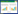文章信息
- 徐芳丽, 王焕, 徐可
- XU Fangli, WANG Huan, XU Ke
- miR-488-3p调控cGAS-STING信号通路对肾小管上皮细胞损伤的影响
- Effects of miR-488-3p on renal tubular epithelial cell injury by regulating the cGAS-STING signaling pathway
- 中国医科大学学报, 2025, 54(11): 982-987
- Journal of China Medical University, 2025, 54(11): 982-987
-
文章历史
- 收稿日期:2024-10-10
- 网络出版时间:2025-11-21 13:04:26
急性肾损伤(acute kidney injury,AKI)是一种常见的临床综合征,在全球住院患者中的发生率约为19.3%~31%[1]。目前AKI的发病机制尚未明确,治疗手段有限。肾小管上皮细胞损伤是AKI中最重要的事件[2]。因此,探讨肾小管上皮细胞损伤的潜在机制对确定AKI的治疗靶点具有重要意义。研究[3]表明,微RNA(micro RNA,miRNA)表达异常与肾小管上皮细胞损伤有关,如miR-488-3p可诱导小鼠肾小球足细胞凋亡[4]。研究[5]发现,抑制环磷酸鸟苷-腺苷酸合成酶(cyclic guanosine monophosphate-adenosine monophosphate synthase,cGAS)-干扰素基因刺激因子(stimulator of interferon gene,STING)通路可缓解小鼠AKI,因此,本研究拟探讨miR-488-3p调控cGAS-STING通路对肾小管上皮细胞损伤的影响及其机制。
1 材料与方法 1.1 材料 1.1.1 细胞人肾小管上皮细胞系HK-2购自上海普诺赛生物工程有限公司。
1.1.2 试剂miR-488-3p抑制剂及其抑制剂对照(inhibitor NC)、cGAS-STING通路激活剂RocA(美国MCE公司);CCK-8试剂盒(济南远达晶美生物科技有限公司);人白细胞介素(interleukin,IL)-10、IL-1β、肿瘤坏死因子α(tumor necrosis factor α,TNF-α)ELISA试剂盒(上海广锐生物科技有限公司);DCFH-DA(上海抚生实业有限公司);丙二醛(malondialdehyde,MDA)、超氧化物歧化酶(superoxide dismutase,SOD)试剂盒(武汉赛培生物科技有限公司);Annexin V-FITC/PI细胞凋亡试剂盒(广州美仑生物科技有限公司);cleaved caspase-3、Bcl-2关联X蛋白(Bcl-2 associated X protein,Bax)、GAPDH、cGAS、STING一抗及二抗(英国abcam公司)。
1.2 方法 1.2.1 细胞分组将HK-2细胞分为对照组、HR组、抑制对照组、miR-488-3p抑制组、RocA组、miR-488-3p抑制+RocA组。对照组HK-2细胞正常培养;HR组HK-2细胞进行缺氧(1%O2+94%N2+5%CO2)处理4 h后再进行复氧(5%CO2+95%O2)处理2 h[6];抑制对照组HK-2细胞转染inhibitor NC 24 h后,缺氧处理4 h,复氧处理2 h;miR-488-3p抑制组HK-2细胞转染miR-488-3p抑制剂24 h后,缺氧处理4 h,复氧处理2 h;RocA组HK-2细胞用25 nmol/L RocA处理24 h后,缺氧处理4 h,复氧处理2 h[7];miR-488-3p抑制+RocA组HK-2细胞转染miR-488-3p抑制剂的同时,还用25 nmol/L RocA处理24 h,再进行缺氧处理4 h,复氧处理2 h。处理结束后收集各组细胞。
1.2.2 miR-488-3p表达水平检测收集各组细胞,用TRIzol试剂提取细胞总RNA,用NanoDrop 2000测定RNA浓度,将RNA逆转录为cDNA后,采用ABI7500系统进行实时定量PCR。以U6作内参照,采用2-∆∆Ct法计算miR-488-3p相对表达量。引物序列:miR-488-3p,正向5’-CGGGGCAGCUCAGUACAG-3’,反向5’-CAGTGCGTGTCGTGGAGT-3’;U6,正向5’-CGCTTCGGCAGCACATATAC-3’,反向5’-AAATATGGAACGCTTCACGA-3’。
