文章信息
- 黄天楚, 黄琬淇, 顾卉, 袁正伟
- HUANG Tianchu, HUANG Wanqi, GU Hui, YUAN Zhengwei
- COLIA1调控小鼠神经细胞迁移在神经管畸形中的作用
- Effect of COLIA1 in regulating mice neurocyte migration in neural tube defects
- 中国医科大学学报, 2021, 50(11): 966-969
- Journal of China Medical University, 2021, 50(11): 966-969
-
文章历史
- 收稿日期:2020-12-14
- 网络出版时间:2021-11-03 14:51
神经管畸形(neural tube defects,NTDs)是胚胎发育过程中神经板无法闭合形成神经管而造成的,全世界约有3%新生儿患有这种缺陷,是导致围产期婴儿死亡的原因之一[1]。NTDs通常分为开放性缺损(颅脊柱裂、无脑畸形和脊髓脊膜膨出)和闭合性缺损(脑膨出、脑膜膨出和隐性脊柱裂)[2]。
细胞迁移是神经管闭合过程中重要的生物学功能之一。随着细胞的迁移会失去其极性和细胞间黏附性,细胞间黏附分子发生改变,使上皮细胞转变成具有侵袭性的间充质细胞,这一过程被称为上皮细胞间质转化(epithelial mesenchymal transition,EMT)[3]。Ⅰ型胶原蛋白α1(collagen type Ⅰα 1,COLIA1)是胶原蛋白家族的成员,广泛分布于骨骼、软骨、皮肤、心脏瓣膜、肺以及结缔组织中[4]。研究[5]显示,COLIA1突变可能会导致胚胎发育过程中的成骨不全。此外,SUN等[6]研究表明,COLIA1可能通过NF-κB信号通路调节恶性星形细胞瘤细胞的侵袭能力,促进肿瘤细胞的迁移。目前,COLIA1与NTDs的关系研究尚未见报道。本研究探讨全反式维甲酸(all-transretinoic acid,ATRA)诱导的小鼠NTDs模型中COLIA1表达对神经细胞迁移活动的影响,旨在为阐明NTDs的发生机制提供新的理论依据。
1 材料与方法 1.1 NTDs模型制备及组织样本收集8~10周龄C57B1/6J雌性小鼠(北京华阜康生物技术有限公司)30只,饲养于中国医科大学附属盛京医院SPF级实验动物房。于前1 d晚合笼,第2天观察雌性小鼠有无阴道栓存在,有阴道栓雌性小鼠单独放一笼,标记为孕0 d(E0)。在E8.5将已孕小鼠随机分为NTDs组[将ATRA(70 mg/kg)溶于橄榄油后,给予一次灌胃处理,n = 15)、对照组(给予同体积的橄榄油灌胃,n = 15)。于E9.5处死,剖宫取出胎鼠。
1.2 细胞培养和转染小鼠神经干细胞C17.2培养于含10%胎牛血清(美国Gibco公司)、1%MEM NEAA(美国Gibco公司)、1%双抗(100 U/mL青霉素、100 μg/mL链霉素)的EMEM(美国Gibco公司)中,于37 ℃,5%CO2的细胞培养箱中培养。培养24 h后转染sh-negative control(sh-NC)、sh-COLIA1 1#、sh-COLIA1 2#、sh-COLIA1 3#(中国吉玛公司)至C17.2细胞。
1.3 Western blotting检测从胚胎和转染后48 h的C17.2细胞中用RIPA提取蛋白质。通过8%的SDS-PAGE电泳分离样品蛋白,然后电转印至PVDF膜上,用5%脱脂牛奶封闭2 h。一抗COLIA1(1∶1 000,美国Cell Signal Technology公司)、E-cadherin(1∶1 000,美国BD公司),Snail(1∶1 000,中国万类公司)、Vimentin(1∶1 000,中国Proteintech公司)、β-actin(1∶1 000,中国Proteintech公司)4 ℃下过夜。第2天用TBST室温洗膜3次,每次10 min,然后在室温下与HRP兔和鼠二抗(1∶5 000,中国Proteintech公司)孵育2 h,ECL试剂发光。
1.4 细胞划痕实验在6孔板中加入约5×105细胞,第2天用枪头进行划痕,PBS洗涤细胞3次去除划下的细胞,加入无血清培养基,在5% CO2的细胞培养箱中培养48 h后拍照。
1.5 细胞Transwell迁移实验转染细胞撤血清饥饿12~24 h后消化细胞,终止消化后离心弃去培养液,PBS洗1~2次后无血清培养基重悬,调整细胞密度为5×105/mL。取细胞悬液100 μL加入8孔Transwell小室的上室,下室加入600 μL含血清的培养基,培养细胞12~48 h。取出Transwell小室,弃去孔中培养液,甲醇固定30 min,将小室适当风干。0.1%结晶紫染色20 min,用棉签轻轻擦掉上层未迁移细胞,PBS洗3次。显微镜(×200)下随机5个视野计数细胞。
1.6 统计学分析利用SPSS 26.0软件进行统计分析,GraphPad8.0统计软件作图,计量资料以x±s表示;组间比较采用独立样本t检验,P < 0.05为差异有统计学意义。
