文章信息
- 王思涵, 李瑶涵, 于月新, 白雪
- WANG Sihan, LI Yaohan, YU Yuexin, BAI Xue
- HOMA-IR水平对多囊卵巢综合征患者IVF-ET助孕后临床结局的预测价值
- Predictive value of HOMA-IR levels on the clinical outcomes of patients with polycystic ovary syndrome undergoing in vitro fertilization-embryo transfer
- 中国医科大学学报, 2025, 54(12): 1107-1113
- Journal of China Medical University, 2025, 54(12): 1107-1113
-
文章历史
- 收稿日期:2025-02-16
- 网络出版时间:2025-12-15 14:24:36
多囊卵巢综合征(polycystic ovary syndrome,PCOS)是育龄期女性最常见的生殖内分泌紊乱疾病,患病率为5%~10%,影响女性的整个生命周期[1]。胰岛素抵抗在PCOS的发病机制中起着核心作用[2],PCOS炎症状态被认为与高胰岛素血症、胰岛素抵抗和肥胖相互作用,这种相互作用可能进一步加剧PCOS患者的代谢紊乱和生殖功能障碍[3]。体外受精-胚胎移植(in vitro fertilization-embryo transfer,IVF-ET)助孕是用于调整生活方式和药物治疗后仍无法受孕的PCOS患者一种三线治疗方法 [4]。已有研究[5]表明接受IVF-ET助孕的PCOS女性临床流产率高于健康对照组。影响PCOS患者IVF-ET助孕的代谢异常因素可能导致多次胚胎移植失败,进而造成不良的临床妊娠结局。因此,PCOS患者在IVF-ET助孕前调整体内代谢水平至稳定状态至关重要。
高胰岛素-正常血糖钳夹是胰岛素抵抗评价的金标准,但成本高昂且技术复杂,临床上不常使用[6]。临床上常用稳态模型评估胰岛素抵抗指数(homeostasis model assessment of insulin resistance,HOMA-IR)来评估胰岛素抵抗[7-8]。HOMA-IR值越高,表明胰岛素抵抗越严重。高水平HOMA-IR会引起胚胎种植率降低,从而导致临床妊娠率降低[9],研究[10]显示,HOMA-IR水平与患者IVF-ET助孕结果相关,PCOS患者IVF-ET助孕后的临床妊娠率随着HOMA-IR升高而降低。HOMA-IR被认为是评估胰岛素抵抗关键的预测性指标,但未明确HOMA-IR的临界值[11]。目前,尚未明确预测IVF-ET助孕后PCOS患者临床妊娠和活产结局的HOMA-IR的临界值。本研究探讨HOMA-IR与PCOS患者IVF-ET助孕后临床妊娠和活产结局的相关性,及其对PCOS患者IVF-ET助孕后临床结局的预测价值。
1 材料与方法 1.1 临床资料及分组收集2018年6月至2023年1月于中国人民解放军北部战区总医院行IVF-ET助孕的PCOS患者的临床资料。纳入标准:(1)年龄≤40岁;(2)IVF-ET助孕进行单胚胎移植;(3)符合PCOS诊断标准[12]。排除标准:(1)近半年服用二甲双胍等降糖药物或其他激素类药物;(2)患有卵巢功能减退、甲状腺功能障碍等内分泌疾病;(3)子宫发育畸形以及卵巢肿瘤等器质性疾病;(4)夫妻中任一方存在染色体异常或第3代试管婴儿辅助;(5)多胎妊娠和胎儿畸形。共纳入563例。根据是否临床妊娠和活产,将患者分为妊娠组(n = 457)与未妊娠组(n = 106),以及活产组(n = 341)与非活产组(n = 116)。本研究未对患者实施任何额外的干预或治疗措施,不会给患者带来额外的风险。本研究已获得我院医学伦理委员会批准[Y(2024)275号]。
1.2 检测指标收集患者的一般资料,包括患者病案号、IVF-ET编号、年龄、体重指数(body mass index,BMI)、抗缪勒管激素(anti-Müllerian hormone,AMH)、空腹血糖(fasting plasma glucose,FPG)、空腹胰岛素(fasting insulin,FINS)等,并计算HOMA-IR值,HOMA-IR=FPG(mmol/L)×FINS(mIU/L)/22.5。月经周期第2~5天激素指标,包括孕酮(progesterone,P)、雌二醇(estradiol,E2)、泌乳素(prolactin,PRL)、卵泡刺激素(follicle stimulating hormone,FSH)、黄体生成素(luteinizing hormone,LH)、睾酮(testosterone,T),计算LH/FSH比值。促排卵及实验室胚胎培育情况,控制性超促排卵方案(controlled ovarian hyperstimulation,COH)包括长方案、改良长方案、拮抗剂方案和黄体期方案。体外受精(in vitro fertilization,IVF),卵胞质内单精子注射(intracytoplasmic sperm injection,ICSI),获卵数,受精率,优质胚胎率,移植胚胎类型,移植时的子宫内膜厚度。
1.3 统计学分析使用SPSS 26.0软件进行统计分析。不符合正态分布的计量资料采用M(P25~P75)表示,2组间比较采用Wilcoxon秩和检验。计数资料采用率(%)表示,2组间比较采用χ2检验或Fisher确切概率法。采用多因素logistic回归分析临床妊娠和活产结局的影响因素。利用受试者操作特征(receive operating characteristic,ROC)曲线评估HOMA-IR水平对PCOS患者IVF-ET助孕后临床妊娠和活产结局的预测价值。P < 0.05为差异有统计学意义。
