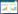文章信息
- 方虹, 鲁林
- FANG Hong, LU Lin
- miR-200c-3p靶向调控Notch1表达对异位子宫内膜间质细胞增殖、凋亡和迁移的影响
- Effect of miR-200c-3p on the proliferation, apoptosis, and migration of ectopic endometrial stromal cells through targeted regulation of the Notch1 expression
- 中国医科大学学报, 2025, 54(12): 1101-1106, 1113
- Journal of China Medical University, 2025, 54(12): 1101-1106, 1113
-
文章历史
- 收稿日期:2024-10-24
- 网络出版时间:2025-12-15 12:16:27
2. 武汉市中医医院骨科,武汉 430000
2. Department of Orthopedics, Wuhan Traditional Chinese Medicine Hospital, Wuhan 430000, China
子宫内膜异位症(endometriosis,EMT)可导致育龄女性盆腔疼痛和不孕症。研究[1]显示,异位子宫内膜间质细胞(ectopic endometrial stromal cell,EESC)的增殖、迁移和侵袭特性与恶性肿瘤细胞相似,且EMT中存在上皮-间质转化[2]。然而,EESC增殖、迁移的分子机制仍不完全清楚,因此亟需进一步探讨其分子机制并制定更加有效的治疗策略。
微RNA(microRNA,miRNA)可通过调控靶基因表达,参与细胞增殖、凋亡和迁移等生物学过程,miR-200c-3p可抑制肿瘤细胞上皮-间质转化、侵袭和血管生成[3]。研究[4]显示,EMT患者异位子宫内膜中miR-200c-3p表达降低,上调其表达可抑制子宫内膜细胞侵袭。而Notch1在EMT患者异位内膜组织中的表达高于在位内膜[5],抑制Notch1表达可降低子宫内膜干细胞增殖和迁移[6]。TargetScan软件预测发现,miR-200c-3p与Notch1存在互补序列,且有研究[7]证实Notch1是miR-200c-3p的靶标。本研究探讨了在EESC中过表达miR-200c-3p并上调Notch1对细胞增殖、凋亡、迁移和侵袭的影响,旨在明确miR-200c-3p能否通过调控Notch1影响EESC恶性生物学行为。
1 材料与方法 1.1 组织来源选取2022年1月至2023年1月就诊于湖北省中医院的30例EMT患者,平均年龄(35.20±2.26)岁,从卵巢子宫内膜异位囊肿的囊壁中获取异位的子宫内膜组织;选取同期就诊于湖北省中医院的30例育龄无EMT妇女,平均年龄(36.48±2.19)岁,获取其正常子宫内膜组织。本研究获得湖北省中医院伦理委员会批准,所有研究对象签署知情同意书。
1.2 主要试剂Annexin V-FITC/PI细胞凋亡检测试剂盒购自上海碧云天生物技术股份有限公司;兔源一抗Notch1、波形蛋白(Vimentin)、E-钙黏蛋白(E-cadherin)、N-钙黏蛋白(N-cadherin)和GAPDH均购自英国abcam公司;miR-200c-3p模拟物(miR-200c-3p mimics)及其对照(miR-NC)、Notch1过表达质粒(pcDNA3.1-Notch1)及其对照(pcDNA3.1-NC)均购自广州市锐博生物科技有限公司;QuikChange Site-Directed Mutagenesis试剂盒购自美国安捷伦科技有限公司。
1.3 原代细胞培养新鲜的异位子宫内膜组织用PBS冲洗3次,并用微型剪刀切碎。然后,用Ⅱ型胶原酶(1 mg/mL)消化组织60 min,随后通过100 μm和40 μm孔径的尼龙网过滤分离EESC。分离出的EESC置于DMEM/F12培养基[内含10%胎牛血清(fetal bovine serum,FBS)、100 U/mL双抗)],于37 ℃、5% CO2培养箱中培养。第3次传代后使用抗Vimentin(1∶1 000)和抗E-cadherin(1∶1 000)抗体通过免疫荧光对EESC的纯度进行测定,选取纯度 > 99%的EESC进行后续实验[8]。
1.4 细胞转染将EESC以5×104/mL的密度接种到6孔板中,并在37 ℃下培养过夜。将EESC分为Control组(常规培养)、miR-NC组(转染阴性对照NC mimic)、miR-200c-3p-OE组(转染miR-200c-3p mimic)、miR-200c-3p-OE+pc-NC组(共转染miR-200c-3p mimic与pcDNA3.1空载体)、miR-200c-3p-OE+pc-Notch1组(共转染miR-200c-3p mimic与pcDNA3.1-Notch1载体)。使用lipofectamine 2000转染,48 h后通过实时定量PCR评估转染效率。
