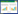文章信息
- 付怀秀, 于翔, 康宏向, 梁洁, 陈鹏, 沈本剑, 金义光, 熊力, 毛建平
- FU Huai-xiu, YU Xiang, KANG Hong-xiang, LIANG Jie, CHEN Peng, SHEN Ben-jian, JIN Yi-guang, XION Li, MAO Jian-ping
- Photosan脂质立方液晶纳米光敏剂的制备及光动力杀伤效应研究
- Preparation of Photosensitizer-loaded Cubic Liquid Crystalline and Its Photodynamic Therapy Effects
- 中国生物工程杂志, 2015, 35(3): 35-41
- China Biotechnology, 2015, 35(3): 35-41
- http://dx.doi.org/10.13523/j.cb.20150305
-
文章历史
- 收稿日期:2014-12-30
- 修回日期:2015-01-07
2. 中国人民解放军军事医学科学院放射与辐射医学研究所 北京 100850;
3. 中南大学湘雅二医院 长沙 410083
2. Institute of Radiation Medicine, Academy of Military Medical Science, Beijing 100850, China;
3. Department of General Surgery, Second Xiang Hospital, Central South University, Changsha 410083, China
光动力疗法(PDT)是除手术、放疗和化疗外的一种肿瘤微创新疗法,它能够特异性杀死肿瘤细胞,减小对正常组织的损害,其原理是光敏剂特异性聚集在肿瘤细胞,在光的作用下引起光化学反应,产生具有细胞毒性的单线态氧或氧自由基等杀伤肿瘤细胞及损伤肿瘤微血管等[1, 2],从而达到消灭肿瘤的目的。PDT正逐渐成为浅表肿瘤的选择性治疗方法,已经被用于治疗多种皮肤肿瘤[3]和头颈部肿瘤[4]等。Wyss等[5]报道PDT被用作复发性乳腺癌的临床微创疗法具有不良反应小,患者满意度高等优点。光敏剂作为PDT发挥效应的关键因素之一,在PDT发展中起着重要的作用。传统的第一代光敏剂具有靶向性差、生物利用度低、毒副作用大等缺点,药物的靶向性已经是研究影响PDT杀伤肿瘤效果的重点[6]。增强光敏剂的光动力学效应和降低其毒副作用的光敏剂纳米粒制备研究,近年来也有不少文献报道[7, 8]。立方液晶纳米粒的内部是由两亲性材料与水形成的双连续网络结构,具有独特的内部双水道结构,且具有载药量大、靶向性强、毒副作用小等优点[9]。Photosan是德国生产的第一代光敏剂,成分为血卟啉寡聚物的混合物,其溶液稳定性不好,另外该光敏剂在体内滞留的时间较长,患者需长时间避光[10]。为了提高Photosan的靶向性和安全性,本实验将Photosan与立方液晶纳米粒结合,制备Photosan立方液晶纳米粒,针对处方辅料GMO和P407对细胞的安全浓度进行探索,筛选出对细胞有效的安全浓度范围获得合适的处方并制备了药物新剂型,通过体外培养人肝L-02细胞和人肝癌HepG2细胞,探讨Photosan立方液晶对细胞的暗毒性和光动力效应。
1 材料与方法 1.1 材 料 1.1.1 细胞株人肝L-02细胞和人肝癌细胞系HepG2由本实验室冻存。
1.1.2 主要仪器恒温CO2细胞培养箱(Thermo,美国),酶标仪(Biotek,美国),光学显微镜(Olympus,日本),DM2500P偏光显微镜(Leica,德国),Malvern粒径仪(Malvern,英国),S-4800型扫描电子显微镜(日立,日本),半导体激光器(波长657nm,功率密度30mW/cm2)。
1.1.3 主要试剂Photosan(SeeLab,德国),单油酸甘油酯(纯度≥90%,批号4012188923,Danisc,丹麦),泊洛沙姆407(BASF,德国),DMEM培养基(Gibco,美国),FBS(四季青,中国),0.25% Trypsin-EDTA solution(Sigma,美国),[3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium,inner salt,MTS](Promega,美国),磷酸盐缓冲液(PBS,pH 7.2~7.4)。
1.2 方 法 1.2.1 Photosan立方液晶制备称取0.022g GMO于1ml无水乙醇中超声溶解形成溶液A;0.010g 泊洛沙姆407和5mg Photosan溶于50ml超纯水,磁力加热搅拌器搅拌,获得溶液B;将200μl 溶液A悬滴入溶液B,在此过程中使用磁力加热搅拌器搅拌,直至形成液晶相溶液,0.2μm过滤器过滤除菌。
1.2.2 Photosan立方液晶的粒径和形貌表征取少量样品置于载玻片上,在偏光显微镜下观察其双折射性[11]。将Photosan立方液晶超声10min,使其均匀分散,用Malvern粒径仪进行粒径尺寸和分布检测。取少量样品置于硅片上,样品干燥后放入离子溅射仪中镀金,用扫描电子显微镜进行扫描并拍照。
