文章信息
- 王洪秀, 张倩, 王玲杰, 唐科志
- WANG Hong-xiu, ZHANG Qian, WANG Ling-jie, TANG Ke-zhi
- 链格孢菌毒素合成相关基因研究进展
- Advances in Genetic Research Related to the Synthesis of Alternaria alternata Toxins
- 中国生物工程杂志, 2015, 35(11): 92-98
- China Biotechnology, 2015, 35(11): 92-98
- http://dx.doi.org/10.13523/j.cb.20151113
-
文章历史
- 收稿日期: 2015-06-11
- 修回日期: 2015-06-25
链格孢菌在自然界中广泛分布在植物、土壤、有机质中,致病型的链格孢菌对寄主植物产生毒素。链格孢菌产生的毒素分为非寄主选择性毒素(nonspecific toxin,NHST) 和寄主选择性毒素(host-selective toxin,HST)。NHST种类很多,包括链格孢酸(alternaric acid)、细链格孢毒素(tentoxin)、细链格孢酮酸(tenuazonic acid)等,它们在致病过程中常起着毒力因子(virulence factor)的作用,加重病害[1]。 HST是决定链格孢菌寄生范围和寄主高度专化性的致病因子(pathogenecity factor),通常是具有不同结构的低分子质量次级代谢产物,只对具有毒素受体(receptor)的相应寄主品种具有毒性。产HST的链格孢属真菌及其寄主范围、毒素名称、产毒基因等详见表 1。
| 链格孢菌致病型 | 寄主范围 | HST | HST基因 | 基因产物 | CD染色体 | 作用位点 |
| 橘致病型 | 桔及其杂种 | ACT-毒素Ⅰ和Ⅱ | ACTT基因 | ACT-毒的EDA部分 | ﹤2.0 | 质膜 |
| 粗柠檬致病型 | 粗柠檬和兰谱 | ACR-毒素Ⅰ | ACRT基因 | ACR-毒素的聚酮化合物 | ﹤1.5 | 线粒体 |
| 日本梨致病型 | 日本梨 | AK-毒素Ⅰ和Ⅱ | AKT基因 | AK-毒素的EDA部分 | ﹤2.0 | 质膜 |
| 苹果致病型 | 苹果 | AM-毒素Ⅰ、Ⅱ和Ⅲ | AMT基因 | AM-毒素的环状缩酚酸肽 | ﹤1.8 | 质膜和叶绿体 |
| 草莓致病型 | 草莓 | AF-毒素Ⅰ、Ⅱ和Ⅲ | AFT基因 | AF-毒素的EDA部分 | 1.0 | 质膜 |
| 番茄致病型 | 番茄 | AAL-毒素Ta和Tb | ALT基因 | AAL-毒素的聚酮化合物 | 1.0 | 神经酰胺合酶 |
| 烟草致病型 | 烟草 | AT-毒素 | 未知 | 未知 | 未知 | 线粒体 |
| 芸苔链格孢 | 白菜 | AB毒素:腐败菌素B、高腐败菌素B、腐败菌素B2和脱甲基腐败菌素 | 未知 | 未知 | 未知 | 质膜和叶绿体 |
| 细极链格孢 | 木豆 | AP毒素 | 未知 | 未知 | 未知 | 线粒体 |
控制HST合成的基因组区域不是简单的遗传位点,而是由特殊的复制、重组事件形成的大而复杂的DNA区域[1]。这些区域内,链格孢菌参与HST毒素生物合成的基因位于基因组的相同位点共同表达,这些基因被称为基因簇[12](gene cluster)。1999年首先在日本梨致病型中得到了参与AK-毒素生物合成的基因簇,随后在草莓致病型、橘致病型、苹果致病型、番茄致病型、粗柠檬致病型都发现了合成HST的基因簇[2, 3, 5, 13, 14, 15]。
HST基因簇的特点包括具有类转座子、由多拷贝基因组成、位于CD染色体上。
某一通路的基因聚集成基因簇,提高了这一通路的全部基因在单一事件中发生水平基因转移(horizontal gene transfer,HGT)的可能性[12]。在AFT和AMT基因簇区域发现很多具有不完整转座酶(transposase)ORF的类转座子序列(transposon-like sequence),它们是失活的转座子化石[4]。Tsuge等[1]认为,AFT、AMT、AAL等基因簇在形成过程中发生了HGT,其中AAL基因簇起源于镰刀菌属(Fusarium),并通过HGT从镰刀菌属转移到链格孢菌番茄致病型中。
链格孢菌不同致病型的HST毒素生物合成基因都有多个拷贝,在敲除毒素合成基因的实验中发现,多拷贝的毒素合成基因是链格孢菌产生足够毒素以表现完全致病性的前提[2, 3, 16, 17]。HST基因簇上毒素合成基因的复制对链格孢菌获得足够的毒力和有效的侵染寄主植物可能具有非常重要的意义[3]。
2 CD染色体除烟草致病型未知外,链格孢菌其他6种致病型菌株的HST生物合成基因簇都集中在一个小于2.0Mb的小染色体上[2, 3, 4, 5, 15, 18, 19]。链格孢菌草莓致病型NAF8菌株缺失了编码AFT基因的小染色体后,虽然在培养基上能正常生长和产孢,但却失去了产毒素和致病能力[15, 20],这种小染色体被定义为超数染色体(supernumerary)或CD染色体,这种染色体的不稳定性不会影响真菌在培养基上的生长或孢子的萌发,但会影响真菌在寄主植物上的致病力[20]。