1.5 实时定量PCR从正常子宫内膜组织、异位子宫内膜组织以及各组EESC中提取总RNA,并将其反转录为cDNA,使用SYBR Green Mix进行实时定量PCR。引物序列如下:miR-200c-3p,正向5’-CAGTGCGTGTCGTGGAGT-3’,反向5’-GGTAATACTGCCGGGTAAT-3’;U6,正向5’-GCTTCGGCAGCACATATACTAAAAT-3’,反向5’-CGCTTCACGAATTTGCGTGTCAT-3’;Notch1,正向5’-TCCAACTGCGACACCAACCC-3’,反向5’-CCCAGCGAGCACTCATCCAC-3’;GAPDH,正向5’-CTTTGGTATCGTGGAAGGACTC-3’和反向5’-GTAGAGGCAGGGATGATGTTCT-3’。以U6或GAPDH为内参基因,采用2-ΔΔCt法计算相对表达量。所有实验重复3次。
1.6 MTT转染48 h后,将各组EESC接种到96孔板(5×104/mL,100 μL/孔)中并在37 ℃下孵育过夜。按照MTT法进行相应操作,孵育结束后在490 nm波长下检测吸光度(absorbance,A),并评估细胞活力。
1.7 流式细胞术收集细胞,按照凋亡检测试剂盒分别添加5 μL Annexin V-FITC、5 μL PI在室温下避光染色15 min。使用FACSCalibur流式细胞仪(美国碧迪医疗生物技术有限公司)分析凋亡细胞(早期+晚期凋亡细胞的百分比)。
1.8 Transwell实验转染48 h后,将各组EESC(2×104/孔,100 μL)接种到无血清DMEM的Transwell板的上室中(Transwell上室膜用Matrigel预包被30 min,仅用于侵袭试验)。将600 μL含有10% FBS的DMEM培养基接种到下室中。在37 ℃下孵育24 h后取出小室,固定、染色后洗涤,于小室晾干后用Ⅸ73荧光倒置显微镜(日本奥林巴斯株式会社)观察细胞迁移及侵袭情况。
1.9 Western blotting用RIPA裂解液提取总蛋白,BCA法定量。于蛋白变性后行SDS-PAGE凝胶电泳、转膜、封闭。结束后,添加对应一抗[Notch1(1∶1 000)、Vimentin(1∶1 000)、E-cadherin(1∶500)、N-cadherin(1∶5 000)和GAPDH(1∶2 500)] 于4 ℃孵育过夜,次日将膜与二抗(1∶2 000)于室温下孵育1 h,随后曝光检测。
1.10 双萤光素酶报告基因实验使用TargetScan软件(版本7.1;http://www.target-scan.org/vert_71/)预测miR-200c-3p与Notch1之间的结合序列。通过扩增含有miR-200c-3p靶序列的Notch1 3’非翻译区(untranslated region,UTR),并将其整合到pmirGLO载体中,以构建野生型(wild type,WT)-Notch1报告载体。使用QuikChange Site-Directed Mutagenesis试剂盒构建突变型(mutant type,MUT)-Notch1报告载体。将EESC在37 ℃下孵育24 h后,用lipofectamine 2000将WT-Notch1、MUT-Notch1报告载体分别与miR-200c-3p mimics或miR-NC共转染到EESC中。48 h后检测萤光素酶活性。
1.11 统计学分析采用SPSS 25.0软件进行统计分析,数据以x±s表示。多组比较采用单因素方差分析和SNK-q检验;组间比较采用t检验。P < 0.05为差异有统计学意义。
2 结果 2.1 子宫内膜组织中miR-200c-3p和Notch1表达与正常子宫内膜组织(1.00±0.09,1.00±0.13,0.22±0.04)比较,异位子宫内膜组织miR-200c-3p表达(0.57±0.06)降低,Notch1 mRNA(2.89±0.21)和蛋白表达(0.61±0.07)升高(P < 0.05),见图 1。
|
| 1, normal endometrial tissue; 2, ectopic endometrial tissue. 图 1 Western blotting检测子宫内膜组织中Notch1蛋白表达结果 Fig.1 Notch1 expression in endometrial tissue by Western blotting |
2.2 各组EESC中miR-200c-3p和Notch1的表达比较
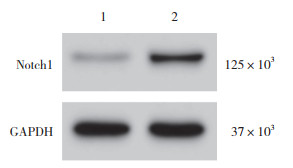
与Control组、miR-NC组比较,miR-200c-3p-OE组miR-200c-3p表达水平升高,Notch1 mRNA和蛋白表达降低(P < 0.05);与miR-200c-3p-OE组、miR-200c-3p-OE+pc-NC组比较,miR-200c-3p-OE+pc-Notch1组Notch1 mRNA和蛋白表达升高(P < 0.05)。见图 2、表 1。
|
