2) Key Laboratory for Space Bioscience and Biotechnology, Institute of Special Environmental Biophysics, School;
3) Department of Life Sciences, Natural History Museum, London SW7 5BD, United Kingdom;
4) School of Sciences, Tibet University, Lhasa 850000, China;
5) Research Department for Limnology, University of Innsbruck, Mondseestrasse 9, A-5310 Mondsee
The ciliate order Euplotida Small & Lynn, 1985 is characterized by its sparse but well-developed ventral cirri arranged in frontal, ventral, and transverse groups (Fan et al., 2013; Jiang et al., 2013; Gao et al., 2017; Huang et al., 2011, 2012, 2018; Lian et al., 2018). Eupotids have been the subject of numerous recent investigations of their morphology, morphogenesis and systematics (Shao et al., 2010a, b; Yi et al., 2012; Liu et al., 2017; Luo et al., 2014, 2018; Wang et al., 2017a, b; Yan et al., 2018; Zhao et al., 2017, 2018). Uronychidae is a distinctive family in the Euplotida that is characterized by its highly developed paroral. Uronychia binucleata was first reported by Young (1922). It is characterized by its medium-sized, oval to almost rectangular body, two macronuclear nodules, high numbers of basal bodies in its dorsal kineties and characteristic buccal apparatus (Song and Wilbert, 1997).
Divisional morphogenesis of Uronychia has been studied for several species and appears to be highly conserved among all congeners for which data are available (Taylor, 1928; Wilbert and Kahan, 1981; Hill, 1990; Wilbert, 1995; Song, 1996; Song et al., 2004; Shen et al., 2009; Shi et al., 2017; Song and Shao, 2017). However, the development of the leftmost frontal cirrus and paroral in Uronychia has not been described in detail.
Uronychia binucleata is a rare species, and its morphogenesis was partially described by Hill (1990) (misidentified as U. transfuga) and Song (1996) (misidentified as U. uncinata). The rediscovery of U. binucleatain in coastal waters of northern China provided the opportunity to investigate its morphogenetic process in detail, including, for the first time in Uronychia, the formation of the leftmost frontal cirrus and the paroral.
2 Materials and Methods 2.1 Sampling, Culturing, and IsolationUronychia binucleata was collected from Yellow Sea coastal waters next to a sewage outfall at a beach near Zhanqiao Pier, (120˚32΄E, 36˚06΄N), Qingdao, China, on December 6, 2014. The water temperature was about 6.5⊥ and the salinity was 25%-28%. Individual U. binucleata cells were isolated in the laboratory using micropipettes. Cultures were established at 10-15⊥ using boiled sea water and adding small scuticociliates as the food source (Shao et al., 2007).
2.2 MorphogenesisProtargol staining (Wilbert, 1975) was used to reveal the ciliature and nuclear apparatus. Counts and measurements of stained specimens were performed at a magnification of 1000℅. Drawings of protargol-stained cells were made with the help of a camera lucida (Yan et al., 2016, 2017). To illustrate the changes occurring during morphogenetic processes, old (parental) ciliary structures are depicted by contour whereas new structures are shaded black (Chen et al., 2018; Lu et al., 2018).
2.3 Terminology and SystematicsTerminology is mainly according to Song et al. (2004). Systematics is according to Lynn (2008), Gao et al. (2016) and Lyu et al. (2018).
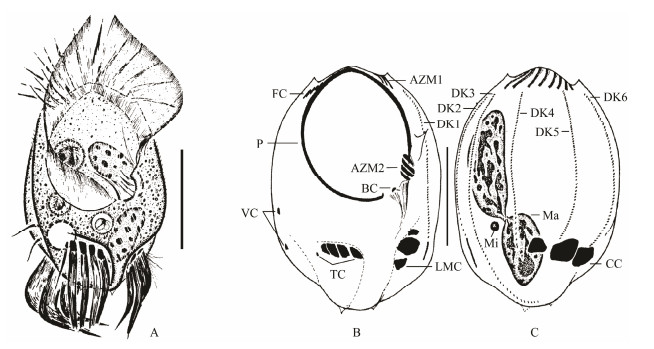
3 Results and DiscussionThe morphology of the present population matches almost exactly with that described by Song and Wilbert (1997), hence no further details are documented here (Figs. 1A-C; 4A, B; Table 1).