2 结果 2.1 妊娠组与未妊娠组患者临床指标比较结果显示,妊娠组与未妊娠组年龄、BMI、HOMA-IR、受精数、优质胚胎数和优质胚胎率比较差异有统计学意义(P < 0.05),见表 1。
| Item | Total(n = 563) | Pregnancy group(n = 457) | Non-pregnancy group(n = 106) | Z/χ2 | P |
| Age(year) | 32(30-34) | 31(30-34) | 33(31-36) | 2.251 | < 0.001 |
| Duration of infertility(year) | 4(2-5) | 4(2-5) | 3(2-5) | 0.570 | 0.902 |
| Types of infertility [n (%)] | 0.757 | 0.384 | |||
| Primary infertility | 353(62.7) | 291(63.6) | 62(58.4) | ||
| Secondary infertility | 210(37.3) | 166(36.4) | 44(41.6) | ||
| BMI(kg/m2) | 25.7(22.5-28.6) | 25.0(22.0-28.3) | 27.5(25.7-29.2) | 3.123 | < 0.001 |
| P(ng/mL) | 0.18(0.10-0.30) | 0.18(0.10-0.29) | 0.19(0.12-0.35) | 1.100 | 0.178 |
| E2(pg/mL) | 38.17(30.22-48.21) | 38.51(30.51-48.10) | 38.81(29.85-47.24) | 0.457 | 0.985 |
| PRL(μIU/mL) | 287.5(224.9-357.0) | 287.6(226.8-356.9) | 280.5(214.4-358.0) | 0.766 | 0.600 |
| FSH(mIU/mL) | 5.56(4.78-6.65) | 5.56(4.75-6.65) | 5.52(4.98-6.56) | 0.779 | 0.578 |
| LH(mIU/mL) | 7.56(5.32-10.28) | 7.56(5.32-10.28) | 7.55(5.63-9.73) | 0.810 | 0.529 |
| LH/FSH | 1.33(0.90-1.83) | 1.32(0.89-1.84) | 1.37(0.98-1.76) | 0.820 | 0.513 |
| T(ng/mL) | 0.65(0.54-0.75) | 0.65(0.54-0.75) | 0.65(0.52-0.76) | 0.602 | 0.861 |
| AMH(ng/mL) | 6.49(4.37-9.35) | 6.47(4.37-9.18) | 6.54(3.89-9.05) | 1.030 | 0.239 |
| HOMA-IR | 2.97(2.14-4.11) | 2.72(2.05- 3.65) | 4.11(3.35-5.20) | 4.434 | < 0.001 |
| Fertilization protocols [n (%)] | 0.992 | 0.319 | |||
| IVF | 331(58.7) | 276(58.8) | 55(52.4) | ||
| ICSI | 232(41.3) | 181(41.2) | 51(47.6) | ||
| Ovulation induction protocols [n (%)] | 0.707 | 0.872 | |||
| Antagonist protocol | 213(37.8) | 175(38.2) | 38(35.8) | ||
| Luteal-phase protocol | 21(3.7) | 16(3.5) | 5(4.7) | ||
| Long protocol | 83(14.8) | 64(14.0) | 19(17.9) | ||
| Modified long protocol | 246(43.7) | 202(44.3) | 44(41.6) | ||
| Number of oocytes retrieved | 17(12-24) | 17(13-24) | 17(9-25) | -1.398 | 0.162 |
| Number of fertilized oocytes | 13(8-19) | 13(9-19) | 11(7-17) | -2.167 | 0.030 |
| Number of high-quality embryos | 5(3-9) | 6(3-9) | 4(2-8) | -3.168 | < 0.001 |
| Fertilization rate | 0.76(0.63-0.87) | 0.76(0.64-0.88) | 0.72(0.57-0.86) | 1.187 | 0.119 |
| High-quality embryo rate | 0.46(0.27-0.66) | 0.47(0.28-0.68) | 0.40(0.24-0.60) | 1.427 | 0.034 |
| Types of embryos transferred [n (%)] | 0.156 | 0.068 | |||
