2) College of Chemistry and Chemical Engineering, Ocean University of China, Qingdao 266100, China;
3) College of Marine Geosciences, Ocean University of China, Qingdao 266100, China
Frequent oil spill practices in drilling and transportation are major pollution sources in seawater and marine sediment (Bordoloi and Konwar, 2009; Wang et al., 2018). There are several methods for treating oil pollution in seawater, such as adsorption, chemical dispersion, and microbial degradation. However, the complicated sediment environment has caused a major challenge for marine oil pollution removal (Samelak et al., 2020). Due to the hydrophobicity and high viscosity of crude petroleum hydrocarbon contaminants, they bind tightly to the sediment matrix, resulting in low bioavailability during remediation (Liu et al., 2018). In order to solve this problem, people added surfactants into the remediation system to make much more dispersion and increase its degradation rate (Atlas, 1995; Jemil et al., 2016; Shi et al., 2021). Biosurfactants and synthetic surfactants are conventional species being utilized in many aspects. Of these different forms, biosurfactants are expected to become a substitute for their green and biodegradability (Marchant and Banat, 2012). Studies have already indicated that glycolipid biosurfactants such as rhamnolipids can stably promote the emulsification and dispersion of crude oil, thereby enhancing its biodegradation (Head et al., 2006; Nikolopoulou et al., 2013). Rhamnolipids surfactants have been widely reported to promote the biodegradation of petroleum hydrocarbons (Lai et al., 2009; Wan et al., 2011). Furthermore, it can be used in various scenes, such as wastewater and polluted marine environment, to accelerate the degradation of crude oil, diesel, and polycyclic aromatic hydrocarbons (Whang et al., 2008; Sponza and Gok, 2010; Nikolova et al., 2021). However, these methods have different effects on petroleum hydrocarbons with different carbon chain lengths, which results in limited total degradation capacity of hydrocarbons by biosurfactants (Chen et al., 2013). Till now, people still have no effective treatment measure on the ocean floor.
On the other hand, microbial fuel cells (MFCs) are efficient bioremediation electrochemical technology (Sathe et al., 2021). The MFCs can accelerate the degradation of petroleum and diesel during the metabolism of microorganisms by electro-catalysis. It has been reported that the MFCs can not only screen specific bacteria with degradation ability in petroleum pollution remediation but also generate power through consuming pollutants by anode electrogenic bacteria (Morris and Jin, 2012; Li et al., 2019; Zhang et al., 2022).
Marine sediment microbial fuel cells (MSMFCs) are one kind of MFCs on ocean floor, in which their anodes and cathodes are located in marine sediment and seawater respectively. Driven by the potential difference at the sediment-water interface, electrons from the anode bacteria are transferred to the cathode through an external circuit while the current is generated (Zhou et al., 2018; Li et al., 2022). Our project group has utilized the MSMFCs to degrade organic carbon and petroleum pollutants with good results (Guo et al., 2018). However, how to keep the MSMFCs with a higher power and higher degradability petroleum pollutant still remains a giant task for its practical application (Zhang et al., 2015).
Combining the emulsification and solubilization of rhamnolipids with the anodic bio-electrochemical catalytic effect in the MSMFCs will bring pioneering results to obtain higher removal efficiency of petroleum with higher power output. Therefore, authors add different concentrations of rhamnolipids into oil-containing sediment to construct the novel anodes of the MSMFCs in this paper, and prove that rhamnolipids addition greatly improve anode electrochemical performance and cell power density. Its synergistic effect in MSMFCs significantly improves oil degradation efficiency in marine sediment.
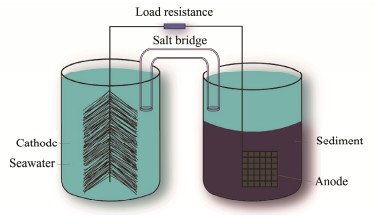
2 Experiment 2.1 Construction of the MSMFCsIn this experiment, the titanium wire (Φ1 mm) passed through carbon felt (3 cm × 3 cm × 0.15 cm) as an anode, in which the carbon felt was treated as followings: impurities on the surface of carbon felt were removed by ultrasonic cleaning in acetone, and then hydroxyl and other oxygen-containing functional groups were introduced by air burning (400℃) in muffle furnace (Li et al., 2015). Carbon fiber brush was utilized as cathode because its much larger surface area will keep its electrochemical stability. The experimental device was a double chamber with a shared big-size cathode to eliminate the potential influence of cathode, which made the experimental results more accurate.