1.2.3 细胞培养和传代人肝L-02细胞和人肝癌HepG2细胞培养在含10% 胎牛血清DMEM 细胞培养基中(青霉素100U/ml,链霉素100U/ml),置于37℃,5% CO2培养箱培养。细胞每2~3天传代。
1.2.4 MTS法探索处方辅料对细胞的安全浓度实验分组:空白对照组、阴性对照组和实验组。空白对照组:无细胞培养,加各种试剂;阴性对照组:细胞无药物孵育,无光照;实验组GMO浓度分别为6.875μg/ml、13.75μg/ml、27.5μg/ml、55μg/ml、110μg/ml,P407浓度分别为0.2μg/ml、2μg/ml、20μg/ml、200μg/ml。每组设3个复孔。取对数生长期细胞接种于96孔板,细胞密度1×105cells/ml,每孔100μl,孵育12h后吸去培养基。给药,孵育4h,吸去溶液,PBS洗两遍,每孔加100μl培养基,继续孵育24h,加入20μl MTS,1h后酶标仪测定490nm波长处的OD值。按照以下公式计算细胞存活率:细胞存活率=(实验组OD值-空白对照组OD值)/(阴性对照组OD值-空白对照组OD值)×100%。相同实验重复3次。
1.2.5 Photosan和Photosan立方液晶的细胞毒性和光动力效应实验组分给药组、光照组和PDT组。给药组包括立方液晶空白组、Photosan组和Photosan立方液晶组;PDT组包括Photosan-PDT组和Photosan立方液晶-PDT组;光照组:细胞给予光照(5J/cm2),无药物孵育。立方液晶空白药物除不含Photosan外,所含其他成分与Photosan立方液晶相同,以GMO浓度表示为17.6μg/ml;Photosan和Photosan立方液晶浓度均为20μg/ml。具体步骤见1.2.4 。完全避光,给药4h后,吸去溶液,PBS洗两遍,加入100μl细胞培养基,光照组和PDT组给予光照,继续孵育24h。相同实验重复3次。
1.2.6 统计学分析采用Graphpad Prism 5软件进行统计学分析,结果以均数±标准差表示,P<0.05为差异具有统计学意义。
2 结 果 2.1 Photosan立方液晶制备得到的立方液晶空白溶液呈淡蓝色乳光;Photosan溶液呈棕红色,透明;Photosan立方液晶溶液呈棕红色乳光。
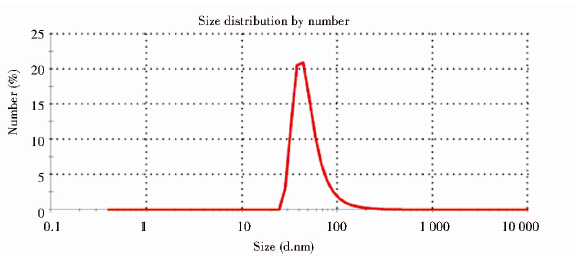
2.2 Photosan立方液晶表征油/水两相系在偏光显微镜下显示为层状液晶,见图 1(a);Photosan立方液晶显示如图 1(b),偏光为暗视野,无双折射性,说明Photosan立方液晶呈现各向同性。Malvern粒径仪测得Photosan立方液晶平均粒径大小为52.42nm,多分散系数(polydispersity index,PDI)为0.528(图 2)。Photosan立方液晶扫描电子显微镜图像可见其形成均匀正方体颗粒(图 3)。

|
| 图 1 层状液晶(a)和Photosan立方液晶(b)偏光显微镜图像 Fig. 1 Polarized photomicrographs of Lamellar phase (a) and Photosan cubic phase (b) |
|
| 图 2 Photosan立方液晶纳米粒径粒分布图 Fig. 2 Diameter distribution of Photosan cubosomes |
|
| 图 3 Photosan立方液晶纳米粒的扫描电子显微镜图像 Fig. 3 Photographs of Photosan cubosomes by scanning electron microscope |
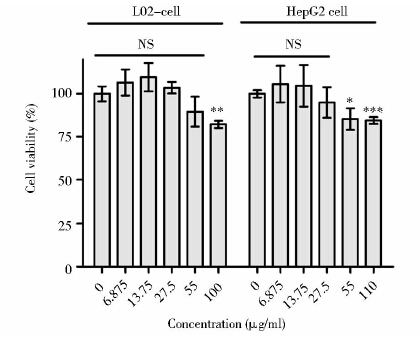
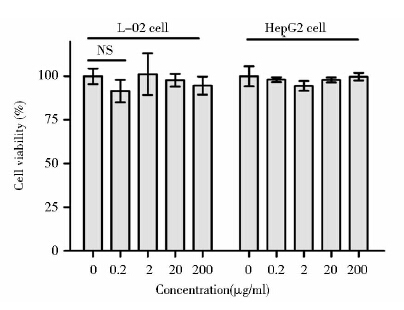
GMO≥110μg/ml时L-02细胞存活率与对照组相比差异有显著统计学意义( P<0.01),说明其在该浓度范围内对L-02细胞有毒;GMO≥55μg/ml时HepG2细胞存活率与对照组相比差异有统计学意义(P< 0.05),说明其在该浓度范围内对HepG2细胞有毒;GMO浓度在6.875~27.5μg/ml时,L-02细胞和HepG2细胞的存活率与对照组相比差异无统计学意义,说明其在该浓度范围内对这两种细胞均没有毒性,属于安全浓度范围(图 4)。P407浓度在0.2~200μg/ml时,L-02细胞和HepG2细胞存活率与对照组相比较差异无统计学意义,说明其在该浓度范围对L-02细胞和HepG2细胞无毒(图 5)。20μg/ml Photosan立方液晶中GMO含量为16.7μg/ml和P407含量为40μg/ml均在其安全浓度范围内。
|
| 图 4 不同浓度GMO对L-02和HepG2细胞的毒性作用 Fig. 4 Cytotoxicity of multiple concentration GMO on L-02 cells and HepG2 cells analyzed by MTS assay Each data was calculated from three independent replicates,and was presented as mean ± SD. NS: not significant; * P<0.05; ** P<0.01; *** P<0.001; compared with the control group(student t-test) |
