文章信息
- 孙佳莹, 付茜, 辛雨, 张嵚垚, 马淑梅
- SUN Jiaying, FU Xi, XIN Yu, ZHANG Qinyao, MA Shumei
- QT离散度对川崎病患儿发生冠状动脉损伤及其预后的预测价值
- Predictive value of QT dispersion for coronary artery injury and its prognosis in children with Kawasaki disease
- 中国医科大学学报, 2023, 52(3): 193-198
- Journal of China Medical University, 2023, 52(3): 193-198
-
文章历史
- 收稿日期:2022-10-13
- 网络出版时间:2023-03-16 09:51:06
2. 中国医科大学附属盛京医院心血管内科,沈阳 110004
2. Department of Cardiology, Shengjing Hospital of China Medical University, Shenyang 110004, China
川崎病(Kawasaki disease,KD)又称为小儿皮肤黏膜淋巴结综合征[1],男性发病率较高,男女比例约为1.5∶1,发病年龄6个月~5岁[2],成人少见[3],治疗上以阿司匹林、免疫球蛋白、糖皮质激素等药物治疗为主[4-5]。KD常表现为中小动脉系统性血管炎性病变,可导致冠状动脉损伤,进而造成心肌受损,这是儿童获得性心脏病最常见的原因[6]。已有研究发现,部分KD患儿临床有效治疗后仍可发生冠状动脉损伤,导致心肌电位分布改变和复极变化。心电图QT间期主要反映心室复极情况,其大小在不同导联存在差异,QT离散度(QT dispersion,QTd)反映心室肌复极的不均一性[7],因此可作为检测恶性心律失常的指标[8]。本研究探讨心电图检查中QTd对KD患儿冠状动脉损伤及其预后的预测价值。
1 材料与方法 1.1 研究对象收集2018年1月至2021年7月中国医科大学附属盛京医院收治的急性期KD患儿的临床资料。纳入标准:符合2019年美国心脏病协会KD诊断和治疗指南[9]。排除标准:患有心源性休克、严重肝肾功能不全、全身免疫性疾病、恶性肿瘤、血液系统疾病患儿。共纳入1 743例。其中男1 039例,女704例,平均年龄(3.68±2.29)岁。本研究获得中国医科大学附属盛京医院伦理委员会批准(2018PS507K)。
1.2 检查方法所有患儿均在入院后药物治疗前完善相关化验、心电图及心脏彩超检查;生化标志物测定(电化学发光、化学发光、酶法测定等)由我院检验科完成。心电图采用常规心电图仪描记同步12导联体表心电图,增益10 mm/mV,纸速25 mm/s,电极位置常规放置,每一导联记录4~6个心动周期,获得清晰且平稳图像后测量。排除有干扰、T波无法可靠测量、低振幅或T波U波融合的心电图,患儿不配合时常规镇静。QT间期测量选择窦性心律时波形清晰的3个心动周期测量后计算平均值。QTd为12导联心电图中最大和最小QT间期的差值。以上指标均由同一位心电医生使用标准数字卡尺(中国航空航天)测量完成。心脏彩超检查选择儿童尺寸传感器彩色多普勒超声诊断仪,患儿安静状态下呈平卧位,≤5岁患儿冠状动脉内径 > 3 mm、> 5岁患儿冠状动脉内径 > 5 mm、某处冠状动脉内径超过临近冠状动脉内径的1.5倍诊断为冠状动脉损伤[10]。
1.3 随访所有纳入患儿均出院1周后门诊复查,完善血常规、心电图、心脏彩超等检查,发病后1个月内复查(1次/周),发病2个月、6个月、12个月时复查1次。患儿于2022年7月随访完成,中位随访时间为11(9~14)个月。随访记录事件包括冠状动脉损伤、心肌梗死、心肌炎、心包炎、心内膜炎、KD复发。
1.4 统计学分析应用SPSS 23.0软件进行统计分析,计量资料采用x±s表示,2组比较采用独立样本t检验。计数资料采用率(%)表示,组间比较采用χ2检验。受试者操作特征(receiver operating characteristic,ROC)曲线分析QTd对冠状动脉损伤和主要不良心脏事件的预测能力和最佳截断值。生存分析采用Kaplan-Meier生存曲线法,生存率比较采用log-rank检验。logistic回归分析KD患儿发生冠状动脉损伤的独立危险因素,Cox回归分析评价QTd对患儿预后的影响。P < 0.05为差异有统计学意义。
