-circRNA0001380高表达提示急性前壁STEMI患者心力衰竭发生风险增加。当截断值为0.012 436时,其预测灵敏度为96.8%,特异度为90.0%,见图 2。以上结果提示Hsa-circRNA0001380可能是急性前壁STEMI患者发生心力衰竭的潜在生物学标志物。
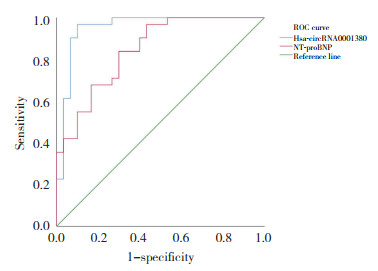
|
| 图 2 Hsa-circRNA0001380及NT-proBNP对急性前壁STEMI患者心力衰竭的预测价值 Fig.2 Predictive value of Hsa-circRNA0001380 and NT-proBNP for heart failure in patients with acute anterior-wall STEMI |
3 讨论
STEMI由冠状动脉粥样硬化斑块破裂或糜烂引起,是冠状动脉粥样硬化性心脏病的一个严重类型。STEMI后心力衰竭是导致心源性死亡及致残率增加的重要原因之一,常导致患者预后不良[9-12]。研究[13]显示,急性STEMI后新发心力衰竭的发生率为10%~45%。急性前壁STEMI常并发血流动力学紊乱及左心室收缩功能明显受损,发生心血管不良事件风险增高[14],对此类患者早期监测心功能、指导治疗及评估临床预后具有重要意义。
circRNA是通过自剪切产生的连续闭合、环状非编码RNA[3-5],其特点为高稳定性,且进化保守,在转录后层面调控基因表达,在多种疾病的生理和病理过程中均发挥作用[15]。研究[16-18]显示,circRNA可作为诊断AMI及判断预后的生物标志物。自噬相关环状RNA(autophagy-related circular RNA,ACR)是由自噬基因编码的circRNA,通过抑制自噬保护心肌细胞,并减少心肌细胞凋亡,减轻心肌缺血/再灌注损伤,缩小心肌梗死面积[19]。有研究[20]发现,心肌梗死后小鼠模型circRNA CDR1as及miR-7a表达水平上调,且circRNA CDR1as可能通过调控miR-7a的靶标而促进AMI的发生发展。血清中circRNA水平增高与AMI后的心力衰竭发生相关。还有研究[21]发现,circRNA MICRA表达水平与心肌梗死后左心室收缩功能障碍程度有一定的关联,MICRA水平较低的心肌梗死患者更易出现LVEF降低。既往研究[22]表明,Hsa-circRNA0001380在活动性肺结核患者外周血单核细胞中表达下调,可作为活动性肺结核诊断的潜在生物标志物。然而,Hsa-circRNA0001380在急性前壁STEMI及随后的心力衰竭发展中的诊断价值尚未见报道。
本研究结果显示,急性前壁STEMI患者外周血中Hsa-circRNA0001380表达上调,该结果与circRNA可作为AMI良好生物标志物的观点一致,支持Hsa-circRNA0001380可能是急性前壁STEMI的诊断性生物标志物。此外,血清Hsa-circRNA0001380水平还与急性前壁STEMI患者心功能相关。本研究还发现,急性前壁STEMI伴发心力衰竭患者血清Hsa-circ-RNA0001380水平高于无心力衰竭患者。而且,Hsacirc-RNA0001380水平与LVEDV呈正相关,与LVEF呈负相关。外周血Hsa-circRNA0001380可能成为急性前壁STEMI并发左心室收缩功能障碍的潜在生物标志物。为了进一步评估循环Hsa-circRNA0001380对急性前壁STEMI后心力衰竭的诊断价值,本研究进一步绘制了ROC曲线,结果表明,Hsa-circ-RNA0001380的AUC为0.953,灵敏度为96.8%,特异度为90.0%,较传统的心功能指标NT-proBNP有更好的诊断价值。
外周血Hsa-circRNA0001380在急性前壁STEMI中的具体作用机制尚不明确,其可能的调控效应如下:Hsa-circRNA0001380可与Hsa-miR-622、Hsa-miR-136-5p等微RNA结合,调控下游基因表达,参与细胞内生物学过程[23]。此外,Hsa-circRNA0001380具有m6A修饰位点,参与调节RNA稳定性、定位及转录效率等RNA生命过程,并与其他分子相互作用,调控细胞内分布及下游基因表达,进而影响心血管系统生理学及病理学过程[24]。目前关于Hsa-circ-RNA0001380参与AMI后心力衰竭发生发展的分子机制罕有报道,有待于深入探究。本研究中,急性前壁STEMI患者外周血中Hsa-circRNA0001380表达水平增加,且与心肌梗死后心功能参数存在相关性,ROC曲线证实其对急性前壁STEMI后心力衰竭具有预测价值,因此,推测Hsa-circRNA0001380可能成为AMI后心力衰竭潜在的新的治疗靶点。
综上所述,本研究首次发现外周血Hsa-circRNA0001380可作为急性前壁STEMI患者接受急诊PCI术后发生心力衰竭的潜在的诊断标志物,但其能否成为急性STEMI可能的治疗靶点仍有待深入研究。关于Hsa-circRNA0001380的生物学功能、具体作用机制与临床应用前景,亟待开展大规模多中心随机对照研究。
| [1] |
CHOI SH, KWON SW. Mechanical complications of ST segment elevation myocardial infarction: are they tangible?[J]. J Thorac Dis, 2022, 14(7): 2458-2460. DOI:10.21037/jtd-22-705 |
| [2] |
TALAAT A, ABOU EL EZZ M, EL GHANY MMA, et al. Value of peri-procedural lung ultrasound in predicting heart failure or left ventricular systolic dysfunction within 3 months in STEMI patients undergoing primary PCI[J]. Cor Vasa, 2025, 67(3): 350-359. DOI:10.33678/cor.2025.006 |