| 1, Control group; 2, miR-NC group; 3, miR-200c-3p-OE group; 4, miR-200c-3p-OE+pc-NC group; 5, miR-200c-3p-OE+pc-Notch1 group. 图 2 Western blotting各组EESC中Notch1蛋白表达 Fig.2 Notch1 expression of EESC in each group by Western blotting |
| Group | n | miR-200c-3p | Notch1 mRNA | Notch1 protein |
| Control | 6 | 1.00±0.12 | 1.01±0.10 | 0.43±0.05 |
| miR-NC | 6 | 0.98±0.10 | 1.03±0.11 | 0.44±0.06 |
| miR-200c-3p-OE | 6 | 2.75±0.191),2) | 0.42±0.071),2) | 0.18±0.031),2) |
| miR-200c-3p-OE+pc-NC | 6 | 2.81±0.16 | 0.40±0.06 | 0.17±0.03 |
| miR-200c-3p-OE+pc-Notch1 | 6 | 2.79±0.17 | 0.85±0.093),4) | 0.36±0.043),4) |
| 1)P < 0.05 vs. Control group;2)P < 0.05 vs. miR-NC group;3)P < 0.05 vs. miR-200c-3p-OE group;4)P < 0.05 vs. miR-200c-3p-OE+pc-NC group. | ||||
2.3 各组EESC活力比较
与Control组、miR-NC组比较,miR-200c-3p-OE组细胞活力降低(P < 0.05);与miR-200c-3p-OE组、miR-200c-3p-OE+pc-NC组比较,miR-200c-3p-OE+pc-Notch1组细胞活力升高(P < 0.05)。见表 2。
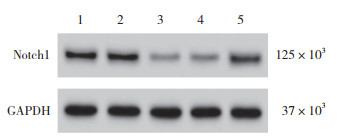
| Group | n | Cell viability | Apoptosis rate(%) |
| Control | 6 | 0.81±0.10 | 3.22±0.40 |
| miR-NC | 6 | 0.84±0.09 | 3.17±0.38 |
| miR-200c-3p-OE | 6 | 0.52±0.071),2) | 19.69±0.721),2) |
| miR-200c-3p-OE+pc-NC | 6 | 0.49±0.06 | 20.03±0.75 |
| miR-200c-3p-OE+pc-Notch1 | 6 | 0.73±0.083),4) | 8.81±0.563),4) |
| 1)P < 0.05 vs. Control group;2)P < 0.05 vs. miR-NC group;3)P < 0.05 vs. miR-200c-3p-OE group;4)P < 0.05 vs. miR-200c-3p-OE+pc-NC group. | |||
2.4 各组EESC凋亡水平比较
与Control组、miR-NC组比较,miR-200c-3p-OE组细胞凋亡率升高(P < 0.05);与miR-200c-3p-OE组、miR-200c-3p-OE+pc-NC组比较,miR-200c-3p-OE+pc-Notch1组细胞凋亡率降低(P < 0.05)。见图 3、表 2。
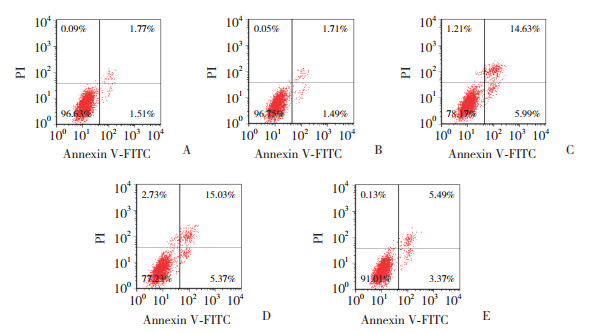
|
| A, Control group; B, miR-NC group; C, miR-200c-3p-OE group; D, miR-200c-3p-OE+pc-NC group; E, miR-200c-3p-OE+pc-Notch1 group. 图 3 各组EESC凋亡情况 Fig.3 Apoptosis status of EESC in each group |
2.5 各组EESC迁移和侵袭能力比较
与Control组、miR-NC组比较,miR-200c-3p-OE组细胞迁移和侵袭数降低(P < 0.05);与miR-200c-3p-OE组、miR-200c-3p-OE+pc-NC组比较,miR-200c-3p-OE+pc-Notch1组细胞迁移和侵袭数升高(P < 0.05)。见图 4、表 3。
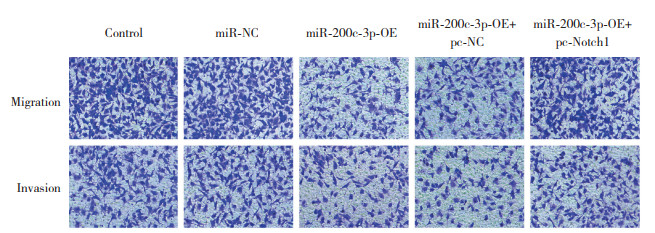
|
| 图 4 各组EESC迁移和侵袭情况结晶紫染色×200 Fig.4 Migration and invasion of EESC in each group Crystal violet staining×200 |
| Group | n | Cell migration number | Cell invasion number |
| Control | 6 | 125.35±15.82 | 92.80±10.75 |