Natural sediment and seawater used in the experiments were obtained from the Jiaozhou Bay nearby Licun River in Qingdao, China. Four samples of oil-sediment by mixing uniformly were put into 1 L beakers, in which each contained 500 g of sediment and 5 g of petroleum. Then 0.025, 0.1 and 0.2 g of rhamnolipids were added into the sediment in the three beakers respectively and stirred well. Therefore, three kinds of battery anodes containing 50, 200 and 400 mg kg−1 rhamnolipids were constructed respectively. As shown in Fig.1 the treated carbon felt was buried about 5 cm deep in the sediment as the anode, the carbon brush was fixed in the seawater of the chamber as cathode, a load resistor of 1000 Ω was connected between the cathode and anode, and the double chamber was connected by a salt bridge to form an ion channel. Battery devices containing 50, 200, 400 mg kg−1 rhamnolipids in sediment were denoted as MSMFC-50, MSMFC-200, MSMFC-400, respectively, and the blank as MSMFC-0.
|
Fig. 1 Schematic diagram of the MSMFCs experimental set-up. |
The contact angles of carbon felt anodes immersed in the different rhamnolipids concentrations were determined using a dynamic contact angle analyzer. The microbial community in the anode biofilm of these cells was analyzed by 16S rDNA high-throughput sequencing. The anodic polarization curves and power density curves were measured with universal meters. The anodic Tafel curves and cyclic voltammetry curves were measured by the Electrochemical Workstation (Shanghai Chenhua Instruments Limited Company, CHI660E), where the anode was used as the working electrode, a Pt foil was used as the counter electrode, and a saturated calomel electrode (SCE) as the reference electrode.
Soxhlet extraction was used to analyze the amount of oil degradation (Mao et al., 2015). During long-term discharge operation, 40 g of sediment was collected from the device every 16 d in a surface dish to dry naturally and ground into fine powder. 10 g of dry sediment was accurately weighed for Soxhlet extraction, and the extracted sample was concentrated by rotary evaporator. The oil content in the sediment was calculated and the degradation rate was derived from its change in content along the experiment.
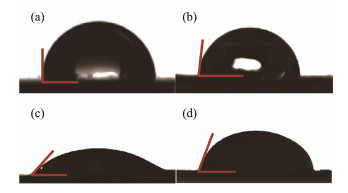
3 Results and Discussion 3.1 Anode Hydrophilicity and Microbial Community Analysis in MSMFCs Sediment 3.1.1 Anode hydrophilicity with different concentrations of rhamnolipidsThe contact angles of the anode surface embedded in different concentrations of rhamnolipids environment are shown in Fig.2, from which the Figs.2a – 2d corresponded to the contact angles of 90˚, 84˚, 48˚ and 69˚, respectively. The results indicate that the contact angle of carbon felt surface becomes smaller when the anode material is in the sea mud environment with rhamnolipids. And it presents the smallest contact angle among them when its concentration is 200 mg kg−1. This is because rhamnolipids are amphiphilic molecules containing hydrophilic and hydrophobic groups (Biktasheva et al., 2022). They can improve the interaction between the electrode material and water, which would be beneficial to the attachment of bacteria. This will facilitate the aggregation of electricity producing bacteria and petroleum degrading bacteria.
|
Fig. 2 Contact angle of different anodes. (a), MSMFC-0; (b), MSMFC-50; (c), MSMFC-200; (d), MSMFC-400. |
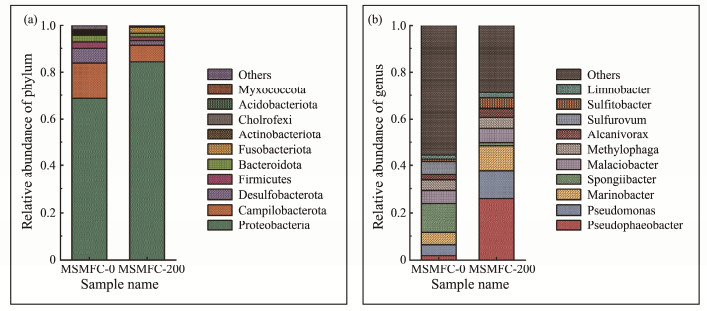
The anodes as electron acceptors can promote the degradation of organic matter in sediment, and microbial dominant species on the anode surface affects the degradation ratio and the electricity production (Wang et al., 2012). The microbial community analysis on the anodes is shown in Fig.3 and Table 1, which indicates that the Chao1 index, Pielou_e index and Shannon index in MSM FC-200 are higher than MSMFC-0. The results show that the solubilization and electrochemical synergy increases the richness and diversity of microbial community in the sediment.
