文章信息
- 刘园园, 才奇博, 曲妍, 杨秀静, 关荣春, 刘灿君
- LIU Yuanyuan, CAI Qibo, QU Yan, YANG Xiujing, GUAN Rongchun, LIU Canjun
- PCSK9基因rs562556多态性对2型糖尿病合并急性心肌梗死患者PCI术后主要不良心血管事件的预测价值
- Predictive value of PCSK9 gene rs562556 polymorphism for major adverse cardiovascular events after pci in patients with type 2 diabetes mellitus complicated by acute myocardial infarction
- 中国医科大学学报, 2025, 54(10): 889-895
- Journal of China Medical University, 2025, 54(10): 889-895
-
文章历史
- 收稿日期:2024-09-26
- 网络出版时间:2025-10-15 16:02:48
2. 齐齐哈尔医学院附属第三医院心血管科,黑龙江 齐齐哈尔 161000
2. Department of Cardiology, The Third Affiliated Hospital of Qiqihar Medical University, Qiqihar 161000, China
急性心肌梗死(acute myocardial infarction,AMI)的发病率和死亡率在全球范围内居高不下,严重威胁公共健康[1-3]。AMI的危险因素包括2型糖尿病(type 2 diabetes mellitus,T2DM)、高胆固醇血症、高血压和年龄等[4-5]。AMI的首选治疗方案是经皮冠状动脉介入治疗(percutaneous coronary intervention,PCI)[6],但在T2DM患者中发生术后复发性主要不良心血管事件(major adverse cardiovascular event,MACE)的风险极高[7]。研究[8]发现,基因多态性与AMI患者PCI术后发生MACE相关。蛋白转化酶枯草杆菌素/酶切蛋白9型(proprotein converase subtilisin/keexin type 9,PCSK9)是由PCSK9基因编码的丝氨酸蛋白酶,PCSK9基因多态性与心血管事件发生密切相关[9],其中PCSK9 R93C变异与中国汉族人群早发性心肌梗死风险显著降低相关[10]。目前尚未见PCSK9基因多态性与T2DM合并AMI患者PCI术后MACE的相关报道。本研究拟探讨T2DM合并AMI患者PCSK9基因rs562556多态性与PCI术后发生MACE的关系,并构建其临床预测模型,为早期预测MACE的发生提供可靠方法。
1 材料与方法 1.1 研究对象及分组选取2019年1月至2021年12月齐齐哈尔医学院附属第三医院接受PCI的T2DM合并AMI患者97例。中位年龄53(45~79)岁,其中男49例,女48例。纳入标准:符合《急性心肌梗死诊断和治疗指南》 [11]诊断标准,确诊为AMI并入住监护病房;符合WHO制定的T2DM诊断标准,即空腹血糖(fasting plasma glucose,FPG)≥7.0 mmol/L或餐后2 h血糖≥11.1 mmol/L,糖化血红蛋白≥6.5%,出现AMI症状至入院时间≤12 h;年龄 < 80岁;1个月内未服用降脂类药物;精神状态和认知功能正常;临床资料和随访资料完整。排除标准:由1型糖尿病、妊娠糖尿病及其他内分泌疾病引起的继发性糖尿病;年龄 < 18岁;消化系统疾病、肝功能异常和肺部疾病;免疫功能异常;恶性肿瘤。
根据2年随访期间是否发生MACE,将97例患者分为非MACE组(n = 57)和MACE组(n = 40)。收集患者的一般资料,包括年龄、性别、体重指数(body mass index,BMI)、高血压病史、血脂异常病史、冠状动脉粥样硬化性心脏病(简称冠心病)病史和吸烟史等。记录患者入院诊断[ST段抬高型心肌梗死(ST-segment elevation myocardial infarction,STEMI)或非ST段抬高型心肌梗死(non-STEMI,NSTEMI)]、收缩压、舒张压、心率、支架数量和支架直径等。本研究已获得齐齐哈尔医学院附属第三医院医学伦理委员会批准,审批号为(齐)伦审[2021] 100号。所有患者签署知情同意书。
1.2 方法 1.2.1 生化指标检测入院后第2天清晨抽取患者空腹静脉血5 mL,采用全自动生化仪检测FPG、肌酐、总胆固醇(total cholesterol,TC)、甘油三酯(triglyceride,TG)、高密度脂蛋白胆固醇(high-density lipoprotein cholesterol,HDL-C)和低密度脂蛋白胆固醇(low-density lipoprotein cholesterol,LDL-C)水平。用免疫投射比浊法检测超敏C反应蛋白(hypersensitivity C reactive protein,hs-CRP)水平。用电化学发光免疫法定量测定血清N末端脑钠肽前体(N-terminal pro-brain natriuretic peptide,NT-proBNP)水平。用电化学发光法定量检测血清心肌肌钙蛋白T(cardiac troponin T,cTnT)水平。