| miR-NC | 6 | 128.40±16.13 | 93.36±11.20 |
| miR-200c-3p-OE | 6 | 60.26±9.271),2) | 45.42±6.941),2) |
| miR-200c-3p-OE+pc-NC | 6 | 58.93±8.89 | 42.90±7.37 |
| miR-200c-3p-OE+pc-Notch1 | 6 | 101.34±13.153),4) | 80.15±9.403),4) |
| 1)P < 0.05 vs. Control group;2)P < 0.05 vs. miR-NC group;3)P < 0.05 vs. miR-200c-3p-OE group;4)P < 0.05 vs. miR-200c-3p-OE+pc-NC group. | |||
2.6 各组EESC上皮-间质转化相关蛋白表达比较
与Control组、miR-NC组比较,miR-200c-3p-OE组E-cadherin蛋白水平升高,Vimentin、N-cadherin蛋白水平降低(P < 0.05);与miR-200c-3p-OE组、miR-200c-3p-OE+pc-NC组比较,miR-200c-3p-OE+pc-Notch1组E-cadherin蛋白水平降低,Vimentin、N-cadherin蛋白水平升高(P < 0.05)。见图 5、表 4。
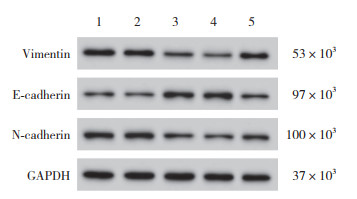
|
| 1, Control group; 2, miR-NC group; 3, miR-200c-3p-OE group; 4, miR-200c-3p-OE+pc-NC group; 5, miR-200c-3p-OE+pc-Notch1 group. 图 5 各组EESC上皮-间质转化相关蛋白的Western blotting检测结果 Fig.5 Western blotting results for epithelial-mesenchymal transition-related proteins of EESC in each group |
| Group | n | Vimentin | E-cadherin | N-cadherin |
| Control | 6 | 0.55±0.07 | 0.23±0.04 | 0.79±0.09 |
| miR-NC | 6 | 0.59±0.06 | 0.20±0.03 | 0.82±0.10 |
| miR-200c-3p-OE | 6 | 0.24±0.041),2) | 0.61±0.061),2) | 0.43±0.061),2) |
| miR-200c-3p-OE+pc-NC | 6 | 0.20±0.04 | 0.63±0.07 | 0.42±0.07 |
| miR-200c-3p-OE+pc-Notch1 | 6 | 0.43±0.053),4) | 0.39±0.053),4) | 0.71±0.093),4) |
| 1)P < 0.05 vs. Control group;2)P < 0.05 vs. miR-NC group;3)P < 0.05 vs. miR-200c-3p-OE group;4)P < 0.05 vs. miR-200c-3p-OE+pc-NC group. | ||||
2.7 miR-200c-3p与Notch1的靶向关系验证
miR-200c-3p与Notch1之间存在结合序列,见图 6。共转染miR-200c-3p mimics(0.34±0.05)较共转染miR-NCWT-Notch1(1.01±0.08)相对萤光素酶活性降低(P < 0.05);MUT-Notch1的相对萤光素酶活性在共转染miR-NC(1.00±0.09)和miR-200c-3p mimics(0.98±0.10)的细胞中无统计学差异(P > 0.05)。

|
| 图 6 TargetScan软件预测的miR-200c-3p与Notch1的结合序列 Fig.6 Binding sequence between miR-200c-3p and Notch1 predicted by TargetScan software |
3 讨论
EMT是育龄女性慢性盆腔痛、不孕症的主要原因[9]。ESC在一系列激素、生长因子、趋化因子和炎症介质的作用下表现出更强的侵袭性和迁移性。因此,积极探索ESC迁移、侵袭相关分子机制,对提高治疗有效性有重要意义。miR-200c-3p作为肿瘤抑制基因,调控多种肿瘤细胞的异常增殖、凋亡、侵袭和迁移[10]。既往研究[11]表明,miR-200c-3p在EMT患者异位子宫内膜组织中表达降低,其过表达可抑制EESC的增殖、迁移及上皮-间质转化,但其在EMT发病机制中的作用尚未完全明确。