|
Fig. 3 Phylum (a) and genus (b) distributions of the microbial communities on different anodes. |
|
|
Table 1 The alpha diversity analysis of microbial community on different anodes |
As shown in Fig.3a at the phylum level, the most abundant phylum in MSMFC-200 is Proteobacteria, contributing 84.3% of the total microbial community, followed by the phyla Campilobacterota (6.0%), and Desulfobacterota (2.2%). Some studies have found that Proteobacteria was not only the dominant bacteria for oil degradation, but also able to produce electron transfer mediators for different electron acceptors to promote electricity production (Sette et al., 2007; Krishna and Mohan, 2016). Because the anode surface of MSMFC- 200 was enriched with diversity bacteria, it may produce higher power density and oil degradation rates. Campilobacterota exist in anaerobic environments and are able to participate in the oxidation of sulfides in biological systems (Carrier et al., 2020).
The genera on anode biofilms vary in different devices (Fig.3b). The species richness of MSMFC-200 is significantly higher than MSMFC-0. Among the anode microbial genera of MSMFC-200, Pseudophaeobacter has the highest percentage of 26.0%. Liu et al. (2022) have found that there were a large number of oil-degrading bacteria such as Pseudophaeobacter in the area with oil spill. Pseudomonas belonging to Proteobacteria is the predominantly reported organism in the biodegradation of phenolic compounds, which can form thick biofilms and generate mediator to promote electron transfer (Read et al., 2010). Marinobacter plays an important role in the degradation of crude oil in marine environment (Jamal, 2022), accounting for 10.5% in MSMFC-200 and 5.1% in MSM FC-0. This result means that the addition of rhamnolipids into the sediment can cause higher percentage of electricity-producing and petroleum-degrading bacteria, which would cause higher electricity-producing capacity and degradation rate.
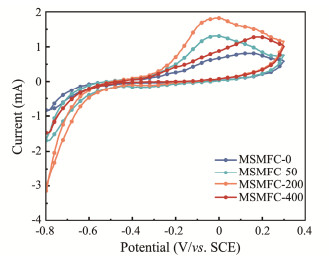
3.2 Anode Electrochemical Performance Analysis 3.2.1 Cyclic voltammetry analysisThe cyclic voltammetry curves of anodes were shown in Fig.4, and the specific capacitances were calculated to get the Table 2 according to Eq. (1) (Han et al., 2022)
| $ C = \frac{1}{{s \cdot A \cdot \Delta V}}\int_{{V_0}}^{{V_0} + \Delta V} {i{\text{d}}V}, $ | (1) |
|
Fig. 4 Cyclic voltammetry curves of different anodes. |
|
|
Table 2 Specific capacitance and oxidation peak of different anodes |
where C is the specific capacity (F m−2); s the sweep speed (V s−1); A the area (m2); ΔV the potential window (V); and i the current (A).
Different anodes in these curves present different capacitance performance. The specific capacitances of MSMFC-0, MSMFC-50, MSMFC-200, and MSMFC-400 by calculation are 127.37, 319.04, 401.45 and 208.80 F m−2, respectively, and the highest oxidation peaks and capacitance value are observed for MSMFC-200 in Fig.4, with the most negative peak potential. The results demonstrate that rhamnolipids can significantly improve anodic capacitive properties and electrochemical activity, which is closely related with its higher percentage of electrogenic bacteria on its surface.
3.2.2 Tafel curves and electron transfer kineticsThe strongly polarized region of the Tafel curve conforms to the Tafel linear extrapolation Eq. (2) (Rajesh et al., 2015):
| $ \eta = a + b\log \left| i \right|, $ | (2) |
where η is the overpotential (mV), and i the exchange current density (mA m−2).