2 结果 2.1 COLIA1在ATRA致畸小鼠模型中的表达Western blotting检测E9.5小鼠胚胎组织COLIA1的表达结果显示,对照组、NTDs组COLIA1表达分别为1.132±0.118,0.875±0.106。与对照组比较,NTDs组COLIA1表达显著降低(P < 0.05),COLIA1的表达对NTDs的形成可能起到抑制作用。见图 1。
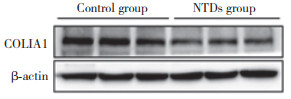
|
| 图 1 E9.5天小鼠胚胎正常组和NTDs组COLIA1的表达 Fig.1 Expression of COLIA1 in E9.5 mice embryos between control and NTDs groups |
2.2 对照组和NTDs组E-cadherin、Vimentin和Snail表达比较
结果显示,与对照组比较,NTDs组Vimentin和Snail表达降低,而E-cadherin表达上升(均P < 0.05)。说明在NTDs胚胎中细胞迁移能力降低,EMT进程减慢,推测COLIA1可能抑制神经管闭合中迁移活动以及EMT的发生。见图 2、表 1。
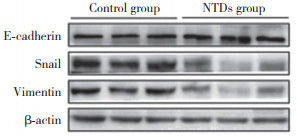
|
| 图 2 对照组和NTDs组E-cadherin、Vimentin和Snail的表达 Fig.2 Expression of E-cadherin, Vimentin, and Snail between control and NTDs groups |
| Group | E-cadherin | Vimentin | Snail |
| Control | 0.428 9±0.132 | 0.624 3±0.331 | 0.374 1±0.164 |
| NTDs | 0.749 2±0.0311) | 0.370 5±0.3431) | 0.278 1±0.0631) |
| 1)P < 0.05 vs control group. | |||
2.3 COLIA1对神经细胞迁移活动的影响
为了探究COL1A对神经细胞迁活动的影响,分别在C17.2细胞中转染sh-NC、sh-COLIA1 1#、sh-COLIA1 2#、sh-COLIA1 3#载体48 h后进行Western blotting检测,结果显示沉默COLIA1时,沉默2链效果更明显,因此后续实验应用sh-COLIA1 2#载体。结果显示,与对照组比较,sh-COLIA1 2#组E-cadherin表达明显上升(P < 0.05),而Snail、Vimentin表达明显下降(P < 0.05),提示COLIA1能够促进EMT的发生,见表 2、图 3。此外,细胞划痕以及Transwell迁移实验结果显示沉默COLIA1可明显抑制神经细胞的迁移(图 4)。
| Group | E-cadherin | Vimentin | Snail |
| Sh-NC | 0.432 5±0.152 | 0.563 8±0.122 | 0.983 3±0.153 |
| Sh-COLIA1 2# | 0.605 9±0.12311) | 0.369 0±0.2931) | 0.255 5±0.1741) |
| 1)P < 0.05 vs sh-NC group. | |||
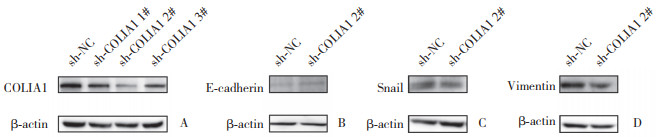
|
| A,COLIA1 expression;B,E-cadherin expression;C,Snail expression;D,Vimentin expression. 图 3 转染COLIA1后各组E-cadherin、Vimentin和Snail表达比较 Fig.3 Comparison of E-cadherin, Vimentin, and Snail expression after transfecting sh-NC and sh-COLIA1 |
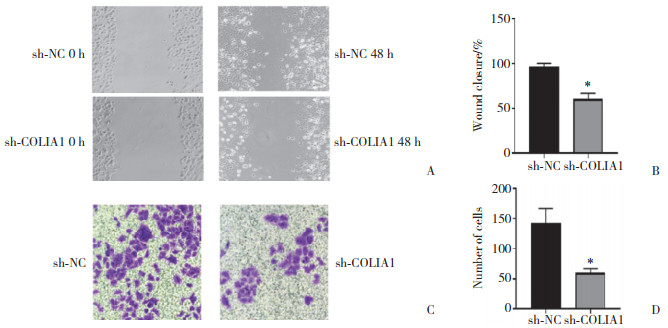
|
| A, effect of transfected cells scratch test after 48 h(×200); B, statistical analysis results of scratch test after transfection; C, effect of transfected cells migration(×200); D, statistical analysis results of migration after transfection. * P < 0.05 vs sh-NC group. 图 4 迁移和划痕试验检测转染后C17.2细胞迁移功能的改变 Fig.4 Effect of migration and scratch test of C17.2 cells after transfection |