本研究发现异位子宫内膜组织中miR-200c-3p表达低于正常子宫内膜组织,表明miR-200c-3p水平的降低参与了EMT的发病机制。miR-200c-3p mimics可抑制EESC增殖、侵袭和迁移。上皮细胞标志物E-cadherin的丢失和间质细胞标志物N-cadherin和Vimentin的增加能够促进细胞转移[12]。ZUBRZYCKA等[13]发现与上皮-间质转化相关的miRNA可作为EMT的新分子生物标志物。XUE等[14]的研究显示miR-223可抑制EMT中上皮-间质转化相关分子的表达,降低细胞迁移、侵袭和增殖。本研究发现miR-200c-3p mimics处理后N-cadherin和Vimentin表达降低,E-cadherin表达升高,证实miR-200c-3p可抑制EESC恶性生物学行为。
miRNA能够通过与靶mRNA的3’ UTR结合,参与EMT的发生发展[15]。本研究发现Notch1是miR-200c-3p的靶点,并受miR-200c-3p负调控。研究[5]表明EMT患者异位子宫内膜组织中Notch1表达上调。本研究发现异位子宫内膜组织中Notch1表达较高;且EESC中miR-200c-3p的过表达导致Notch1的mRNA和蛋白表达降低。为确定miR-200c-3p是否通过直接靶向Notch1影响EESC增殖、迁移和侵袭,本研究在EESC中过表达miR-200c-3p和Notch1,结果发现,Notch1表达升高可逆转miR-200c-3p-OE对EESC恶性生物学行为的抑制作用。LUO等[16]认为miR-34c-5p可通过靶向抑制Notch1表达,促进E-cadherin表达,下调N-cadherin和Vimentin表达,抑制EMT中上皮-间质转化过程以及细胞侵袭和迁移;这进一步为miR-200c-3p通过靶向Notch1抑制EMT进展提供了可能的证据。
综上所述,本研究首次提出miR-200c-3p可能通过靶向抑制Notch1表达抑制EESC恶性生物学行为,可为EMT的治疗提供新方向。然而,后续还需要扩大样本量,纳入体内实验做进一步探讨。
| [1] |
NOTHNICK WB, CUI W, FALCONE T, et al. Prefoldin-5 expression is elevated in eutopic and ectopic endometriotic epithelium and mo- dulates endometriotic epithelial cell proliferation and migration in vitro[J]. Int J Mol Sci, 2024, 25(4): 2390. DOI:10.3390/ijms25042390 |
| [2] |
MARTÍN-LEYVA A, PEINADO FM, OCÓN-HERNÁNDEZ O, et al. Environmental exposure to persistent organic pollutants and its association with endometriosis risk: implications in the epithelial-mesenchymal transition process[J]. Int J Mol Sci, 2024, 25(8): 4420. DOI:10.3390/ijms25084420 |
| [3] |
PONTEMEZZO E, FOGLIO E, VERNUCCI E, et al. miR-200c-3p regulates epitelial-to-mesenchymal transition in epicardial mesothelial cells by targeting epicardial follistatin-related protein 1[J]. Int J Mol Sci, 2021, 22(9): 4971. DOI:10.3390/ijms22094971 |
| [4] |
DONG L, ZHANG L, LIU H, et al. Circ_0007331 knock-down suppresses the progression of endometriosis via miR-200c-3p/HiF-1α axis[J]. J Cell Mol Med, 2020, 24(21): 12656-12666. DOI:10.1111/jcmm.15833 |
| [5] |
汤孟冬, 张浩然, 崔文杰, 等. 去整合素-金属蛋白酶17、Notch1及α-平滑肌动蛋白在子宫内膜异位症组织中的表达及意义[J]. 安徽医药, 2023, 27(11): 2181-2185, 2334. DOI:10.3969/j.issn.1009-6469.2023.11.013 |
| [6] |
METODIEV D, PARVANOV D, RUSEVA M, et al. NOTCH1- and CD117-positive stem cells in human endometriosis and adenomyosis lesions[J]. Diagnostics (Basel), 2024, 14(15): 1642. DOI:10.3390/diagnostics14151642 |
| [7] |