1.2.3 细胞活力检测将HK-2细胞以5×103/孔的密度接种至96孔板,对各组细胞进行相应处理后,每孔加入10 μL CCK-8溶液。37℃避光孵育3 h后,用酶标仪测量450 nm处的吸光度,并计算细胞活力。
1.2.4 IL-10、IL-1β、TNF-α水平检测收集各组HK-2细胞培养液,严格按照ELISA试剂盒说明书检测上清液中IL-10、IL-1β、TNF-α水平。
1.2.5 活性氧(reactive oxygen species,ROS)平均荧光强度、MDA、SOD水平检测收集各组HK-2细胞,PBS洗涤3次后,加入10 μmol/L DCFH-DA混合均匀,在37 ℃下孵育30 min。用PBS洗涤细胞3次,以充分去除未渗透细胞的DCFH-DA,在荧光显微镜下观察HK-2细胞中ROS平均荧光强度变化。按照试剂盒说明书检测HK-2细胞中MDA、SOD水平。
1.2.6 细胞凋亡率检测收集各组HK-2细胞,用胰蛋白酶消化,PBS洗涤后,加入5 μL Annexin V-FITC和5 μL PI,避光孵育15 min后,用流式细胞仪检测细胞凋亡情况。
1.2.7 cleaved caspase-3、Bax、cGAS、STING蛋白表达水平检测收集各组HK-2细胞,RIPA裂解并提取总蛋白。BCA法测定蛋白浓度,取45 μg等量蛋白进行电泳分离,转至PVDF膜。用5%脱脂牛奶阻断膜1 h,TBST洗涤膜3次后,将膜与一抗[cleaved caspase-3(1∶4 000稀释)、Bax(1∶6 000稀释)、cGAS(1∶4 000稀释)、GAPDH(1∶3 000稀释)、STING(1∶5 000稀释)在4 ℃下孵育过夜。用TBST洗涤膜3次,加入二抗(1∶10 000稀释)]室温下孵育1 h,用TBST洗涤3次,ECL显色。使用ImageJ软件对图像进行量化。
1.3 统计学分析用Prism 8.0软件进行统计分析。计量资料以x±s表示,采用单因素方差分析和事后SNK-q检验进行多组比较。P < 0.05为差异有统计学意义。
2 结果 2.1 各组HK-2细胞中miR-488-3p表达及细胞活力比较与对照组(1.00±0.00,99.43%±0.35%)比较,HR组细胞中miR-488-3p表达(1.96±0.17)升高,细胞活力(67.51%±3.12%)降低(P < 0.05);与HR组或抑制对照组(2.01±0.18,66.82%±3.15%)比较,miR-488-3p抑制组细胞中miR-488-3p表达(1.23±0.09)降低,细胞活力(82.29%±3.78%)升高(P < 0.05);与HR组比较,RocA组细胞中miR-488-3p表达(1.98±0.16)差异无统计学意义(P > 0.05),细胞活力(54.95%±2.67%)降低(P < 0.05);与miR-488-3p抑制组比较,miR-488-3p抑制+RocA组细胞中miR-488-3p表达(1.26±0.11)差异无统计学意义(P > 0.05),细胞活力(71.36%±3.31%)降低(P < 0.05)。
2.2 抑制miR-488-3p表达对HK-2细胞IL-10、IL-1β、TNF-α水平的影响与对照组比较,HR组细胞上清液中IL-10水平降低,IL-1β、TNF-α水平升高(P < 0.05);与HR组、抑制对照组比较,miR-488-3p抑制组细胞上清液中IL-10水平升高,IL-1β、TNF-α水平降低(P < 0.05);与HR组比较,RocA组细胞上清液中IL-10水平降低,IL-1β、TNF-α水平升高(P < 0.05);与miR-488-3p抑制组比较,miR-488-3p抑制+RocA组细胞上清液中IL-10水平降低,IL-1β、TNF-α水平升高(P < 0.05)。见表 1。
| Group | IL-10 | IL-1β | TNF-α |
| Control | 76.65±3.14 | 26.78±1.32 | 115.56±5.69 |
| HR | 39.73±1.751) | 59.93±2.461) | 20.34±10.881) |
| Inhibition control | 40.26±1.98 | 60.28±2.51 | 205.51±11.01 |
| miR-488-3p inhibition | 65.93±3.082),3) | 34.45±1.682),3) | 136.56±6.232),3) |