|
Fig. 1 Uronychia binucleata in vivo (A) and after protargol staining (B, C) (modified from Song and Wilbert, 1997). A, Ventral view of a representative individual. B and C, Ventral (B) and dorsal (C) views of the general infraciliature. AZM1-2 = adoral zone of membranelles 1, 2; BC = buccal cirrus; CC = caudal cirri; DK1-6 = dorsal kineties 1-6; FC = frontal cirri; LMC = left marginal cirri; VC = ventral cirri; Ma = macronuclei; Mi = micronucleus; TC = transverse cirri; P = paroral. Scale bar = 50 μm. |
|
|
Table 1 Morphometric characterization of Uronychia binucleata |
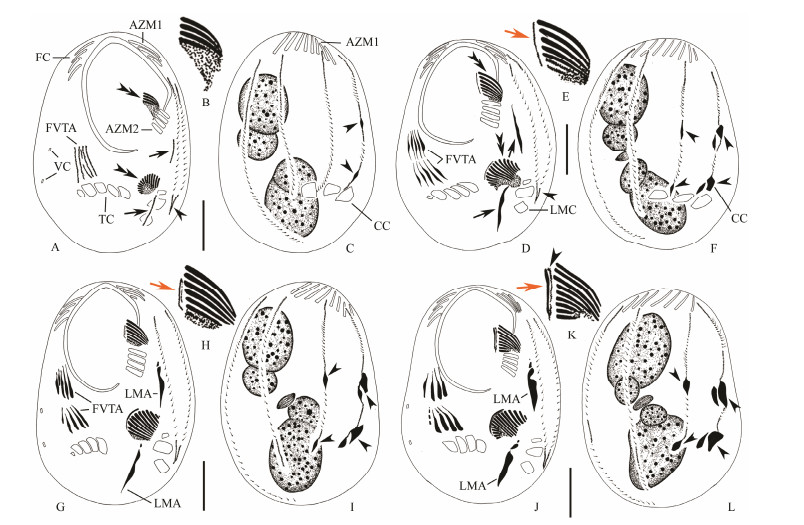
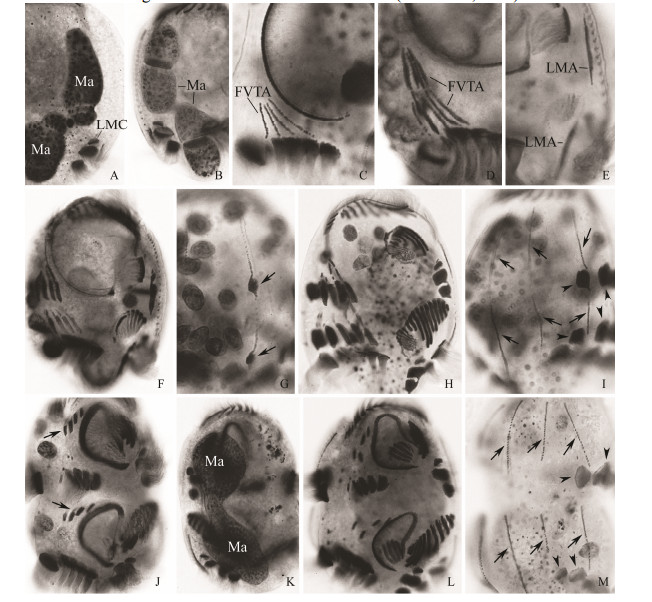
Stomatogenesis Morphogenesis starts with the appearance of a small patch of kinetosomes, the opisthe* soral primordium (OOP) within a subsurface or subcortical pouch positioned between cytostome and left marginal cirri (Fig. 2A). At the same time, the proter*s oral primordium (POP) appears, also subcortically, slightly anterior of the parental adoral zone of membranelles (AZM2) (Figs. 2A, B). Both the OOP and POP develop by rapid proliferation of kinetosomes (Fig. 2D). Subsequently, two groups in the OOP, with eleven and five membranelles respectively, and two groups in the POP, each with five membranelles, differentiate and migrate to the surface of the cell (Figs. 2G, J; 3A, E, G, I).