MAO XQ, JI T, LIU AG, et al. ELK4-mediated lncRNA SNHG22 promotes gastric cancer progression through interacting with EZH2 and regulating miR-200c-3p/Notch1 axis[J]. Cell Death Dis, 2021, 12(11): 957. DOI:10.1038/s41419-021-04228-z |
| [8] |
ZHANG ZY, WANG YQ, ZENG LQ, et al. miR-218-5p in endometrial microenvironment prevents the migration of ectopic endometrial stromal cells by inhibiting LASP1[J]. Reprod Biol Endocrinol, 2022, 20(1): 64. DOI:10.1186/s12958-022-00928-z |
| [9] |
王煜宁, 张颐. 人工智能在子宫内膜异位症诊断中的应用[J]. 中国医科大学学报, 2025, 54(5): 385-389, 413. DOI:10.12007/j.issn.0258-4646.2025.05.001 |
| [10] |
GARRIDO-CANO I, ADAM-ARTIGUES A, LAMEIRINHAS A, et al. Delivery of miR-200c-3p using tumor-targeted mesoporous silica nanoparticles for breast cancer therapy[J]. ACS Appl Mater Interfaces, 2023, 15(32): 38323-38334. DOI:10.1021/acsami.3c07541 |
| [11] |
徐超逸, 陈海燕, 陈赛玲, 等. CircRNA_000809通过miR-200c-3p对子宫内膜异位症间质细胞的增殖、迁移及上皮间质转化的影响[J]. 温州医科大学学报, 2022, 52(7): 532-538. DOI:10.3969/j.issn.2095-9400.2022.07.003 |
| [12] |
XIE YK, KONG WM, ZHAO XL, et al. Metformin inhibits the estrogen-mediated epithelial-mesenchymal transition of ectopic endometrial stromal cells in endometriosis[J]. In Vivo, 2023, 37(6): 2490-2497. DOI:10.21873/invivo.13356 |
| [13] |
ZUBRZYCKA A, MIGDALSKA-SĘK M, JĘDRZEJCZYK S, et al. Circulating miRNAs related to epithelial-mesenchymal transitions (EMT) as the new molecular markers in endometriosis[J]. Curr Issues Mol Biol, 2021, 43(2): 900-916. DOI:10.3390/cimb43020064 |
| [14] |
XUE Y, LIN XY, SHI TT, et al. miRNA-223 expression in patient-derived eutopic and ectopic endometrial stromal cells and its effect on epithelial-to-mesenchymal transition in endometriosis[J]. Clinics (Sao Paulo), 2022, 77: 100112. DOI:10.1016/j.clinsp.2022.100112 |
| [15] |
LIU Y, XIE CM, LI T, et al. PCGEM1 promotes cell proliferation and migration in endometriosis by targeting miR-124-3p-mediated ANTXR2 expression[J]. BMC Womens Health, 2023, 23(1): 104. DOI:10.1186/s12905-023-02250-1 |
| [16] |
LUO YJ, WANG DD, CHEN SL, et al. The role of miR-34c-5p/Notch in epithelial-mesenchymal transition (EMT) in endometriosis[J]. Cell Signal, 2020, 72: 109666. DOI:10.1016/j.cellsig.2020.109666 |
 2025, Vol. 54
2025, Vol. 54