1.2.2 基因检测通过人类基因组计划网站(http://grch37.ensembl.org/)查阅人类PCSK9基因rs562556单核苷酸多态性(single nucleotide polymorphism,SNP)位点信息。PCSK9基因rs562556的等位基因型为G/A,最小等位基因频率(minor allele frequencies,MAF)为0.13。根据等位基因型G/A,基因分型可分为野生型GG、杂合突变型GA和纯合突变型AA,遗传模型可分为显性模型(AA vs. GG+AG)和隐性模型(GG vs. AA+AG)。
入院后第2天清晨抽取患者空腹静脉血5 mL,3 000 r/min离心10 min,取上清进行PCSK9基因SNP检测。利用基因组DNA提取试剂盒(美国Sigma-Aldrich公司)提取样本的DNA,用紫外分光光度法测定DNA纯度,用PCR进行PCSK9基因扩增。PCR反应条件:94 ℃ 3 min,94 ℃20 s、56 ℃ 30 s,72 ℃ 30 s,循环40次。PCSK9基因rs562556引物序列如下:正向5’-ATCACGCTCCCCTTTGGAAGTGCTC-3’,反向5’-GCCGATGAGCTCACAGGCACATGTC-3’。将PCR产物送至生工生物工程(上海)股份有限公司进行纯化测序。
1.2.3 血浆PCSK9水平检测入院后第2天清晨抽取患者空腹静脉血5 mL,3 000 r/min离心10 min后取上清。采用Human Proprotein Convertase 9/PCSK9 ELISA试剂盒(杭州联科生物技术股份有限公司)检测血浆PCSK9水平。根据标准曲线计算血浆PCSK9水平。
1.2.4 随访随访并记录患者PCI术后2年内发生MACE的情况。MACE的定义为心血管死亡、靶血管相关心肌梗死和缺血驱动的靶血管重建[12]。
1.3 统计学分析采用SPSS 23.0和SPSSAU软件进行统计分析。符合和不符合正态分布的计量资料分别用x±s和M(P25~P75)表示,2组比较采用Student’s t检验或非参数秩和检验。计数资料均采用率(%)表示,组间比较采用χ2检验。采用SHEsis在线分析软件(http://analysis.bio-x.cn/)进行Hardy-Weinberg(H-W)平衡检验,P > 0.05认为基因型符合H-W平衡,说明该研究群体具有代表性。采用Spearman相关性检验分析PCSK9基因rs562556基因型与血浆PCSK9水平的关系。采用Kaplan-Meier曲线分析PCSK9基因rs562556多态性对T2DM合并AMI患者PCI术后2年内发生MACE的影响。用多因素logistic回归分析影响MACE的危险因素。用受试者操作特征(receiver operator characteristic,ROC)曲线分析rs562556多态性和主要临床因素对T2DM合并AMI患者PCI术后发生MACE的预测价值。P < 0.05为差异有统计学意义。
2 结果 2.1 2组患者一般临床资料比较与非MACE组患者比较,MACE组患者年龄、心率、肌酐、NT-proBNP、LDL-C和PCSK9水平均明显升高,有高血压病史和冠心病史的患者明显增多,且舒张压显著降低,差异均有统计学意义(均P < 0.05),见表 1。
| Item | Non-MACE group(n = 57) | MACE group(n = 40) | Z/χ2/t | P |
| Age(year) | 49.0(47-52) | 67.5(58-76) | 7.765 | < 0.001 |
| Male [n(%)] | 29(51) | 20(50) | 0.007 | 0.932 |
| BMI(kg/m2) | 23.5±2.6 | 23.9±2.5 | 0.864 | 0.390 |
| STEMI [n(%)] | 42(73.7) | 30(75.0) | 0.023 | 0.878 |
| SBP(mmHg) | 129(105-140) | 128(111-147) | 0.850 | 0.395 |
| DBP(mmHg) | 76.0(72.0-83.0) | 68.5(61.5-75.5) | 4.885 | < 0.001 |
| Heart rate(beats/min) | 80(73-87) | 95(75-104) | 3.567 | < 0.001 |
| Hypertension [n(%)] | 30(52.6) | 29(72.5) | 3.894 | 0.048 |
| Dyslipidemia [n(%)] | 25(43.9) | 16(40.0) | 0.143 | 0.705 |
| CHD [n(%)] | 10(17.5) | 16(40.0) | 6.042 | 0.014 |
| Smoking [n(%)] | 30(52.6) | 19(47.5) | 0.248 | 0.619 |
| Number of stents ≥2 [n(%)] | 17(29.8) | 10(25.0) | 0.272 | 0.602 |
| Stent diameter(mm) | 3(2-4) | 3(2-4) | 0.746 | 0.456 |
| TC(mmol/L) | 3.91±0.24 | 3.92±0.25 | 0.054 | 0.957 |
| TG(mmol/L) | 1.59±0.26 | 1.51±0.26 | 1.442 | 0.153 |
| HDL-C(mmol/L) | 1.01±0.19 | 1.07±0.31 | 1.101 | 0.274 |
| LDL-C(mmol/L) | 2.73±0.50 | 3.20±0.64 | 4.042 | < 0.001 |
