文章信息
- 吕品, 谭悦
- LÜ Pin, TAN Yue
- 抑郁症患者血清TBARS和8-OHdG水平及其与病情严重程度和认知功能的关系
- Serum levels of TBARS and 8-OHdG in patients with depression and their relationship with disease severity and cognitive function
- 中国医科大学学报, 2025, 54(11): 1000-1004
- Journal of China Medical University, 2025, 54(11): 1000-1004
-
文章历史
- 收稿日期:2024-12-31
- 网络出版时间:2025-11-21 13:20:04
抑郁症是精神障碍的一种,患者表现为持续性心情低落、兴趣减退,部分严重患者会出现自残、自杀行为。认知功能障碍是抑郁症患者的常见表现之一,严重影响生活质量[1]。目前,判断抑郁症严重程度以及是否伴随认知功能障碍多通过相应的评分量表,但其涉及范围广、评估流程复杂,部分病情严重的患者无法配合完成,导致漏诊率和误诊率较高[2]。研究[3]提示,氧化应激所致的DNA损伤是造成抑郁症和认知功能障碍发生的共同致病因素,机体氧化应激状态的改变可能用于评估抑郁症患者的病情严重程度和认知功能损害程度。硫代巴比妥酸反应物(thiobarbituric acid reactive substance,TBARS)可反映机体的氧化应激状态,研究[4]发现,其在多种精神疾病患者中呈异常表达。8-羟基脱氧鸟苷(8-hydroxy-2’-deoxyguanosine,8-OHdG)是过量活性氧引起的DNA氧化损伤中最常见的稳定产物,是评估氧化应激致DNA损伤的高效指标[5]。由此推测,TBARS和8-OHdG可能参与了抑郁症和认知功能障碍的发生、发展,但目前相关研究较少。因此,本研究分析了血清TBARS和8-OHdG水平与抑郁症患者病情严重程度和认知功能障碍的关系,为该病的临床诊断提供参考。
1 材料与方法 1.1 研究对象选择2022年6月至2024年6月间我院心理科和精神科收治的150例抑郁症患者为研究对象。其中,男62例,女88例;年龄25~64岁,平均(44.13±5.28)岁。纳入标准:(1)符合抑郁症诊断标准[6],首次确诊;(2)24项汉密尔顿抑郁量表[7](24-item Hamilton Rating Scale for Depression,HAMD-24)评分 > 7分;(3)年龄≥18岁;(4)患者及其家属知晓研究内容并自愿入组。排除标准:(1)依从性差;(2)合并其他严重疾病或精神相关疾病;(3)先天精神发育迟滞;(4)处于妊娠期或哺乳期。另选择同期于我院体检的75例健康志愿者作为对照。其中,男30例,女45例;年龄24~65岁,平均(44.39±5.60)岁。所有志愿者知晓研究内容并自愿入组。2组比较,年龄、性别等均无统计学差异(P > 0.05)。本研究经我院伦理委员会审核批准[(2021)医研伦审第(017)号]。
1.2 血清TBARS和8-OHdG水平检测抑郁症患者于入组次日,健康志愿者于体检当日,采集血样,常规制备血清,于-80 ℃保存。采用酶联免疫吸附试验检测血清TBARS和8-OHdG水平,人TBARS和8-OHdG检测试剂盒均购自江西江蓝纯生物试剂有限公司。
1.3 抑郁症病情严重程度评估和分组由经过培训的精神科医生采用HAMD-24评分评估抑郁症严重程度,分为轻度抑郁症组(HAMD-24评分7~20分,53例)、中度抑郁症组(HAMD-24评分 > 20~35分,62例)和重度抑郁症组(HAMD-24评分 > 35分,35例)。
1.4 认知功能评分和分组由经过培训的精神科医生采用蒙特利尔认知评估量表[8](Montreal Cognitive Assessment Scale,MoCA)评分评估认知功能,分为认知功能正常组(MoCA评分≥26分,83例)和认知功能障碍组(MoCA评分 < 26分,67例)。
1.5 统计学分析采用SPSS 27.0软件分析数据。计量资料符合正态分布,以x±s表示,采用t检验和单因素方差分析进行比较。计数资料以率(%)表示,采用χ2检验进行比较。采用Spearman相关分析法分析抑郁症患者血清TBARS和8-OHdG水平与抑郁症严重程度的相关性,采用Pearson相关分析法分析抑郁症患者血清TBARS和8-OHdG水平与MoCA评分的相关性,采用受试者操作特征(receiver operating characteristic,ROC)曲线评估血清TBARS和8-OHdG诊断抑郁症患者伴发认知功能障碍的价值。P < 0.05为差异有统计学意义。