2 结果ROC曲线分析结果显示,QTd预测KD患儿规范治疗后冠状动脉损伤的最佳截断值为38.5 ms [曲线下面积(area under the curve,AUC)=0.940,95% CI:0.893~0.986,P < 0.001],灵敏度为88.64%,特异度为77.12%。根据最佳截断值将1 743例患儿分为QTd < 38.5组(n = 1 174)和QTd≥38.5组(n = 569)。QTd预测KD患儿主要不良心脏事件的最佳截断值为39.5 ms [AUC =0.867,95% CI:0.792~0.934,P < 0.001],灵敏度为78.57%,特异度为83.71%。根据最佳截断值将1 743例患儿分为QTd < 39.5组(n = 1 211)和QTd≥39.5组(n = 532)。见图 1。
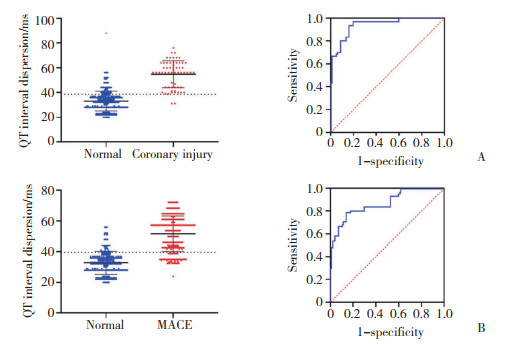
|
| A,coronary injury;B,MACE. MACE,major adverse cardiac events. 图 1 QTd对冠状动脉损伤和发生主要不良心脏事件的散点图和ROC曲线 Fig.1 Scatter diagram and ROC curve of QT dispersion for coronary injury and major adverse cardiac events |
2.1 QTd < 38.5组和QTd≥38.5组各项临床指标比较
结果显示,与QTd < 38.5组比较,QTd≥38.5组患儿年龄、白细胞、C反应蛋白、血肌酐、肌钙蛋白、脑钠肽水平显著增高(均P < 0.05);既往曾患KD患儿更易发生冠状动脉损伤(P < 0.05)。而2组患儿年龄、性别、血压、心率、红细胞、血小板、心脏射血分数等指标比较均无统计学差异(均P > 0.05)。见表 1。
| Item | QTd < 38.5 group(n = 1 174) | QTd≥38.5 group(n = 569) | P |
| Age(x±s,year) | 4.32±2.28 | 3.27±2.22 | 0.036 |
| Male [n(%)] | 764(65.08) | 356(62.57) | 0.494 |
| Heart rate(x±s,beat/min) | 121.74±26.60 | 122.40±28.48 | 0.849 |
| SBP(x±s,mmHg) | 87.36±13.34 | 87.09±14.66 | 0.911 |
| DBP(x±s,mmHg) | 58.74±9.51 | 57.89±9.97 | 0.838 |
| WBC(x±s,109/L) | 13.28±5.46 | 15.28±7.06 | < 0.001 |
| RBC(x±s,g/L) | 4.16±1.72 | 4.27±1.31 | 0.897 |
| Platelet(x±s,109/L) | 163.05±40.50 | 167.37±42.93 | 0.515 |
| CRP(x±s,mg/L) | 57.03±14.87 | 78.63±12.46 | < 0.001 |
| ESR(x±s,mm/h) | 59.24±53.23 | 66.55±57.58 | 0.291 |
| ALT(x±s,U/L) | 32.79±6.65 | 34.04±5.49 | 0.679 |
| AST(x±s,U/L) | 29.17±4.60 | 28.09±4.62 | 0.933 |
| Cre(x±s,μmol/L) | 78.29±16.44 | 127.16±15.80 | < 0.001 |
| Urea(x±s,mmol/L) | 5.83±1.86 | 5.19±2.62 | 0.576 |
| CysC(x±s,mg/L) | 0.98±0.63 | 1.02±0.44 | 0.709 |
| cTnT(x±s,ng/mL) | 0.17±0.06 | 0.34±0.09 | < 0.001 |
| CK-MB(x±s,U/L) | 16.99±4.69 | 19.23±4.24 | 0.077 |
| BNP(x±s,pg/mL) | 287.32±35.31 | 348.07±29.29 | < 0.001 |
| LVEF(x±s,%) | 54.90±12.11 | 50.43±14.85 | 0.392 |
| MV E/A | 1.39±0.83 | 1.36±0.34 | 0.918 |
| Left coronary artery width(x±s,mm) | 2.36±0.26 | 3.49±0.96 | < 0.001 |