| Fresh embryo transfer | 124(22.1) | 111(24.2) | 13(12.2) | ||
| Frozen embryo transfer | 439(77.9) | 346(75.8) | 93(87.8) | ||
| Endometrial thickness on the day of transfer(cm) | 0.95(0.80-1.10) | 0.95(0.80-1.10) | 0.90(0.74-1.10) | 1.380 | 0.168 |
2.2 影响妊娠结局的多因素logistic回归分析
校正了优质胚胎数等已知混杂因素,变量进入模型的标准为P < 0.05。结果显示,年龄、BMI、HOMA-IR是临床妊娠结局的影响因素(P < 0.05),见表 2。
| Variable | COR(95%CI) | AOR(95%CI) |
| Age | 0.872(0.809-0.940)* | 0.872(0.808-0.941)* |
| BMI | 0.890(0.846-0.936)* | 0.925(0.872-0.983)* |
| HOMA-IR | 0.785(0.710-0.869)* | 0.881(0.780-0.996)* |
| Number of high-quality embryos | 1.094(1.035-1.155)* | 1.048(0.979-1.121) |
| High-quality embryo rate | 3.257(1.397-7.591)* | 1.465(0.491-4.371) |
| COR,crude odds ratio;AOR,adjusted odds ratio. *P < 0.05. | ||
2.3 HOMA-IR预测临床妊娠结局的ROC曲线分析
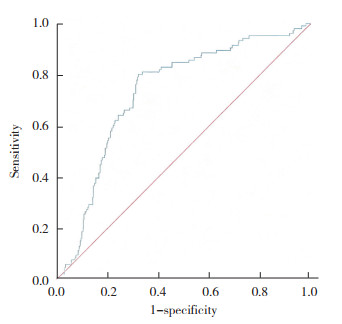
ROC曲线分析结果显示,曲线下面积为0.732(95%CI:0.681~0.783,P < 0.001)。HOMA-IR截断值为3.29,灵敏度为0.802,特异度为0.676,见图 1。
|
| 图 1 HOMA-IR预测临床妊娠结局的ROC曲线 Fig.1 The ROC curve for HOMA-IR in predicting clinical pregnancy outcomes |
2.4 HOMA-IR水平与临床妊娠结局的相关性
根据HOMA-IR的截断值将PCOS患者分为HOMA-IR < 3.29组(n = 328)和HOMA-IR≥3.29组(n = 235),比较2组的临床妊娠率。结果显示,HOMA-IR < 3.29组和HOMA-IR≥3.29组的临床妊娠率分别为93.5%(307/328)、63.8%(150/235),差异有统计学意义(χ2=25.250,P < 0.001)。
2.5 活产组与非活产组患者临床指标比较(表 3)| Item | Live birth group(n = 341) | Non-live birth group(n = 116) | Z/χ2 | P |
| Age(year) | 31(30-33) | 33(30-34) | -3.306 | 0.001 |
| Duration of infertility(year) | 3(2-5) | 4(2-5) | -0.317 | 0.754 |
| Types of infertility [n (%)] | 1.357 | 0.244 | ||
| Primary infertility | 224(65.7) | 67(57.8) | ||
| Secondary infertility | 117(34.3) | 49(42.2) | ||
| BMI(kg/m2) | 24.1(21.5-26.8) | 28.2(25.7-29.5) | -9.531 | < 0.001 |
| P(ng/mL) | 0.19(0.10-0.29) | 0.16(0.10-0.27) | -0.142 | 0.887 |
| E2(pg/mL) | 38.25(30.15-49.20) | 39.19(32.54-47.04) | -0.116 | 0.907 |
| PRL(μIU/mL) | 294.2(231.5-359.0) | 273.8(214.4-355.5) | -1.659 | 0.097 |
| FSH(mIU/mL) | 5.55(4.75-6.64) | 5.56(4.92-6.68) | -0.083 | 0.934 |
| LH(mIU/mL) | 7.56(5.21-10.42) | 7.49(5.50-10.17) | -0.179 | 0.858 |
| LH/FSH | 1.31(0.89-1.85) | 1.35(0.93-1.78) | -0.347 | 0.729 |
| T(ng/mL) | 0.67(0.56-0.77) | 0.62(0.53-0.75) | -1.695 | 0.090 |
| AMH(ng/mL) | 6.53(4.53-9.55) | 6.32(4.18-9.17) | -0.909 | 0.363 |
| HOMA-IR | 2.49(1.95-3.12) | 4.26(3.06-5.65) | -12.251 | < 0.001 |
| Fertilization protocols [n (%)] | 1.315 | 0.252 | ||
| IVF | 211(61.9) | 63(54) | ||