2 结果 2.1 抑郁症患者与健康志愿者血清TBARS和8-OHdG水平的比较抑郁症患者和健康志愿者血清TBARS水平分别为(9.31±1.44)μmol/L和(6.89±1.12)μmol/L,8-OHdG水平分别为(55.94±9.01)ng/L和(27.82±7.63)ng/L。与健康志愿者相比,抑郁症患者血清TBARS和8-OHdG水平均明显升高(分别为t = 12.748,P < 0.05;t = 23.184,P < 0.05)。
2.2 不同病情严重程度的抑郁症患者血清TBARS和8-OHdG水平的比较轻、中、重度抑郁症组患者血清TBARS水平分别为(8.11±1.30)、(9.36±1.42)和(11.04±1.69)μmol/L,8-OHdG水平分别为(45.80±8.02)、(58.54±9.11)和(66.71±10.84)ng/L。重度抑郁症组患者血清TBARS和8-OHdG水平明显高于轻度和中度抑郁症组(均P < 0.05),中度抑郁症组患者血清TBARS和8-OHdG水平明显高于轻度抑郁症组(P < 0.05)。
2.3 抑郁症患者血清TBARS和8-OHdG水平与病情严重程度的相关性Spearman相关分析结果显示,抑郁症患者血清TBARS和8-OHdG水平均与抑郁症严重程度呈正相关(分别为r = 0.615,P < 0.001;r = 0.709,P < 0.001)。
2.4 不同认知功能的抑郁症患者基线资料的比较比较认知功能正常组和认知功能障碍组患者的基线资料,结果显示,认知功能障碍组患者的年龄大于认知功能正常组,高中及高中以上学历的患者比例低于认知功能正常组(P < 0.05)。见表 1。
| Item | n | Cognitive normal group(n = 83) | Cognitive dysfunction group(n = 67) | χ2/t | P |
| Age(year) | 150 | 42.21±5.11 | 46.50±5.94 | 4.753 | < 0.001 |
| Body mass index(kg/m2) | 150 | 23.28±3.49 | 22.91±3.52 | 0.643 | 0.521 |
| Sex [n(%)] | 0.592 | 0.442 | |||
| Male | 62 | 32(38.55) | 30(44.78) | ||
| Female | 88 | 51(61.45) | 37(55.22) | ||
| Degree of education [n(%)] | 5.628 | 0.018 | |||
| Below high school | 56 | 24(28.92) | 32(47.76) | ||
| High school and above | 94 | 59(71.08) | 35(52.24) | ||
| Marital status [n(%)] | 1.720 | 0.423 | |||
| Unmarried | 61 | 33(39.76) | 28(41.79) | ||
| Married | 59 | 36(43.37) | 23(34.33) | ||
| Divorced/widowed | 30 | 14(16.87) | 16(23.88) | ||
| State of the economy [n(%)] | 3.264 | 0.071 | |||
| Good | 74 | 46(55.42) | 28(41.79) | ||
| Worse off | 76 | 37(44.58) | 39(58.21) | ||
| History of smoking [n(%)] | 0.516 | 0.473 | |||
| Yes | 69 | 36(43.37) | 33(49.25) | ||
| No | 81 | 47(56.63) | 34(50.75) | ||
| History of alcohol consumption [n(%)] | 1.650 | 0.199 | |||
| Yes | 63 | 31(37.35) | 32(47.76) | ||
| No | 87 | 52(62.65) | 35(52.24) |
2.5 不同认知功能的抑郁症患者MoCA评分以及血清TBARS和8-OHdG水平的比较