| sCr(μmol/L) | 80.61±15.59 | 110.32±12.79 | 9.929 | < 0.001 |
| FPG(mmoL/L) | 9.61 ± 0.55 | 9.48 ±0.57 | 1.152 | 0.252 |
| cTnT(ng/mL) | 2.60(1.41-3.62) | 2.48(1.60-3.64) | 0.011 | 0.991 |
| hs-CRP(mg/dL) | 17.92(13.14-21.13) | 15.56(11.02-22.92) | 0.048 | 0.962 |
| NT-proBNP(pg/mL) | 1 006(662.65-1 302.65) | 2 204.82(1 510.95-3 168.50) | 6.390 | < 0.001 |
| PCSK9(ng/mL) | 0.30(0.27-0.32) | 0.37(0.31-0.42) | 4.997 | < 0.001 |
| BMI,body mass index;STEMI,ST-segment elevation myocardial infarction;SBP,systolic blood pressure;DBP,diastolic blood pressure;CHD,coronary heart disease;TC,total cholesterol;TG,triglyceride;HDL-C,high-density lipoprotein cholesterol;LDL-C,low-density lipoprotein cholesterol;sCr,serum creatinine;FPG,fasting plasma glucose;cTnT,cardiac troponin T;hs-CRP,hypersensitivity C reactive protein;NT-proBNP,N-terminal pro-brain natriuretic peptide;PCSK9,proprotein converase subtilisin/keexin type 9. | ||||
2.2 2组患者PCSK9基因rs562556基因型与等位基因分布特征
非MACE组和MACE组的PCSK9基因rs562556基因型散布契合H-W遗传平衡(χ2 =5.981,P = 0.051;χ2 =5.879,P = 0.053)。与非MACE组比较,MACE组患者PCSK9基因rs562556基因型AA与等位基因A分布频率均升高,差异有统计学意义(P < 0.05)。见表 2。
| Group | Genotype | Allele | ||||
| AA | AG | GG | A | G | ||
| Non-MACE(n = 57) | 29(50.9) | 23(40.4) | 5(8.7) | 81(71.1) | 33(28.9) | |
| MACE(n = 40) | 30(75.0) | 9(22.5) | 1(2.5) | 69(86.2) | 11(13.8) | |
| χ2 | 6.270 | 6.483 | ||||
| P | 0.043 | 0.011 | ||||
2.3 PCSK9基因rs562556基因型与血浆PCSK9水平的关系
Spearman相关分析结果显示,T2DM合并AMI患者PCSK9基因rs562556基因型AA与血浆PSCK9水平呈正相关(r = 0.61,P < 0.000 1)。
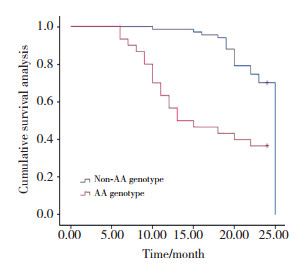
2.4 Kaplan-Meier曲线分析结果PCSK9基因rs562556基因型为AA的T2DM合并AMI患者在PCI术后2年随访期间发生MACE的风险显著增加(P < 0.05),见图 1。
|
| 图 1 Kaplan-Meier曲线分析T2DM合并AMI患者rs562556基因型(AA vs. AG+GG)在2年随访期间发生MACE的差异 Fig.1 Kaplan-Meier curve analysis of the rs562556 genotype (AA vs. AG+GG) in patients with T2DM and AMI indicating MACE-related differences during the 2-year follow-up |
2.5 T2DM合并AMI患者PCI术后发生MACE的危险因素
以PCI术后发生MACE为因变量,以年龄、心率、冠心病史、高血压病史、舒张压、肌酐、NT-proBNP、LDL-C、PCSK9水平和rs562556基因型AA进行多因素logistic回归分析,结果显示,在矫正相关危险因素后,高龄、NT-proBNP和PCSK9水平升高及rs562556基因型AA是T2DM合并AMI患者PCI术后发生MACE的独立危险因素。见表 3。
| Variable | β | SE | Waldχ2 | OR | 95%CI | P |
| Age | 0.019 | 0.003 | 46.391 | 1.019 | 1.013-1.024 | < 0.000 1 |
| NT-proBNP | 0.006 | 0.001 | 39.742 | 1.006 | 1.004-1.008 | < 0.000 1 |
| PCSK9 | 0.010 | 0.002 | 38.491 | 1.110 | 1.007-1.263 | < 0.000 1 |