| ICSI | 130(38.1) | 53(46) | ||
| Ovulation induction protocols [n (%)] | 1.196 | 0.754 | ||
| Antagonist protocol | 122(35.8) | 47(40.5) | ||
| Luteal-phase protocol | 12(3.5) | 5(4.3) | ||
| Long protocol | 48(14.1) | 18(15.5) | ||
| Modified long protocol | 159(46.6) | 46(39.7) | ||
| Number of oocytes retrieved | 17(13-24) | 17(10-23) | -1.084 | 0.277 |
| Number of fertilized oocytes | 13(9-19) | 12(8-18) | -1.756 | 0.080 |
| Number of high-quality embryos | 7(4-10) | 4(2-7) | -8.194 | < 0.001 |
| Fertilization rate | 0.77(0.65-0.88) | 0.75(0.60-0.88) | -1.195 | 0.233 |
| High-quality embryo rate | 0.54(0.37-0.75) | 0.33(0.16-0.56) | -9.068 | < 0.001 |
| Types of embryos transferred [n (%)] | 1.567 | 0.221 | ||
| Fresh embryo transfer | 34(10.0) | 13(11.2) | ||
| Frozen embryo transfer | 307(90.0) | 103(88.7) | ||
| Endometrial thickness on the day of transfer(cm) | 0.95(0.82-1.10) | 0.94(0.77-1.11) | -1.093 | 0.279 |
结果显示,2组年龄、BMI、HOMA-IR、优质胚胎数和优质胚胎率比较差异有统计学意义(P < 0.05)。
2.6 影响活产结局的多因素logistic回归分析(表 4)| Variable | COR (95%CI) | AOR(95%CI) |
| Age | 0.914(0.861-0.972)* | 0.873(0.807-0.947)* |
| BMI | 0.797(0.757-0.839)* | 0.876(0.825-0.932)* |
| HOMA-IR | 0.368(0.305-0.443)* | 0.483(0.397-0.589)* |
| Number of high-quality embryos | 1.164(1.134-1.252)* | 1.054(0.989-1.126) |
| High-quality embryo rate | 33.954(15.225-75.858)* | 9.901(3.221-30.429)* |
| COR,crude odds ratio;AOR,adjusted odds ratio. *P < 0.05. | ||
调整了优质胚胎数等已知混杂因素,变量进入模型的标准为P < 0.1。结果显示,年龄、BMI、优质胚胎率、HOMA-IR是活产结局的影响因素(P < 0.05)。
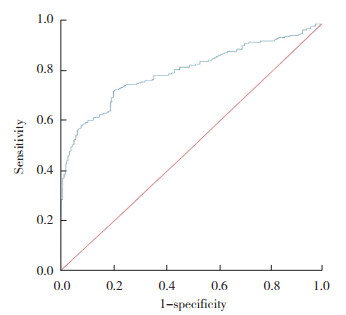
2.7 HOMA-IR预测活产结局的ROC曲线分析ROC曲线分析结果显示,曲线下面积为0.804(95%CI:0.764~0.843,P < 0.001)。HOMA-IR截断值为3.27,灵敏度为0.728,特异度为0.794。见图 2。
|
| 图 2 HOMA-IR预测活产结局的ROC曲线 Fig.2 ROC curve for HOMA-IR in predicting live birth outcomes |
2.8 HOMA-IR水平与活产结局的相关性
根据HOMA-IR的截断值将活产患者分为HOMA-IR < 3.27组(n = 328)和HOMA-IR≥3.27组(n = 129),比较2组的活产率。结果显示,HOMA-IR < 3.27组的活产率(81.0%,266/328)显著高于HOMA-IR≥3.27组(28.6%,37/129),差异有统计学意义(χ2=54.746,P < 0.001)。
3 讨论已有研究[13-14]显示,PCOS患者中HOMA-IR升高与IVF-ET结局呈负相关。可能的多种潜在机制包括:(1)严重的胰岛素抵抗可能破坏下丘脑-垂体-卵巢轴的平衡,导致卵泡发育异常并降低卵母细胞质量,这与本研究中妊娠组、活产组与其对照组比较优质胚胎数及优质胚胎率上存在统计学差异的结果一致;(2)胰岛素抵抗引发的高胰岛素血症可上调卵巢雄激素表达,进一步加剧卵泡闭锁并损害子宫内膜容受性,从而降低胚胎着床率[15];(3)胰岛素抵抗与慢性轻度炎症状态密切相关[16],这种炎症可能干扰母胎界面的免疫微环境,增加早期妊娠丢失风险,进而影响活产结局。可见HOMA-IR可作为PCOS患者IVF-ET助孕后生殖结局的关键预测指标。