研究发现,链格孢菌致病菌株具有含HST基因簇的CD染色体,非致病性菌株则没有CD染色体[21]。实验室条件下,通过原生质体融合获得了番茄致病型和苹果致病型杂交菌株、番茄致病型和草莓致病型杂交菌株,这些融合体具有源于亲本的两种CD染色体,产生两种亲本菌株毒素,对两种亲本菌株的寄主植物都有致病性[19, 22]。原生质体融合实验说明,链格孢菌的CD染色体可以进行种内转移,链格孢菌基因组可以接受和维持外源小染色体。Masunaka等[23]在粗柠檬叶片病斑上分离到一株双寄主特异性链格孢菌,该菌株对粗柠檬和橘都有致病作用,产生ACT毒素和ACR毒素,携带1.05Mb和2.0Mb大小分别编码ACTT和ACRT基因的两种CD染色体。在番茄枯萎病病原尖孢镰刀菌番茄专化型(Fusarium oxysporum f. sp. Lycopersici)和尖孢镰刀菌非致病型菌株(F. oxysporum)的共培养实验中,致病型菌株的2.0Mb的小染色体可以转移到非致病型菌株中,使非致病型菌株具有致病性[24]。这表明,某一致病型菌株产HST的能力或许可以通过种内CD染色体的转移传给非致病菌株或另一个致病型菌株,使非致病型菌株成为致病型菌株或使单一寄主致病型菌株的寄生范围扩大。
Adachi和Tsuge[21]从日本梨叶片上的单个黑色病斑中分离出链格孢菌致病型菌株和非致病型菌株,这表明致病型菌株和非致病型菌株在很小的空间里可以共存和互作。在实验室内观察到链格孢菌不同的致病型菌株之间、致病型与非致病型菌株之间分别都有菌丝融合现象,自然条件下,链格孢菌CD染色体种内转移可能是通过准性生殖世代的菌丝融合完成的[25]。
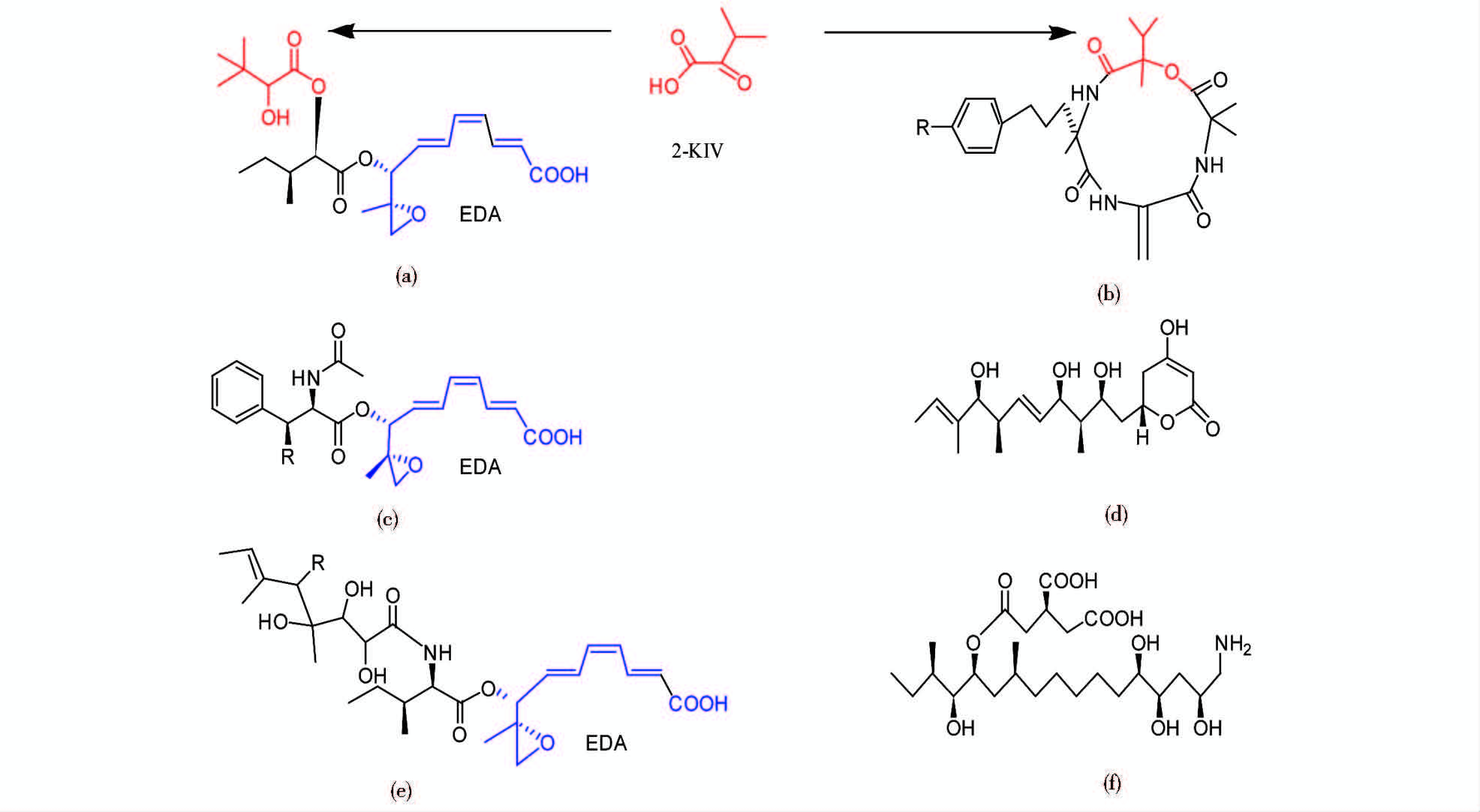
3 毒素及其编码基因HST毒素通常是产HST毒素病原真菌的主要致病因子,毒素的生物活性由其分子结构决定,对于链格孢菌的7种HST,已知结构的有6种(图 1)。毒素在结构上的微小差别可能造成生物活性的巨大差异。例如,AF-毒素与AK-毒素都是相似的三烯酸结构,2′位上功能团不同,造成两种毒素对不同植物产生病害。
对毒素基因的研究为病原菌致病机制的研究提供了分子水平的依据,链格孢菌HST毒素合成基因有许多旁系同源基因,这些基因簇聚集在一个小的CD染色体上,基于同源重组原理的基因敲除(homologous recombination-mediated gene disruption)很难破坏整个功能拷贝。为了解决这个问题,现在多采用RNA沉默来敲除某一基因的功能拷贝。
3.1 AK毒素基因毒素基因和管家基因的系统发生分析都表明日本梨致病型、橘致病型和草莓致病型起源于共同的祖先[26]。AK毒素与ACT、AF毒素结构相似,它们都有9,10-环氧基-8-羟基-9-甲基三烯酸(9,10-epoxy-8-hydroxy-9-methyl-decatrienoic acid moiety,EDA)结构(图 1),在AKT、ACTT、AFT基因簇上控制毒素合成的基因很多是同源基因[27, 28]。目前已知的AKT基因包括AKT1、AKT2、AKT3、AKT4、AKTR、AKTS1,除AKTS1为日本梨致病型特有外,其他基因在橘致病型或草莓致病型中都有同源基因[1, 13, 29]。
为了解链格孢菌日本梨致病型寄主选择性的分子基础,Tanaka等[13]对AK毒素生物合成相关基因进行了鉴定与克隆,他们应用限制性内切核酸酶介导的基因整合技术在5.0kb的区域内成功克隆了2个日本梨致病型菌株产生毒素和致病所必需的基因AKT1和AKT2。AKT1长度为1 734bp,包含5个内含子,编码一种578个氨基酸的羧基活化酶;AKT2长度为786bp,包含1个内含子,编码一种262个氨基酸的蛋白质[13]。AKT3长度为888bp,包含3个内含子,编码的蛋白质具有296个氨基酸,该蛋白质可能属于水合酶∕异构酶家族(hydratase/isomerase enzyme family)[29]。AKTR基因是其他AKT基因表达的调控基因,大小为1 332bp,编码具有444个氨基酸的调节因子,该蛋白质内部有一个亮氨酸拉链(leucine zipper domain),氨基末端有一个DNA结合结构域(zinc binuclear cluster DNA-binding domain),属于典型的真菌Zn(II)2Cys6蛋白家族[29]。AKT7基因包含6个外显子和5个内含子,编码522个氨基酸的细胞色素P450单氧化酶(cytochrome P450 monooxygenase)[30]。AKT7在链格孢菌其他致病型中没有同源基因,不参与EDA的合成,虽然AKT7也是多拷贝的,但是单拷贝突变菌株的EDA和AK毒素产量增加,AKT7超表达菌株的EDA和AK毒素产量显著减少,这可能是细胞色素P450单氧化酶催化EDA或其前体生成某种未知的支路产物所致[30]。