|
Fig. 2 Morphogenesis of Uronychia binucleata during cell division. A-C, Ventral (A, B) and dorsal (C) views to show the formation of the oral primordia (double-arrowheads), the frontoventral-transverse cirral anlagen, anlagen for left marginal cirri (arrows) and caudal cirri (arrowheads). D-F, Ventral (D, E) and dorsal (F) views to mark the appearance of the anlagen for leftmost frontal cirri (arrow in E) and development of the frontoventral-transverse cirral anlagen and oral primordia (double-arrowheads), as well as development of anlagen for left marginal cirri (arrows) and caudal cirri (arrowheads in D, F). G-I, Ventral (G, H) and dorsal (I) view to demonstrate the appearance of the undulating membranes anlagen (arrow) and the development of the caudal cirri (arrowheads). J-L, Ventral (J, K) and dorsal (L) views showing the development of the undulating membranes anlagen (arrowhead), the anlagen for leftmost frontal cirri (arrow) and the anlagen for the caudal cirri (arrowheads). AZM1-2 = adoral zone of membranelles 1, 2; CC = caudal cirri; FC = frontal cirri; FVTA = frontoventral transverse cirral anlagen; LMA = left marginal cirral anlagen; VC = ventral cirri; TC = transverse cirri. Scale bars = 30 μm. |
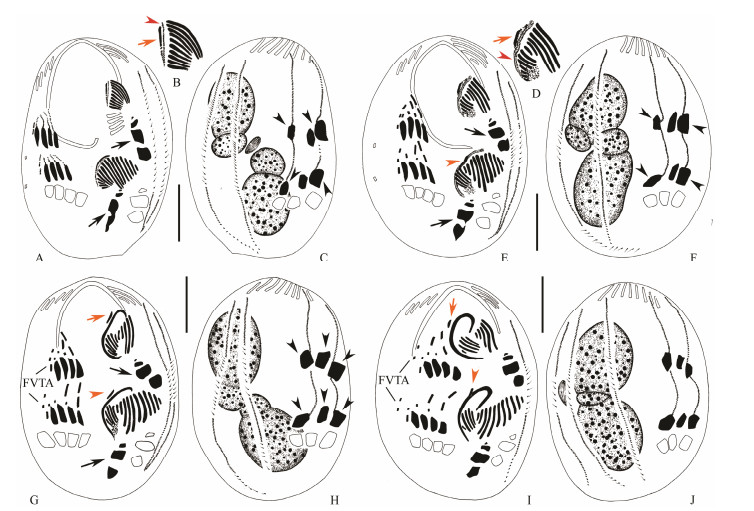
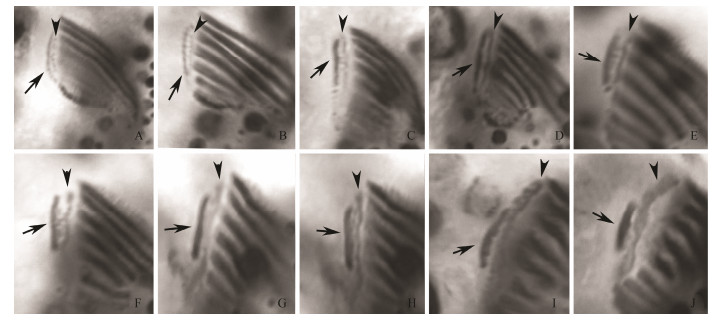
Meanwhile, an additional anlage (anlage for leftmost frontal cirrus, LFCA) appears within the subcortical pouch adjacent to the posterior end of the oral primordium in each divider (Figs. 2D, E). The undulating membranes anlage (UM-anlage) then appears to the left of LFCA and locates between the oral primordium and LFCA in each divider (Figs. 2G, H; 5A). Because we cannot observe the earliest stages of morphogenesis, we were unable to determine whether the UM-anlage and LFCA develop from the same anlage. Later, the UM-anlage and the LFCA begin to lengthen and further develop along the right edge of the pouch opening (Figs. 2J, K; 5B). The anterior portion of the LFCA gradually widens (Figs. 3A, B; 5C-H). Finally the leftmost frontal cirrus develops from the LFCA and the paroral is formed from the UM-anlage (Figs. 3D, E, G; 5I, J).