| Right coronary artery width(x±s,mm) | 2.09±0.31 | 3.33±1.15 | < 0.001 |
| History of KD [n(%)] | 25(2.1) | 31(5.4) | < 0.001 |
| QTd,QT dispersion;SBP,systolic blood pressure;DBP,diastolic blood pressure;WBC,white blood cell;RBC,red blood cell;CRP,C reaction protein;ESR,erythrocyte sedimentation rate;ALT,glutamic pyruvic transaminase;AST,aspartate transaminase;Cre,creatinine;CysC,cystatin C;cTnT,cardiac troponin T;CK-MB,creatine phosphokinase-MB;BNP,brain natriuretic peptide;LVEF,left ventricular ejection fraction;MV E/A,measure the flow velocity of mitral valve orifice;KD,Kawasaki disease. | |||
2.2 冠状动脉损伤预测因子的logistic回归分析
多因素logistic分析结果显示,QTd≥38.5 ms、白细胞、年龄是KD患儿冠状动脉损伤的独立预测因子(均P < 0.05),见表 2。
| Variable | Un-adjusted OR(95% CI) | P | Adjusted OR(95% CI) | P |
| QTd≥38.5 ms | 2.62(1.74-4.28) | < 0.001 | 2.29(1.41-3.87) | 0.006 |
| Age | 1.00(1.00-1.00) | 0.024 | 1.00(0.98-1.00) | 0.025 |
| WBC | 1.01(1.00-1.03) | 0.001 | 1.00(1.00-1.02) | 0.018 |
| CRP | 1.03(0.99-1.05) | 0.286 | - | - |
| Cre | 0.99(0.98-1.00) | 0.037 | 1.00(0.99-1.00) | 0.177 |
| cTnT | 1.06(0.97-1.15) | 0.315 | - | - |
| BNP | 0.99(0.98-1.00) | 0.162 | - | - |
| History of KD | 2.28(1.57-4.03) | 0.001 | 1.54(0.91-1.06) | 0.193 |
| QTd,QT dispersion;WBC,white blood cell;CRP,C reaction protein;Cre,creatinine;cTnT,cardiac troponin T;BNP,brain natriuretic peptide;KD,Kawasaki disease. | ||||
2.3 QTd < 39.5组和QTd≥39.5组主要不良心脏事件发生情况比较
结果显示,2组心律失常、KD复发发生率比较差异有统计学意义(均P < 0.05),而心肌梗死、心肌炎、心包炎、心内膜炎发生率比较均无统计学差异(均P > 0.05)。见表 3。Kaplan-Meier生存分析结果显示,QTd≥39.5组患者发生心律失常、KD复发的风险远高于QTd < 39.5组(均P < 0.001,图 2)。多因素Cox回归分析结果显示,QTd≥39.5 ms、年龄、白细胞、既往KD病史是KD患儿发生主要不良心脏事件的独立预测因子(均P < 0.05),见表 4。
| Item | QTd < 39.5 group(n = 1 211) | QTd≥39.5 group(n = 532) | P |
| Myocardial infarction | 41(3.7) | 19(3.6) | 0.682 |
| Arrhythmia | 30(2.7) | 63(11.8) | < 0.001 |
| Myocarditis | 17(1.5) | 7(1.3) | 0.413 |
| Pericarditis | 8(0.7) | 5(0.9) | 0.439 |