3.2 ACT毒素基因链格孢菌橘致病型的ACTT2,ACTT3和ACTTR与链格孢菌日本梨致病型AKT2,AKT3和AKTTR基因的核酸序列相似度>90%,它们为同源基因,共同参与EDA的生物合成[3, 18, 31]。ACTT2在基因组中以多拷贝形式存在,编码一种水解酶(hydrolase),对ACTT2进行RNA沉默后的菌株彻底失去了产生ACT毒素和致病的能力[3]。链格孢菌橘致病型未发现AKT1和AKT4的同源基因,却有ACTT5和ACTT6基因:ACTT5长为 1 883bp,编码一种酰基辅酶A合成酶(acyl-CoA synthetase);ACTT6长为 897bp,编码一种烯酰辅酶A水合酶(enoyl-CoA hydratase)[3, 32]。链格孢菌日本梨致病型和草莓致病型都有ACTT5和ACTT6这两个基因的同源基因,它们参与编码EDA结构[3, 32]。ACTTS基因是链格孢菌橘致病型特有的基因,ACTTS1和ACTTS4可能编码一种非核糖体肽合成酶[1](nonribosomal peptide synthetase,NRPS)。ACTTS2的大小为1 034bp,编码一种烯酰还原酶[31](enoyl-reductase)。ACTTS3的ORF大小为7 374bp,编码长度约2 457个氨基酸的聚酮合酶(polyketide synthase,PKS),包括β-酮脂酰合成酶(β-ketoacyl synthase)、酰基转移酶(acyl transferase)、甲基转移酶(methyl transferase)、β-酮脂酰还原酶(β-ketoacyl reductase)、磷酸泛酰巯基乙胺(phosphopantetheine)结合结构域[33]。
3.3 AF毒素基因Hatta等[14]确定了长度为1.05Mb的CD染色体的结构,并对390kb的AFT区域进行测序,发现20多个可能的AFT基因,这些基因存在2~7个拷贝,而且这一区域内有很多类转座子序列。链格孢菌草莓致病型AF毒素的合成基因AFT1、AFT3、AFTR分别与链格孢菌日本梨致病型HST合成基因AKT1、AKT3、AKTR的结构非常相似,3对基因中的每一对基因的核酸序列相似度都大于90%,但它们在基因簇中的分布不同[15]。链格孢菌草莓致病型没有AKT2的同源基因,但有AFT8和AFTS1基因[13, 34]。在日本梨致病型和橘致病型中发现AFT8的同源基因,而没有AFTS1的同源基因。AFTS1编码一种醛酮还原酶(aldo-keto reductase),催化AF-毒素Ⅰ的合成,AF-毒素Ⅰ对草莓和日本梨都有致病性,而AF-毒素Ⅱ只对日本梨有致病性,链格孢菌草莓致病型AFTS1基因的突变菌株对草莓没有致病力,只对日本梨有致病力[34]。
3.4 AM毒素基因AM毒素是一种环状四肽,由NRPS经非核糖体途径(non-ribosomal pathway)合成。Johnson等[35]根据真菌NRPS基因的保守区域设计引物,得到一个无内含子序列的环肽合成酶基因,命名为AMT1。AMT1基因位于1.7Mb的CD染色体上,其编码的NRPS有479kDa,包含4个催化结构域。AMT2和AFTS1是同源基因,它们编码的醛酮还原酶(aldo-keto eductase)催化2-酮基异戊酸(2-keto-isovaleric acid,2-KIV )生成AM-毒素和AF-毒素Ⅰ的共同前体(图 1)[34]。Harimoto等[2]使用表达序列标签(expressed sequence tag)分析链格孢菌苹果致病型IFO8984菌株的1.4Mb CD染色体,通过比较基因在产毒和不产毒培养的转录水平,找到产毒时表达上调的21个基因,这21个基因包括AMT1、AMT2,序列分析表明,这些表达上调的基因可能还包括其他AMT基因。IFO8984菌株的CD染色体一侧有一个基因簇包括15个可能的AMT基因,这15个基因以1~4个拷贝的形式存在。其中AMT3有3个拷贝,长为4.3kb,含2个外显子;AMT4长为2.9kb,含3个外显子,其编码的含261个氨基酸的蛋白质与聚酮合酶的硫脂酶结构域相似 [14]。
3.5 ACR毒素基因Masunaka等[23]发现,链格孢菌粗柠檬致病型有特异的1.2~1.5Mb大小的染色体。对链格孢菌粗柠檬致病型HC1菌株的1.5Mb染色体进行大规模测序后发现很多可能的ACRT基因,使用基因敲除(targeted gene disruption)和RNA沉默(RNA silencing)来破坏这些基因,确定了两种ACRT基因的作用。ACRTS1是粗柠檬致病型特有的基因,编码一种可能的羟化酶(hydroxylase)。ACRTS1沉默后的菌株不产毒素,不具有致病性,说明ACRTS1是粗柠檬致病型产毒和致病的必需基因[36]。ACRTS2也是粗柠檬致病型特有的基因,长度为8 338bp,包含7个内含子,编码一种2 513个氨基酸大小的PKS。利用同源重组原理的基因敲除和RNA沉默研究ACRTS2多拷贝的旁系同源基因,发现破坏ACRTS2多拷贝中r 单拷贝后菌株毒素产量减少,ACRTS2沉默后的菌株检测不到ACRTS2转录本,失去产毒和致病能力,说明ACRTS2编码的PKS是粗柠檬致病型产生HST和致病所必需的[36]。虽然ACTTS3和ACRTS2都是PKS基因,但它们参与编码的两种毒素在结构和寄主选择性方面差异显著,ACTTS3和ACRTS2两个基因在编码β-酮脂酰合成酶(KS)区域核苷酸序列相似度为44.9%,揭示出两基因明显属于不同的分支,ACTTS3和ACRTS2基因组序列对比结果也显示两个基因的相似度极低[33, 36]。