本研究结果显示,HOMA-IR、年龄、BMI是临床妊娠结局的独立影响因素;HOMA-IR、年龄、BMI及优质胚胎率是活产结局的独立影响因素(P < 0.05)。LUO等[10]研究指出胰岛素抵抗会降低PCOS患者卵巢敏感性及IVF助孕妊娠结局;CHEN等[17]发现胰岛素抵抗是PCOS患者早期流产的危险因素,与本研究结果一致。本研究结果显示,HOMA-IR对妊娠和活产结局均具有较高预测效能,临床妊娠的最佳截断值为3.29,活产的最佳截断值为3.27。此结果为临床实践提供了精准的量化参考依据。另一方面,不同于SONG等[18]研究仅聚焦临床妊娠结局,本研究同时评估了妊娠与活产两种结局,对生殖成功的评估更为全面。不同研究结果的差异可能源于样本量、种族背景及IVF治疗方案的不同,这也提示需针对特定人群制定个性化的HOMA-IR阈值标准。
本研究明确了HOMA-IR在预测IVF-ET结局中的重要作用,为深入探索胰岛素抵抗相关生殖损伤的分子机制奠定了基础。从临床应用来看,本研究确定的HOMA-IR截断值为PCOS患者IVF-ET术前风险分层提供了实用工具。对于HOMA-IR≥3.29(预测妊娠风险)或HOMA-IR≥3.27(预测活产风险)的患者,可通过生活方式干预、二甲双胍治疗或其他胰岛素增敏治疗优化其代谢状态,进而改善生殖结局。
综上所述,HOMA-IR是评估PCOS患者IVF-ET助孕后临床妊娠及活产结局的可靠预测指标,具有较高的预测效能。HOMA-IR≥3.29时,PCOS患者IVF-ET助孕后的临床妊娠率降低;HOMA-IR≥3.27时,其活产率降低。本研究的不足之处:(1)本研究为单中心回顾性研究,样本均来源于一所医院;(2)可能存在选择偏倚,部分未测量的混杂因素(如详细饮食习惯、体力活动水平)未纳入分析;(3)本研究未探讨HOMA-IR与其他代谢指标(如血脂谱、糖化血红蛋白)对IVF-ET结局的交互作用。未来研究应采用多中心前瞻性设计以扩大样本量、纳入更全面的混杂变量,并通过亚组分析明确HOMA-IR在不同PCOS表型中的作用。
| [1] |
KOLHE JV, CHHIPA AS, BUTANI S, et al. PCOS and depression: common links and potential targets[J]. Reprod Sci, 2022, 29(11): 3106-3123. DOI:10.1007/s43032-021-00765-2 |
| [2] |
RYSSDAL M, VANKY E, STOKKELAND LMT, et al. Immunomodulatory effects of metformin treatment in pregnant women with PCOS[J]. J Clin Endocrinol Metab, 2023, 108(9): e743-e753. DOI:10.1210/clinem/dgad145 |
| [3] |
ZHAI Y, PANG YL. Systemic and ovarian inflammation in women with polycystic ovary syndrome[J]. J Reprod Immunol, 2022, 151: 103628. DOI:10.1016/j.jri.2022.103628 |
| [4] |
COSTELLO MF, GARAD RM, HART R, et al. A review of secon-dand third-line infertility treatments and supporting evidence in women with polycystic ovary syndrome[J]. Med Sci, 2019, 7(7): 75. DOI:10.3390/medsci7070075 |
| [5] |
MU LS, PAN JX, YANG LL, et al. Association between the prevalence of hyperuricemia and reproductive hormones in polycystic ovary syndrome[J]. Reprod Biol Endocrinol, 2018, 16(1): 104. DOI:10.1186/s12958-018-0419-x |
| [6] |
MEEKS KAC, BENTLEY AR, ASSIMES TL, et al. Mendelian randomization analyses suggest a causal role for circulating GIP and IL-1RA levels in homeostatic model assessment-derived measures of β-cell function and insulin sensitivity in Africans without type 2 diabetes[J]. Genome Med, 2023, 15(1): 108. DOI:10.1186/s13073-023-01263-7 |
| [7] |
TRIPATHY D, ALMGREN P, TUOMI T, et al. Contribution of insulin-stimulated glucose uptake and basal hepatic insulin sensitivity to surrogate measures of insulin sensitivity[J]. Diabetes Care, 2004, 27(9): 2204-2210. DOI:10.2337/diacare.27.9.2204 |
| [8] |