| RocA | 31.44±1.432) | 68.45±3.052) | 261.56±1.732) |
| miR-488-3p inhibition+RocA | 53.39±2.174) | 47.55±2.194) | 16.25±8.124) |
| 1) P < 0.05 vs. control group; 2) P < 0.05 vs. HR group; 3) P < 0.05 vs. inhibition control group; 4) P < 0.05 vs. miR-488-3p inhibition group. n=6. | |||
2.3 抑制miR-488-3p表达对HK-2细胞中ROS平均荧光强度、MDA、SOD水平的影响
与对照组比较,HR组细胞中SOD水平降低,ROS平均荧光强度、MDA水平升高(P < 0.05);与HR组、抑制对照组比较,miR-488-3p抑制组细胞中SOD水平升高,ROS平均荧光强度、MDA水平降低(P < 0.05);与HR组比较,RocA组细胞中SOD水平降低,ROS平均荧光强度、MDA水平升高(P < 0.05);与miR-488-3p抑制组比较,miR-488-3p抑制+RocA组细胞中SOD水平降低,ROS平均荧光强度、MDA水平升高(P < 0.05),见图 1、表 2。
|
| A, control group; B, HR group; C, inhibiton control group; D, miR-488-3p inhibition group; E, RocA group; F, miR-488-3p inhibition+RocA group. 图 1 各组HK-2细胞中ROS平均荧光强度DCFH-DA染色×200 Fig.1 The average fluorescence intensity of ROS in HK-2 cells DCFH-DA staining× 200 |
| Group | Arerage fluorescence intensity of ROS | MDA (mmol/mL) | SOD (U/mL) |
| Control | 16.22±0.79 | 1.23±0.14 | 285.69±1.83 |
| HR | 37.75±1.781) | 5.56±0.211) | 167.73±8.261) |
| Inhibition control | 38.05±1.93 | 5.62±0.23 | 169.52±8.29 |
| miR-488-3p inhibition | 21.16±0.972),3) | 2.08±0.102),3) | 251.52±1.212),3) |
| RocA | 44.55±2.032) | 6.72±0.292) | 138.77±6.712) |
| miR-488-3p inhibition+RocA | 30.41±1.464) | 4.01±0.184) | 200.33±9.824) |
| 1) P < 0.05 vs. control group; 2) P < 0.05 vs. HR group; 3) P < 0.05 vs. inhibiton control group; 4) P < 0.05 vs. miR-488-3p inhibition group. n=6. | |||
2.4 抑制miR-488-3p表达对HK-2细胞凋亡的影响
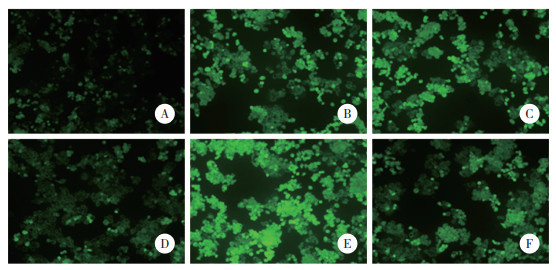
与对照组(3.16%±0.12%)比较,HR组细胞凋亡率(16.88%±0.79%)升高(P < 0.05);与HR组、抑制对照组(17.01%±0.82%)比较,miR-488-3p抑制组细胞凋亡率(6.29%±0.31%)降低(P < 0.05);与HR组比较,RocA组细胞凋亡率(21.22%±1.03%)升高(P < 0.05);与miR-488-3p抑制组比较,miR-488-3p抑制+RocA组细胞凋亡率(10.95%±0.43%)升高(P < 0.05)。见图 2。
|
| A, control group; B, HR group; C, inhibiton control group; D, miR-488-3p inhibition group; E, RocA group; F, miR-488-3p inhibition+RocA group. 图 2 HK-2细胞凋亡的流式细胞术检测结果 Fig.2 Flow cytometry detection results of HK-2 cell apoptosis |