3.6 AAL毒素基因AAL毒素是聚酮化合物,由大型多功能酶PKS合成。Akamatsu等[37]根据真菌PKS基因的保守区域设计引物,克隆出链格孢菌番茄致病型PKS基因片段,证实ALT1编码Ⅰ型PKS,大小为7.8kb。轮枝镰孢菌(Fusarium.verticillioides)伏马菌素(fumonisin)的合成也需要PKS,编码PKS的FUM1基因与链格孢菌番茄致病型ALT1基因非常相似[37, 38, 39, 40]。轮枝镰孢菌伏马菌素的生物合成基因簇有17个基因,其中13个基因与链格孢菌番茄致病型AS-27菌株的AAL毒素合成基因非常相似[41, 42]。AS-27菌株的13个ALT基因分布在CD染色体的两组基因簇上。现在已知AAL毒素合成基因的功能如下:ALT1(FUM1的同源基因)编码Ⅰ型PKS,ALT2(FUM6的同源基因)编码细胞色素P450单氧化酶(cytochrome P450 monooxygenase),ALT3(FUM7的同源基因)编码氨基转移酶(aminotransferase),ALT6(FUM13的同源基因)编码短链脱氢酶(dehydrogenase)或还原酶(reductase),ALT13(FUM21的同源基因)编码转录因子Zn(II)2Cys6[19, 37, 38, 39, 40, 41, 42]。
3.7 AT毒素、AB毒素、AP毒素目前,对AT毒素、AB毒素、AP毒素的研究主要集中在生产、分离纯化、结构鉴定等方面,未见遗传背景方面的报道。AT-毒素在烟草上诱导的细胞死亡是一种程序性细胞死亡,这一过程中类半胱天冬蛋白酶和活性氧信号起着重要作用,其他毒素是否能引起寄主的程序性细胞死亡还不清楚[43]。芸苔链格孢的萌发孢子只在宿主植物的叶片上产AB毒素,在非宿主植物、培养基上不产毒素[44]。AB毒素的分子质量为35kDa,与其他几种链格孢属HST毒素不同,它可能是一种蛋白质,其的产生需要信号分子的激发,诱导孢子产生AB毒素的诱导因子是源于宿主的1.3kDa低聚糖,可能由D-甘露糖和D-半乳糖组成[44, 45]。
4 结 语链格孢菌是自然界中广泛存在、危害严重的植物病原真菌,链格孢菌引起的病害是我国农业生产中的突出问题,给国家和农民带来严重的经济损失,对链格孢菌HST毒素及其合成基因的研究,对于制定长久、有效的病害治理策略具有重大意义。链格孢菌的HST合成基因在真菌生活史中并不重要,但在自然条件下与致病性密切相关,其产物是致病所必需的,基因功能的丧失导致致病性的丧失,阐明它们的结构、特征和功能表达,将增加对真菌致病过程分子机制的认识,为提出更有效的病害控制途径指明了方向。HST毒素基因的研究对深入了解病原与寄主植物的相互关系、认识HST毒素的致病过程至关重要。同时,链格孢菌HST毒素在除草剂、杀菌剂、防病治病、抗性育种等方面具有广阔的应用前景,而对HST遗传背景的研究是毒素生产与合成的关键。
| [1] | Tsuge T, Harimoto Y, Akimitsu K, et al. Host-selective toxins produced by the plant pathogenic fungus Alternaria alternata. FEMS Microbiology Reviews, 2013, 37(1): 44-66. |
| [2] | Harimoto Y, Hatta R, Kodama M, et al. Expression profiles of genes encoded by the supernumerary chromosome controlling AM-toxin biosynthesis and pathogenicity in the apple pathotype of Alternaria alternata. Molecular Plant-Microbe Interactions, 2007, 20(4): 1463-1476. |
| [3] | Miyamoto Y, Masunaka A, Tsuge T, et al. Functional analysis of a multi-copy host-selective ACT-toxin biosynthesis gene in the tangerine pathotype of Alternaria alternata using RNA silencing. Molecular Plant-Microbe Interactions, 2008, 21(12): 1591-1599. |
| [4] | Akamatsu H, Taga M, Kodama M, et al. Molecular karyotypes for Alternaria plant pathogens known to produce host-specific toxins. Current Genetics, 1999, 35(6): 647-656. |
| [5] | Johnson L, Johnson R D, Akamatsu H, et al. Spontaneous loss of a conditionally dispensable chromosome from Alternaria alternata apple pathotype leads to loss of toxin production and pathogenicity. Current Genetics, 2001, 40(2): 65-72. |