| Endocarditis | 12(1.0) | 6(1.1) | 0.746 |
| KD recurrence | 58(5.2) | 142(26.7) | < 0.001 |
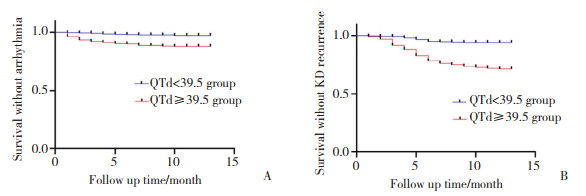
|
| A,survival without arrhythmia;B,survival without KD recurrence. 图 2 2组患者Kaplan-Meier生存分析 Fig.2 Kaplan-Meier survival analysis of the two groups |
| Variable | Un-adjusted HR(95% CI) | P | Multivariate HR(95% CI) | P |
| QTd≥39.5 ms | 3.85(2.11-5.34) | < 0.001 | 1.48(1.20-2.24) | 0.023 |
| Age | 0.97(0.96-0.98) | < 0.001 | 0.98(0.96-1.00) | 0.035 |
| WBC | 0.97(0.94-0.99) | < 0.001 | 0.98(0.97-0.99) | 0.042 |
| CRP | 1.00(1.00-1.00) | < 0.001 | 1.00(0.99-1.00) | 0.267 |
| Creatinine | 1.06(0.97-1.20) | 0.396 | - | - |
| cTnT | 0.95(0.93-1.07) | 0.594 | - | - |
| BNP | 0.99(0.98-1.01) | 0.321 | - | - |
| History of KD | 2.85(1.62-4.27) | < 0.001 | 1.85(1.15-3.01) | 0.011 |
| QTd,QT dispersion;WBC,white blood cell;CRP,C reaction protein;cTnT,cardiac troponin T;BNP,brain natriuretic peptide;KD,Kawasaki disease. | ||||
3 讨论
KD是一种急性自限性疾病,是以全身血管炎性病变为主要病理特征的急性发热性出疹性小儿疾病。临床症状常表现为发热、皮肤改变、眼结膜充血、颈部非脓性淋巴结肿大[11-12]。目前,普遍认为是由感染、氧化应激、遗传易感性和免疫调节失调所致[13-16]。KD患儿心电图上常表现为复极异常,在心电图中表现为QTd延长[16-17]。QTd升高提示心室不均匀复极,与心肌缺血程度有关,可作为潜在的非均匀性心室复极的替代标记物[18]。KD患者QTd延长的确切机制尚不清楚,现有研究认为可能由动作电位持续时间和传导区域变化、局部细胞群的去极化和神经激素因子引起的[19],可能与钙通道和细胞内钙信号传导的基因变异即钙通道电压依赖性β2亚单位基因(CACNB2)的形成相关[20]。KD是一种多系统性血管炎,主要影响中小型动脉,即使没有心内膜和心包改变,冠状动脉微循环水平也可能出现微循环障碍[21]。另一个原因可能与自主神经系统异常有关,在有心脏受累的KD患者中,迷走神经活动受到抑制,从而导致心肌失稳,增加恶性室性心律失常发生的可能性,因此迷走神经张力降低会导致外周血流阻力增加,进而增加心肌耗氧量[22]。
KD常累及全身性中等大小动脉,并倾向于冠状动脉,多于病程中期(发病2~4周后)出现冠状动脉损伤,早期不易发现[23]。KD患儿心肌血流储备较正常儿童低,总冠状动脉阻力较高[24]。如果未能及时诊断和治疗,约15%~25%KD患儿超声心动图上可能出现明显冠状动脉异常,经治疗后有5%患儿发展为冠状动脉瘤,心电图检查可发现异常复极[6]。心肌组织损伤可导致电脉冲传播改变,表现为心肌恢复时间(QT间期持续时间)的区域异质性[25]。已有研究[26]表明QTd增加可能对发生潜在致命性心律失常具有预测价值。KD患儿QTd与冠状动脉损伤和主要不良心脏事件之间的关系尚未明确。本研究结果显示,QTd预测KD患儿发生冠状动脉损伤的最佳截断值为38.5 ms,灵敏度为88.64%,特异度为77.12%。Kaplan-Meier生存分析结果显示,QTd≥39.5 ms的KD患儿发生心律失常、KD复发的可能性更大,提示QTd可能适合作为KD患儿治疗后发生冠状动脉损伤、心律失常及疾病复发的危险分层标准,临床上可通过监测KD患儿心脏异常复极来评估冠状动脉病变情况。