2.5 抑制miR-488-3p表达对各组细胞中cleaved caspase-3、Bax及cGAS-STING通路蛋白表达的影响
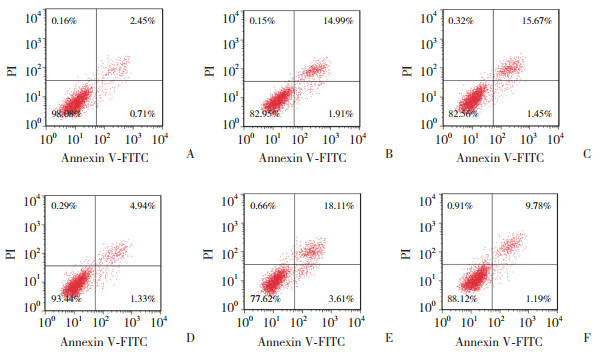
与对照组(0.78±0.07,0.52±0.05,0.36±0.03,0.12±0.01)比较,HR组HK-2细胞中cleaved caspase-3、Bax、cGAS、STING蛋白表达水平(1.89±0.14,1.43±0.12,1.01±0.09,0.72±0.07)升高(P < 0.05);与HR组、抑制对照组(1.91±0.16,1.45±0.13,1.03±0.10,0.70±0.06)比较,miR-488-3p抑制组HK-2细胞中cleaved caspase-3、Bax、cGAS、STING蛋白表达水平(0.97±0.08,0.73±0.07,0.47±0.04,0.28±0.02)降低(P < 0.05);与HR组比较,RocA组HK-2细胞中cleaved caspase-3、Bax、cGAS、STING蛋白表达水平(2.37±0.15,1.92±0.14,1.39±0.12,0.89±0.08)升高(P < 0.05);与miR-488-3p抑制组比较,miR-488-3p抑制+RocA组HK-2细胞中cleaved caspase-3、Bax、cGAS、STING蛋白表达水平(1.51±0.13,1.04±0.09,0.73±0.07,0.51±0.05)升高(P < 0.05)。见图 3。
|
| 1, control group; 2, HR group; 3, inhibition control group; 4, miR-488-3p inhibition group; 5, RocA group; 6, miR-488-3p inhibition+RocA group. 图 3 HK-2细胞中cleaved caspase-3、Bax、cGAS、STING蛋白检测 Fig.3 Detection of cleaved caspase-3, Bax, cGAS, and STING proteins in HK-2 cells |
3 讨论
AKI已被公认为影响全球数百万患者的重大公共卫生问题[8-9]。本研究通过缺氧复氧处理肾小管上皮细胞HK-2模拟体内AKI过程,结果发现炎症反应参与了HK-2细胞的损伤过程,与其他研究[10]结果一致。
研究显示,ROS产生过量引起的氧化应激是肾小管上皮细胞损伤的主要原因[11],SOD可抑制随后的脂质过氧化,而MDA可促进脂质过氧化,从而对细胞造成损伤[12]。本研究中,HR组HK-2细胞中SOD水平低于对照组,ROS平均荧光强度、MDA水平高于对照组,证实了缺氧复氧诱导的HK-2细胞存在氧化应激。ROS参与肾损伤的发生和发展,破坏线粒体功能,促进炎症,加速细胞凋亡[13]。cleaved caspase-3是由caspase-3剪切后形成,被认为是细胞凋亡的标志[14]。抑制Bax表达可改善缺氧复氧诱导的肾小管上皮细胞凋亡[15]。本研究中,HR组HK-2细胞凋亡率、cleaved caspase-3、Bax蛋白表达水平均高于对照组,表明缺氧复氧诱导了HK-2细胞凋亡。提示抑制炎症、氧化应激及细胞凋亡可能是减轻缺氧复氧诱导的HK-2细胞损伤的有效途径。
miR-488可抑制缺氧复氧心肌H9c2细胞氧化应激和细胞凋亡,进而减轻细胞损伤[16]。本研究中,经缺氧复氧处理后的HK-2细胞中miR-488-3p表达上调,于是对缺氧复氧诱导的HK-2细胞进行了下调miR-488-3p表达的处理,结果发现,下调miR-488-3p表达可抑制缺氧复氧诱导的HK-2细胞炎症、氧化应激及细胞凋亡,进而减轻细胞损伤。提示miR-488-3p可能成为治疗AKI的潜在有效靶点之一。
cGAS-STING通路在肾损伤中起着至关重要的作用[17]。如抑制cGAS-STING通路可减少顺铂诱导的人肾小管上皮细胞HKC-8凋亡,缓解炎症,改善氧化应激,缓解AKI[18]。本研究中,与HR组比较,RocA组HK-2细胞中cGAS、STING蛋白表达上调,炎症反应、氧化应激及细胞凋亡增加。为了进一步验证以上结果,本研究用cGAS-STING通路激活剂RocA设置了回复实验,结果显示,RocA逆转了下调miR-488-3p表达对缺氧复氧诱导的HK-2细胞炎症、氧化应激及细胞凋亡的影响。进一步证实cGAS-STING通路的激活参与了缺氧复氧诱导的HK-2细胞损伤过程。