|
Fig. 3 Morphogenesis of Uronychia binucleata during cell division. A-C, Ventral (A, B) and dorsal (C) views showing the development of the undulating membranes anlagen (arrowhead in B), the anlagen for leftmost frontal cirri (arrow in B), left marginal cirri (arrows in A) and caudal cirri (arrowheads in C). D-F, Ventral (D, E) and dorsal (F) views to denote the formation of leftmost frontal cirri in proter (arrow) and opisthe (arrowheads in E), as well as the undulating membranes anlage (arrowhead in D). Note the development of the anlagen for the left marginal cirri (arrows in E) and caudal cirri (arrowheads in F). G and H, Ventral (G) and dorsal (H) views to mark the development of the frontoventral-transverse cirral anlagen and the leftmost frontal cirri in proter (arrow) and opisthe (arrowhead). Note the development of the anlagen for the left marginal cirri (arrows in G) and caudal cirri (arrowheads in H). I and J, Ventral (I) and dorsal (J) views of a middle divider, demonstrating the development of the undulating membranes and the leftmost frontal cirri in proter (arrow) and opisthe (arrowhead). FVTA = frontoventral transverse cirral anlagen. Scale bars = 30 μm. |
|
Fig. 5 Development of leftmost frontal cirrus and paroral in Uronychia binucleata during cell division after protargol staining. A-H. Ventral views to show the formation of the undulating membranes anlagen (arrowheads) and the anlagen for leftmost frontal cirri (arrow). I and J, Ventral views to show the formation of the leftmost frontal cirri. |
During the late stages, the two groups of membranelles in both oral primordia separate. In the proter, the anterior group migrates anteriorly. As in its congeners, the anterior group will replace the posterior five membranelles of the adoral zone of the proter (AZM1), while the parental six membranelles of the adoral zone are retained (Fig. 3Ⅰ). In the opisthe, the anterior group migrates anteriorly and forms 11 membranelles in AZM1 (Fig. 3Ⅰ). In both daughter cells, the posterior groups form the membranelles in AZM2. In the posterior group of new membranelles, the most posterior one (which is also the smallest) moves apart from the other four and reaches its final position as the & buccal cirrus' (Figs. 3Ⅰ; 4L).
|
Fig. 4 Morphogenesis of Uronychia binucleata during cell division after protargol staining. A and B, Ventral (A) and dorsal (B) views of same cell in interphase, marking the marginal cirri and macronuclear nodules. C, Ventral view of an early divider, to show the formation of the frontoventral-transverse cirral anlagen. D, Ventral view of an early divider, to show the development of the frontoventral-transverse cirral anlagen. E, Ventral view to show the appearance of the marginal anlagen. F and H, Ventral views of early dividers to show the development of the frontoventral-transverse cirral anlagen. G and I, Dorsal views, to denote the formation of the caudal cirri (arrows in G, arrowheads in I) and development of dorsal kineties anlagen (arrows in I). J, Ventral view to show the migration of the frontal cirri (arrows). K, Dorsal view to demonstrate the macronuclear nodules. L, Ventral view showing the development of the frontoventral-transverse cirri, undulating membranes, and adoral zone of membranelles. M, Dorsal view to mark the formation of the caudal cirri (arrowheads) and development of dorsal kineties anlagen (arrows). FVTA = frontoventral transverse cirral anlagen; LMA = left marginal cirral anlagen; LMC = left marginal cirri; Ma = macronuclear nodules. |
Development of the somatic ciliature in proter and opisthe At about the same time as the oral primordia are formed, basal bodies for FVT-cirral primordia (CA) develop on the cell surface firstly as four, and then five, streaks (anlagen I-V) anterior of the transverse cirri (Figs. 2A; 4C). No parental ciliary organelles are involved in the formation of these new primordia. Each of the five anlagen extends to its maximum length before they divide into two sets (Figs. 2A, D; 4D). Subsequently the streaks broaden, break apart and develop into distinct cirri as they migrate to their final positions (Figs. 2G, J; 3A, E; 4F). According to their segmentation, cirral anlagen I-V give rise to cirri in the pattern 3:3:2:2:3, respectively, while the leftmost cirrus is formed near the UM-anlage as described above (Figs. 3G, I; 4H).