The exchange current density implies the electron transfer kinetic activity and their results were shown in Fig.5 and Table 3 respectively. Among them, the MSMFC-200 has achieved the highest value, which exchange current density is 38.65 times higher than the MSMFC-0 (1.26 × 10−3 mA m−2). This indicates that the addition of rhamnolipids can improve the electron transfer kinetic activity of the anode. The MSMFC-200 has the highest electron transfer kinetic activity, which is consistent with the results of cyclic voltammetry test (Fig.4). The results suggest that rhamnolipids increase the hydrophilicity of the anode surface, making it easier for microorganisms to adhere and degrade oil, while improving the electron transfer kinetics of the anode.
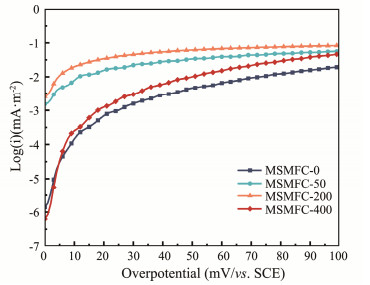
|
Fig. 5 Tafel curves of different anodes. |
|
|
Table 3 Exchange current densities of different anodes |
Polarization curves (Fig.6) show that the slope of MSMFC-200 is significantly lower than that of MSMFC-0, which it presents smaller slope, higher anti-polarization ability and much more stability. MSMFC-0, MSMFC-50, MSMFC-200, and MSMFC-400 achieve the maximum power densities (Pmax) of 66.58, 114.66, 140.24 and 77.80 mW m−2, respectively, in which their internal resistances are 5.60, 3.02, 2.38, and 4.27 Ω. Obviously, the addition of rhamnolipids can reduce the internal resistance and improve the power output of the batteries. This means that the oil can be more fully decomposed and utilized by microorganisms on the anode surface after being dispersed by rhamnolipids. During this period, the higher amount of the electrogenic microorganism and petroleum-degrading bacteria is enriched, and much more electrons produced accordingly, thereby improving its maximum power densities. This is consistent with the results of the microbial community test (Fig.3) and the Tafel test (Fig.5).
|
|
Table 4 Maximum power density of different cells |
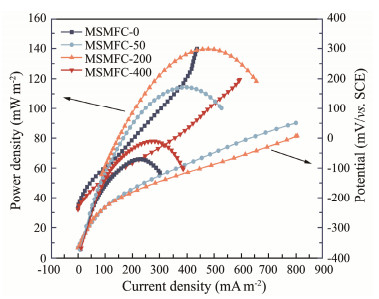
|
Fig. 6 Polarization curves and cell power density curves of different cells. |
The crude petroleum content in the marine sediment during the long-term discharge of the MSMFCs is shown in Fig.7, and it can be clearly seen that the initial contents of oil contaminants of MSMFC-0, MSMFC-50, MSMFC-200 and MSMFC-400 are obtained with 10.20, 10.01, 10.21, and 10.09 g kg−1, respectively. After 64 d of operation, they decrease by 17.55%, 31.07%, 40.06%, and 19.82%, respectively. The degradation ability in the four devices for petroleum was in the following order: MSMFC-200 > MSMFC-50 > MSMFC-400 > MSMFC-0, and the degradation rate of MSMFC-200 was 2.28 times higher than that of MSMFC-0. Combined with the power results (Fig.6), a linear fit analysis of the maximum power density and the amount of oil degradation gives the following relationship and the R2 is 0.9922.
| $ {P_{\max }} = 0.0317\Delta m + 0.0128, $ | (3) |
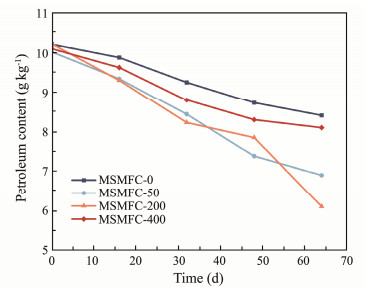
|
Fig. 7 Petroleum contents of different MSMFCs. |
where Pmax is the maximum power density (W m−2), Δm is the amount of oil degradation (g kg−1). Therefore, the maximum output electric energy (Emax) during oil degradation can be expressed by the following equation.
