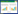文章信息
- 张凤英, 汤永红, 谢言青, 李敏, 蒋莉, 吴娜, 潘兆, 唐颖峰, 袁玲, 洪嫄媛, 刘会, 张平
- ZHANG Fengying, TANG Yonghong, XIE Yanqing, LI Min, JIANG Li, WU Na, PAN Zhao, TANG Yingfeng, YUAN Ling, HONG Yuanyuan, LIU Hui, ZHANG Ping
- 过表达Slc1a2调节Glu/GABA平衡抑制铁死亡改善睡眠剥夺小鼠的认知功能障碍
- Overexpression of Slc1a2 regulates Glu/GABA balance, inhibits ferroptosis and improves cognitive dysfunction in sleep-deprived mice
- 中国医科大学学报, 2025, 54(11): 967-976
- Journal of China Medical University, 2025, 54(11): 967-976
-
文章历史
- 收稿日期:2024-11-27
- 网络出版时间:2025-11-21 13:03:52
2. 衡阳市新安康复医院内科, 湖南 衡阳 421001
2. Department of Internal Medicine, Hengyang Xin'an Rehabilitation Hospital, Hengyang 421001, China
目前,睡眠障碍是普遍存在的问题[1],其中受睡眠剥夺(sleep deprivation,SD)影响的人数约占1/3[2]。短期SD可造成情绪压力、注意力持续时间缩短、学习和记忆障碍以及白天嗜睡增加等问题,而长期SD会显著增加心脏病、高血压、肥胖症、脑卒中、糖尿病和阿尔茨海默病等疾病的风险[3]。研究[4]表明,SD可导致认知功能下降和情绪调节中断。前额叶皮层(prefrontal cortex,PFC)是调节工作记忆、注意力和推理等高级大脑功能的关键区域,其功能和结构的完整性对维持神经心理活动至关重要[5-6]。此外,PFC功能与睡眠质量密切相关,睡眠质量改变可能直接影响其功能[7-8]。然而,SD影响PFC功能的具体机制尚不完全清楚。谷氨酸(glutamate,Glu)是主要的兴奋性神经递质,而γ-氨基丁酸(gamma-aminobutyric acid,GABA)是主要的抑制性神经递质,它们之间的平衡破坏会导致神经发育障碍、神经变性、神经疾病以及急性神经障碍[9]。研究[10]表明,SD导致Glu/GABA代谢失衡,从而影响神经元信号传导和整体大脑功能。Slc1a2基因编码参与Glu转运的兴奋性氨基酸转运蛋白2(excitatory amino acid transporter 2,EAAT2)的表达,其作为一种主要在星形胶质细胞中表达的高效Glu转运蛋白,可调节Glu水平以防止其过度积累引起神经毒性[11-13]。研究[14]表明,Slc1a2在SD大鼠PFC中表达下调,这可能导致Glu/GABA循环紊乱。近年来研究[15]发现睡眠不足导致的认知功能障碍与大脑中铁沉积的增加有关,此外Glu过度积累可导致细胞内谷胱甘肽耗尽并诱导铁死亡[16]。本研究探讨过表达Slc1a2对SD小鼠睡眠时间、认知功能以及PFC中Glu/GABA代谢、GABA能神经元功能和铁死亡的影响,旨在为Slc1a2作为睡眠障碍治疗的潜在靶点提供理论基础。
1 材料与方法 1.1 实验动物与分组130只8周龄SPF级雄性C57BL/6J小鼠购自北京Vital River实验动物技术有限公司,体重20~25 g,于60%~65%湿度、22~25 ℃、12 h昼夜光照循环的环境下饲养,自由摄食饮水。按照随机数字表法将小鼠分为正常睡眠(NS)组、正常睡眠+过表达Slc1a2组(NS+ov-Slc1a2组)、SD组、SD+过表达对照组(SD+ov-NC组)和SD+过表达Slc1a2组(SD+ov-Slc1a2组),每组26只。本研究获得中国医科大学实验动物福利与伦理委员会批准(CMUKT2024130)。
1.2 主要试剂及仪器腺病毒(上海吉凯基因医学科技股份有限公司),异氟烷(美国Sigma-Aldrich公司),多聚甲醛(上海碧云天生物技术股份有限公司),免疫荧光和Western blotting检测使用的抗体以及铁水平检测试剂盒(英国abcam公司),TRIzol(美国Invitrogen公司),第一链cDNA合成试剂盒(上海碧云天生物技术股份有限公司),实时定量PCR试剂盒(南京诺唯赞生物科技股份有限公司),C11-BODIPY探针(美国Sigma-Aldrich公司),丙二醛(malondialdehyde,MDA)水平检测试剂盒(武汉云克隆科技股份有限公司),Medusa小动物EP记录系统[易格生物科技(南京)有限公司],Lunion Stage自动睡眠评分软件(上海麓联智能科技有限公司),XR-XS108型自动化系统(上海鑫软计算机技术有限公司),CFX96实时荧光定量PCR系统(美国Bio-Rad公司),Image Quant LAS 4000C凝胶成像系统(美国GE公司)。
1.3 方法 1.3.1 SD模型制备使用基于旋转杆的自动化系统制备SD小鼠模型[17]。将小鼠置于可自由摄食的笼子中,笼子底部的杆顺时针和逆时针旋转。顺时针和逆时针各3圈交替进行,每旋转6圈暂停18 s使小鼠摄食饮水。每日杆以5 r/min的速度从20:00旋转至次日16:00以防止小鼠睡眠,持续7 d [17]。
1.3.2 小鼠脑内腺病毒注射小鼠腹腔注射戊巴比妥钠(45 mg/kg)麻醉后固定于脑立体定位仪上。暴露头骨,使用微量注射器将5 μL过表达对照或过表达Slc1a2腺病毒(4×1010 PFU/mL,上海吉凯基因医学科技股份有限公司)注射到小鼠双侧PFC中(前囟前1.8 mm,中线向左或右0.3 mm,深度2.5 mm),注射速度为0.2 μL/min,注射后将注射器再固定10 min防止拔出时回流[18]。