3 讨论
神经嵴干细胞具有强大迁移能力和多项分化潜能,是具有迁徙功能的过渡性多能细胞群体[7]。随着神经褶皱开始闭合形成神经管,于神经外胚层和非神经外胚层边界的神经嵴脱层,沿着整个前后体轴广泛迁移,分化形成其他不同结构[7]。在神经板闭合为神经管的过程中,EMT进程参与神经细胞的迁移、游走和定植的调控。在EMT进程中,具有上皮特性E-cadherin随着EMT的发生表达逐渐降低[8]。E-cadherin主要定位于胚胎表皮外胚层细胞和神经板边界,随着细胞的迁移,神经板内陷,中央神经板逐渐与外胚层分离,E-cadherin逐渐失去。因此,E-cadherin表达增高,细胞迁移失败,导致神经管闭合失败[9]。Snail作为EMT转录抑制因子可与编码E-cadherin基因的启动子中保守的E-box序列相结合,使E-cadherin的表达下调,并且研究[10]证实,Snail高表达可促进了神经嵴细胞的迁移,并参与神经管的分层。此外,研究显示在NTDs胎儿中Vimentin表达下调[11]。由此可见,神经细胞的迁移活动对神经管的闭合有重要作用,并且EMT标志物会伴随神经管闭合过程变化,与本研究结果一致。已有研究[12-13]报道转录因子Pax3对于明确神经板边界和促进神经嵴细胞的形成至关重要,并且调控EMT的关键转录因子Snail可作为Pax3的直接靶点。本研究发现,在神经管闭合过程中COLIA1能够引起EMT标志物E-cadherin、Snail和Vimentin表达的变化,进而调控细胞迁移,但这些EMT标志物是否为COLIA1的直接靶点以及具体的调控机制还有待进一步研究论证。
COLIA1可作为成骨标志物参与成骨的形成,还可作为哺乳动物最丰富的细胞外间质(extracellular matrix,ECM)之一[14]。而ECM周期性重塑对细胞的迁移活动同样至关重要。已有大量研究证实COLIA1调控细胞迁移活动。COLIA1可促进胃癌细胞的迁移和侵袭,COLIA1过表达诱导TGF-β信号通路激活,进而促进EMT进程[15]。本研究在NTDs模型中发现COLIA1表达降低且伴随EMT相关指标异常。另外,当沉默C17.2细胞的COLIA1表达后,细胞的迁移活动降低,促进EMT进程标志物表达下降。由此表明,COLIA1表达降低可能抑制了神经管发育过程中神经细胞的迁移活动。
综上所述,COLIA1能够促进细胞的迁移活动以及EMT进程,进而减少NTDs的发生。COLIA1有望成为治疗NTDs的新靶点,然而关于COLIA1如何具体调控EMT进程中的关键分子以及EMT上游分子(PAX3等)与COLIA1之间的关系尚不明确,有待于进一步研究论证。
| [1] |
ENDALIFER ML, DIRESS G. Epidemiology and determinant factors of neural tube defect: narrative review[J]. Surg Neurol Int, 2020, 11: 81. DOI:10.25259/sni_84_2020 |
| [2] |
MCCOMB JG. A practical clinical classification of spinal neural tube defects[J]. Childs Nerv Syst, 2015, 31(10): 1641-1657. DOI:10.1007/s00381-015-2845-9 |
| [3] |
LAMOUILLE S, XU J, DERYNCK R. Molecular mechanisms of epithelial-mesenchymal transition[J]. Nat Rev Mol Cell Biol, 2014, 15(3): 178-196. DOI:10.1038/nrm3758 |
| [4] |
LU Y, ZHANG S, WANG Y, et al. Molecular mechanisms and clinical manifestations of rare genetic disorders associated with type I collagen[J]. Intractable Rare Dis Res, 2019, 8(2): 98-107. DOI:10.5582/irdr.2019.01064 |
| [5] |
YIN XJ, DU Y, ZHANG H, et al. Identification of a de novo fetal variant in osteogenesis imperfecta by targeted sequencing-based noninvasive prenatal testing[J]. J Hum Genet, 2018, 63(11): 1129-1137. DOI:10.1038/s10038-018-0489-9 |
| [6] |