|
| 图 5 不同浓度P407对L-02细胞和HepG2细胞的毒性作用 Fig. 5 Cytotoxicity of multiple concentration P407 on L-02 cells and HepG2 cells analyzed by MTS assay Each data was calculated from three independent replicates,and was presented as mean ± SD. NS: not significant; compared with the control group(student t-test) |
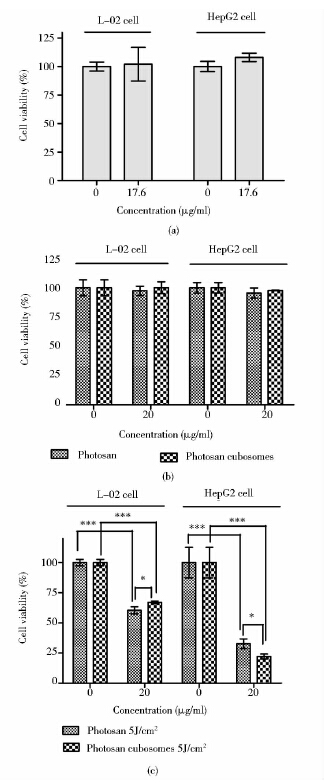
如图 6所示,立方液晶空白(图 6a)、Photosan和Photosan立方液晶(图 6b)作用于L-02细胞和HepG2细胞,L-02细胞和HepG2细胞的存活率与对照组相比差异均无统计学意义(P>0.05),说明上述药物在给定的浓度对细胞增殖的抑制作用不明显,即立方液晶空白在给定的浓度对L-02细胞和HepG2细胞无毒,Photosan和Photosan立方液晶在20μg/ml时无暗毒性。光照组与对照组相比细胞存活率差异也无统计学意义,说明该光照剂量对细胞的增殖无抑制作用(P>0.05)。
|
| 图 6 立方液晶空白(a)、Photosan和Photosan立方液晶(b)对L-02细胞和HepG2细胞的毒性作用和光动力杀伤效应(c) Fig. 6 Cytotoxicity and Photodynamic effects (c) of the blank cubosomes(a),Photosan and Photosan cubosomes(b) respectively on L-02 cells and HepG2 cells analyzed by MTS assay Each data retrieved from three independent replicates,and was presented as mean ±SD. * P<0.05; ** P<0.01; *** P<0.001 (Student t-test) |
Photosan-PDT和Photosan立方液晶-PDT作用于L-02细胞和HepG2细胞,结果如图 6(c)所示,实验组细胞的存活率与对照组相比差异均具有统计学意义(P<0.05),说明PDT组对L-02细胞和HepG2细胞的增殖有抑制作用。Photosan-PDT组与Photosan立方液晶-PDT组相比,L-02细胞的存活率后者高于前者,且差异具有统计学意义(P<0.05),而HepG2细胞的存活率后者低于前者,且差异具有统计学意义(P<0.05),说明Photosan立方液晶-PDT对L-02细胞的增殖抑制作用低于Photosan-PDT,而对HepG2细胞的增殖抑制作用高于Photosan-PDT。另外,当Photosan立方液晶-PDT组浓度为20μg/ml时,HepG2细胞的存活率明显高于L-02细胞的存活率,说明Photosan立方液晶-PDT虽然对人正常肝细胞的增殖有一定抑制作用,但对人肝癌细胞的增殖具有更显著的抑制作用。总之,Photosan脂质立方液晶纳米粒对人正常肝细胞安全性较好,而对人肝癌细胞的光动力杀伤效应优于Photosan。
3 讨 论为了增强抗肿瘤作用,Tong Z S等[12]将PDT与阿霉素联合使用,体内实验证明可增强抗乳腺癌作用。国际上对纳米药物的研究促进了光敏剂在提高PDT生物效应方面的发展,国内外已成功研制出多种光敏剂的纳米药物载体,如脂质体[13, 14]、纳米粒子[8, 15, 16]和微球[17]等。本研究以立方液晶纳米粒为载体,制备靶向性更强、安全性更好的新型纳米光敏剂。
在制备立方液晶纳米粒的过程中,考察其处方辅料作用于细胞的安全浓度,并参考文献获得合适的立方液晶相溶液处方[11, 18, 19],成功制备了立方液晶纳米粒。通过粒径仪测定Photosan立方液晶的粒径大小,证实该纳米粒的粒径大小属于纳米级别。据报道,粒径大小对药物靶向性具有不同影响,粒径小于150nm 的Tiosens脂质体可以抑制80%~90%肿瘤的生长,而更大粒径则达不到显著效果[20],在本课题中Photosan立方液晶的粒径小于150nm,为杀伤肿瘤细胞提供了很好的制备方法。PDI表示粒径分布的均一程度,该纳米粒为0.528,说明本次实验所制备的纳米微粒较为分散。另外,立方液晶相是指一定浓度的两亲性脂质分散在水中自发形成的“蜂窝状”结构,具有热力学稳定的脂质双层膜[11],该生物膜处于动态平衡中,更反映了立方液晶相的微观结构类似于生物膜,有利于促进药物的吸收,因该胶体分散系稳定性强,可以保护其包载的药物减少外界因素的影响[21]。