1.3.3 各组小鼠处理方法SD组、SD+ov-NC组和SD+ov-Slc1a2组小鼠按1.3.1方法建立SD模型,NS组和NS+ov-Slc1a2组小鼠仅置于装置中而不进行SD。NS+ov-Slc1a2组、SD+ov-NC组和SD+ov-Slc1a2组于SD模型建立前7 d按1.3.2方法进行腺病毒注射,1次/d,持续7 d。SD造模结束后,每组随机选取10只小鼠进行睡眠时间检测。每组余下的16只小鼠中随机选择10只依次进行旷场试验、Y迷宫实验和新物体识别测试。行为学检测结束后,每组16只小鼠中随机选取10只小鼠用于全细胞膜片钳记录,随机选取3只小鼠灌注取脑用于免疫荧光检测,随机选取3只小鼠收集PFC组织进行Western blotting、实时定量PCR和铁死亡检测。
1.3.4 睡眠时间检测将小鼠用2.5%(质量分数)异氟烷麻醉后置于脑立体定位仪中,将4根电极线分别插入左额叶(前囟前3.0 mm,中线向左0.8 mm,深度3.5 mm)、右额叶(前囟前3.0 mm,中线向右0.8 mm,深度3.5 mm)、左顶叶(前囟后2.0 mm,中线向左2.5 mm,深度1.8 mm)和右顶叶(前囟后2.0 mm,中线向右2.5 mm,深度1.8 mm)记录脑电图,将1对绝缘电极插入颈部肌肉记录肌电图,使用Medusa小动物EP记录系统以1 000 Hz的采样率记录脑和肌肉电活动。使用Lunion Stage自动睡眠评分软件将睡眠状态分为清醒、快速眼动(rapid eye movement,REM)睡眠和非快速眼动(non-rapid eye movement,NREM)睡眠。清醒的特征是脑电图振幅低、频率高的同时肌肉张力升高。REM睡眠通过脑电图中的高θ(6~10 Hz)功率和肌肉力量的缺失识别。NREM睡眠的特征是脑电图显示高电压和低频(1~4 Hz)信号同时肌电图活动减少[19]。在12 h内记录各组小鼠每小时的清醒情况,并统计每隔3 h的NREM睡眠时间、REM睡眠时间和觉醒时间。
1.3.5 行为学检测在旷场实验中,将小鼠放置在1个分为4个象限的方形竞技场中,在标准照明下评估它们在中心区域的运动和时间,持续10 min[20],记录运动的总距离和中心区域停留时间。Y迷宫实验涉及3只手臂,在训练阶段阻断1只手臂,小鼠可以探索2只张开的手臂10 min,1 h后打开阻断的手臂并让小鼠探索所有手臂5 min以检验他们对新打开手臂的偏好[21],记录小鼠的平均速度并计算新臂选择指数:新臂选择指数(%)=在新臂中时间/(在新臂中时间+在旧臂中时间)×100。在新物体识别测试中,小鼠首先适应包含2个相同物体的装置5 min,间隔2 h后用新物体替换其中1个旧物体,并在5 min内检测小鼠对每个物体的探索以计算识别指数,即在新对象中花费的时间与测试总持续时间的比值[19]
1.3.6 全细胞膜片钳记录小鼠用异氟烷深入麻醉后断头取脑,将大脑浸入预冷的充氧解剖液中,用冷冻切片机将大脑切成厚300 μm的切片。将切片转移到人工脑脊液中,30 ℃孵育1 h。在持续灌流人工脑脊液、31 ℃以及前置电压-70 mV条件下记录微型兴奋性突触后电流(miniature excitatory postsynaptic currentsm,mEPSC)。
1.3.7 免疫荧光检测小鼠异氟烷麻醉后依次用生理盐水和4%(质量分数)多聚甲醛灌注,提取完整大脑组织并在4%多聚甲醛中固定48 h,用30%(质量分数)蔗糖溶液脱水后切割成40 µm的脑切片。用含0.01%(体积分数)Triton X-100和10%(体积分数)山羊血清的PBS封闭后,Slc1a2、胶原纤维酸性蛋白(glial fibrillary acidic protein,GFAP)和GABA一抗4 ℃孵育切片过夜。PBS洗涤切片3次,于室温下用荧光二抗孵育1 h,DAPI染色液复染细胞核,于荧光显微镜下观察并拍照。计算Slc1a2+GFAP+细胞比例和GABA阳性细胞率。Slc1a2+GFAP+细胞比例(%)= Slc1a2阳性细胞数/GFAP阳性细胞数×100;GABA阳性细胞率(%)= GABA阳性细胞数/DAPI阳性细胞数×100。
1.3.8 Western blotting检测用含1%(体积分数)PMSF的RIPA裂解液裂解PFC组织样本,BCA法检测提取蛋白的浓度,将蛋白样本加入SDS-聚丙烯酰胺凝胶并进行电泳分离,将凝胶上分离的蛋白转移到PVDF膜上。5%(体积分数)脱脂牛奶室温封闭1 h,Slc1a2和α-tubulin一抗4 ℃孵育过夜,TBST洗膜后室温下用二抗孵育1 h,TBST洗去多余抗体并进行ECL发光成像,使用ImageJ软件定量分析Slc1a2蛋白的相对表达。
1.3.9 实时定量PCR检测用TRIzol法提取PFC组织总RNA,使用第一链cDNA合成试剂盒将RNA转化为cDNA。使用实时定量PCR试剂盒进行检测。以α-tubulin为内参,采用2-ΔΔCt法计算目的基因Glul、Slc6a1和Abat mRNA的相对表达量。
1.3.10 铁死亡检测收集的新鲜PFC组织切成厚10 μm的冰冻切片后用C11-BODIPY探针孵育,DAPI染核后于荧光显微镜下观察并拍照。ImageJ软件计算荧光强度,活性氧(reactive oxygen species,ROS)水平为绿色荧光强度与红色荧光强度的比值。Fe2+和MDA水平的检测严格按照相关试剂盒说明书进行操作。
1.4 统计学分析采用GraphPad Prism 8.0处理数据。符合正态分布的计量资料以x±s表示,2组间比较采用t检验,多组间比较采用单因素方差分析,两两比较采用LSD-t检验(方差齐)和Dunnett T3检验(方差不齐)。P < 0.05为差异有统计学意义。
2 结果 2.1 SD模型评估频谱分析结果显示,SD组小鼠相较于NC组表现出易醒性以及REM和NREM睡眠时间减少,表明SD模型建立成功。见图 1。
|
| A, NS group; B, SD group. EEG, electroencephalogram; EMG, electromyography; WEAK, awakening; NREM, non-rapid eye movement; REM, rapid eye movement. 图 1 小鼠脑电图、肌电图和睡眠状态分析 Fig.1 Analysis of electroencephalograms, electromyograms, and sleep status in mice |
2.2 各组小鼠PFC中Slc1a2表达情况