认知功能正常组和认知功能障碍组患者MoCA评分分别为(28.01±1.79)和(21.47±3.64)分,血清TBARS水平分别为(8.43±1.37)和(10.40±1.68)μmol/L,血清8-OHdG水平分别为(51.27±8.98)和(61.73±9.46)ng/L。与认知功能正常组相比,认知功能障碍组症患者MoCA评分明显降低,血清TBARS和8-OHdG水平明显升高(P < 0.05)。
2.6 抑郁症患者血清TBARS和8-OHdG水平与MoCA评分的相关性Pearson相关分析结果显示,抑郁症患者血清TBARS和8-OHdG水平均与MoCA评分呈负相关(分别为r = -0.350,P < 0.001;r = -0.414,P < 0.001)。
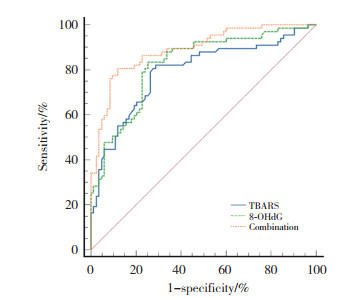
2.7 血清TBARS和8-OHdG水平对抑郁症患者认知功能障碍的诊断价值以患者血清TBARS和8-OHdG水平为检验变量,绘制其诊断抑郁症患者伴发认知功能障碍的ROC曲线。结果显示,患者血清TBARS和8-OHdG单独诊断的曲线下面积(area under the curve,AUC)分别为0.791(95%CI:0.717~0.853)和0.825(95%CI:0.754~0.882),截断值分别为9.42 μmol/L和57.97 ng/L。二者联合诊断时,AUC为0.891(95%CI:0.829~0.936),明显优于血清TBARS和8-OHdG单独诊断(分别为Z二者联合-TBARS=2.911,P = 0.004;Z二者联合-8-OHdG=2.575,P = 0.010)。见表 2、图 1。
| Indicator | AUC | Cutoff value | 95%CI | Sensitivity(%) | Specificity(%) | Youden index |
| TBARS | 0.791 | 9.42 μmol/L | 0.717-0.853 | 82.09 | 71.08 | 0.532 |
| 8-OHdG | 0.825 | 57.97 ng/L | 0.754-0.882 | 83.58 | 74.70 | 0.583 |
| Combination | 0.891 | - | 0.829-0.936 | 80.60 | 87.95 | 0.686 |
|
| 图 1 血清TBARS和8-OHdG水平诊断抑郁症患者认知功能障碍的ROC曲线 Fig.1 ROC curve of serum TBARS and 8-OHdG levels for diagnosing cognitive dysfunction in patients with depression |
3 讨论
抑郁症是一种常见的精神疾病。认知功能障碍是抑郁症常见的并发症,并且随着抑郁症严重程度的增加,患者认知功能损害更加严重,会对患者的语言能力、执行功能等多方面产生损害,还会进一步加重患者的抑郁和焦虑情绪,对患者的日常工作和生活造成严重的影响[2]。
TBARS是脂质过氧化形成的产物,可表征由过量氧自由基造成的细胞膜损伤程度,已被广泛用于指示机体的氧化应激状态[9]。研究[10]指出,精神分裂症伴抑郁症患者血清TBARS水平明显升高,TBARS水平与病情严重程度以及是否合并抑郁症明显相关,可作为早期诊断精神分裂症伴抑郁症的生物学指标。8-OHdG为活性氧诱导的鸟嘌呤碱基损伤,在碱基第8位添加1个羟基形成,其通常作为DNA氧化损伤和氧化应激程度的指标[11]。研究显示,8-OHdG水平升高可激活神经细胞的氧化应激反应,抑制其正常的生物学功能,加重神经元损伤,是缺血性脑卒中患者脑卒中后抑郁的独立危险因素,其水平变化可反映患者抑郁的严重程度。有研究[12]构建了约束应激小鼠模型,通过用药阻止8-OHdG水平升高后,小鼠因应激诱导的抑郁样行为明显改善。本研究结果显示,抑郁症患者血清TBARS和8-OHdG水平明显升高,且不同病情严重程度的患者血清TBARS和8-OHdG水平有统计学差异,提示了TBARS和8-OHdG水平与抑郁症的发生和病情恶化明显相关,可能作为反映抑郁症病情进展的血清学指标。