本研究通过MTS方法证实所制备的Photosan立方液晶对L-02细胞和HepG2细胞无暗毒性。当Photosan立方液晶浓度为20μg/ml,光照剂量为5J/cm2时,Photosan-PDT和Photosan立方液晶-PDT对正常肝细胞和肝癌细胞增殖均产生一定抑制作用,而Photosan立方液晶-PDT对正常肝细胞的抑制作用低于Photosan-PDT,说明Photosan立方液晶的安全性优于Photosan;Photosan立方液晶-PDT对肝癌细胞增殖的抑制作用高于Photosan-PDT,说明在相同条件下Photosan立方液晶比Photosan的光动力杀伤效应强,且Photosan立方液晶-PDT对人肝癌细胞的增殖的抑制作用明显高于人正常肝细胞,显示了Photosan立方液晶具有较好的靶向性。Photosan立方液晶更好的靶向性和安全性的特点表明其具有良好的临床应用价值,为临床用药拓展了新思路。本课题将继续在该研究的基础上进一步优化条件,如光敏剂的浓度、功率密度、光照剂量和分散体系稳定性处方等,并对Photosan立方液晶纳米粒对肿瘤细胞光动力杀伤效果及作用机制进行后续研究。
| [1] | Chen B, Roskams T, de Witte P A. Antivascular tumor eradication by hypericin-mediated photodynamic therapy. Photochem Photobiol, 2002, 76(5): 509-513. |
| [2] | Harrod-Kim P. Tumor ablation with photodynamic therapy: introduction to mechanism and clinical applications. J Vasc Interv Radiol, 2006, 17(9): 1441-1448. |
| [3] | Triesscheijn M, Baas P, Schellens J H, et al. Photodynamic therapy in oncology. Oncologist, 2006, 11(9): 1034-1044. |
| [4] | Yano T, Hatogai K, Morimoto H, et al. Photodynamic therapy for esophageal cancer. Ann Transl Med, 2014, 2(3): 29. |
| [5] | Wyss P, Schwarz V, Dobler-Girdziunaite D, et al. Photodynamic therapy of locoregional breast cancer recurrences using a chlorin-type photosensitizer. Int J Cancer, 2001, 93(5): 720-724. |
| [6] | Rkein A M, Ozog D M. Photodynamic therapy. Dermatol Clin, 2014, 32(3):415-425. |
| [7] | Liu Z T, Xiong L, Liu Z P, et al. In vivo and in vitro evaluation of the cytotoxic effects of Photosan-loaded hollow silica nanoparticles on liver cancer. Nanoscale Res Lett, 2014, 9(1):319. |
| [8] | Pei D, Xiong L, Lin L, et al. Cytotoxicity of hollow silica nanoparticles loaded with photosensitizes on huh-7 cells. Pak J Pharm Sci, 2014, 27(3 Suppl): 719-722. |
| [9] | Lopes L B, Lopes J L C, Oliveira D C R, et al. Liquid crystalline phases of monoolein and water for topical delivery of cyclosporin A: Characterization and study of in vitro and in vivo delivery. European Journal of Pharmaceutics and Biopharmaceutics, 2006, 63(2): 146-155. |
| [10] | Maier A, Tomaselli F, Matzi V, et al. Comparison of 5-aminolaevulinic acid and porphyrin photosensitization for photodynamic therapy of malignant bronchial stenosis: a clinical pilot study. Lasers Surg Med, 2002, 30(1): 12-17. |
| [11] | Wu H, Li J, Zhang Q, et al. A novel small Odorranalectin-bearing cubosomes: Preparation, brain delivery and pharmacodynamic study on amyloid-β25-35-treated rats following intranasal administration. European Journal of Pharmaceutics and Biopharmaceutics, 2012, 80(2): 368-378. |