| $ {E_{\max }} = {P_{\max }} \cdot S \cdot t, $ | (4) |
where Emax is the maximum output power per hour (kWh),
|
|
Table 5 Petroleum degradation rate of different MSMFCs |
Large droplets of crude petroleum in natural sediment have poor contact with microorganisms on the anode surface, resulting in low bioavailability and degradation rates (Fig.8a). Emulsifying and solubilizing effect of rhamnolipids make the solubility of insoluble crude oil into much finer droplets and disperse them into pore water for full contact with anode, which facilitates its microbial degradation (Fig.8b). In terms of cell structure, the rhamnolipids help anodic bacterial biofilm to contact much more petroleum droplets, thus allowing the finer droplets to enter the cellular 'digestion' channel. As shown in the Fig.8c, phospholipid molecules in the bacterial membrane have a similar structure and polarity as like rhamnolipids, so that stronger affinity and adsorption between oil and membrane caused the solubilization of oil into the bacterial cell. Meanwhile, rhamnolipids would change the cell membrane permeability of petroleum-degrading bacteria and accelerate the trans-membrane transportation of alkanes and polycyclic aromatic hydrocarbons (PAHs) (Mihelcic et al., 1993).
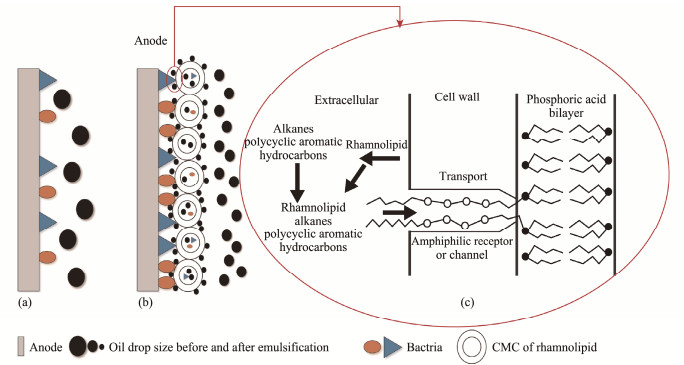
|
Fig. 8 Mechanisms of accelerated degradation of petroleum contaminants in sediments by MSMFCs. (a), model of petroleum degradation by blank sediments; (b), model of petroleum degradation by sediments with rhamnolipids; (c), much finer oil and its processes into cell membrane. |
On the other hand, the natural electric potential difference between aerobic seawater and anaerobic sediment greatly accelerates the anodic electrons consumption through external circuit, thus forming an electrocatalytic effect. This electrocatalytic effect will screen and domesticate anode surface microorganisms, so that the proportion of dominant species of electricity-producing and oildegrading bacteria such as Pseudophaeobacter, Pseudomonas, and Marinobacter increases significantly. Therefore, the degradation rate of oil increases by 2.28 times compared to natural sediments.
MSMFC-200 has the highest electrochemical performance and petroleum degradation rate, which is attributed to its critical micelle concentration (CMC). Yin et al. have found that the CMC of rhamnolipids was 50 mg L−1 with surface tension of 29.3 mN m−1 (Qiang et al., 2009). Its higher concentration than CMC may promote degradation. However, when its concentration is much higher than CMC, rhamnolipids themselves are preferentially utilized by microorganisms, resulting in inhibited degradation of other organic matter (Kosaric, 2001). In addition, excessive concentrations of rhamnolipids can also increase the excess solubility of petroleum hydrocarbons into sediment and increase their toxicity on microbial species (Marecik et al., 2012). Therefore, the MSMFCs at its concentration of 200 mg kg−1 has the strongest synergistic effect and highest bioavailability.
4 ConclusionsAddition of 200 mg kg−1 rhamnolipids into sediment domesticates the bacterial community on the anode surface, and screens priority proportion of electrogenic microorganisms (e.g., Proteobacteria) and petroleum degrading bacteria (Pseudophaeobacter, Pseudomonas and Marinobacter, etc). Specifically, it also significantly increases anode electrochemical performance, with its specific capacitance (401.45 F m−2) being 2.50 times higher than that of the blank (127.37 F m−2) and peak current reaching 1.80 mA. Meanwhile, its anodic exchange current density (4.87 × 10−2 mA m−2) is 38.65 times higher than its blank, resulting in a higher electron transfer kinetic activity. Rhamnolipids not only boosts its maximum power density (140.24 mW m−2) higher than the blank (66.58 mW m−2), but also enhance oil degradation rate about 2.28 times higher than the blank (17.55%) after 64 days of MSMFCs operation. Both emulsification and solubilization effect of rhamnolipids and electrochemical catalytic effect by MSMFCs play an important role in promoting the bacterial decomposition and utilization of petroleum and higher performance of MSMFCs. This method provides a new practical guidance for the in-situ degradation of crude oil pollution and long-term power production from batteries on the seafloor.