综上所述,QTd对评估KD患儿是否发生冠状动脉损伤具有预测价值。当QTd≥38.5 ms时,应警惕患儿冠状动脉损伤的发生;当QTd≥39.5 ms时应警惕患儿主要不良心脏事件的发生。本研究为单中心回顾性分析,具有一定局限性,今后应进行多中心研究来进一步论证。
| [1] |
KAWASAKI T. Acute febrile mucocutaneous syndrome with lymphoid involvement with specific desquamation of the fingers and toes in children[J]. Arerugi, 1967, 16(3): 178-222. |
| [2] |
SHARMA C, GANIGARA M, GALEOTTI C, et al. Multisystem inflammatory syndrome in children and Kawasaki disease: a critical comparison[J]. Nat Rev Rheumatol, 2021, 17(12): 731-748. DOI:10.1038/s41584-021-00709-9 |
| [3] |
SHOWERS CR, MAURER JM, KHAKSHOUR D, et al. Case of adult-onset Kawasaki disease and multisystem inflammatory syndrome following SARS-CoV-2 vaccination[J]. BMJ Case Rep, 2022, 15(7): e249094. DOI:10.1136/bcr-2022-249094 |
| [4] |
BECK RA, SPIVA S. Corticosteroids for the treatment of Kawasaki disease in children[J]. Am Fam Physician, 2023, 107(1): 20-21. |
| [5] |
GORELIK M, CHUNG SA, ARDALAN K, et al. 2021 American college of rheumatology/vasculitis foundation guideline for the management of Kawasaki disease[J]. Arthritis Care Res (Hoboken), 2022, 74(4): 538-548. DOI:10.1002/acr.24838 |
| [6] |
NOVAL RIVAS M, ARDITI M. Kawasaki disease: pathophysiology and insights from mouse models[J]. Nat Rev Rheumatol, 2020, 16(7): 391-405. DOI:10.1038/s41584-020-0426-0 |
| [7] |
APOORVA M, ATKAR C. A study on qt dispersion before and after thrombolysis in acute myocardial infarction, and its prognostic implications[J]. J Assoc Physicians India, 2022, 70(4): 11-12. |
| [8] |
ODENING KE, VAN DER LINDE HJ, ACKERMAN MJ, et al. Electromechanical reciprocity and arrhythmogenesis in long-QT syndrome and beyond[J]. Eur Heart J, 2022, 43(32): 3018-3028. DOI:10.1093/eurheartj/ehac135 |
| [9] |
Correction to: diagnosis, treatment, and long-term management of Kawasaki disease: a scientific statement for health professionals from the American heart association[J]. Circulation, 2019, 140 (5): e181-e184. DOI: 10.1161/CIR.0000000000000703.
|
| [10] |
NABAVIZADEH SH, MEHDIZADEGAN N, MOEINI M, et al. Assessing the association of Kawasaki disease symptoms and echocardiographic findings[J]. Pediatr Cardiol, 2022, 43(4): 816-820. DOI:10.1007/s00246-021-02792-x |
| [11] |
YENER GO, KISAARSLAN AP, ULU K, et al. Differences and similarities of multisystem inflammatory syndrome in children, Kawasaki disease and macrophage activating syndrome due to systemic juvenile idiopathic arthritis: a comparative study[J]. Rheumatol Int, 2022, 42(5): 879-889. DOI:10.1007/s00296-021-04980-7 |
| [12] |
WASEEM M, SHARIFF MA, TEIN TAY E, et al. Multisystem inflammatory syndrome in children[J]. J Emerg Med, 2022, 62(1): 28-37. DOI:10.1016/j.jemermed.2021.07.070 |
| [13] |
YANAGAWA H, NAKAMURA Y, YASHIRO M, et al. A nationwide incidence survey of Kawasaki disease in 1985-1986 in Japan[J]. J Infect Dis, 1988, 158(6): 1296-1301. DOI:10.1093/infdis/158.6.1296 |
| [14] |
ISHIKAWA T, SEKI K. The association between oxidative stress and endothelial dysfunction in early childhood patients with Kawasaki disease[J]. BMC Cardiovasc Disord, 2018, 18(1): 30. DOI:10.1186/s12872-018-0765-9 |
| [15] |
YALE SH, TEKINER H, YALE ES. Tomisaku Kawasaki and Kawasaki disease[J]. Childs Nerv Syst, 2022, 38(2): 233-235. DOI:10.1007/s00381-020-04784-3 |
| [16] |
PATEL JM. Multisystem inflammatory syndrome in children (MIS-C)[J]. Curr Allergy Asthma Rep, 2022, 22(5): 53-60. DOI:10.1007/s11882-022-01031-4 |
| [17] |
HIGHAM PD, CAMPBELL RW. QT dispersion[J]. Br Heart J, 1994, 71(6): 508-510. DOI:10.1136/hrt.71.6.508 |
| [18] |
STANKOVIC I, PUTNIKOVIC B, JANICIJEVIC A, et al. Myocardial mechanical and QTc dispersion for the detection of significant coronary artery disease[J]. Eur Heart J Cardiovasc Imag, 2015, 16(9): 1015-1022. DOI:10.1093/ehjci/jev029 |
| [19] |
HIGHAM PD, FURNISS SS, CAMPBELL RW. QT dispersion and components of the QT interval in ischaemia and infarction[J]. Br Heart J, 1995, 73(1): 32-36. DOI:10.1136/hrt.73.1.32 |
| [20] |
OYAMADA J, SHIMIZU C, KIM J, et al. Bifid T waves on the ECG and genetic variation in calcium channel voltage-dependent beta 2 subunit gene (CACNB2) in acute Kawasaki disease[J]. Congenit Heart Dis, 2019, 14(2): 213-220. DOI:10.1111/chd.12696 |
| [21] |
LIM EJ, ARIS IM, CHOO J, et al. Association between coronary artery measurements and retinal microvasculature in children with new onset of Kawasaki disease[J]. Sci Rep, 2019, 9(1): 16714. DOI:10.1038/s41598-019-53220-3 |
| [22] |
KIKUCHI Y, SATO Y, ICHIHASHI K, et al. Autonomic function in Kawasaki disease with myocardial infarction: usefulness of monitoring heart rate variability[J]. Pediatr Int, 2003, 45(4): 407-409. DOI:10.1046/j.1442-200x.2003.01742.x |
| [23] |
AE R, MADDOX RA, ABRAMS JY, et al. Kawasaki disease with coronary artery lesions detected at initial echocardiography[J]. J Am Heart Assoc, 2021, 10(7): e019853. DOI:10.1161/JAHA.120.019853 |
| [24] |
TUAN SH, LI MH, HSU MJ, et al. Cardiopulmonary function, exercise capacity, and echocardiography finding of pediatric patients with Kawasaki disease: an observational study[J]. Medicine, 2016, 95(2): e2444. DOI:10.1097/MD.0000000000002444 |
| [25] |
HUNG Y, LIN WH, LIN CS, et al. The prognostic role of QTc interval in acute myocarditis[J]. Acta Cardiol Sin, 2016, 32(2): 223-230. DOI:10.6515/acs20150226a |
| [26] |
MOGHADAM EA, HAMZEHLOU L, MOAZZAMI B, et al. Increased QT interval dispersion is associated with coronary artery involvement in children with Kawasaki disease[J]. Oman Med J, 2020, 35(1): e88. DOI:10.5001/omj.2020.06 |
 2023, Vol. 52
2023, Vol. 52