BUCHANAN TA, WATANABE RM, XIANG AH. Limitations in surrogate measures of insulin resistance[J]. J Clin Endocrinol Metab, 2010, 95(11): 4874-4876. DOI:10.1210/jc.2010-2167 |
| [9] |
HUANG WL, LIU C, DING L, et al. The effect of β-cell dysfunction on reproductive outcomes of PCOS undergoing IVF or ICSI embryo transfer cycles: a retrospective cohort study[J]. Front Endocrinol, 2024, 15: 1327041. DOI:10.3389/fendo.2024.1327041 |
| [10] |
LUO ZY, WANG LL, WANG YZ, et al. Impact of insulin resistance on ovarian sensitivity and pregnancy outcomes in patients with polycystic ovary syndrome undergoing IVF[J]. J Clin Med, 2023, 12(3): 818. DOI:10.3390/jcm12030818 |
| [11] |
ORTIZ-FLORES AE, LUQUE-RAMÍREZ M, FERNÁNDEZ-DURÁN E, et al. Diagnosis of disorders of glucose tolerance in wo-men with polycystic ovary syndrome (PCOS) at a tertiary care center: fasting plasma glucose or oral glucose tolerance test?[J]. Metabolism, 2019, 93: 86-92. DOI:10.1016/j.metabol.2019.01.015 |
| [12] |
Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS)[J]. Hum Reprod, 2004, 19(1): 41-47. DOI:10.1093/humrep/deh098 |
| [13] |
SEPPÄ S, KUIRI-HÄNNINEN T, HOLOPAINEN E, et al. Management of endocrine disease: diagnosis and management of primary amenorrhea and female delayed puberty[J]. Eur J Endocrinol, 2021, 184(6): R225-R242. DOI:10.1530/EJE-20-1487 |
| [14] |
FEDELI V, UNFER V, DINICOLA S, et al. Inositol restores appropriate steroidogenesis in PCOS ovaries both in vitro and in vivo experimental mouse models[J]. Cells, 2024, 13(14): 1171. DOI:10.3390/cells13141171 |
| [15] |
MOGHETTI P, TOSI F. Insulin resistance and PCOS: chicken or egg?[J]. J Endocrinol Invest, 2021, 44(2): 233-244. DOI:10.1007/s40618-020-01351-0 |
| [16] |
RUEBEL M, SHANKAR K, GADDY D, et al. Maternal obesity is associated with ovarian inflammation and upregulation of early growth response factor 1[J]. Am J Physiol Endocrinol Metab, 2016, 311(1): E269-E277. DOI:10.1152/ajpendo.00524.2015 |
| [17] |
CHEN YH, GUO JY, ZHANG QW, et al. Insulin resistance is a risk factor for early miscarriage and macrosomia in patients with polycystic ovary syndrome from the first embryo transfer cycle: a retrospective cohort study[J]. Front Endocrinol, 2022, 13: 853473. DOI:10.3389/fendo.2022.853473 |
| [18] |
SONG HX, YU ZQ, LI P, et al. HOMA-IR for predicting clinical pregnancy rate during IVF[J]. Gynecol Endocrinol, 2022, 38(1): 33-38. DOI:10.1080/09513590.2021.1952976 |
 2025, Vol. 54
2025, Vol. 54