AcknowledgementThis work was financially supported by the National Natural Science Foundation of China (No. 22075262).
Author Contributions
Luyao Hou: methodology, investigation, data curation, writing original draft, formal analysis. Xuerong Zai: conceptualization, methodology, resources, supervision, project administration. Yao Meng: methodology, investigation, data curation, formal analysis. Shengkang Liang: conceptualization, writing-review and editing. Yali Cao: software. Yan Chen: methodology. Huaijing Zhang: formal analysis. Xiang Huang: resources. Yubin Fu: conceptualization, resources, writing - review and editing.
Data Availability
The data and references presented in this study are available from the corresponding author upon reasonable request.
Declarations
Ethics Approval and Consent to Participate
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for Publication
Informed consent for publication was obtained from all participants.
Conflict of Interests
The authors declare that they have no conflict of interests.
Atlas, R. M., 1995. Petroleum biodegradation and oil spill bioremediation. Marine Pollution Bulletin, 31: 178-182. DOI:10.1016/0025-326X(95)00113-2 (  0) 0) |
Biktasheva, L., Gordeev, A., Selivanovskaya, S., and Galitskaya, P., 2022. Di- and mono-rhamnolipids produced by the Pseudomonas putida PP021 isolate significantly enhance the degree of recovery of heavy oil from the Romashkino Oil Field (Tatarstan, Russia). Processes, 10: 779-789. DOI:10.3390/pr10040779 (  0) 0) |
Bordoloi, N. K., and Konwar, B. K., 2009. Bacterial biosurfactant in enhancing solubility and metabolism of petroleum hydrocarbons. Journal of Hazardous Materials, 170: 495-505. DOI:10.1016/j.jhazmat.2009.04.136 (  0) 0) |
Carrier, V., Svenning, M. M., Grundger, F., Niemann, H., Dessandier, P. A., Panieri, G., et al., 2020. The impact of methane on microbial communities at marine arctic gas hydrate bearing sediment. Frontiers in Microbiology, 11: 1932-1951. DOI:10.3389/fmicb.2020.01932 (  0) 0) |
Chen, Q. G., Bao, M. T., Fan, X. N., Liang, S. K., and Sun, P. Y., 2013. Rhamnolipids enhance marine oil spill bioremediation in laboratory system. Marine Pollution Bulletin, 71: 269-275. DOI:10.1016/j.marpolbul.2013.01.037 (  0) 0) |
Guo, M., Zai, X. R., Li, T., Zhang, H. J., Zhao, Y. G., Zhao, M. G., et al., 2018. Effect of amino acid addition in marine sediment on electrochemical performance in microbial fuel cells. Fuel Cells, 18: 518-525. DOI:10.1002/fuce.201800021 (  0) 0) |
Han, Y. K., Zai, X. R., and Fu, Y. B., 2022. Effect of oxygen-containing groups on electrochemical and marineelectric field response properties of carbon fiber electrode. IEEE Sensors Journal, 22: 13864-13871. DOI:10.1109/JSEN.2022.3172364 (  0) 0) |
Head, I. M., Jones, D. M., and Roling, W. F. M., 2006. Marine microorganisms make a meal of oil. Nature Reviews Microbiology, 4: 173-182. DOI:10.1038/nrmicro1348 (  0) 0) |
Jamal, M. T., 2022. Enrichment of potential halophilic marinobacter consortium for mineralization of petroleum hydrocarbons and also as oil reservoir indicator in Red Sea, Saudi Arabia. Polycyclic Aromatic Compounds, 42: 400-411. DOI:10.1080/10406638.2020.1735456 (  0) 0) |
Jemil, N., Ben, A. H., Hmidet, N., and Nasri, M., 2016. Characterization and properties of biosurfactants produced by a newly isolated strain bacillus methylotrophicus DCS1 and their applications in enhancing solubility of hydrocarbon. World Journal of Microbiology & Biotechnology, 32: 13. (  0) 0) |
