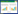文章信息
- 王常兰, 程美嘉, 边欢欢, 邹丹
- WANG Changlan, CHENG Meijia, BIAN Huanhuan, ZOU Dan
- 葛根素改善酒精暴露/戒断所致雌性小鼠认知障碍的神经炎症相关机制研究
- Neuroinflammation-related mechanism of puerarin in improving cognitive impairment induced by alcohol exposure/withdrawal in female mice
- 中国医科大学学报, 2025, 54(11): 988-994
- Journal of China Medical University, 2025, 54(11): 988-994
-
文章历史
- 收稿日期:2025-01-02
- 网络出版时间:2025-11-21 13:11:14
2. 大阪大学医学部医学统计学教研室, 大阪 565-0871;
3. 北京京煤集团总医院检验科, 北京 102300
2. Department of Medical Statistics, Faculty of Medicine, Osaka University, Osaka 565-0871, Japan;
3. Clinical Laboratory, Beijing Jingmei Group General Hospital, Beijing 102300, China
海马结构是大脑边缘系统中对酒精敏感的区域,大量饮酒会导致认知障碍[1-2]。海马结构内DG-CA3-CA1突触回路的突触可塑性与学习记忆密切相关。CA3神经元通过苔藓纤维接收来自DG的输入,并通过Schaffer侧支连接至CA1[3]。CA1和CA3具有不同的功能,参与特定信息的处理。CA1参与维持短期记忆,而CA3参与空间或情景记忆快速形成的过程[4]。
文献[5]报道,酒精戒断期的脑损伤较饮酒期更为严重。酒精依赖患者可能在最后一次饮酒后6~24 h内出现酒精戒断综合征 [6]。最新的动物模型研究[7]表明,与雄性相比,雌性大鼠更容易出现酒精诱发的神经毒性反应。雌性小鼠在酒精戒断期间的中枢神经系统(central nervous system,CNS)损伤更为严重[8]。葛根素能穿透血脑屏障进入大脑[9]。在多种CNS疾病(如缺血性脑血管病、阿尔茨海默病等)中发挥神经保护作用。本课题组前期研究[10]发现,50 mg /kg葛根素腹腔注射可改善50%(v/v)酒精诱导的小鼠的认知功能障碍。推测葛根素改善酒精诱导的认知功能障碍与海马结构中炎性细胞因子的表达水平有关。本研究通过探讨50 mg/kg葛根素对52%(v/v)(以下用52%表示)酒精暴露/戒断小鼠认知功能障碍的影响,揭示小鼠海马结构中促炎性细胞因子白细胞介素(interleukin,IL)-6、肿瘤坏死因子α(tumor necrosis factor α,TNF-α)和IL-1β在此过程中的作用。以期为葛根素改善酒精暴露及急性酒精戒断引起的认知功能障碍提供新的研究思路。
1 材料与方法 1.1 实验动物及分组昆明种雌性小鼠120只(8周龄,体重30 g±5 g)购自辽宁长生生物技术股份有限公司(SCXK2020-0001)。小鼠在(20±2)℃、光暗循环12 h条件下自由饲养1周,以适应环境。将120只小鼠随机分为4组,分别为对照组、52%酒精组、葛根素+52%酒精组、生理盐水(normal saline,NS)+52%酒精组。除对照组外,其他3组小鼠均给予52%酒精灌胃[10 mL/(kg·d)],葛根素+52%酒精组和NS+52%酒精组小鼠分别在酒精灌胃前1 h给予葛根素或NS腹腔注射[20 mL/(kg·d)],连续28 d。将停止给药后20、40、60 h设为酒精戒断时间。本研究获得沈阳医学院动物伦理委员会批准(SYYXY2022061402)。
1.2 Morris水迷宫(Morris water maze,MWM)测试MWM实验由定位航行和空间探索2部分组成。前者于实验第24~28天进行;后者分别于实验第29、30、31天进行。空间探索实验结束后立即处死小鼠。实验方法参考文献[11]。