| [6] | Akimitsu K, Tsuge T, Kodama M, et al. Alternaria host-selective toxins: determinant factors of plant disease. Journal of General Plant Pahtology, 2014, 80(2):109-122. |
| [7] | 康子腾,姜黎明,罗义勇,等. 植物病原链格孢属真菌的致病机制研究进展. 生命科学,2013, 25(9):909-914. Kang Z T, Jiang L M, Luo Y Y, et al. The research advances of mechanism of pathogenicity of Alternaria phytopathogenic fungi. Chinese Bulletin of Life Sciences, 2013, 25(9):909-914. |
| [8] | 赵圆,王玲杰,王雪峰,等. 杂柑褐斑病的病原鉴定.果树学报,2014,31(2) :292-295. Zhao Y, Wang L J, Wang X F, et al. Identification of the pathogenic fungus causing brown spot in two tangerine hybrid varieties. Journal of Fruit Science, 2014, 31(2):292-295. |
| [9] | 王玲杰,王洪秀,赵圆,等.柑橘褐斑病菌毒素研究进展. 中国南方果树,2014, 43(3) :60-63. Wang L J, Wang H X,Zhao Y, et al. The research progress of toxin produced by pathogeny of Citrus Brown Spot. South China Fruits, 2014, 43(3):60-63. |
| [10] | 马振国,董金皋,樊慕贞. 芸薹链格孢致病毒素研究Ⅱ——AB-毒素对白菜叶片细胞PAL、POD、SOD活性的影响. 河北农业大学学报,2002,25(8):60-65. Ma Z G, Dong J G, Fan M Z. Study on toxin of Alternaria brassicae Ⅱ——effect of AB-toxin on PAL, PO, SOD activity in leave cells of Brassica chinensis. Journal of Agricultural University of Hebei, 2002,25(8):60-65. |
| [11] | 万佐玺,强胜,李扬汉. 链格孢菌寄主选择性毒素的研究现状. 湖北民族学院学报(自然科学版),2001,19(4):169-175. Wan Z X, Oiang S, Li Y H. Study on the status guo of Alternaria alternata specific toxins. JournaI of Hubei Institute for NationaIities (Nat Sci), 2001,19(4):169-175. |
| [12] | Rosewich U L, Kistler H C. Role of horizontal gene transfer in the evolution of fungi. Annual Review of Phytopathology, 2000, 38(4): 325-363. |
| [13] | Tanaka A, Shiotani H, Yamamoto M, et al. Insertional mutagenesis and cloning of the genes required for biosynthesis of the host-specific AK-toxin in the Japanese pear pathotype of Alternaria alternata. Molecular Plant-Microbe Interactions, 1999, 12(8): 691-702. |
| [14] | Hatta R, Shinjo A, Ruswandi S, et al. DNA transposon fossils present on the conditionally dispensable chromosome controlling AF-toxin biosynthesis and pathogenicity of Alternaria alternata. Journal of General Plant Pahtology, 2006, 72(5): 210-219. |
| [15] | Hatta R, Ito K, Hosaki Y, et al. A conditionally dispensable chromosome controls host-specific pathogenicity in the fungal plant pathogen Alternaria alternata. Genetics, 2002, 161(4): 59-70. |