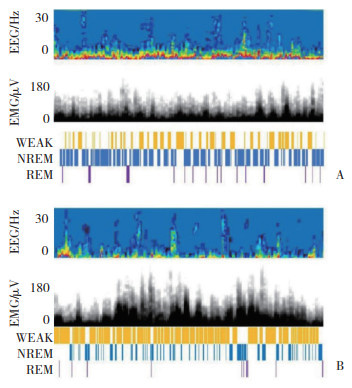
结果显示,与NS组相比,NS+ov-Slc1a2组小鼠PFC中Slc1a2+GFAP+细胞比例升高(P < 0.05),SD组和SD+ov-NC组小鼠Slc1a2+GFAP+细胞比例降低(均P < 0.05)。与SD组和SD+ov-NC组相比,SD+ov-Slc1a2组小鼠Slc1a2+GFAP+细胞比例升高(均P < 0.05)。Western blotting检测结果显示,与NS组相比,NS+ov-Slc1a2组小鼠PFC中Slc1a2蛋白表达升高(P < 0.05),SD组和SD+ov-NC组小鼠PFC中Slc1a2蛋白表达降低(均P < 0.05)。与SD组和SD+ov-NC组相比,SD+ov-Slc1a2组小鼠PFC中Slc1a2蛋白表达升高(均P < 0.05)。见图 2、3,表 1。
|
| 图 2 免疫荧光检测各组小鼠PFC中Slc1a2+GFAP+细胞比例×200 Fig.2 Proportion of Slc1a2+GFAP+ cells in the PFC of mice in each group by immunofluorescence ×200 |

|
| 图 3 Western blotting检测各组小鼠PFC中Slc1a2蛋白表达 Fig.3 Slc1a2 protein expression in the PFC of mice in each group by Western blotting |
| Group | n | Slc1a2+GFAP+ cell (%) | Slc1a2 protein |
| NS | 3 | 44.03±4.60 | 0.52±0.04 |
| NS+ov-Slc1a2 | 3 | 64.82±5.221) | 0.76±0.051) |
| SD | 3 | 5.37±0.761) | 0.15±0.021) |
| SD+ov-NC | 3 | 5.27±1.071) | 0.18±0.061) |
| SD+ov-Slc1a2 | 3 | 24.67±6.952),3) | 0.45±0.042),3) |
| F | 57.86 | 52.99 | |
| P | < 0.001 | < 0.001 | |
| 1) compared with the NS group, P < 0.05;2) compared with the SD group, P < 0.05;3) compared with the SD+ov-NC group, P < 0.05. | |||
2.3 各组小鼠睡眠时间比较
与NS组相比,NS+ov-Slc1a2组小鼠NREM和REM睡眠时间增加,觉醒时间减少(均P < 0.05),SD组和SD+ov-NC组小鼠NREM和REM睡眠时间减少,觉醒时间增加(均P < 0.05)。与SD组和SD+ov-NC组相比,SD+ov-Slc1a2组小鼠NREM和REM睡眠时间增加,觉醒时间减少(均P < 0.05)。见表 2。
| Group | n | NREM | REM | Awakening time |
| NS | 10 | 92.17±6.44 | 15.79±2.84 | 72.36±5.23 |
| NS+ov-Slc1a2 | 10 | 105.21±5.051) | 19.79±2.281) | 55.00±6.461) |
| SD | 10 | 50.38±7.051) | 7.35±1.921) | 102.74±5.541) |
| SD+ov-NC | 10 | 55.1±7.181) | 7.44±2.271) | 105.9±5.211) |
| SD+ov-Slc1a2 | 10 | 71.37±7.552),3) | 10.52±2.362),3) | 89.13±7.482),3) |
| F | 71.44 | 27.82 | 66.26 | |
| P | < 0.001 | < 0.001 | < 0.001 | |
| 1) compared with the NS group, P < 0.05;2) compared with the SD group, P < 0.05;3) compared with the SD+ov-NC group, P < 0.05. | ||||
2.4 各组小鼠Glu和GABA代谢酶表达情况
实时定量PCR检测结果显示,与NS组相比,NS+ov-Slc1a2组小鼠PFC中Glul、Slc6a1和Abat mRNA表达升高(均P < 0.05),SD组和SD+ov-NC组小鼠PFC中Glul、Slc6a1和Abat mRNA表达降低(均P < 0.05);与SD组和SD+ov-NC组相比,SD+ov-Slc1a2组小鼠PFC中Glul、Slc6a1和Abat mRNA表达升高(均P < 0.05)。见表 3。
| Group | n | Glul mRNA | Slc6a1 mRNA | Abat mRNA |
| NS | 3 | 1.01±0.05 | 0.98±0.05 | 1.01±0.08 |
| NS+ov-Slc1a2 | 3 | 2.48±0.271) | 2.82±0.611) | 1.75±0.261) |
| SD | 3 | 0.52±0.131) | 0.34±0.041) | 0.29±0.051) |
| SD+ov-NC | 3 | 0.52±0.091) | 0.35±0.031) | 0.30±0.081) |
| SD+ov-Slc1a2 | 3 | 0.65±0.112),3) | 0.52±0.062),3) | 0.59±0.062),3) |
| F | 56.67 | 166.10 | 241.80 | |
| P | < 0.001 | < 0.001 | < 0.001 | |
| 1) compared with the NS group, P < 0.05;2) compared with the SD group, P < 0.05;3) compared with the SD+ov-NC group, P < 0.05. | ||||
2.5 各组小鼠PFC中GABA能神经元功能变化