1.3 Western blotting(WB)方法参考文献[11]。提取小鼠海马总蛋白并上样(30 μg)后,行12% SDS-PAGE电泳、转膜,抗体孵育后,发光显影。一抗为IL-6抗体(DF6087,美国Affinity公司)、TNF-α抗体(NBP1-19532SS,美国Novusbio公司)、IL-1β抗体(#12242,美国CST公司)和β-actin抗体(AC038,中国爱博泰克生物科技有限公司)。二抗为辣根过氧化物酶标记的羊抗兔IgG(E-AB-1003,中国伊莱瑞特生物科技股份有限公司)。β-actin作内参照。
1.4 免疫组织化学(immunohistochemistry,IHC)染色方法参考文献[11]。一抗同1.3。二抗为EnVision TM试剂盒(KIT-9901,中国迈新生物技术开发有限公司)。
1.5 统计学分析采用GraphPad Prism 9.0软件进行统计学分析。计量资料以x±s表示,组间比较采用单因素方差分析和Tukey’s t检验。P < 0.05为差异有统计学意义。
2 结果 2.1 葛根素对酒精戒断20 h小鼠认知功能及海马结构中IL-6、TNF-α、IL-1β表达的影响MWM结果显示,在酒精戒断20 h时,52%酒精组和NS+52%酒精组的目标象限距离百分比和时间百分比显著低于对照组和葛根素+52%酒精组(均P < 0.01)。见表 1。WB结果显示,与对照组和葛根素+52%酒精组相比,52%酒精组和NS+52%酒精组小鼠海马结构中IL-6[F(3,16)=30.13,P < 0.01]、TNF-α [F(3,16)=13.21,P < 0.01]和IL-1β表达水平均显著降低[F(3,16)=10.26,P < 0.01]。见图 1A。IHC结果显示,与对照组和葛根素+52%酒精组比较,52%酒精组和NS+52%酒精组小鼠海马透明层(stratum lucidum,SL)区IL-6 [F(3,16)=16.72,P < 0.01]、CA1区TNF-α [F(3,16)=10.36,P < 0.01]和IL-1β [F(3,16)=7.709,P < 0.01]的表达均显著降低。见图 1B。
| Item | Control group | 52% alcohol group | Puerarin+52% alcohol group | NS+52% alcohol group | F (3, 36) | P |
| The target quadrant distance percentage | ||||||
| Alcohol withdrawal 20 h | 32.57±7.84 | 16.67±8.801),2) | 32.46±10.48 | 15.03±11.681),2) | 9.66 | < 0.01 |
| Alcohol withdrawal 40 h | 37.95±9.54 | 18.60±6.871),2) | 33.94±10.17 | 14.01±7.421),2) | 18.16 | < 0.01 |
| Alcohol withdrawal 60 h | 33.04±9.56 | 15.02±7.961),2) | 31.55±6.67 | 18.50±4.581),2) | 15.05 | < 0.01 |
| The target quadrant time percentage | ||||||
| Alcohol withdrawal 20 h | 33.27±7.77 | 17.55±8.301),3) | 32.17±10.57 | 12.63±12.521),2) | 10.85 | < 0.01 |
| Alcohol withdrawal 40 h | 39.13±10.34 | 17.70±7.971),2) | 34.24±9.68 | 12.08±7.571),2) | 20.84 | < 0.01 |
| Alcohol withdrawal 60 h | 33.7±10.32 | 14.77±9.191),2) | 32.14±7.94 | 17.38±7.171),2) | 12.59 | < 0.01 |