SUN S, WANG Y, WU Y, et al. Identification of COLIA1 as an invasion-related gene in malignant astrocytoma[J]. Int J Oncol, 2018, 53(6): 2542-2554. DOI:10.3892/ijo.2018.4568 |
| [7] |
MÉNDEZ-MALDONADO K, VEGA-LÓPEZ GA, AYBAR MJ, et al. Neurogenesis from neural crest cells: molecular mechanisms in the formation of cranial nerves and Ganglia[J]. Front Cell Dev Biol, 2020, 8: 635. DOI:10.3389/fcell.2020.00635 |
| [8] |
PYRGAKI C, LIU A, NISWANDER L. Grainyhead-like 2 regulates neural tube closure and adhesion molecule expression during neural fold fusion[J]. Dev Biol, 2011, 353(1): 38-49. DOI:10.1016/j.ydbio.2011.02.027 |
| [9] |
ZOHN IE, LI Y, SKOLNIK EY, et al. p38 and a p38-interacting protein are critical for downregulation of E-cadherin during mouse gastrulation[J]. Cell, 2006, 125(5): 957-969. DOI:10.1016/j.cell.2006.03.048 |
| [10] |
TUCKER RP. Neural crest cells: a model for invasive behavior[J]. Int J Biochem Cell Biol, 2004, 36(2): 173-177. DOI:10.1016/S1357-2725(03)00243-7 |
| [11] |
ARONNE MP, EVRARD SG, MIROCHNIC S, et al. Prenatal ethanol exposure reduces the expression of the transcriptional factor Pax6 in the developing rat brain[J]. Ann N Y Acad Sci, 2008, 1139(1): 478-498. DOI:10.1196/annals.1432.006 |
| [12] |
PLOUHINEC JL, ROCHE DD, PEGORARO C, et al. Pax3 and Zic1 trigger the early neural crest gene regulatory network by the direct activation of multiple key neural crest specifiers[J]. Dev Biol, 2014, 386(2): 461-472. DOI:10.1016/j.ydbio.2013.12.010 |
| [13] |
HONG CS, SAINT-JEANNET JP. Znf703, a novel target of Pax3 and Zic1, regulates hindbrain and neural crest development in Xenopus[J]. Genesis, 2017, 55: e23082. DOI:10.1002/dvg.23082 |
| [14] |
LI H, CHANG HM, SHI Z, et al. The p38 signaling pathway mediates the TGF-β1-induced increase in typeⅠcollagen deposition in human granulosa cells[J]. Faseb J, 2020, 34(11): 15591-15604. DOI:10.1096/fj.202001377r |
| [15] |
GUO Y, LU GC, MAO HH, et al. miR-133b suppresses invasion and migration of gastric cancer cells via the COLIA1/TGF-β axis[J]. Oncotargets Ther, 2020, 13: 7985-7995. DOI:10.2147/OTT.S249667 |
 2021, Vol. 50
2021, Vol. 50