The anlagen of the left marginal cirri are formed de novo and separately on the cell surface near the old AZM2 and the marginal cirri (Figs. 2A; 4E). These anlagen then enlarge and segment to form the new left marginal cirri (Figs. 2D, G, J; 3A, E, G, I). On the dorsal side, the proliferation of new basal bodies occurs at two levels within each of the six old kineties. The development of these anlagen seems to occur progressively from right to left (Figs. 2A, C, D, F, G, I; 4M). Two caudal cirri are formed at the posterior end of the rightmost anlage while the second primordium from the right gives rise to the third caudal cirrus (Figs. 2I, L; 3C, F, H, J; 4G, I).
3.2 Noteworthy Morphogenetic Features in UronychiaMorphogenesis in five Uronychia species, including U. xinjiangensis, U. multicirrus, U. transfuga, U. binucleata, and U. setigera, has been investigated using silvers staining methods (Dembowska, 1926; Taylor, 1928; FaurëFaurfremiet 1964; Wilbert and Kahan, 1981; Hill, 1990; Wilbert, 1995; Song, 1996; Song et al., 2004; Shen et al., 2009; Chen et al., 2017; Shi et al., 2017). The pattern of morphogenesis is highly conserved among these congeners and the same general pattern was also observed for U. binucleata in the present study. The mode of development of leftmost frontal cirrus and paroral, however, was unknown in Uronychia and is described here for the first time. In U. binucleata, the anlage for the leftmost frontal cirrus is formed to the right of the UM-anlage and is earlier than the formation of the UM-anlage. This contrasts with the process in U. multicirrus, in which this anlage develops anterior of the UM-anlage after the formation of the latter (Shen et al., 2009).
AcknowledgementsThis work is supported by the National Natural Science Foundation of China (Nos. 31872190, 31702009), the Key Laboratory of Mariculture (KLM), Ministry of Education, Ocean University of China (OUC) and the Innovation Team of Team of Higher Learning Insritutions of Tianjin (No. TD13-5089). We also thank Prof. Weibo Song (OUC) for his kind help and suggestions during drafting the manuscript.
Chen, X. M., Lu, X., Luo, X., Jiang, J., Shao, C., Al-Rasheid, K. A. S., Warren, A. and Song, W. B., 2017. The diverse morphogenetic patterns in spirotrichs and philasterids: Researches based on five-year-projects supported by IRCN-BC and NSFC. European Journal of Protistology, 61: 439-452. DOI:10.1016/j.ejop.2017.05.003 (  0) 0) |
Chen, L. Y., Wu, W. N., El-Serehy, H. A., Hu, X. and Clamp, J. C., 2018. Morphology, morphogenesis, and phylogeny of Anteholosticha intermedia (Ciliophora, Urostylida) population from the United States. European Journal of Protistology, 65: 1-15. DOI:10.1016/j.ejop.2018.04.003 (  0) 0) |
Dembowska, W. S., 1926. Studies on the regeneration of Protozoa. II. Regeneration of the ciliary apparatus in some marine Hypotricha. Journal of Experimental Zoology, 43: 485-504. (  0) 0) |
Fan, Y., Warren, A., Al-Farraj, S. A., Chen, X. M. and Shao, C., 2013. Morphology and SSU rRNA gene-based phylogeny of two Diophrys-like ciliates from northern China, with notes on morphogenesis of Pseudodiophrys nigricans (Protozoa, Ciliophora). Journal of Morphology, 274: 395-403. DOI:10.1002/jmor.20097 (  0) 0) |
Faur'-Fremiet, E., 1964. Les ciliés hypotrichs retrocursits. Archives de Zoologie Expérimentale et Générale, 104: 65-74. (  0) 0) |
Gao, F., Huang, J. A., Zhao, Y., Li, L. F., Liu, W., Miao, M., Zhang, Q., Li, J., Yi, Z., El-Serehy, H. A., Warren, A. and Song, W. B., 2017. Systematic studies on ciliates (Alveolata, Ciliophora): Progress and achievements based on molecular information. European Journal of Protistology, 61: 409-423. DOI:10.1016/j.ejop.2017.04.009 (  0) 0) |