| [12] | Tong Z S, Miao P T, Liu T T, et al. Enhanced antitumor effects of BPD-MA-mediated photodynamic therapy combined with adriamycin on breast cancer in mice. Acta Pharmacol Sin, 2012, 33(10): 1319-1324. |
| [13] | Temizel E, Sagir T, Ayan E, et al. Delivery of lipophilic porphyrin by liposome vehicles: preparation and photodynamic therapy activity against cancer cell lines. Photodiagnosis Photodyn Ther, 2014, 11(4): 537-545. |
| [14] | Derycke A S, de Witte P A. Liposomes for photodynamic therapy. Adv Drug Deliv Rev, 2004, 56(1): 17-30. |
| [15] | Deng X, Xiong L, Lin L, et al. Photosan-II loaded hollow silica nanoparticles: preparation and its effect in killing for QBC939 cells. Photodiagnosis Photodyn Ther, 2013, 10(4): 460-469. |
| [16] | Haedicke K, Kozlova D, Grafe S, et al. Multifunctional calcium phosphate nanoparticles for combining near-infrared fluorescence imaging and photodynamic therapy. Acta Biomater, 2015, 14:197-207 |
| [17] | Wang W, Xu D, Wei X, et al. Magnetic-luminescent YbPO4:Er,Dy microspheres designed for tumor theranostics with synergistic effect of photodynamic therapy and chemotherapy. Int J Nanomedicine, 2014, 9: 4879-4891. |
| [18] | Turchiello R, Vena F, Maillard P, et al. Cubic phase gel as a drug delivery system for topical application of 5-ALA, its ester derivatives and m-THPC in photodynamic therapy (PDT). Journal of Photochemistry and Photobiology B: Biology, 2003, 70(1): 1-6. |
| [19] | Evenbratt H, Jonsson C, Faergemann J, et al. In vivo study of an instantly formed lipid–water cubic phase formulation for efficient topical delivery of aminolevulinic acid and methyl-aminolevulinate. International Journal of Pharmaceutics, 2013, 452(1-2): 270-275. |
| [20] | Meerovich G, Meerovich I, Lukyanets E, et al. Influence of liposome size on accumulation in tumor and therapeutic efficiency of liposomal near-IR photosensitizer for PDT based on aluminum hydroxide tetra-3-phenylthiophthalocyanine. NSTI Nanotech, 2008, 2: 41-44. |
| [21] | Shah J C, Sadhale Y, Chilukuri D M. Cubic phase gels as drug delivery systems. Adv Drug Deliv Rev, 2001, 47(2-3): 229-250. |
 2015, Vol. 35
2015, Vol. 35