| [16] | Ruswandi S R, Kitani K, Akimitsu K, et al. Structural analysis of cosmid clone pcAFT-2 carrying AFT10-1encoding an acyl-CoA dehydrogenase involved in AF-toxin production in the strawberry pathotype of Alternaria alternata. Journal of General Plant Pahtology, 2005, l71(2): 107-116. |
| [17] | Harimoto Y, Tanaka T, Kodama M, et al. Multiple copies of AMT2 are prerequisite for the apple pathotype of Alternaria alternata to produce enough AM-toxin for expressing pathogenicity. Journal of General Plant Pahtology, 2008, 74(3): 222-229. |
| [18] | Masunaka A, Tanaka A, Tsuge T, et al. Distribution and characterization of AKT homologs in the tangerine pathotype of Alternaria alternata. Phytopathology, 2000, 90(3): 762-768. |
| [19] | Akagi Y, Akamatsu H, Otani H, et al. Horizontal chromosome transfer: a mechanism for the evolution and differentiation of a plant pathogenic fungus. Eukaryot Cell, 2009, 8(19): 1732-1738. |
| [20] | Covert S F. Supernumerary chromosomes in filamentous fungi. Current Genetics, 1998, 33(5): 311-319. |
| [21] | Adachi Y, Tsuge T. Coinfection by different isolates of Alternaria alternata in single black spot lesions of Japanese pear leaves. Phytopathology, 1994, 84(12): 447-451. |
| [22] | Akagi Y, Taga M, Yamamoto M, et al. Chromosome constitution of hybrid strains constructed by protoplast fusion between the tomato and strawberry pathotypes of Alternaria alternata. Journal of General Plant Pahtology, 2009, 75(6): 101-109. |
| [23] | Masunaka A, Ohtani K, Peever T L, et al. An isolate of Alternaria alternata that is pathogenic to both tangerines and rough lemon and produces two hostselective toxins, ACT- and ACR-toxins. Phytopathology, 2005, 95(3): 241-247. |
| [24] | Ma L J, Van Der Does H C, Borkovich K A. Comparative genomics reveals mobile pathogenicity chromosomes in Fusarium. Nature, 2000, 464(7287): 367-373. |
| [25] | Huang S L, Itoh Y, Kohmoto K, et al. Hyphal anastomosis and complementary growth of fused cells in Alternaria alternata. Mycoscience, 1996, 37(11): 1-13. |
| [26] | Stewart J E, Timmer L W, Lawrence C B, et al. Discord between morphological and phylogenetic species boundaries: incomplete lineage sorting and recombination results in fuzzy species boundaries in an asexual fungal pathogen. BMC Evolutionary Biology, 2015, 14(1): 38-46. |