Gao, F., Warren, A., Zhang, Q., Gong, J., Miao, M., Sun, P., Xu, D., Huang, J. A., Yi, Z. and Song, W. B., 2016. The all-databased evolutionary hypothesis of ciliated protists with a revised classification of the phylum Ciliophora (Eukaryota, Alveolata). Scientific Reports, 6: 24874. DOI:10.1038/srep24874 (  0) 0) |
Hill, B. F., 1990. Uronychia transfuga (O. F. Müller, 1786) Stein, 1859 (Ciliophora, Hypotrichida, Uronychidae): Cortical structure and morphogenesis during division.. Journal of Protozoology, 37: 99-107. DOI:10.1111/j.1550-7408.1990.tb05877.x (  0) 0) |
Huang, J. A., Dunthorn, M. and Song, W. B., 2012. Expanding character sampling for the molecular phylogeny of euplotid ciliates (Protozoa, Ciliophora) using three markers, with a focus on the family Uronychiidae. Molecular Phylogenetics and Evolution, 63: 598-605. DOI:10.1016/j.ympev.2012.02.007 (  0) 0) |
Huang, J. A., Yi, Z., Miao, M., Al-Rasheid, K. A. S. and Song, W. B., 2011. Phylogenetic relationships within the genus Aspidisca (Protozoa, Ciliophora, Euplotida) revealed by ITS15. 8S-ITS2 region sequences. Chinese Journal of Oceanology and Limnology, 29: 277-283. DOI:10.1007/s00343-011-0127-2 (  0) 0) |
Huang, J. B., Zhang, T. T., Zhang, Q. Q., Li, Y., Warren, A., Pan, H. B. and Yan, Y., 2018. Further insights into the highly derived haptorids (Ciliophora, Litostomatea): Phylogeny based on multigene data. Zoologica Scripta. DOI:10.1111/zsc.12269 (  0) 0) |
Jiang, J., Huang, J., Li, J., Al-Rasheid, K. A. S., Al-Farraj, S. A., Lin, X. and Hu, X., 2013. Morphology of two marine euplotids (Ciliophora: Euplotida), Aspidisca fusca Kahl, 1928 and A. hexeris Quennerstedt, 1869, with notes on their small subunit rRNA gene sequences. European Journal of Protistology, (49): 634-643. (  0) 0) |
Lian, C., Luo, X., Fan, X., Huang, J. A., Yu, Y., Bourland, W. and Song, W. B., 2018. Morphological and molecular redefinition of Euplotes platystoma Dragesco & Dragesco-Kernéis, 1986 and Aspidisca lynceus (Müller, 1773) Ehrenberg, 1830, with reconsideration of a & well-known* Euplotes ciliate, Euplotes harpa Stein, 1859 (Ciliophora, Euplotida). Journal of Eukaryotic Microbiology, 65: 531-543. DOI:10.1111/jeu.12499 (  0) 0) |
Liu, W., Jiang, J., Xu, Y., Pan, X., Qu, Z., Luo, X., El-Serehy, H. A., Warren, A., Ma, H. and Pan, H., 2017. Diversity of freeliving marine ciliates (Alveolata, Ciliophora): Faunal studies in coastal waters of China during the years 2011-2016. European Journal of Protistology, 61: 424-438. DOI:10.1016/j.ejop.2017.04.007 (  0) 0) |
Lu, X., Huang, J. A., Shao, C. and Berger, H., 2018. Morphology, cell-division, and phylogeny of Schmidingerothrix elongata spec. nov. (Ciliophora, Hypotricha), and brief guide to hypotrichs with Gonostomum-like oral apparatus. European Journal of Protistology, 62: 24-42. (  0) 0) |
Luo, X., Hu, X. and Suzuki, T., 2014. Microscopic investigation of three species of Diophrys (Ciliophora, Euplotida, Uronychiidae) from Japan, including Diophrys peculiaris nov. spec. European Journal of Protistology, 50: 496-508. DOI:10.1016/j.ejop.2014.08.003 (  0) 0) |
Luo, X. T., Yan, Y., Shao, C., Bourland, W. and Song, W. B., 2018. Evolution of strongylidiid ciliates: Molecular, morphological and ontogenetic data support strongylidiids as a member in Dorsomarginalia (Protozoa, Ciliophora) and reactivation of the family Strongylidiidae Faurë-Fremiet, 1961. Zoological Journal of the Linnean Society. DOI:10.1093/zoolinnean/zly001 (  0) 0) |
Lynn, D. H., 2008. The Ciliated Protozoa: Characterization, Classi- fication, and Guide to the Literature. 3rd edition. Springer, Dordrecht, 1-605.