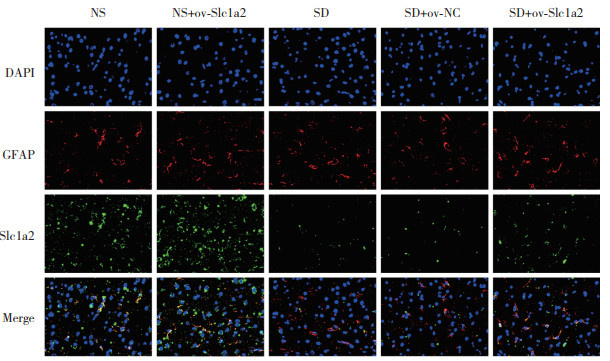
结果显示,与NS组相比,NS+ov-Slc1a2组小鼠PFC中mEPSC频率和振幅、GABA阳性细胞率升高(均P < 0.05),SD组和SD+ov-NC组小鼠PFC中mEPSC频率和振幅、GABA阳性细胞率降低(均P < 0.05);与SD组和SD+ov-NC组相比,SD+ov-Slc1a2组小鼠PFC中mEPSC频率和振幅、GABA阳性细胞率升高(均P < 0.05)。见图 4、表 4。
|
| 图 4 免疫荧光检测各组小鼠PFC中GABA阳性细胞×200 Fig.4 GABA positive cells in the PFC of mice in each group by immunofluorescence ×200 |
| Group | Frequency of mEPSC (Hz, n=10) | Amplitude of mEPSC (pA, n=10) | GABA-positive cells rate (%, n=3) |
| NS | 4.92±0.24 | 18.67±1.24 | 89.49±2.86 |
| NS+ov-Slc1a2 | 5.13±0.071) | 21.04±1.231) | 94.19±3.071) |
| SD | 2.11±0.361) | 10.61±1.301) | 12.36±2.51) |
| SD+ov-NC | 2.17±0.321) | 10.43±1.241) | 12.79±1.791) |
| SD+ov-Slc1a2 | 4.73±0.542),3) | 16.28±1.172),3) | 58.90±3.892),3) |
| F | 164.70 | 111.60 | 520.80 |
| P | < 0.001 | < 0.001 | < 0.001 |
| 1) compared with the NS group, P < 0.05;2) compared with the SD group, P < 0.05;3) compared with the SD+ov-NC group, P < 0.05. | |||
2.6 各组小鼠PFC中铁死亡比较
结果显示,与NS组相比,NS+ov-Slc1a2组小鼠PFC中脂质ROS、Fe2+和MDA水平降低(均P < 0.05),SD组和SD+ov-NC组小鼠PFC中脂质ROS、Fe2+和MDA水平升高(均P < 0.05)。与SD组和SD+ov-NC组相比,SD+ov-Slc1a2组小鼠PFC中脂质ROS、Fe2+和MDA水平降低(均P < 0.05)。见图 5、表 5。
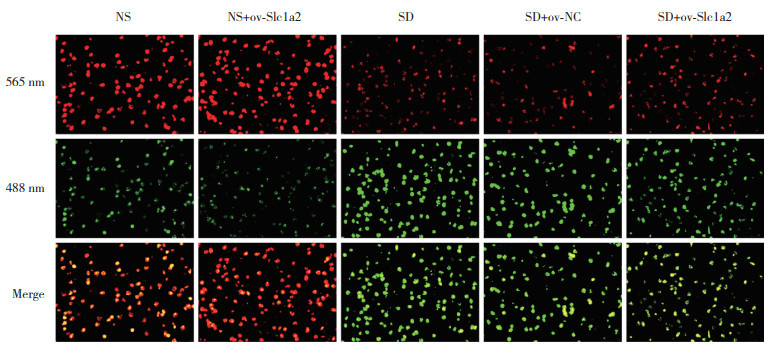
|
| 565 nm, excitation light is 565 nm, red; 488 nm, excitation light is 488 nm, green. 图 5 C11-BODIPY探针荧光染色检测各组小鼠PFC中脂质ROS阳性细胞表达×200 Fig.5 Expression of lipid ROS positive cells in the PFC of mice in each group by C11-BODIPY probe fluorescence staining ×200 |
| Group | n | ROS | Fe2+(μmol/g) | MDA(μmol/mg) |
| NS | 10 | 0.66±0.06 | 2.15±0.13 | 4.37±0.26 |
| NS+ov-Slc1a2 | 10 | 0.33±0.081) | 1.80±0.121) | 3.57±0.281) |
| SD | 10 | 1.91±0.131) | 4.78±0.181) | 8.12±0.441) |
| SD+ov-NC | 10 | 1.99±0.131) | 4.84±0.141) | 8.46±0.321) |
| SD+ov-Slc1a2 | 10 | 1.41±0.082),3) | 3.20±0.172),3) | 6.83±0.352),3) |
| F | 74.39 | 918.60 | 430.50 | |
| P | < 0.001 | < 0.001 | < 0.001 | |
| 1) compared with the NS group, P < 0.05;2) compared with the SD group, P < 0.05;3) compared with the SD+ov-NC group, P < 0.05. | ||||
2.7 各组小鼠认知功能比较
旷场实验结果显示,与NS组相比,NS+ov-Slc1a2组小鼠中心区域停留时间增加(P < 0.05),SD组和SD+ov-NC组小鼠中心区域停留时间减少(均P < 0.05)。与SD组和SD+ov-NC组相比,SD+ov-Slc1a2组小鼠中心区域停留时间增加(均P < 0.05)。各组小鼠总距离比较无统计学差异(均P > 0.05)。新物体识别测试结果显示,与NS组相比,NS+ov-Slc1a2组小鼠识别指数增加(P < 0.05),SD组和SD+ov-NC组小鼠识别指数减少(均P < 0.05)。与SD组和SD+ov-NC组相比,SD+ov-Slc1a2组小鼠识别指数增加(均P < 0.05)。Y迷宫实验结果显示,与NS组相比,NS+ov-Slc1a2组小鼠新臂选择指数增加(P < 0.05),SD组和SD+ov-NC组小鼠新臂选择指数减少(均P < 0.05);与SD组和SD+ov-NC组相比,SD+ov-Slc1a2组小鼠新臂选择指数增加(均P < 0.05)。各组小鼠平均速度比较无统计学差异(均P > 0.05)。见表 6。
| Group | n | Open field experiment | New object recognition test[recognition index (%)] | Y maze experiment | ||
| Total distance (m) | Stay time in the central area (s) | Average speed (cm/s) | Novel arm choice index (%) | |||