| [3] |
VERDUCI L, TARCITANO E, STRANO S, et al. CircRNAs: role in human diseases and potential use as biomarkers[J]. Cell Death Dis, 2021, 12: 468. DOI:10.1038/s41419-021-03743-3 |
| [4] |
ZHOU QL, BOECKEL JN, YAO JH, et al. Diagnosis of acute myocardial infarction using a combination of circulating circular RNA cZNF292 and clinical information based on machine learning[J]. MedComm, 2023, 4(3): e299. DOI:10.1002/mco2.299 |
| [5] |
MARINESCU MC, LAZAR AL, MARTA MM, et al. Non-coding RNAs: prevention, diagnosis, and treatment in myocardial ischemia-reperfusion injury[J]. Int J Mol Sci, 2022, 23(5): 2728. DOI:10.3390/ijms23052728 |
| [6] |
WU JH, LI CZ, LEI Z, et al. Comprehensive analysis of circRNA miRNA-mRNA regulatory network and novel potential biomarkers in acute myocardial infarction[J]. Front Cardiovasc Med, 2022, 9: 850991. DOI:10.3389/fcvm.2022.850991 |
| [7] |
SALGADO-SOMOZA A, ZHANG L, VAUSORT M, et al. The circular RNA MICRA for risk stratification after myocardial infarction[J]. Int J Cardiol Heart Vasc, 2017, 17: 33-36. DOI:10.1016/j.ijcha.2017.11.001 |
| [8] |
WANG L, WANG CH, SUN ZQ, et al. Knockdown of Mmu-circ-0001380 attenuates myocardial ischemia/reperfusion injury via modulating miR-106b-5p/Phlpp2 axis[J]. J Cardiovasc Transl Res, 2023, 16(5): 1064-1077. DOI:10.1007/s12265-023-10383-9 |
| [9] |
BUTLER J, HAMMONDS K, TALHA KM, et al. Incident heart failure and recurrent coronary events following acute myocardial infarction[J]. Eur Heart J, 2025, 46(16): 1540-1550. DOI:10.1093/eurheartj/ehae885 |
| [10] |
GAO WW, YU LF, SHE JJ, et al. Cardio-cerebral infarction: a narrative review of pathophysiology, treatment challenges, and prognostic implications[J]. Front Cardiovasc Med, 2025, 12: 1507665. DOI:10.3389/fcvm.2025.1507665 |
| [11] |
HARRINGTON J, BUTLER J. Heart failure after myocardial infarction: glass emptier than full[J]. Eur J Heart Fail, 2023, 25(8): 1225-1227. DOI:10.1002/ejhf.2961 |
| [12] |
LI XW, SHANG CM, XU CY, et al. Development and comparison of machine learning-based models for predicting heart failure after acute myocardial infarction[J]. BMC Med Inform Decis Mak, 2023, 23(1): 165. DOI:10.1186/s12911-023-02240-1 |
| [13] |
LIANG JK, ZHANG ZH. Predictors of in-hospital heart failure in patients with acute anterior wall ST-segment elevation myocardial infarction[J]. Int J Cardiol, 2023, 375: 104-109. DOI:10.1016/j.ijcard.2023.01.002 |
| [14] |