| [27] | Nakatsuka S, Goto T, Itoh Y, et al. Chemical Studies on Structures and Host-specificities of ACT-toxins Produced by Alternaria alternata Tangerine Pathotype Causing Citrus Brown Spot Disease. Nagoya: 31st Symposium on the Chemistry of Natural Products, 1989: 671-676. |
| [28] | Huang F, Fu Y S, Nie D, et al. Identification of a novel phylogenetic lineage of Alternaria alternata causing citrus brown spot in China. Fungal Biology,2015,12(10):320-330. |
| [29] | Tanaka A, Tsuge T. Structural and functional complexity of the genomic region controlling AK-toxin biosynthesis and pathogenicity in the Japanese pear pathotype of Alternaria alternata. Molecular Plant-Microbe Interactions, 2000, 13(8): 975-986. |
| [30] | Takaoka S, Kurata M, Harimoto Y, et al. Complex regulation of secondary metabolism controlling pathogenicity in the phytopathogenic fungus Alternaria alternate. New Phytologist , 2015, 202(4): 1297-1309. |
| [31] | Ajiro N, Miyamoto Y, Masunaka A. Role of the host-selective ACT-toxin synthesis gene ACTTS2 encoding an enoyl-reductase in pathogenicity of the tangerine pathotype of Alternaria alternata. Phytopathology, 2010, 100(12): 120-126. |
| [32] | Miyamoto Y, Isshiki Y, Honda A. Function of genes encoding acyl-CoA synthetase and enoyl-CoA hydratase for host-selective ACT-toxin biosynthesis in the tangerine pathotype of Alternaria alternata. Phytopathology, 2009, 99(4): 369-377. |
| [33] | Miyamoto Y, Masunaka A, Tsuge T. ACTTS3 encoding a polyketide synthase is essential for the biosynthesis of ACT-toxin and pathogenicity in the tangerine pathotype of Alternaria alternata. Molecular Plant-Microbe Interactions, 2010, 23(4): 406-414. |
| [34] | Itoh K, Tanaka T, Hatta R, et al. Dissection of the host range of the fungal plant pathogen Alternaria alternata by modification of secondary metabolism. Molecular Microbiology, 2004, 52(3): 399-411. |
| [35] | Johnson R D, Johnson L, Itoh Y, et al. Cloning and characterization of a cyclic peptide synthetase gene from Alternaria alternata apple pathotype whose product is involved in AM-toxin synthesis and pathogenicity. Molecular Plant-Microbe Interactions, 2000, 13(6): 742-753. |