| NS | 10 | 17.03±1.87 | 56.50±5.52 | 59.87±3.10 | 16.73±1.16 | 50.86±5.46 |
| NS+ov-Slc1a2 | 10 | 17.93±0.91 | 61.90±2.511) | 64.01±2.811) | 16.88±0.50 | 64.22±4.311) |
| SD | 10 | 16.41±2.89 | 27.10±1.851) | 38.02±2.701) | 16.70±1.34 | 33.74±2.001) |
| SD+ov-NC | 10 | 16.24±2.27 | 27.80±1.691) | 37.00±3.211) | 16.50±1.33 | 32.31±3.101) |
| SD+ov-Slc1a2 | 10 | 15.81±3.02 | 44.30±3.402),3) | 53.35±4.472),3) | 17.17±1.59 | 45.41±2.672),3) |
| F | 0.39 | 165.90 | 107.20 | 0.43 | 64.38 | |
| P | 0.76 | < 0.001 | < 0.001 | 0.73 | < 0.001 | |
| 1) compared with the NS group, P < 0.05, 2) compared with the SD group, P < 0.05, 3) compared with the SD+ov-NC group, P < 0.05. | ||||||
3 讨论
SD会对个体的情绪和认知功能产生不利影响,目前临床治疗面临挑战[22-23]。Slc1a2是主要表达于星形胶质细胞质膜上的Glu转运体,主要发挥维持细胞外Glu浓度的作用,可缩短突触处的Glu瞬时现象,从而抑制突触和突触外Glu受体的激活并将兴奋性传递到睡眠和觉醒促进神经元,最终控制睡眠-觉醒行为[24]。研究[14]表明,Slc1a2在SD大鼠PFC中表达下调,且增强其表达可改善SD诱导的大鼠躁狂样行为。本研究使用基于螺旋杆的自动化系统建立小鼠SD模型,发现SD小鼠NREM和REM睡眠时间减少,觉醒时间增加,表明SD小鼠模型建立成功。本研究结果显示,SD模型小鼠PFC中Slc1a2+GFAP+细胞比例降低,且小鼠PFC中Slc1a2蛋白表达下调,与既往研究[14]结果一致。此外,本研究发现在SD小鼠PFC中过表达Slc1a2可增加NREM和REM睡眠时间,减少觉醒时间,表明PFC中异位过表达Slc1a2可改善SD小鼠睡眠障碍。
研究[25-26]表明,SD影响大脑内的神经活动,特别是改变PFC区域抑制控制和激活之间的平衡。Slc1a2在Glu转运和神经信号转导中发挥关键作用,而且由于Glu和GABA分别是主要的兴奋性和抑制性神经递质,Glu代谢和GABA合成之间的平衡对大脑功能至关重要[27]。Glul是氮代谢途径中的关键中心酶之一,其通过ATP依赖性反应催化Glu和铵转化为谷氨酰胺[28]。Slc6a1编码位于突触前神经元和神经胶质上的GABA转运体1,该转运体通过钠氯依赖的再摄取机制高效清除突触间释放的GABA[29]。GABA转氨酶Abat是参与GABA分解代谢的关键酶,位于线粒体基质内并催化GABA的氨基转移到α-酮戊二酸,形成琥珀酸半醛并随后转化为三羧酸循环中的关键中间体琥珀酸[30]。Glul、Slc6a1和Abat表达可间接反应Glu代谢和GABA合成之间的平衡。本研究发现SD小鼠PFC中Glul、Slc6a1和Abat mRNA表达下调,而在PFC中过表达Slc1a2可部分逆转SD小鼠PFC中Glul、Slc6a1和Abat mRNA表达,表明过表达Slc1a2可改善SD小鼠PFC中Glu/GABA代谢失衡。
GABA是神经系统中主要的抑制性神经递质,其活性导致神经元超极化,抑制放电并在神经网络中发挥抑制作用[31]。mEPSC对应于由单个Glu囊泡的突触前释放引起的反应,mEPSC振幅增加反映了突触后传递的加强,而频率增加反映了功能性突触数量或突触前释放概率的增加[32]。本研究发现SD小鼠PFC中mEPSC频率和振幅降低,而过表达Slc1a2可恢复SD小鼠PFC中mEPSC频率和振幅,表明过表达Slc1a2能改善SD小鼠PFC中的兴奋性神经信号传递。GABA能神经元对睡眠产生及其调节至关重要[33]。本研究发现SD小鼠PFC中GABA阳性细胞率降低,而过表达Slc1a2增加SD小鼠PFC中GABA阳性细胞率,表明过表达Slc1a2可能改善SD小鼠PFC中的GABA能神经元功能。铁死亡是铁和脂质氧化产物依赖性调节的一种细胞死亡形式,以脂质过氧化、抗氧化酶失活和铁稳态失衡为特征,并与多种脑部疾病有关[34]。谷胱甘肽消耗导致脂质过氧化物积累从而诱导铁死亡[35]。Glu过度积累会导致谷胱甘肽耗竭,进而诱导铁死亡[36]。本研究发现SD小鼠PFC中脂质ROS、Fe2+和MDA水平升高,而过表达Slc1a2降低SD小鼠PFC中脂质ROS、Fe2+和MDA水平,表明过表达Slc1a2可能改善SD小鼠PFC中的铁死亡。
研究[37]表明,SD影响大脑神经网络的结构和功能,动态降低信号处理速度,导致患者反应时间延长和精神状态降低,从而损害注意力、工作记忆、记忆巩固、警觉性、判断力、决策力等认知能力。本研究采用行为测试研究Slc1a2在SD诱导的认知障碍中的作用。旷场实验结果显示,过表达Slc1a2不影响SD小鼠的运动功能,但增加SD小鼠在中心区域的停留时间。此外,新物体识别测试结果显示,过表达Slc1a2增加SD小鼠的识别指数。这些结果表明过表达Slc1a2可能改善SD小鼠焦虑行为和认知能力。Y迷宫结果显示,PFC中过表达Slc1a2增加SD小鼠新臂选择指数,而对小鼠的平均速度无影响,表明过表达Slc1a2改善SD小鼠空间记忆损伤。
综上所述,在PFC中异位过表达Slc1a2能明显改善SD小鼠睡眠障碍,减轻SD对PFC中的兴奋性神经信号传递和GABA能神经元功能的破坏,并改善SD小鼠认知障碍和焦虑行为,其作用机制可能与改善PFC中Glu/GABA代谢失衡并抑制铁死亡有关。然而,Slc1a2不同亚型的作用、参与调节SD的Slc1a2相关的分子通路对神经递质的调节以及对SD诱导的认知和情绪问题的影响仍需进一步研究。