综上所述,本研究发现下调miR-488-3p表达能够抑制缺氧复氧诱导的HK-2细胞炎症、氧化应激及细胞凋亡,进而减轻细胞损伤,其机制可能与抑制cGAS-STING通路激活有关。本研究结果可能为AKI的治疗提供新的参考依据。
| [1] |
XIANG Y, FU Y, WU W, et al. Autophagy in acute kidney injury and maladaptive kidney repair[J]. Burns Trauma, 2023, 11: tkac059. DOI:10.1093/burnst/tkac059 |
| [2] |
袁琦, 简禄勇, 郭华慧, 等. 自噬抑制急性肾损伤诱导急性肺损伤的分子机制[J]. 中国医科大学学报, 2024, 53(6): 501-508. DOI:10.12007/j.issn.0258-4646.2024.06.004 |
| [3] |
GAN XG, WANG ZH, XU HT. Mechanism of miRNA-141-3p in calcium oxalate-induced renal tubular epithelial cell injury via NLRP3-mediated pyroptosis[J]. Kidney Blood Press Res, 2022, 47(5): 300-308. DOI:10.1159/000521795 |
| [4] |
揭育祯, 丁宁, 谢琳, 等. 同型半胱氨酸上调miR-488-3p表达诱导MPC-5小鼠肾小球足细胞凋亡[J]. 细胞与分子免疫学杂志, 2022, 38(9): 801-806. DOI:10.13423/j.cnki.cjcmi.009470 |
| [5] |
LIU S, GAO XT, YIN YL, et al. Silk fibroin peptide self-assembled nanofibers delivered naringenin to alleviate cisplatin-induced acute kidney injury by inhibiting mtDNA-cGAS-STING pathway[J]. Food Chem Toxicol, 2023, 177(1): 113844-113854. DOI:10.1016/j.fct.2023.113844 |
| [6] |
陈文凤, 桂伟. 微小RNA-137对人肾小管上皮细胞缺氧/复氧损伤的影响[J]. 中国临床药理学杂志, 2021, 37(18): 2416-2422. DOI:10.13699/j.cnki.1001-6821.2021.18.009 |
| [7] |
张士伟, 程慎令, 邢启峰. 茯苓酸通过抑制cGAS-STING信号通路减轻溃疡性结肠炎大鼠结肠上皮细胞损伤[J]. 免疫学杂志, 2023, 39(8): 672-680. DOI:10.13431/j.cnki.immunol.j.20230087 |
| [8] |
JI XZ, CHU LJ, SU D, et al. MRPL12-ANT3 interaction involves in acute kidney injury via regulating MPTP of tubular epithelial cells[J]. iScience, 2023, 26(5): 106656-106677. DOI:10.1016/j.isci.2023.106656 |
| [9] |
HUANG YB, JIANG L, LIU XQ, et al. Melatonin alleviates acute kidney injury by inhibiting NRF2/Slc7a11 axis-mediated ferroptosis[J]. Oxid Med Cell Longev, 2022, 2022(1): 4776243-4776266. DOI:10.1155/2022/4776243 |
| [10] |
朱冬菊, 吴祥, 祝司霞. 白藜芦醇对缺氧复氧致肾小管上皮细胞内质网应激和炎性损伤的影响[J]. 中华中医药学刊, 2023, 41(5): 42-44, 264. DOI:10.13193/j.issn.1673-7717.2023.05.010 |