认知功能障碍的发生与年龄、遗传、慢性疾病等多种因素有关。目前,抑郁症患者易并发认知功能障碍的生物学机制尚未完全明确。研究[13]显示,机体的氧化应激损伤是导致抑郁症患者认知功能损害的重要影响因素。TBARS和8-OHdG作为反映氧化应激程度的指标,其水平变化同样与认知功能障碍的发生密切相关。有研究[14]构建了认知功能损害大鼠模型,发现其突触体组分中TBARS水平明显升高,且随着损伤时间的增加而逐渐升高。研究[15]显示,轻度认知障碍患者和阿尔茨海默病患者血清8-OHdG水平呈明显升高趋势,且8-OHdG水平与MoCA评分呈明显负相关。本研究结果与上述研究结果相似。本研究中,抑郁症伴发认知功能障碍的患者血清TBARS和8-OHdG水平明显高于抑郁症未并发认知功能障碍的患者,相关性分析结果显示,抑郁症患者血清TBARS和8-OHdG水平均与MoCA评分呈负相关,提示血清TBARS和8-OHdG水平变化可反映抑郁症患者的认知功能损害程度。ROC曲线分析结果显示,血清TBARS和8-OHdG水平诊断抑郁症患者认知功能障碍的AUC分别为0.791和0.825,提示二者可能作为临床诊断抑郁症患者是否伴发认知功能障碍的辅助指标。当二者联合诊断时,AUC为0.891,特异度为87.95%,较二者单独诊断效能更高。本研究明确了血清TBARS和8-OHdG水平诊断抑郁症患者认知功能障碍的截断值,当血清TBARS水平高于9.42 μmol/L或8-OHdG水平高于57.97 ng/L时,患者伴发认知功能障碍的风险较高,临床上应当多加关注,及时调整治疗方案。此外,本研究发现,认知功能正常组与认知功能障碍组患者比较,年龄和文化程度有统计学差异,但本研究未评估年龄和文化程度对患者认知功能的影响。后续将进行进一步的分析,深入评估二者对抑郁症患者的影响。
综上所述,抑郁症患者血清TBARS和8-OHdG水平均明显升高,其与患者的病情严重程度和认知功能密切相关,检测二者血清水平对判断病情进展以及是否并发认知功能障碍有一定的临床应用价值。但本研究样本量较小,结果可能有一定的偏倚。后续将扩大样本量,通过多中心研究进行验证。同时深入分析TBARS和8-OHdG参与抑郁症发生和认知障碍的机制,为临床抑郁症和认知功能障碍的诊疗提供依据。
| [1] |
黄颖林, 孟祥伟, 张改娟, 等. 新疆塔城地区抑郁症住院患者的认知功能状况及其影响因素[J]. 中国医科大学学报, 2021, 50(4): 341-344. DOI:10.12007/j.issn.0258-4646.2021.04.011 |
| [2] |
黄颖林, 孟祥伟, 张珍珍, 等. 认知功能对抑郁症住院患者治疗反应的预测[J]. 中国医科大学学报, 2021, 50(5): 439-443. DOI:10.12007/j.issn.0258-4646.2021.05.011 |
| [3] |
TONG T, HAO C, SHEN J, et al. Electroacupuncture ameliorates chronic unpredictable mild stress-induced depression-like behavior and cognitive impairment through suppressing oxidative stress and neuroinflammation in rats[J]. Brain Res Bull, 2024, 206: 110838. DOI:10.1016/j.brainresbull.2023.110838 |
| [4] |
SOARES FAC, FILHO NAK, BERETTA BFS, et al. Thiobarbituric acid reactive substances in dogs with spontaneous hypercortisolism[J]. Domest Anim Endocrinol, 2021, 77: 106634. DOI:10.1016/j.domaniend.2021.106634 |
| [5] |
GORIUC A, COJOCARU KA, LUCHIAN I, et al. Using 8-hydroxy- 2'-deoxiguanosine (8-OHdG) as a reliable biomarker for assessing periodontal disease associated with diabetes[J]. Int J Mol Sci, 2024, 25(3): 1425. DOI:10.3390/ijms25031425 |
| [6] |
中华医学会, 中华医学会杂志社, 中华医学会全科医学分会, 等. 抑郁症基层诊疗指南(2021年)[J]. 中华全科医师杂志, 2021, 20(12): 1249-1260. DOI:10.3760/cma.j.cn114798-20211020-00778 |
| [7] |