| [1] |
VGONTZAS A, PAVLOVIĆ J, BERTISCH S. Sleep symptoms and disorders in episodic migraine: assessment and management[J]. Curr Pain Headache Rep, 2023, 27(10): 511-520. DOI:10.1007/s11916-023-01160-z |
| [2] |
LIU Y, WHEATON AG, CHAPMAN DP, et al. Prevalence of healthy sleep duration among adults: United States, 2014[J]. MMWR Morb Mortal Wkly Rep, 2016, 65(6): 137-141. DOI:10.15585/mmwr.mm6506a1 |
| [3] |
ESTRADA C, CUENCA L, CANO-FERNANDEZ L, et al. Voluntary exercise reduces plasma cortisol levels and improves transitory memo-ry impairment in young and aged Octodon degus[J]. Behav Brain Res, 2019, 373: 112066. DOI:10.1016/j.bbr.2019.112066 |
| [4] |
SICLARI F, VALLI K, ARNULF I. Dreams and nightmares in healthy adults and in patients with sleep and neurological disorders[J]. Lancet Neurol, 2020, 19(10): 849-859. DOI:10.1016/S1474-4422(20)30275-1 |
| [5] |
HISER J, KOENIGS M. The multifaceted role of the ventromedial prefrontal cortex in emotion, decision making, social cognition, and psychopathology[J]. Biol Psychiatry, 2018, 83(8): 638-647. DOI:10.1016/j.biopsych.2017.10.030 |
| [6] |
SHALLICE T, CIPOLOTTI L. The prefrontal cortex and neurological impairments of active thought[J]. Annu Rev Psychol, 2018, 69: 157-180. DOI:10.1146/annurev-psych-010416-044123 |
| [7] |
TOSSELL K, YU X, GIANNOS P, et al. Somatostatin neurons in prefrontal cortex initiate sleep-preparatory behavior and sleep via the preoptic and lateral hypothalamus[J]. Nat Neurosci, 2023, 26: 1805-1819. DOI:10.1038/s41593-023-01430-4 |
| [8] |
HONG J, LOZANO DE, BEIER KT, et al. Prefrontal cortical regulation of REM sleep[J]. Nat Neurosci, 2023, 26(10): 1820-1832. DOI:10.1038/s41593-023-01398-1 |
| [9] |
SEARS SM, HEWETT SJ. Influence of glutamate and GABA transport on brain excitatory/inhibitory balance[J]. Exp Biol Med (Maywood), 2021, 246(9): 1069-1083. DOI:10.1177/1535370221989263 |
| [10] |
YU X, LI W, MA Y, et al. GABA and glutamate neurons in the VTA regulate sleep and wakefulness[J]. Nat Neurosci, 2019, 22(1): 106-119. DOI:10.1038/s41593-018-0288-9 |
| [11] |
ZHOU Y, HASSEL B, EID T, et al. Axon-terminals expressing EAAT2 (GLT-1; Slc1a2) are common in the forebrain and not limited to the hippocampus[J]. Neurochem Int, 2019, 123: 101-113. DOI:10.1016/j.neuint.2018.03.006 |
| [12] |
PAJARILLO E, DIGMAN A, NYARKO-DANQUAH I, et al. Astrocytic transcription factor REST upregulates glutamate transporter EAAT2, protecting dopaminergic neurons from manganese-induced excitotoxicity[J]. J Biol Chem, 2021, 297(6): 101372. DOI:10.1016/j.jbc.2021.101372 |
| [13] |
JIANG LL, ZHU B, ZHAO YJ, et al. Membralin deficiency dysregu-lates astrocytic glutamate homeostasis leading to ALS-like impairment[J]. J Clin Invest, 2019, 129(8): 3103-3120. DOI:10.1172/JCI127695 |