(  0) 0) |
Lyu, Z., Wang, J., Huang, J. A., Warren, A. and Shao, C., 2018. Multigene-based phylogeny of Urostylida (Ciliophora, Hypotrichia), with establishment of a novel family. Zoologica Scripta, 47: 243-254. DOI:10.1111/zsc.12267 (  0) 0) |
Shao, C., Song, W. B., Yi, Z., Gong, J., Li, J. Q. and Lin, X. F., 2007. Morphogenesis of the marine spirotrichous ciliate, Trachelostyla pediculiformis (Cohn, 1866) Borror, 1972 (Ciliophora, Stichotrichia), with consideration of its phylogenetic position. European Journal of Protistology, 43: 255-264. DOI:10.1016/j.ejop.2007.01.004 (  0) 0) |
Shao, C., Ma, H. G., Gao, S., Al-Rasheid, K. A. S. and Song, W. B., 2010a. Reevaluation of cortical developmental patterns in Euplotes (s. l.), including a morphogenetic redescription of E. charon (Protozoa, Ciliophora, Euplotida). Chinese Journal of Oceanology and Limnology, 28: 593-602. (  0) 0) |
Shao, C., Zhang, Q., Al-Rasheid, K. A. S., Warren, A. and Song, W. B., 2010b. Ontogenesis and molecular phylogeny of the marine ciliate Diophryopsis hystrix: Implications for the systematics of the Diophrys-complex (Ciliophora, Spirotrichea, Euplotida). Journal of Eukaryotic Microbiology, 57: 33-39. DOI:10.1111/j.1550-7408.2009.00442.x (  0) 0) |
Shen, Z., Shao, C., Gao, S., Lin, X. F., Li, J. Q., Hu, X. Z. and Song, W. B., 2009. Description of the rare marine ciliate, Uronychia multicirrus Song, 1997 (Ciliophora; Euplotida) based on morphology, morphogenesis and SS rRNA gene sequence. Journal of Eukaryotic Microbiology, 56: 296-304. DOI:10.1111/j.1550-7408.2009.00409.x (  0) 0) |
Shi, X., Liu, G., Wang, C. and Hu, X., 2017. Description of a new brackish water ciliate, Uronychia xinjiangensis n. sp. (Ciliophora, Euplotida) based on morphology, morphogenesis and molecular phylogeny. Acta Protozoologica, 56: 303-315. (  0) 0) |
Song, W. B., 1996. Morphogenetic studies on Uronychia uncinata (Protozoa, Ciliophora) during its asexual division. Acta Oceanologica Sinica, 15: 93-99. (  0) 0) |
Song, W. B. and Shao, C., 2017. Ontogenetic Patterns of Hypotrich Ciliates. Science Press, Beijing, 1-498 (in Chinese).
(  0) 0) |
Song, W. B. and Wilbert, N., 1997. Morphological investigations on some free living ciliates (Protozoa, Ciliophora) from China sea with description of a new hypotrichous genus, Hemigastrostyla nov. gen. Archiv für Protistenkunde, 148: 413-444. DOI:10.1016/S0003-9365(97)80020-6 (  0) 0) |
Song, W. B., Wilbert, N., Chen, Z. and Shi, X., 2004. Considerations on the position of Uronychia and related euplotids based on the data of ontogeny and 18S rRNA gene sequence analyses, with morphogenetic redescription of Uronychia setigera Calkins, 1902 (Ciliophora: Euplotida). Acta Protozoologica, 43: 313-328. (  0) 0) |
Taylor, C. V., 1928. Protoplasmic reorganization in Uronychia uncinata n. sp., during binary fission and regeneration. Physiological Zoology, 1: 1-26. (  0) 0) |
Wang, P., Wang, Y. R., Wang, C. D., Zhang, T. T., Al-Farraj, S. A. and Gao, F., 2017a. Further consideration on the phylogeny of ciliated protists: Analyses using both mitochondrial and nuclear data with focus on the extremely confused class Phyllopharyngea (Protista, Ciliophora). Molecular Phylogenetics and Evolution, 112: 96-106. DOI:10.1016/j.ympev.2017.04.018 (  0) 0) |
Wang, Y. R., Wang, Y. Y., Sheng, Y., Huang, J. B., Chen, X., Al-Rasheid, K. A. S. and Gao, S., 2017b. A comparative study of genome organization and epigenetic mechanisms in model ciliates, with an emphasis on Tetrahymena, Paramecium and Oxytricha. European Journal of Protistology, 61: 376-387. DOI:10.1016/j.ejop.2017.06.006 (  0) 0) |
Wilbert, N., 1975. Eine verbesserte technik der Protargolimpr gnation für ciliaten. Mikrokosmos, 64: 171-179. (  0) 0) |
Wilbert, N., 1995. Benthic ciliates of salt lakes. Acta Protozoologica, 34: 271-288. (  0) 0) |
Wilbert, N. and Kahan, D., 1981. Ciliates of solar lake on the Red Sea shore. Archiv für Protistenkunde, 124: 70-95. DOI:10.1016/S0003-9365(81)80004-8 (  0) 0) |
Yan, Y., Xu, Y., Al-Farraj, S. A., Al-Rasheid, K. A. S. and Song, W. B., 2016. Morphology and phylogeny of three trachelocercids (Protozoa, Ciliophora, Karyorelictea), with description of two new species and insight to the evolution of the family Trachelocercidae. Zoological Journal of the Linnean Society, 177: 306-319. DOI:10.1111/zoj.12364 (  0) 0) |
Yan, Y., Rogers, A. J., Gao, F. and Katz, L. A., 2017. Unusual features of non-dividing somatic macronuclei in the ciliate class Karyorelictea. European Journal of Protistology, 61: 399-408. DOI:10.1016/j.ejop.2017.05.002 (  0) 0) |
Yan, Y., Fan, Y., Luo, X., El-Serehy, H. A., Bourland, W. and Chen, X. R., 2018. New contribution to the species-rich genus Euplotes: Morphology, ontogeny and systematic position of two species (Ciliophora; Euplotia). European Journal of Protistology, 64: 20-39. DOI:10.1016/j.ejop.2018.03.003 (  0) 0) |
Yi, Z., Katz, L. and Song, W. B., 2012. Assessing whether alpha-tubulin sequences are suitable for phylogenetic reconstruction of Ciliophora with insights into its evolution in euplotids. PLoS One, 7: e40635. DOI:10.1371/journal.pone.0040635 (  0) 0) |
Young, D. B., 1922. A contribution to the morphology and physiology of the genus Uronychia. Journal of Experimental Zoology, 36: 353-395. (  0) 0) |
Zhao, X. L., Wang, Y. Y., Wang, Y. R., Liu, Y. and Gao, S., 2017. Tetrahymena histone methyltransferase TXR1 is required for both H3 and H3.3 lysine 27 methylation. Science China, Life Sciences, 60: 264-270. DOI:10.1007/s11427-016-0183-1 (  0) 0) |
Zhao, Y., Yi, Z., Warren, A. and Song, W. B., 2018. Species delimitation for the molecular taxonomy and ecology of a widely distributed microbial eukaryotes genus Euplotes (Alveolata, Ciliophora). Proceedings of the Royal Society B: Biological Sciences, 285: 20172159. DOI:10.1098/rspb.2017.2159 (  0) 0) |
 2019, Vol. 18
2019, Vol. 18