| [36] | Izumi Y, Ohtani K, Miyamoto Y, et al. A polyketide synthase gene, ACRTS2 , is responsible for biosynthesis of host-selective ACR-toxin in the rough lemon pathotype of Alternaria alternate. Molecular Plant-Microbe Interactions, 2012, 25(11):1419-1429. |
| [37] | Akamatsu H, Otani H, Kodama M. Characterization of a gene cluster for host-specific AAL-toxin biosynthesis in the tomato pathotype of Alternaria alternate. Fungal Genet Newsl, 2003, 50(suppl): 355. |
| [38] | Proctor R H, Desjardins A E, Plattner R D, et al. A polyketide synthase gene required for biosynthesis of fumonisin mycotoxins in Gibberella fujikuroi mating population A. Fungal Genetics and Biology, 1999, 27(2): 100-112. |
| [39] | Seo J A, Proctor R H, Plattner R D. Characterization of four clustered and coregulated genes associated with fumonisin biosynthesis in Fusarium verticillioides. Fungal Genetics and Biology, 2003, 34(2): 155-165. |
| [40] | Yamagishi D, Akamatsu H, Otani H, et al. Pathological evaluation of host-specific AAL-toxins and fumonisin mycotoxins produced by Alternaria and Fusarium species. Journal of General Plant Pahtology, 2006, 72(6): 323-326. |
| [41] | Proctor R H, Brown D W, Plattner R D, et al. Co-expression of 15 contiguous genes delineates a fumonisin biosynthetic gene cluster in Gibberella moniliformis. Fungal Genetics and Biology, 2003, 38(5): 237-249. |
| [42] | Waalwijk C, Van Der Lee T, De Vries I, et al. Synteny in toxigenic Fusarium species: the fumonisin gene cluster and the mating type region as examples. European Journal of Plant Pathology, 2004, 110(3): 533-544. |
| [43] | Yakimova E T, Yordanova Z P, Slavov S, et al. Alternaria alternata AT toxin induces programmed cell death in tobacco. Journal of Phytopathology-phytopathologische Zeitschrift, 2009, 157(10):592-601. |
| [44] | Otani H, Kohnobe A, Kodama M, et al. Involvement of Host Factors in the Production of a Protein Host-specific Toxin by Alternaria brassicicola. In: Kohmoto K, Yoder O C. Molecular Genetics of Host-specific Toxin in Plant Disease. Dordrecht: Kluwer Academic Publishers, 1998: 63-69. |
| [45] | Oka K, Akamatsub H, Kodamab M, et al. Host-specific AB toxin production by germinating spores of Alternaria brassicicola is induced by a host-derived oligosaccharide. Physiological and Molecular Plant Pathology, 2015, 66(1):12-19. |
 2015, Vol. 35
2015, Vol. 35