| [14] |
LEEM KH, KIM SA, PARK HJ. Antimania-like effect of Panax ginseng regulating the glutamatergic neurotransmission in REM-sleep deprivation rats[J]. Biomed Res Int, 2020, 2020: 3636874. DOI:10.1155/2020/3636874 |
| [15] |
CHEN B, CHEN ZH, LIU MJ, et al. Inhibition of neuronal ferroptosis in the acute phase of intracerebral hemorrhage shows long-term cerebroprotective effects[J]. Brain Res Bull, 2019, 153: 122-132. DOI:10.1016/j.brainresbull.2019.08.013 |
| [16] |
JIANG T, CHENG H, SU JJ, et al. Gastrodin protects against glutamate-induced ferroptosis in HT-22 cells through Nrf2/HO-1 signaling pathway[J]. Toxicol In Vitro, 2020, 62: 104715. DOI:10.1016/j.tiv.2019.104715 |
| [17] |
ZHANG SY, ZHANG Y, ZHENG YG, et al. Dexmedetomidine attenuates sleep deprivation-induced inhibition of hippocampal neurogenesis via VEGF-VEGFR2 signaling and inhibits neuroinflammation[J]. Biomed Pharmacother, 2023, 165: 115085. DOI:10.1016/j.biopha.2023.115085 |
| [18] |
TENG SS, ZHEN FH, WANG L, et al. Control of non-REM sleep by ventrolateral medulla glutamatergic neurons projecting to the preo-ptic area[J]. Nat Commun, 2022, 13: 4748. DOI:10.1038/s41467-022-32461-3 |
| [19] |
ZHU JP, CHEN C, LI Z, et al. Overexpression of Sirt6 ameliorates sleep deprivation induced-cognitive impairment by modulating glutamatergic neuron function[J]. Neural Regen Res, 2023, 18(11): 2449-2458. DOI:10.4103/1673-5374.371370 |
| [20] |
ZAHRA A, JIANG JX, CHEN YN, et al. Memantine rescues prenatal citalopram exposure-induced striatal and social abnormalities in mice[J]. Exp Neurol, 2018, 307: 145-154. DOI:10.1016/j.expneurol.2018.06.003 |
| [21] |
SUN XY, LI LJ, DONG QX, et al. Rutin prevents tau pathology and neuroinflammation in a mouse model of Alzheimer's disease[J]. J Neuroinflammation, 2021, 18(1): 131. DOI:10.1186/s12974-021-02182-3 |
| [22] |
KRAUSE AJ, BEN SIMON E, MANDER BA, et al. The sleep-deprived human brain[J]. Nat Rev Neurosci, 2017, 18: 404-418. DOI:10.1038/nrn.2017.55 |
| [23] |
KANG ZM, LIN YQ, SU CS, et al. Hsp70 ameliorates sleep deprivation-induced anxiety-like behavior and cognitive impairment in mice[J]. Brain Res Bull, 2023, 204: 110791. DOI:10.1016/j.brainresbull.2023.110791 |
| [24] |
BRIGGS C, HIRASAWA M, SEMBA K. Sleep deprivation distinctly alters glutamate transporter 1 apposition and excitatory transmission to orexin and MCH neurons[J]. J Neurosci, 2018, 38(10): 2505-2518. DOI:10.1523/JNEUROSCI.2179-17.2018 |
| [25] |
ROSCHEL H, GUALANO B, OSTOJIC SM, et al. Creatine supplementation and brain health[J]. Nutrients, 2021, 13(2): 586. DOI:10.3390/nu13020586 |
| [26] |