| [11] |
DENG WM, WEI XL, XIE ZW, et al. Inhibition of PLK3 attenuates tubular epithelial cell apoptosis after renal ischemia-reperfusion injury by blocking the ATM/P53-mediated DNA damage response[J]. Oxid Med Cell Longev, 2022, 2022(1): 4201287-4201298. DOI:10.1155/2022/4201287 |
| [12] |
YANG B, WANG G, LI Y, et al. Hydroxycitric acid prevents hype-roxaluric-induced nephrolithiasis and oxidative stress via activation of the Nrf2/Keap1 signaling pathway[J]. Cell Cycle, 2023, 22(17): 1884-1899. DOI:10.1080/15384101.2023.2247251 |
| [13] |
WANG J, MA R, WANG Y, et al. rhMYDGF alleviates I/R-induced kidney injury by inhibiting inflammation and apoptosis via the Akt pathway[J]. Transplantation, 2023, 107(8): 1729-1739. DOI:10.1097/tp.0000000000004497 |
| [14] |
ZHAI MY, HAN MM, HUANG X, et al. Dexmedetomidine protects human renal tubular epithelial HK-2 cells against hypoxia/reoxyge-nation injury by inactivating endoplasmic reticulum stress pathway[J]. Cell J, 2021, 23(4): 457-464. DOI:10.22074/cellj.2021.7220 |
| [15] |
ZHANG BH, LIU H, YUAN Y, et al. Knockdown of TRIM8 protects HK-2 cells against hypoxia/reoxygenation-induced injury by inhibi-ting oxidative stress-mediated apoptosis and pyroptosis via PI3K/Akt signal pathway[J]. Drug Des Devel Ther, 2021, 15(1): 4973-4983. DOI:10.2147/DDDT.S333372 |
| [16] |
赵荫涛, 裴小鑫, 刘源, 等. 下调miR-488靶向促进Jag1表达抑制缺氧复氧心肌H9c2细胞损伤的机制研究[J]. 中华医学遗传学杂志, 2021, 38(12): 1199-1203. DOI:10.3760/cma.j.cn511374-20200915-00669 |
| [17] |
SUN C, SHI H, ZHAO X, et al. The activation of cGAS-STING in acute kidney injury[J]. J Inflamm Res, 2023, 16(1): 4461-4470. DOI:10.2147/jir.s423232 |
| [18] |
QI JY, LUO Q, ZHANG QY, et al. Yi-Shen-Xie-Zhuo formula allevia-tes cisplatin-induced AKI by regulating inflammation and apoptosis via the cGAS/STING pathway[J]. J Ethnopharmacol, 2023, 309(1): 116327-116337. DOI:10.1016/j.jep.2023.116327 |
 2025, Vol. 54
2025, Vol. 54