| 1) P < 0.01 vs. control group; 2) P < 0.01, 3) P < 0.05 vs. puerarin+52% alcohol group. | ||||||
|
| A, Western blotting results; B, IHC results. #P < 0.05, ##P < 0.01 vs. control group; *P < 0.05, **P < 0.01. A, 52% alcohol; P, puerarin; NS, normal saline. 图 1 酒精戒断20 h时WB、IHC结果 Fig.1 Results from WB and IHC at the 20th hour of alcohol withdrawal |
2.2 葛根素对酒精戒断40 h小鼠认知功能及海马结构中IL-6、TNF-α、IL-1β表达的影响
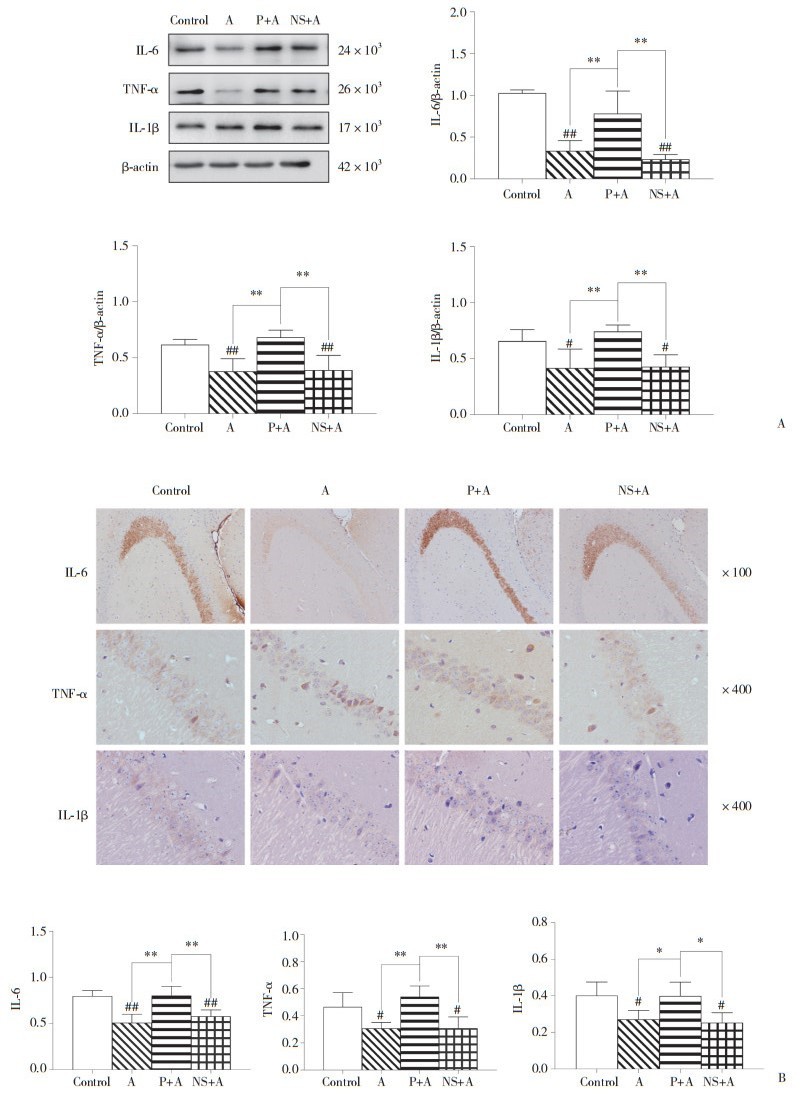
酒精戒断40 h时,MWM结果显示,52%酒精组和NS+52%酒精组的目标象限距离百分比和时间百分比均显著低于对照组和葛根素+52%酒精组(均P < 0.01)。见表 1。WB结果显示,与对照组和葛根素+52%酒精组相比,52% 酒精组和NS+52%酒精组小鼠海马结构中仅IL-1β表达水平显著降低[F(3,16)= 6.96,P < 0.01]。IHC结果与WB结果一致[F(3,16)= 6.143,P < 0.01]。见图 2。
|
| A, Western blotting results; B, IHC results. #P < 0.05 vs. control group; *P < 0.05, **P < 0.01. A, 52% alcohol; P, puerarin; NS, normal saline. 图 2 酒精戒断40 h时WB和IHC结果 Fig.2 Results from WB and IHC at the 40th hour of alcohol withdrawal |
2.3 葛根素对酒精戒断60 h小鼠认知功能及海马结构中IL-6、TNF-α、IL-1β表达的影响
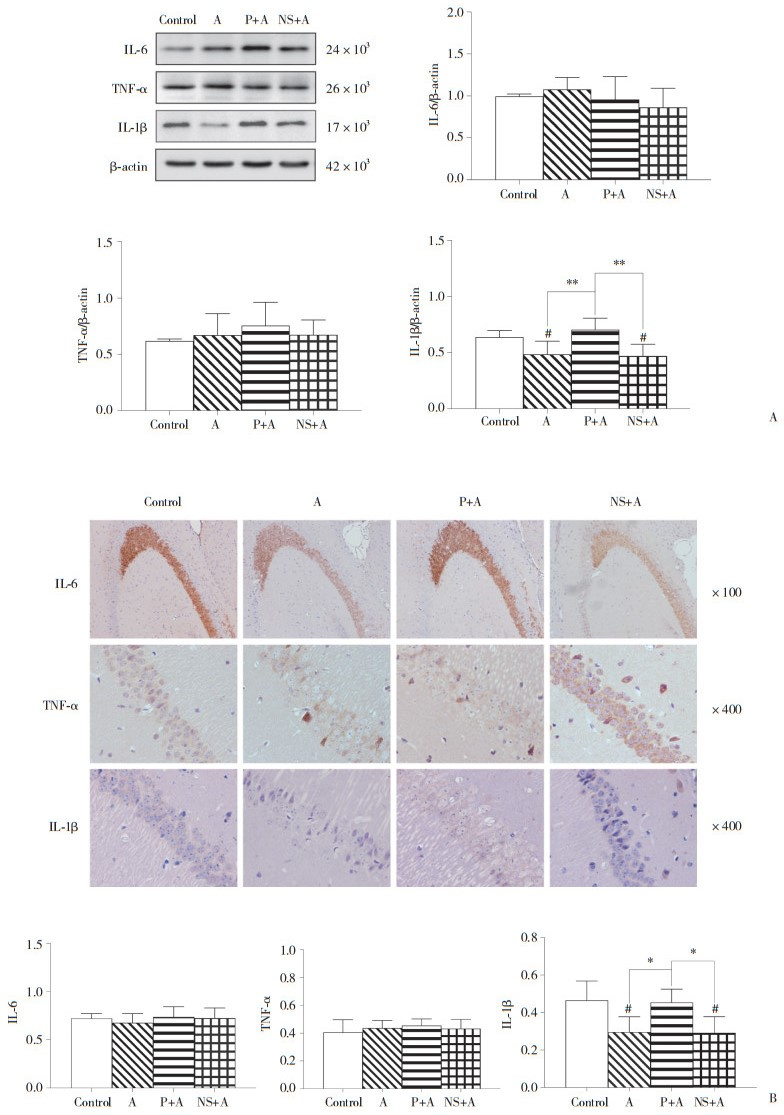
酒精戒断60 h时,MWM结果显示,52%酒精组和NS+52%酒精组的目标象限距离百分比和时间百分比均显著低于对照组和葛根素+52%酒精组(均P < 0.01),见表 1。但WB和IHC结果显示,各组小鼠3种炎性细胞因子表达水平无统计学差异(均P > 0.05)。见图 3。
|
| A, Western blotting results; B, IHC results. A, 52% alcohol; P, puerarin; NS, normal saline. 图 3 酒精戒断60 h时WB和IHC结果 Fig.3 Results from WB and IHC at the 60th hour of alcohol withdrawal |
3 讨论
长期酗酒可导致认知功能障碍,酒精对CNS的影响取决于其暴露的模式和剂量[12]。药代动力学研究[13]表明,葛根素具有半衰期短、消除快等优点。葛根素对慢性酒精中毒小鼠空间学习记忆障碍具有保护作用[10]。本研究结果显示,在小鼠酒精戒断20 h、40 h、60 h时的MWM空间探索实验中,52%酒精组和NS+52%酒精组的目标象限距离百分比和时间百分比均较对照组和葛根素+52%酒精组明显缩短,提示葛根素预处理可改善酒精暴露小鼠的认知功能。
慢性饮酒能诱导小胶质细胞释放IL-6、TNF-α、IL-1β等炎性细胞因子,破坏突触可塑性,导致神经炎症,促进神经元变性和死亡[14]。雌性大鼠在青春期至成年早期的长期高浓度酒精暴露会导致与海马形态改变相关的长期情绪和记忆缺陷[15]。青少年间歇性酒精暴露的认知缺陷在女性中尤其明显,且在酒精戒断后长期存在[7]。