ZHAO R, ZHANG XX, FEI NB, et al. Decreased cortical and subcortical response to inhibition control after sleep deprivation[J]. Brain Imag Behav, 2019, 13(3): 638-650. DOI:10.1007/s11682-018-9868-2 |
| [27] |
ANDERSEN JV, SCHOUSBOE A, VERKHRATSKY A. Astrocyte energy and neurotransmitter metabolism in Alzheimer's disease: integration of the glutamate/GABA-glutamine cycle[J]. Prog Neurobiol, 2022, 217: 102331. DOI:10.1016/j.pneurobio.2022.102331 |
| [28] |
WANG S, LIN RX, TUMUKUNDE E, et al. Glutamine synthetase contributes to the regulation of growth, conidiation, Sclerotia development, and resistance to oxidative stress in the fungus Aspergillus flavus[J]. Toxins, 2022, 14(12): 822. DOI:10.3390/toxins14120822 |
| [29] |
KASSABIAN B, FENGER CD, WILLEMS M, et al. Intrafamilial variability in SLC6A1-related neurodevelopmental disorders[J]. Front Neurosci, 2023, 17: 1219262. DOI:10.3389/fnins.2023.1219262 |
| [30] |
ZHANG MX, ZHONG HT, CAO T, et al. Gamma-aminobutyrate transaminase protects against lipid overload-triggered cardiac injury in mice[J]. Int J Mol Sci, 2022, 23(4): 2182. DOI:10.3390/ijms23042182 |
| [31] |
PATEL JC, SHERPA AD, MELANI R, et al. GABA co-released from striatal dopamine axons dampens phasic dopamine release through autoregulatory GABAA receptors[J]. Cell Rep, 2024, 43(3): 113834. DOI:10.1016/j.celrep.2024.113834 |
| [32] |
SAKIMOTO Y, SHINTANI A, YOSHIURA D, et al. A critical period for learning and plastic changes at hippocampal CA1 synapses[J]. Sci Rep, 2022, 12(1): 7199. DOI:10.1038/s41598-022-10453-z |
| [33] |
ZHAO QC, MACI M, MILLER MR, et al. Sleep restoration by optogenetic targeting of GABAergic neurons reprograms microglia and ameliorates pathological phenotypes in an Alzheimer's disease mo-del[J]. Mol Neurodegener, 2023, 18(1): 93. DOI:10.1186/s13024-023-00682-9 |
| [34] |
BAO WD, PANG P, ZHOU XT, et al. Loss of ferroportin induces memory impairment by promoting ferroptosis in Alzheimer's disease[J]. Cell Death Differ, 2021, 28(5): 1548-1562. DOI:10.1038/s41418-020-00685-9 |
| [35] |
YUAN M, WANG F, SUN TQ, et al. Vitamin B6 alleviates chronic sleep deprivation-induced hippocampal ferroptosis through CBS/GSH/GPX4 pathway[J]. Biomed Pharmacother, 2024, 174: 116547. DOI:10.1016/j.biopha.2024.116547 |
| [36] |
YANG SX, XIE ZP, PEI TT, et al. Salidroside attenuates neuronal ferroptosis by activating the Nrf2/HO1 signaling pathway in Aβ1-42-induced Alzheimer's disease mice and glutamate-injured HT22 cells[J]. Chin Med, 2022, 17(1): 82. DOI:10.1186/s13020-022-00634-3 |
| [37] |
KHAN MA, AL-JAHDALI H. The consequences of sleep deprivation on cognitive performance[J]. Neurosciences, 2023, 28(2): 91-99. DOI:10.17712/nsj.2023.2.20220108 |
 2025, Vol. 54
2025, Vol. 54