YILDIZ I, RENCÜZOĞULLARı I, KARABAĞ Y, et al. Predictors of left ventricular ejection function decline in young patients with ST-segment elevation myocardial infarction[J]. Rev Assoc Med Bras, 2022, 68(6): 802-807. DOI:10.1590/1806-9282.20220033 |
| [15] |
LONG Q, LV BJ, JIANG SJ, et al. The landscape of circular RNAs in cardiovascular diseases[J]. Int J Mol Sci, 2023, 24(5): 4571. DOI:10.3390/ijms24054571 |
| [16] |
XU CR, JIA ZW, CAO XF, et al. Hsa-circ-0007059 promotes apoptosis and inflammation in cardiomyocytes during ischemia by targeting microRNA-378 and microRNA-383[J]. Cell Cycle, 2022, 21(10): 1003-1019. DOI:10.1080/15384101.2022.2040122 |
| [17] |
XUAN LN, LUO HS, WANG S, et al. Circulating CCRR serves as potential novel biomarker for predicting acute myocardial infarction[J]. Frigid Zone Med, 2024, 4(3): 137-151. DOI:10.1515/fzm-2024-0015 |
| [18] |
JU J, LI XM, ZHAO XM, et al. Circular RNA FEACR inhibits ferroptosis and alleviates myocardial ischemia/reperfusion injury by interacting with NAMPT[J]. J Biomed Sci, 2023, 30(1): 45. DOI:10.1186/s12929-023-00927-1 |
| [19] |
ZHOU LY, ZHAI M, HUANG Y, et al. The circular RNA ACR attenuates myocardial ischemia/reperfusion injury by suppressing autophagy via modulation of the Pink1/FAM65B pathway[J]. Cell Death Differ, 2019, 26(7): 1299-1315. DOI:10.1038/s41418-018-0206-4 |
| [20] |
GENG HH, LI R, SU YM, et al. The circular RNA Cdr1as promotes myocardial infarction by mediating the regulation of miR-7a on its target genes expression[J]. PLoS One, 2016, 11(3): e0151753. DOI:10.1371/journal.pone.0151753 |
| [21] |
WANG YK, WANG HQ, ZHANG LP, et al. A novel identified circular RNA, circSnap47, promotes heart failure progression via regulation of miR-223-3p/MAPK axis[J]. Mol Cell Biochem, 2023, 478(3): 459-469. DOI:10.1007/s11010-022-04523-z |
| [22] |
LUO HL, PENG Y, LUO H, et al. Circular RNA hsa-circ-0001380 in peripheral blood as a potential diagnostic biomarker for active pulmonary tuberculosis[J]. Mol Med Rep, 2020, 21(4): 1890-1896. DOI:10.3892/mmr.2020.10992 |
| [23] |
CAI RP, XU YL, REN YL, et al. microRNA-136-5p protects cardiomyocytes from coronary microembolization through the inhibition of pyroptosis[J]. Apoptosis, 2022, 27(3/4): 206-221. DOI:10.1007/s10495-022-01712-5 |
| [24] |
ZHOU C, MOLINIE B, DANESHVAR K, et al. Genome-wide maps of m6A circRNAs identify widespread and cell-type-specific methy-lation patterns that are distinct from mRNAs[J]. Cell Rep, 2017, 20(9): 2262-2276. DOI:10.1016/j.celrep.2017.08.027 |
 2026, Vol. 55
2026, Vol. 55