Kosaric, N., 2001. Biosurfactants and their application for soil bioremediation. Food Technology and Biotechnology, 39: 295-304. (  0) 0) |
Krishna, K. V., and Mohan, S. V., 2016. Selective enrichment of electrogenic bacteria for fuel cell application: Enumerating microbial dynamics using MiSeq platform. Bioresource Technology, 213: 146-154. DOI:10.1016/j.biortech.2016.03.117 (  0) 0) |
Lai, C. C., Huang, Y. C., Wei, Y. H., and Chang, J. S., 2009. Biosurfactant-enhanced removal of total petroleum hydrocarbons from contaminated soil. Journal of Hazardous Materials, 167: 609-614. DOI:10.1016/j.jhazmat.2009.01.017 (  0) 0) |
Li, B., Xu, D., Feng, L., Liu, Y., and Zhang, L., 2022. Advances and prospects on the aquatic plant coupled with sediment microbial fuel cell system. Environmental Pollution, 297: 11. (  0) 0) |
Li, W. W., Li, R. P., Li, C. Y., and Zhang, L., 2015. Surface characterization and electrical property of carbon fibers modified by air oxidation. Surface and Interface Analysis, 47: 325-330. DOI:10.1002/sia.5711 (  0) 0) |
Li, X. L., Zheng, R. Y., Zhang, X. W., Liu, Z. W., Zhu, R. Y., Zhang, X. Y., et al., 2019. A novel exoelectrogen from microbial fuel cell: Bioremediation of marine petroleum hydrocarbon pollutants. Journal of Environmental Management, 235: 70-76. DOI:10.1016/j.jenvman.2019.01.007 (  0) 0) |
Liu, L., Fan, H., and Zhao, J., 2018. Progress of biosurfactant in EOR. Oilfield Chemistry, 35: 738-743. (  0) 0) |
Liu, S. X., Sun, R., Cai, M. M., Kong, Y., Gao, Y., Zhang, T. H., et al., 2022. Petroleum spill bioremediation by an indigenous constructed bacterial consortium in marine environments. Ecotoxicology and Environmental Safety, 241: 113796-113804. DOI:10.1016/j.ecoenv.2022.113796 (  0) 0) |
Mao, Z., Chen, J., and Li, W., 2015. Research on the extraction method to determine total petroleum hydrocarbons in contaminated sea sand. Shandong Chemical Industry, 44: 57-61. (  0) 0) |
Marchant, R., and Banat, I. M., 2012. Microbial biosurfactants: Challenges and opportunities for future exploitation. Trends in Biotechnology, 30: 558-565. DOI:10.1016/j.tibtech.2012.07.003 (  0) 0) |
Marecik, R., Wojtera-Kwiczor, J., Lawniczak, L., Cyplik, P., Szulc, A., Piotrowska-Cyplik, A., et al., 2012. Rhamnolipids increase the phytotoxicity of diesel oil towards four common plant species in a terrestrial environment. Water Air and Soil Pollution, 223: 4275-4282. DOI:10.1007/s11270-012-1190-9 (  0) 0) |
Mihelcic, J. R., Lueking, D. R., Mitzell, R. J., and Stapleton, J. M., 1993. Bioavailability of sorbed- and separate-phase chemicals. Biodegradation, 4: 141-153. DOI:10.1007/BF00695116 (  0) 0) |
Morris, J. M., and Jin, S., 2012. Enhanced biodegradation of hydrocarbon-contaminated sediments using microbial fuel cells. Journal of Hazardous Materials, 213: 474-477. (  0) 0) |
Nikolopoulou, M., Eickenbusch, P., Pasadakis, N., Venieri, D., and Kalogerakis, N., 2013. Microcosm evaluation of autochthonous bioaugmentation to combat marine oil spills. New Biotechnology, 30: 734-742. DOI:10.1016/j.nbt.2013.06.005 (  0) 0) |
Nikolova, C. N., Ijaz, U. Z., Magill, C., Kleindienst, S., Joye, S. B., and Gutierrez, T., 2021. Response and oil degradation activities of a Northeast Atlantic bacterial community to biogenic and synthetic surfactants. Microbiome, 9: 1143-1159. (  0) 0) |
Qiang, J., