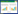文章信息
- 赵正, 王晓凤, 李茂新, 杨雪
- ZHAO Zheng, WANG Xiaofeng, LI Maoxin, YANG Xue
- 表面活性蛋白A在星形胶质细胞和小胶质细胞中的表达及炎症调节作用
- Surfactant protein A expression in astrocytes and microglia and its regulatory effects on inflammation
- 中国医科大学学报, 2021, 50(5): 418-424
- Journal of China Medical University, 2021, 50(5): 418-424
-
文章历史
- 收稿日期:2020-09-14
- 网络出版时间:2021-05-17 9:21
多发性硬化是一种中枢神经系统慢性炎症性脱髓鞘疾病,其发病机制与异常的自身免疫反应有关[1]。星形胶质细胞和小胶质细胞是中枢神经系统中的常驻免疫细胞,正常情况下处于静止状态,在病理条件下被激活并参与中枢神经系统的免疫反应调节[2-3]。在多发性硬化发病过程中,外周异常增多的自身反应性T淋巴细胞和其他炎症细胞通过血脑屏障进入中枢神经系统,激活小胶质细胞和星形胶质细胞表面受体介导的信号通路,进而产生各种促炎性细胞因子和趋化因子,参与多发性硬化的病理改变[4-5]。小胶质细胞和星形胶质细胞都表达Toll样受体4(Toll-like receptor 4,TLR4)[6-7],活化后的TLR4能促发细胞内炎症级联反应并激活核转录因子κB(nuclear factor-κB,NF-κB),进而调节多种促炎性细胞因子的表达。因此,通过TLR4/NF-κB通路介导的炎症反应在多发性硬化的病理生理过程中至关重要[8]。
表面活性蛋白A(surfactant protein A,SPA)在肺组织和肺外多个组织器官中表达并发挥免疫炎症调节作用[9]。研究[10-12]发现SPA能结合TLR4,并能抑制TLR4与脂多糖(lipopolysaccharide,LPS)结合,从而减弱其引发的NF-κB活化级联反应。然而,SPA是否在星形胶质细胞和小胶质细胞中表达,是否通过TLR4信号通路参与调节中枢神经系统的免疫炎症反应目前尚未见报道。本研究探讨SPA在星形胶质细胞和小胶质细胞中的表达及其炎症调节作用。
1 材料与方法 1.1 实验动物、细胞及主要试剂清洁级健康6~8周Lewis雌性大鼠10只,体质量180~220 g,购自中国医科大学实验动物中心。大鼠单笼饲养并自由进食专用饲料及蒸馏水,室温18~22 ℃,相对湿度40%~60%,保持垫料清洁及空气畅通并维持12 h昼夜节律,适应性饲养2周后用于实验。人星形胶质细胞及人小胶质细胞(cat.nos.1800和1900)购自美国ScienCell Research Laboratories公司,应用含10%胎牛血清的DMEM培养基在37 ℃、5% CO2的细胞培养箱中培养。
DMEM培养基和胎牛血清购自美国Gibco公司;人SPA购自美国Abcam公司;山羊SPA抗体、兔胶质纤维酸性蛋白(glial fibrillary acidic protein,GFAP)抗体、兔小胶质细胞特异性蛋白抗体钙离子(ionized calcium binding adapter molecule 1,Iba-1)、兔TLR4抗体及兔NF-κB抗体购自美国Santa公司;驴抗兔和驴抗山羊荧光二抗购自美国Invitrogen公司;驴血清、DAB染色剂及LPS购自美国Sigma公司;肿瘤坏死因子-α(tumor necrosis factor-α,TNF-α)和白细胞介素-1β(interleukin-1β,IL-1β)ELISA试剂盒购自美国R & D公司。
1.2 方法 1.2.1 星形胶质细胞及小胶质细胞中SPA检测 1.2.1.1 大鼠脑组织切片星形胶质细胞及小胶质细胞中SPA检测采用免疫荧光双染法,水合氯醛麻醉大鼠后迅速断头取脑组织,置于4%多聚甲醛中固定、石蜡包埋并切片(厚度4 μm)。切片二甲苯脱蜡,梯度乙醇水化;PBS洗涤切片5 min,3次,滴加5% BSA封闭,室温下孵育30 min;滴加SPA和GFAP混合液或SPA和Iba-1一抗混合液,保湿盒内4 ℃孵育过夜。恢复至室温,PBS洗涤切片5 min,3次,滴加杂二抗混合液,常温下于避光保湿盒内孵育2 h;PBS避光下洗涤切片5 min,3次,Hoechst 33258染核3 min;PBS避光下洗涤切片5 min,3次,加入防淬灭剂封片,用正置荧光显微镜观察星形胶质细胞及小胶质细胞中SPA的表达,实验重复3次。
1.2.1.2 体外人星形胶质细胞及小胶质细胞细胞中SPA检测采用免疫组织化学染色法,未经处理的人星形胶质细胞和小胶质细胞爬片,于盖玻片上生长3 d后进行免疫组织化学染色。切片4%多聚甲醛固定30 min,PBS洗涤5 min,3次,然后置于3% H2O2室温下孵育15 min;PBS洗涤5 min,3次,加入正常山羊血清,室温下孵育30 min;滴加SPA一抗,保湿盒内4 ℃孵育过夜;切片放至室温后PBS洗涤5 min,3次,滴加二抗,室温下孵育30 min;PBS洗涤切片5 min,3次,滴加SP试剂,室温下孵育30 min;PBS洗涤切片5 min,3次,光学显微镜下DAB显色;苏木素核复染5 min,1%盐酸乙醇中分化10 s,自来水冲洗返蓝20 min;梯度乙醇脱水,二甲苯中透明,中性树胶封片,未滴加一抗的切片作为对照,观察SPA在体外星形胶质细胞及小胶质细胞中的表达情况,实验重复3次。
1.2.2 体外细胞培养和分组(1)按照不同浓度LPS处理分组,使用含不同浓度(1、5、10 μg/mL)LPS培养液分别培养人星形胶质细胞及小胶质细胞24 h,并以0 μg/mL LPS作为对照组。(2)按照LPS处理不同时间分组,应用含有10 μg/mL LPS的培养液分别培养人星形胶质细胞及人小胶质细胞2、4、8、16、24 h,以10 μg/mL LPS的培养液培养0 h作为对照,检测各组细胞中SPA表达情况。实验重复3次。(3)按照不同处理方式分组,LPS组(5 μg/mL LPS的培养液培养细胞8 h)、LPS+SPA组(5 μg/mL LPS和0.5 μg/mL SPA的培养液培养细胞8 h)、SPA组(0.5 μg/mL SPA的培养液培养细胞8 h)、阴性对照组(未加入任何处理因素的培养基培养)。检测各组细胞中TLR4和NF-κB p65蛋白表达以及各组培养液中TNF-α和IL-1β的水平,实验重复3次。
1.2.3 Western blotting检测细胞中SPA、TLR4和NF-κB p65的表达将人星形胶质细胞和小胶质细胞以2×106/孔接种至6孔板中,按照1.2.1中分组方式处理。各组细胞用RIPA裂解液(250 μL)冰上裂解20 min,4 ℃ 12 000 r/min离心15 min;取上清行SDS-PAGE电泳。电泳结束后将凝胶中蛋白质转移至硝酸纤维素膜上。用5%脱脂奶粉封闭液内,室温封闭2 h,用TBST液洗膜5 min,3次;加入一抗,4 ℃冰箱摇床上孵育过夜。TBST漂洗膜3遍,加二抗,室温孵育2h。将PVDF膜放入显影仪面板上,加入200 μL显影液,暗室中显影、定影、洗片。利用Scion Image(美国Scion Corporation公司)软件检测各蛋白条带灰度值,将各蛋白和β-actin灰度值比值作为蛋白相对表达量,实验重复3次。
1.2.4 ELISA法检测细胞培养液中TNF-α和IL-1β的表达取各组细胞,4 ℃ 12 000 r/min离心5 min,取上清液,按照ELISA检测试剂盒说明书检测TNF-α和IL-1β的表达,实验重复3次。
1.3 统计学分析应用SPSS 22.0软件进行统计分析。计量资料采用x±s表示。正态分布的数据组间比较应用one-way ANOVA分析;非正态分布数据组间比较应用Mann Whitney U检验。P < 0.05为差异有统计学意义。
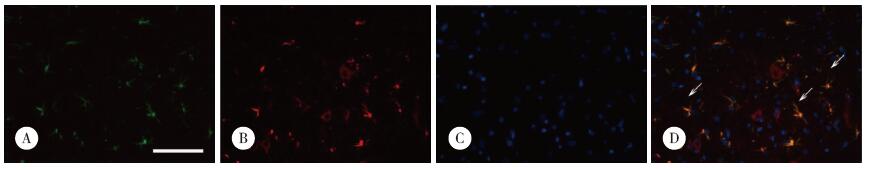
2 结果 2.1 大鼠脑组织星形胶质细胞中SPA表达大鼠脑组织中免疫荧光双重染色共定位结果显示,大鼠星形胶质细胞中可见SPA表达(图 1),而小胶质细胞未见SPA表达(图 2)。
|
| A, GFAP; B, SPA; C, Hoechst 33258;D, merged. SPA+ cells are stained red, GFAP+ cells are stained green, and the nucleus are stained blue by Hoechst 33258. GFAP+/SPA+ astrocytes are costained yellow (indicated by the arrow). Scale bar = 50 μm. 图 1 SPA和GFAP双重免疫荧光染色 Fig.1 Double-immunofluorescence staining for SPA and GFAP detection |
|
| A, Iba-1;B, SPA; C, Hoechst 33258;D, merged. SPA+ cells are stained red, Iba-1+ cells are stained green, and the nucleus are stained blue by Hoechst 33258. SPA and Iba-1 do not show co-localization signal (indicated by the arrow). Scale bar = 50 μm. 图 2 SPA和Iba-1双重免疫荧光染色 Fig.2 Double-immunofluorescence staining for SPA and Iba-1 detection |
2.2 体外培养人星形胶质细胞和人小胶质细胞中SPA表达
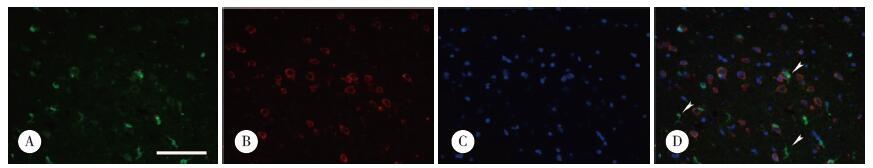
免疫组化结果(图 3)显示,人星形胶质细胞的胞质和胞核中均可见SPA表达(图 3A),与大鼠脑组织不同,人小胶质细胞的胞质中可见SPA表达(图 3C)。
|
| A, fixed human astrocytes were stained for SPA; B, fixed human astrocytes were stained with secondary antibody only; C, fixed human microglial cells were stained for SPA; D, fixed human microglial cells were stained with secondary antibody only. scale bar = 10 μm. 图 3 体外培养人星形胶质细胞和小胶质细胞中SPA免疫组织化学染色结果 Fig.3 Immunohistochemical staining of SPA in human astrocytes and microglial cells |
2.3 LPS处理对体外培养的人星形胶质细胞和人小胶质细胞中SPA表达的影响
应用不同浓度(0~10 μg/mL)LPS处理细胞24 h后的检测结果显示,人星形胶质细胞和人小胶质细胞中SPA水平均随LPS浓度上升而增高。与对照组(0 μg/mL LPS)比较,1、5 μg/mL LPS处理组人星形胶质细胞中SPA表达水平无统计学差异(P > 0.05),而10 μg/mL LPS处理组SPA水平显著升高(P < 0.05);1 μg/mL LPS处理组人小胶质细胞中SPA水平无统计学差异(P > 0.05),而5、10 μg/mL LPS处理组SPA水平显著升高(P < 0.05),且10 μg/mL LPS处理组SPA水平较1、5 μg/mL LPS处理组显著升高(P < 0.05)。同时,采用10 μg/mL LPS分别处理人星形胶质细胞和人小胶质细胞2、4、8、16、24 h结果显示,2种细胞都没有出现时间依赖性的变化趋势。见图 4、表 1、表 2。
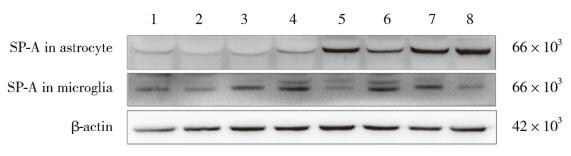
|
| 1, 0 μg/mL LPS for 24 h; 2, 1 μg/mL LPS for 24 h; 3, 5 μg/mL LPS for 24 h; 4, 10 μg/mL LPS for 24 h; 5, 10 μg/mL LPS for 2 h; 6, 10 μg/mL LPS for 4 h; 7, 10 μg/mL LPS for 8 h; 8, 10 μg/mL LPS for 16 h. 图 4 不同浓度和不同时间LPS处理的人星形胶质细胞和小胶质细胞中SPA的表达 Fig.4 SPA expression in human astrocytes and microglia cells exposed to varying LPS doses, or to 10 µg/ml LPS for varying durations |
| Group | SPA expression | |
| Astrocyte | Microglia | |
| 0 μg/mL LPS | 0.226 2±0.011 5 | 0.242 5±0.008 6 |
| 1 μg/mL LPS | 0.267 4±0.016 7 | 0.268 8±0.015 8 |
| 5 μg/mL LPS | 0.315 1±0.025 4 | 0.548 0±0.038 31) |
| 10 μg/mL LPS | 0.471 1±0.039 01) | 0.793 6±0.052 61), 2) |
| 1)P < 0.05 vs 0 μg/mL LPS group;2)P < 0.05 vs 1,5 μg/mL LPS groups. | ||
| Group | SPA expression | |
| Astrocyte | Microglia | |
| 2 h group | 0.982 2±0.049 8 | 0.530 2±0.015 6 |
| 4 h group | 0.720 3±0.025 0 | 1.039 8±0.087 8 |
| 8 h group | 1.223 3±0.090 3 | 0.738 5±0.039 8 |
| 16 h group | 1.164 0±0.073 7 | 0.407 1±0.010 6 |
| 24 h group | 0.471 1±0.039 0 | 0.793 6±0.052 6 |
2.4 SPA抑制LPS诱导的TLR4/NF-κB信号通路活化
结果显示,与阴性对照组比较,LPS组星形胶质细胞中TLR4和NF-κB p65表达水平均显著升高(P < 0.05);与LPS组比较,LPS+SPA组TLR4和NF-κB p65水平均显著下降(P < 0.05)。而SPA组与阴性对照组比较TLR4与NF-κB p65表达均没有统计学差异(均P > 0.05)。小胶质细胞与星形胶质细胞培养的实验结果一致,SPA同样抑制了LPS诱导的TLR4和NF-κB p65表达,见图 5、表 3。
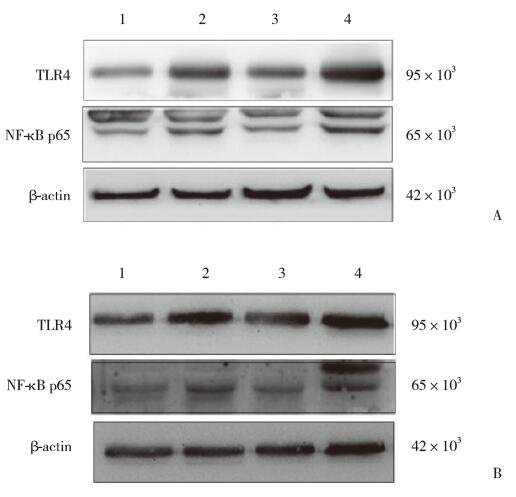
|
| A, astrocyte; B, microglia. 1, SPA group; 2, SPA+LPS group; 3, control group; 4, LPS group. 图 5 各组人星形胶质细胞和小胶质细胞中TLR4和NF-κB p65表达 Fig.5 TLR4 and NF-κB p65 proteins in human astrocytes and microglia |
| Group | TLR4 | NF-κB p65 | |||
| Astrocyte | Microglia | Astrocyte | Microglia | ||
| Control | 0.554 7±0.023 2 | 1.232 4±0.091 3 | 0.734 3±0.044 4 | 0.781 9±0.045 7 | |
| LPS | 1.460 8±0.097 31) | 2.171 8±0.137 51) | 1.484 3±0.075 11) | 1.705 5±0.123 01) | |
| SPA | 0.516 1±0.025 0 | 1.055 0±0.086 9 | 0.542 5±0.017 4 | 0.640 7±0.020 2 | |
| SPA+LPS | 0.862 2±0.038 92) | 1.614 4±0.107 82) | 1.114 3±0.083 12) | 1.145 0±0.099 32) | |
| 1)P < 0.05 vs control group;2)P < 0.05 vs LPS group. | |||||
2.5 SPA抑制LPS诱导的TNF-α和IL-1β的产生
结果显示,与阴性对照组比较,LPS组的人星形胶质细胞和人小胶质细胞培养液中的TNF-α和IL-1β水平均显著升高(P < 0.05)。人星形胶质细胞培养液中,LPS+SPA组与LPS组比较IL-1β显著减少(P < 0.05),而2组间TNF-α水平没有统计学差异(P > 0.05);而在人小胶质细胞培养液中LPS+SPA组TNF-α和IL-1β水平较LPS组均明显下降(P < 0.05)。SPA组与阴性对照组TNF-α和IL-1β水平在2种细胞培养液中均无统计学差异(P > 0.05)。见表 4。
| Group | TNF-α | IL-1β | |||
| Astrocyte | Microglia | Astrocyte | Microglia | ||
| Control | 364.33±18.98 | 937.04±85.70 | 149.10±9.426 | 155.98±10.96 | |
| LPS | 1 625.40±145.271) | 2 451.00±157.861) | 457.98±20.051) | 581.73±19.781) | |
| SPA | 448.18±29.39 | 1 156.67±83.91 | 136.62±10.35 | 174.02±12.12 | |
| SPA+LPS | 1 281.68±94.01 | 1 811.14±99.812) | 248.11±13.432) | 400.61±23.772) | |
| 1)P < 0.05 vs control group;2)P < 0.05 vs LPS group. | |||||
3 讨论
多发性硬化及实验性自身免疫性脑脊髓炎病理过程的发生发展与中枢神经系统的固有免疫细胞——星形胶质细胞和小胶质细胞的免疫调节作用息息相关。在多发性硬化及实验性自身免疫性脑脊髓炎的发病过程中,外周异常活化的免疫细胞(CD4+T细胞等)穿过受损的血脑屏障侵入中枢神经系统,结合并激活静止状态下的星形胶质细胞和小胶质细胞。活化后的胶质细胞通过多种炎症信号通路产生并释放促炎性细胞因子及趋化因子等介质,发挥抗原提呈等多种免疫功能参与神经免疫的调控[13-14]。本研究采用免疫荧光双染方法观察SPA在大鼠脑内星形胶质细胞和小胶质细胞中的表达,结果发现在大鼠脑组织星形胶质细胞胞质中可见SPA表达,而小胶质细胞不表达;但在体外培养的人星形胶质细胞和小胶质细胞中都观察到SPA表达,这可能与SPA在不同物种间的表达差异有关。近年来,研究[9-10]发现多种组织器官的炎症疾病中都可检测到SPA水平变化,认为SPA水平可反映炎症反应的严重程度。
LPS是一种炎症激活物质,常用于建立体外实验的炎症模型。本研究结果证实在星形胶质细胞和小胶质细胞中LPS以剂量依赖方式促进SPA表达,参与两种胶质细胞相关的炎症反应调节。研究[12-16]证实SPA能通过特异性受体介导并调节下游的炎症信号通路,SPA能直接与细胞表面的TLR4结合,并能抑制TLR4与LPS的结合,从而减弱其引发的NF-κB活化级联反应。TLR4是LPS最适合的结合受体之一[17],TLR4在星形胶质细胞和小胶质细胞表面表达丰富,能够介导胶质细胞的活化和炎性细胞因子表达,是2种胶质细胞发挥免疫功能的重要受体蛋白。研究[17-18]证实,在LPS刺激下,胶质细胞表达的TLR4及其下游NF-κB水平都随LPS浓度增加而升高。由此推测SPA可能参与调节2种胶质细胞的TLR4及其下游的转录因子NF-κBp65的表达。本研究结果显示,LPS刺激能使人星形胶质细胞和小胶质细胞中表达的TLR4和NF-κB p65水平升高,并促进了炎性细胞因子TNF-α和IL-1β的释放,而加入外源性SPA能抑制LPS诱导的TLR4和NF-κB p65表达以及炎性细胞因子释放。单独加入SPA并不能产生促进或抑制TLR4和NF-κB活化的作用。因此认为在中枢神经系统炎症反应过程中,SPA能够抑制星型胶质细胞和小胶质细胞中TLR4/NF-κB活化,减少下游炎性细胞因子释放,并通过这种途径抑制两种胶质细胞的免疫炎症反应。
SPA在中枢神经系统中作用于星形胶质细胞和小胶质细胞的机制可能十分复杂:(1)SPA是TLR4的配体,SPA有可能通过结合TLR4来抑制其活化,影响炎症信号通路激活而抑制炎症反应;(2)SPA又能结合LPS,它亦或通过与LPS相互作用干扰其结合TLR4而产生抑制炎症反应作用[19];(3)SPA还是多个受体(TLR2、CD14和gp-340等)的内源性配体,这些受体都能促发炎症级联反应,LPS诱导SPA表达升高有可能反过来通过结合这些抗体调节了其下游信号通路[20-21]。SPA在人星形胶质细胞和小胶质细胞中表现出的抗炎症作用可能是以上3种途径综合作用的结果,但这些推论仍需要进一步研究来证实。
综上所述,SPA在大鼠脑组织星形胶质细胞以及人星形胶质细胞和小胶质细胞中表达,推测SPA是通过调节星型胶质细胞和小胶质细胞的TLR4信号通路参与中枢神经系统的免疫炎症调节。本研究为SPA在中枢神经系统中的免疫调控作用提供了新的证据。
| [1] |
POPESCU BFG, LUCCHINETTI CF. Pathology of demyelinating diseases[J]. Annu Rev Pathol, 2012, 7: 185-217. DOI:10.1146/annurev-pathol-011811-132443 |
| [2] |
FILES DK, JAUSURAWONG T, KATRAJIAN R, et al. Multiple sclerosis[J]. Prim Care: Clin Off Pract, 2015, 42(2): 159-175. DOI:10.1016/j.pop.2015.01.007 |
| [3] |
MORENO M, GUO F, MILLS KO E, et al. Origins and significance of astrogliosis in the multiple sclerosis model, MOG peptide EAE[J]. J Neurol Sci, 2013, 333(1-2): 55-59. DOI:10.1016/j.jns.2012.12.014 |
| [4] |
PYKA-FOŚCIAK G, ZEMŁA J, LIS GJ, et al. Changes in spinal cord stiffness in the course of experimental autoimmune encephalomyelitis, a mouse model of multiple sclerosis[J]. Arch Biochem Biophys, 2020, 680: 108221. DOI:10.1016/j.abb.2019.108221 |
| [5] |
LUNDGAARD I, OSORIO MJ, KRESS BT, et al. White matter astrocytes in healthy and disease[J]. Neurol Sci, 2014, 276: 161-173. DOI:10.1016/j.neuroscience.2013.10.050 |
| [6] |
YAO LL, KAN EM, LU J, et al. Toll-like receptor 4 mediates microglial activation and production of inflammatory mediators in neonatal rat brain following hypoxia: role of TLR4 in hypoxic microglia[J]. J Neuroinflammation, 2013, 10(1): 785. DOI:10.1186/1742-2094-10-23 |
| [7] |
WU YQ, LI W, ZHOU C, et al. Ketamine inhibits lipopolysaccharide-induced astrocytes activation by suppressing TLR4/NF-κB pathway[J]. Cell Physiol Biochem, 2012, 30(3): 609-617. DOI:10.1159/000341442 |
| [8] |
DEERHAKE ME, BISWAS DD, BARCLAY WE, et al. Pattern recognition receptors in multiple sclerosis and its animal models[J]. Front Immunol, 2019, 10: 2644. DOI:10.3389/fimmu.2019.02644 |
| [9] |
NAYAK A, DODAGATTA-MARRI E, TSOLAKI AG, et al. An insight into the diverse roles of surfactant proteins, SP-A and SP-D in innate and adaptive immunity[J]. Front Immunol, 2012, 3: 131. DOI:10.3389/fimmu.2012.00131 |
| [10] |
赵正, 杨雪. 肺表面活性蛋白A在肺外疾病免疫炎性反应中的作用研究进展[J]. 疑难病杂志, 2020, 19(4): 423-427, 432. DOI:10.3969/j.issn.1671-6450.2020.04.023 |
| [11] |
SENDER V, LANG L, STAMME C. Surfactant protein-A modulates LPS-induced TLR4 localization and signaling via β-arrestin 2[J]. PLoS One, 2013, 8(3): e59896. DOI:10.1371/journal.pone.0059896 |
| [12] |
YAMADA C, SANO H, SHIMIZU T, et al. Surfactant protein A directly interacts with TLR4 and MD-2 and regulates inflammatory cellular response. Importance of supratrimeric oligomerization[J]. J Biol Chem, 2006, 281(31): 21771-21780. DOI:10.1074/jbc.M513041200 |
| [13] |
NAPOLI I, NEUMANN H. Protective effects of microglia in multiple sclerosis[J]. Exp Neurol, 2010, 225(1): 24-28. DOI:10.1016/j.expneurol.2009.04.024 |
| [14] |
LUNDGAARD I, OSÓRIO MJ, KRESS BT, et al. White matter astrocytes in health and disease[J]. Neuroscience, 2014, 276: 161-173. DOI:10.1016/j.neuroscience.2013.10.050 |
| [15] |
LIU J, HU FQ, WANG GR, et al. Lipopolysaccharide-induced expression of surfactant proteins A_1 and A_2 in human renal tubular epithelial cells[J]. J Inflamm, 2013, 10(1): 1-8. DOI:10.1186/1476-9255-10-2 |
| [16] |
BHATTI F, BALL G, HOBBS R, et al. Pulmonary surfactant protein a is expressed in mouse retina by Müller cells and impacts neovascularization in oxygen-induced retinopathy[J]. Investig Ophthalmol Vis Sci, 2014, 56(1): 232-242. DOI:10.1167/iovs.13-13652 |
| [17] |
PATRA MC, SHAH M, CHOI S, et al. Toll-like receptor-induced cytokines as immunotherapeutic targets in cancers and autoimmune diseases[J]. Semin Cancer Biol, 2020, 64: 61-82. DOI:10.1016/j.semcancer.2019.05.002 |
| [18] |
LEITNER GR, WENZEL TJ, MARSHALL N, et al. Targeting toll-like receptor 4 to modulate neuroinflammation in central nervous system disorders[J]. Expert Opin Ther Targets, 2019, 23(10): 865-882. DOI:10.1080/14728222.2019.1676416 |
| [19] |
KUMAR V. Toll-like receptors in the pathogenesis of neuroinflammation[J]. J Neuroimmunol, 2019, 332: 16-30. DOI:10.1016/j.jneuroim.2019.03.012 |
| [20] |
AUTILIO C, PÉREZ-GIL J. Understanding the principle biophysics concepts of pulmonary surfactant in health and disease[J]. Arch Dis Child Fetal Neonatal Ed, 2019, 104(4): F443-F451. DOI:10.1136/archdischild-2018-315413 |
| [21] |
JAKEL A, QASEEM AS, KISHORE U, et al. Ligands and receptors of lung surfactant proteins SP-A and SP-D[J]. Front Biosci Landmark Ed, 2013, 18: 1129-1140. DOI:10.2741/4168 |
 2021, Vol. 50
2021, Vol. 50