LIN CH, PARK C, MCINTYRE RS. Early improvement in HAMD-17 and HAMD-7 scores predict response and remission in depressed patients treated with fluoxetine or electroconvulsive therapy[J]. J Affect Disord, 2019, 253: 154-161. DOI:10.1016/j.jad.2019.04.082 |
| [8] |
BRUIJNEN CJWH, DIJKSTRA BAG, WALVOORT SJW, et al. Psychometric properties of the Montreal Cognitive Assessment (MoCA) in healthy participants aged 18-70[J]. Int J Psychiatry Clin Pract, 2020, 24(3): 293-300. DOI:10.1080/13651501.2020.1746348 |
| [9] |
HSIAO SY, KUNG CT, SU CM, et al. Impact of oxidative stress on treatment outcomes in adult patients with sepsis: a prospective study[J]. Medicine, 2020, 99(26): e20872. DOI:10.1097/MD.0000000000020872 |
| [10] |
王津鑫, 陈衍华, 王娟. 精神分裂症伴抑郁症患者血清iNOS、TBARS水平及其与疾病严重程度的相关性研究[J]. 精神医学杂志, 2023, 36(3): 246-250. DOI:10.3969/j.issn.2095-9346.2023.03.005 |
| [11] |
ORFANAKOS K, ALIFIERIS CE, VERIGOS EK, et al. The predictive value of 8-hydroxy-deoxyguanosine (8-OHdG) serum concentrations in irradiated non-small cell lung carcinoma (NSCLC) patients[J]. Biomedicines, 2024, 12(1): 134. DOI:10.3390/biomedicines12010134 |
| [12] |
YASUDA T, KASHIMA Y. A soy protein enzymatic digest mitigates Nrf2-related oxidative stress and attenuates depression-like behavior in a mouse model of sub-chronic restraint stress[J]. Heliyon, 2024, 10(6): e27826. DOI:10.1016/j.heliyon.2024.e27826 |
| [13] |
JI N, LEI M, CHEN Y, et al. How oxidative stress induces depression?[J]. ASN Neuro, 2023, 15: 17590914231181037. DOI:10.1177/17590914231181037 |
| [14] |
ANSARI MA, RAO MS, AL-JARALLAH A. Insights into early pathogenesis of sporadic Alzheimer's disease: role of oxidative stress and loss of synaptic proteins[J]. Front Neurosci, 2024, 17: 1273626. DOI:10.3389/fnins.2023.1273626 |
| [15] |
CAO X, CHEN P. Changes in serum amyloid A (SAA) and 8-OHdG in patients with senile early cognitive impairment[J]. Med Sci Monit, 2020, 26: e919586. DOI:10.12659/MSM.919586 |
 2025, Vol. 54
2025, Vol. 54