| rs562556 genotype AA | 0.521 | 0.114 | 21.042 | 1.684 | 1.348-2.104 | < 0.000 1 |
2.6 ROC曲线分析rs562556基因多态性对T2DM合并AMI患者PCI术后发生MACE的预测价值
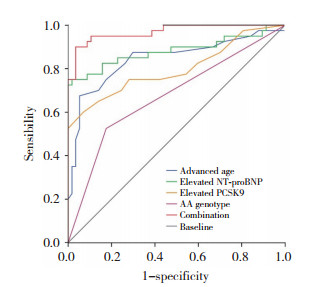
用ROC曲线分析各危险因素对T2DM合并AMI患者PCI术后2年发生MACE的预测准确度,结果显示,高龄、NT-proBNP和PCSK9水平升高及rs562556基因多态性(AA型)对PCI术后2年发生MACE具有良好的预测价值。在PCI术后2年随访期内,rs562556基因型AA联合高龄和NT-proBNP、PCSK9高水平对MACE的预测价值比单独指标的预测价值更高。见图 2和表 4。
|
| 图 2 ROC曲线分析rs562556基因型AA对T2DM合并AMI患者PCI术后发生MACE的预测价值 Fig.2 ROC curve analysis of rs562556 genotype AA for MACE prediction post-PCI in patients with T2DM and AMI |
| Indicator | AUC | 95%CI | Sensitivity(%) | Specificity(%) | Youden index |
| rs562556 genotype AA | 0.675 | 0.563-0.787 | 52.5 | 82.5 | 0.350 |
| Advanced age | 0.845 | 0.757-0.932 | 67.5 | 94.7 | 0.622 |
| Elevated NT-proBNP | 0.882 | 0.802-0.963 | 75.0 | 98.2 | 0.732 |
| Elevated PCSK9 | 0.798 | 0.701-0.896 | 60.0 | 93.0 | 0.530 |
| Combination | 0.969 | 0.938-1.000 | 90.0 | 96.5 | 0.865 |
3 讨论
AMI是T2DM患者死亡的常见原因,血管功能障碍是糖尿病性心肌病的主要致病因素[13-15]。T2DM是引起动脉粥样硬化性血栓形成的重要因素[16]。胰岛素抵抗和血糖升高可加速动脉粥样硬化的发展,增加发生AMI的风险[17]。T2DM诱导动脉粥样硬化和血栓形成的机制主要是由于持续高血糖导致脂质代谢紊乱,促进脂质沉积,导致机体处于微炎症状态,促进血栓形成[18]。T2DM患者脂质代谢异常是导致糖尿病患者血管并发症的最重要原因[19]。高TG和低HDL-C水平是诊断心血管疾病的重要标志[20]。高LDL-C是发生心血管疾病的重要危险因素[21]。降脂治疗可降低冠状动脉疾病患者发生心血管事件的风险[22]。研究[23]表明,患者在接受最大剂量他汀类药物治疗后,PCSK9抑制剂仍能降低患者血浆LDL-C水平,而且对胰岛素抵抗和血栓等也具有治疗作用[24]。PCSK9还能通过降解LDL受体调节循环LDL-C水平,是改善AMI患者脂蛋白谱和心血管预后的有效靶点[25]。此外,PCSK9还与葡萄糖代谢有关。临床试验表明,PCSK9抑制剂治疗糖尿病更有效[26]。PCSK9能调节葡萄糖耐受不良、胰岛素抵抗、腹部肥胖、炎症和高血压[27]。但PCSK9是否与T2DM合并AMI患者PCI术后发生MACE有关尚不清楚。本研究发现,PCSK9可能与T2DM合并AMI患者PCI术后发生MACE的风险有关。
基因多态性是指同一基因核苷酸排列顺序在不同个体中出现差异,可影响生理代谢过程,进而引发相关疾病。研究[28]发现,PCSK9基因突变与LDL-C水平的变化有关。中国人群中PCSK9基因E670G多态性与心血管疾病的发生有关[29]。除了E670G多态性,PCSK9基因rs562556变异也是一种功能获得突变。rs562556 G等位基因与重症疟疾患者生存率相关[30]。此外,PCSK9还被发现参与调控血管内皮功能、氧化应激反应及炎症反应,可能在心血管不良事件中发挥重要作用[31]。因此,本研究探讨了rs562556基因多态性与T2DM合并AMI患者PCI术后发生MACE的关系,结果发现,PCSK9基因rs562556基因型AA的T2DM合并AMI患者在PCI术后2年内发生MACE的风险显著增加。高龄、NT-proBNP和PCSK9水平升高及rs562556基因型AA是T2DM合并AMI患者PCI术后发生MACE的独立危险因素,并对PCI术后发生MACE具有良好的预测价值,PCSK9基因单个SNP位点rs562556的等位基因A频率越高的患者,PCI术后发生MACE的概率更高。目前已有多种PCSK9抑制剂用于临床,在降低LDL-C及减少心血管事件方面疗效显著[32]。然而,尚缺乏针对不同PCSK9基因多态性的个体化治疗方案。未来基因分型有望用于风险分层和指导PCSK9抑制剂的精准使用,值得进一步探索。
综上所述,本研究通过比较不同AMI患者外周血PCSK9水平、血脂水平及PCSK9基因rs562556多态性,提出T2DM合并AMI患者血浆PCSK9水平明显升高,且rs562556基因型AA的患者PCI术后发生MACE的风险更高,高龄、NT-proBNP和PCSK9水平升高以及rs562556基因型AA对PCI术后2年内发生MACE具有良好的预测价值,为临床早期干预治疗T2DM合并AMI患者PCI术后MACE的发生提供了可靠的参考。
| [1] |
WELT FGP, BATCHELOR W, RICHARD SPEARS J, et al. Reperfusion injury in patients with acute myocardial infarction: jacc scientific statement[J]. J Am Coll Cardiol, 2024, 83(22): 2196-2213. DOI:10.1016/j.jacc.2024.02.056 |
| [2] |
蔡琳琳, 马晶茹. 组织蛋白酶D在急性ST段抬高型心肌梗死后心血管事件中的作用[J]. 中国医科大学学报, 2024, 53(10): 929-933. DOI:10.12007/j.issn.0258‐4646.2024.10.010 |
| [3] |
LU Y, LI SX, LIU Y, et al. Sex-specific risk factors associated with first acute myocardial infarction in young adults[J]. JAMA Netw Open, 2022, 5(5): e229953. DOI:10.1001/jamanetworkopen.2022.9953 |
| [4] |
DANCHIN N, FARNIER M, ZELLER M, et al. Long-term outcomes after acute myocardial infarction in patients with familial hypercholesterolemia: the French registry of acute ST-elevation and non-ST-elevation myocardial infarction program[J]. J Clin Lipidol, 2020, 14(3): 352-360. DOI:10.1016/j.jacl.2020.03.008 |
| [5] |
MA J, WANG MH, WU P, et al. Predictive effect of triglyceride-glucose index on no-reflow phenomenon in patients with type 2 diabetes mellitus and acute myocardial infarction undergoing primary percutaneous coronary intervention[J]. Diabetol Metab Syndr, 2024, 16(1): 67. DOI:10.1186/s13098-024-01306-y |
| [6] |
ZHANG Y, CAO HX, JIANG P, et al. Cardiac rehabilitation in acute myocardial infarction patients after percutaneous coronary intervention: a community-based study[J]. Medicine, 2018, 97(8): e9785. DOI:10.1097/MD.0000000000009785 |
| [7] |
ZHOU MG, LIU J, HAO YC, et al. Prevalence and in-hospital outcomes of diabetes among patients with acute coronary syndrome in China: findings from the Improving Care for Cardiovascular Disease in China-Acute Coronary Syndrome Project[J]. Cardiovasc Diabetol, 2018, 17(1): 147. DOI:10.1186/s12933-018-0793-x |
| [8] |
DU PQ, LI XG, LI DD, et al. PEAR1, PON1, CYP2C19, CYP1A2 and F2R polymorphisms are associated with MACE in clopidogrel-treated patients with acute coronary syndrome undergoing percutaneous coro-nary intervention[J]. Pharmgenomics Pers Med, 2024, 17: 611-621. DOI:10.2147/PGPM.S490030 |
| [9] |
GAI MT, ADI D, CHEN XC, et al. Polymorphisms of rs2483205 and rs562556 in the PCSK9 gene are associated with coronary artery disease and cardiovascular risk factors[J]. Sci Rep, 2021, 11(1): 11450. DOI:10.1038/s41598-021-90975-0 |
| [10] |
YANG L, PU T, ZHANG Y, et al. The R93C variant of PCSK9 reduces the risk of premature MI in a Chinese Han population[J]. Front Genet, 2022, 13: 875269. DOI:10.3389/fgene.2022.875269 |
| [11] |
DAI WB, REN JY, HU ST, et al. The safety and efficacy of indobufen or aspirin combined with clopidogrel in patients with acute myocar-dial infarction after percutaneous coronary intervention[J]. Platelets, 2024, 35(1): 2364748. DOI:10.1080/09537104.2024.2364748 |
| [12] |
BARBATO E, TOTH GG, JOHNSON NP, et al. A prospective na-tural history study of coronary atherosclerosis using fractional flow reserve[J]. J Am Coll Cardiol, 2016, 68(21): 2247-2255. DOI:10.1016/j.jacc.2016.08.055 |
| [13] |
LEE SH, KIM HK, AHN JH, et al. Prognostic impact of hypercoa- gulability and impaired fibrinolysis in acute myocardial infarction[J]. Eur Heart J, 2023, 44(19): 1718-1728. DOI:10.1093/eurheartj/ehad088 |
| [14] |
ZHANG Y, DING XS, HUA B, et al. Predictive effect of triglyceri- de-glucose index on clinical events in patients with type 2 diabetes mellitus and acute myocardial infarction: results from an observational cohort study in China[J]. Cardiovasc Diabetol, 2021, 20(1): 43. DOI:10.1186/s12933-021-01236-3 |
| [15] |
AVAGIMYAN A, FOGACCI F, POGOSOVA N, et al. Diabetic cardiomyopathy: 2023 update by the international multidisciplinary board of experts[J]. Curr Probl Cardiol, 2024, 49(1 Pt A): 102052. DOI:10.1016/j.cpcardiol.2023.102052 |
| [16] |
CHEN J, LI WW, CAO JZ, et al. Risk factors for carotid plaque formation in type 2 diabetes mellitus[J]. J Transl Med, 2024, 22(1): 18. DOI:10.1186/s12967-023-04836-7 |
| [17] |
NATH S, MOHANTY R, PATNAIK U. Insulin resistance in non diabetic individuals with acute myocardial infarction and its relationship with acute phase reactants[J]. Int J Acad Med, 2020, 6(1): 22. DOI:10.4103/ijam.ijam_9_19 |
| [18] |
POZNYAK A, GRECHKO AV, POGGIO P, et al. The diabetes mellitus-atherosclerosis connection: the role of lipid and glucose metabolism and chronic inflammation[J]. Int J Mol Sci, 2020, 21(5): 1835. DOI:10.3390/ijms21051835 |
| [19] |
钟森杰, 李静, 王陵军, 等. 基于痰瘀相关理论探讨冠心病脂质浸润机制及痰瘀同治的研究进展[J]. 中国中药杂志, 2023, 48(6): 1431-1437. DOI:10.19540/j.cnki.cjcmm.20221126.502 |
| [20] |
CHENG BW, YI YM, CHEN MT, et al. TG/HDL-C ratio is positively associated with risk and severity of CHD among NAFLD patients: a case control study[J]. Front Endocrinol, 2024, 15: 1383489. DOI:10.3389/fendo.2024.1383489 |
| [21] |
MHAIMEED O, BURNEY ZA, SCHOTT SL, et al. The importance of LDL-C lowering in atherosclerotic cardiovascular disease prevention: lower for longer is better[J]. Am J Prev Cardiol, 2024, 18: 100649. DOI:10.1016/j.ajpc.2024.100649 |