IL-6由CNS胶质细胞产生,CNS中IL-6水平的改变会损害小鼠认知功能并降低小鼠对新事物的反应能力[16]。CNS中IL-6缺乏会损害小鼠海马依赖的学习和记忆功能[17]。本研究中,WB结果显示,慢性酒精染毒后,在酒精戒断20 h时,52%酒精组和NS+52%酒精组小鼠海马结构中IL-6的表达水平明显低于对照组和葛根素+52%酒精组。在酒精戒断40 h、60 h后,逐渐恢复正常。苔藓纤维连接DG和海马,所有纤维终止于CA3和CA2区之间交界处,形成CA3特定的SL区。本研究中,IHC结果显示,在酒精戒断20 h时,52%酒精组和NS+52%酒精组海马SL区IL-6表达明显低于相应的对照组和葛根素+52%酒精组,52%酒精组与NS+52%酒精组SL区IL-6表达无统计学差异。提示慢性过度饮酒后的急性酒精戒断可降低海马SL区IL-6的表达,但给予50 mg/kg葛根素腹腔注射预处理可阻止IL-6表达的下降。
TNF-α是一种由星形胶质细胞、小胶质细胞和某些神经元合成的促炎性细胞因子,在神经毒性、突触传递和突触可塑性中发挥重要作用。在慢性酒精蒸汽暴露模型中,大鼠血清TNF-α和IL-1β在酒精戒断第1天显著降低[18]。对于酒精连续暴露4 d的大鼠模型,在酒精戒断的第2、7、14天,从海马结构和内嗅皮质分离的小胶质细胞中促炎性细胞因子TNF-α、IL-6和IL-1β的表达显著降低[19]。
本研究中,WB与IHC结果显示,慢性高浓度酒精暴露28 d的小鼠在酒精戒断20 h时,海马结构中TNF-α水平明显下降,40 h、60 h时逐渐恢复。葛根素预处理可阻止酒精戒断第20 h海马结构内TNF-α水平的降低。乙醇在神经胶质细胞中可氧化为乙醛,且2种化合物均可抑制脑中TNF-α的分泌。50 mg/kg葛根素预处理可预防急性酒精戒断引起的海马CA1区TNF-α表达水平降低。
IL-1β是一种由脑内胶质细胞产生的强效促炎性细胞因子,在海马依赖性突触可塑性和长期记忆的维持中起重要作用。IL-1β在海马依赖的记忆形成过程中遵循倒U型模式,即生理水平的IL-1β有利于记忆形成,但偏离生理范围会导致记忆损害。本研究发现,在酒精戒断20 h、40 h时,52%酒精组和NS+52% 酒精组IL-1β表达显著低于对照组和葛根素+52%酒精组;葛根素维持了酒精戒断后小鼠海马结构中IL-1β的表达水平;在酒精戒断后,IL-1β在海马内表达水平的恢复速度慢于IL-6和TNF-α。
综上所述,本研究发现,慢性酒精染毒雌性小鼠在急性酒精戒断20 h时,葛根素通过增加海马结构中IL-6、TNF-α和IL-1β的表达水平改善了小鼠的认知功能。而未进行葛根素预处理的酒精灌胃小鼠,在酒精戒断60 h时,IL-6、TNF-α和IL-1β的表达水平有所恢复,但小鼠的认知功能却尚未完全恢复,因此,推测酒精引起的认知障碍可能还有脑内其他神经结构和信号通路参与,有待进一步研究。
| [1] |
JIANG CH, ZHANG Y, TANG XL, et al. IL-6 and IL-1β upregulation and tau protein phosphorylation in response to chronic alcohol exposure in the mouse hippocampus[J]. Neuroreport, 2021, 32(10): 851-857. DOI:10.1097/WNR.0000000000001661 |
| [2] |
边欢欢, 程美嘉, 吴仪, 等. 酒精饮料对小鼠认知功能的影响及其机制[J]. 中国医科大学学报, 2023, 52(3): 230-235. DOI:10.12007/j.issn.0258-4646.2023.03.007 |
| [3] |
DOMÍNGUEZ-SALA E, ANDREU-CERVERA A, MARTÍN-CLIMENT P, et al. Properties of the epileptiform activity in the cingulate cortex of a mouse model of LIS1 dysfunction[J]. Brain Struct Funct, 2022, 227(5): 1599-1614. DOI:10.1007/s00429-022-02458-1 |
| [4] |
YANG QH, YAN CX, SUN YH, et al. Extracellular matrix remodeling alleviates memory deficits in Alzheimer's disease by enhancing the astrocytic autophagy-lysosome pathway[J]. Adv Sci, 2024, 11(31): 2400480. DOI:10.1002/advs.202400480 |
| [5] |
MACHT V, DE CASTRO S, VETRENO RP. Impact of neuroimmune system activation by adolescent binge alcohol exposure on adult neurobiology[J]. Adv Exp Med Biol, 2025, 1473: 179-208. DOI:10.1007/978-3-031-81908-7_9 |
| [6] |
TIGLAO SM, MEISENHEIMER ES, OH RC. Alcohol withdrawal syndrome: outpatient management[J]. Am Fam Physician, 2021, 104(3): 253-262. |
| [7] |
MACHT V, ELCHERT N, CREWS F. Adolescent alcohol exposure produces protracted cognitive-behavioral impairments in adult male and female rats[J]. Brain Sci, 2020, 10(11): 785. DOI:10.3390/brainsci10110785 |
| [8] |
SHARMA A, BRENNER M, WANG P. Potential role of extracellular CIRP in alcohol-induced Alzheimer's disease[J]. Mol Neurobiol, 2020, 57(12): 5000-5010. DOI:10.1007/s12035-020-02075-1 |
| [9] |
LIU S, CAO XL, LIU GQ, et al. The in silico and in vivo evaluation of puerarin against Alzheimer's disease[J]. Food Funct, 2019, 10(2): 799-813. DOI:10.1039/c8fo01696h |
| [10] |
BIAN HH, WU Y, CUI ZG, et al. Study on the autophagy-related mechanism of puerarin in improving the cognitive impairment induced by alcohol in female mice[J]. Brain Inj, 2022, 36(1): 137-145. DOI:10.1080/02699052.2022.2037712 |
| [11] |
XING Y, ZOU D. Ethanol-induced cognitive dysfunction is associa-ted with alterations in the mammalian target of rapamycin signalling pathway in the hippocampus of male mice[J]. Neuroreport, 2018, 29(14): 1230-1237. DOI:10.1097/WNR.0000000000001104 |
| [12] |
ROBERTO M, NELSON TE, UR CL, et al. Long-term potentiation in the rat hippocampus is reversibly depressed by chronic intermittent ethanol exposure[J]. J Neurophysiol, 2002, 87(5): 2385-2397. DOI:10.1152/jn.2002.87.5.2385 |
| [13] |
WANG SG, ZHANG SM, WANG SP, et al. A comprehensive review on Pueraria: insights on its chemistry and medicinal value[J]. Biomed Pharmacother, 2020, 131: 110734. DOI:10.1016/j.biopha.2020.110734 |
| [14] |
BARNETT A, DAVID E, ROHLMAN A, et al. Adolescent binge alcohol enhances early Alzheimer's disease pathology in adulthood through proinflammatory neuroimmune activation[J]. Front Pharmacol, 2022, 13: 884170. DOI:10.3389/fphar.2022.884170 |
| [15] |
Oliveira AC, Pereira MC, Santana LN, et al. Chronic etha-nol exposure during adolescence through early adulthood in female rats induces emotional and memory deficits associated with morphological and molecular alterations in hippocampus[J]. J Psychopharmacol, 2015, 29(6): 712-724. DOI:10.1177/0269881115581960 |
| [16] |
ROBERTS AJ, KHOM S, BAJO M, et al. Increased IL-6 expression in astrocytes is associated with emotionality, alterations in central amygdala GABAergic transmission, and excitability during alcohol withdrawal[J]. Brain Behav Immun, 2019, 82: 188-202. DOI:10.1016/j.bbi.2019.08.185 |
| [17] |
GRUOL DL, CALDERON D, HUITRON-RESENDIZ S, et al. Impact of elevated brain IL-6 in transgenic mice on the behavioral and neurochemical consequences of chronic alcohol exposure[J]. Cells, 2023, 12(18): 2306. DOI:10.3390/cells12182306 |
| [18] |
SANCHEZ-ALAVEZ M, NGUYEN W, MORI S, et al. Time course of microglia activation and brain and blood cytokine/chemokine levels following chronic ethanol exposure and protracted withdrawal in rats[J]. Alcohol, 2019, 76: 37-45. DOI:10.1016/j.alcohol.2018.07.005 |
| [19] |
PENG H, NIXON K. Microglia phenotypes following the induction of alcohol dependence in adolescent rats[J]. Alcohol Clin Exp Res, 2021, 45(1): 105-116. DOI:10.1111/acer.14504 |
 2025, Vol. 54
2025, Vol. 54