| [22] |
SKEIK N, NOWARIAK ME, SMITH JE, et al. Lipid-lowering the- rapies in peripheral artery disease: a review[J]. Vasc Med, 2021, 26(1): 71-80. DOI:10.1177/1358863X20957091 |
| [23] |
SABATINE MS. PCSK9 inhibitors: clinical evidence and implementation[J]. Nat Rev Cardiol, 2019, 16(3): 155-165. DOI:10.1038/s41569-018-0107-8 |
| [24] |
QI ZY, HU L, ZHANG JJ, et al. PCSK9(proprotein convertase subtilisin/kexin 9) enhances platelet activation, thrombosis, and myocardial infarct expansion by binding to platelet CD36[J]. Circulation, 2021, 143(1): 45-61. DOI:10.1161/CIRCULATIONAHA.120.046290 |
| [25] |
XU JN, WANG TT, SHU H, et al. Insight into the role of PCSK9 in glucose metabolism[J]. Clin Chim Acta, 2023, 547: 117444. DOI:10.1016/j.cca.2023.117444 |
| [26] |
NEJABAT M, HADIZADEH F, ALMAHMEED W, et al. Effects of PCSK9 inhibitors on cancer, diabetes, and cardiovascular diseases[J]. Drug Discov Today, 2025, 30(3): 104316. DOI:10.1016/j.drudis.2025.104316 |
| [27] |
MELENDEZ QM, KRISHNAJI ST, WOOTEN CJ, et al. Hypercholesterolemia: the role of PCSK9[J]. Arch Biochem Biophys, 2017, 625-626: 39-53. DOI:10.1016/j.abb.2017.06.001 |
| [28] |
SHIRAHAMA R, ONO T, NAGAMATSU S, et al. Coronary artery plaque regression by a PCSK9 antibody and rosuvastatin in double-heterozygous familial hypercholesterolemia with an LDL receptor mutation and a PCSK9 V4I mutation[J]. Intern Med, 2018, 57(24): 3551-3557. DOI:10.2169/internalmedicine.1060-18 |
| [29] |
LI YY, WANG H, YANG XX, et al. PCSK9 gene E670G polymorphism and coronary artery disease: an updated meta-analysis of 5, 484 subjects[J]. Front Cardiovasc Med, 2020, 7: 582865. DOI:10.3389/fcvm.2020.582865 |
| [30] |
FEDORYAK O, ARAMA C, DIARRA I, et al. Association of the rs562556 PCSK9 gene polymorphism with reduced mortality in severe malaria among Malian children[J]. Can J Infect Dis Med Microbiol, 2020, 2020: 9340480. DOI:10.1155/2020/9340480 |
| [31] |
DUTKA M, ZIMMER K, ĆWIERTNIA M, et al. The role of PCSK9 in heart failure and other cardiovascular diseases-mechanisms of action beyond its effect on LDL cholesterol[J]. Heart Fail Rev, 2024, 29(5): 917-937. DOI:10.1007/s10741-024-10409-7 |
| [32] |
ABDULJABBAR MH. PCSK9 inhibitors: focus on evolocumab and its impact on atherosclerosis progression[J]. Pharmaceuticals, 2024, 17(12): 1581. DOI:10.3390/ph17121581 |
 2025, Vol. 54
2025, Vol. 54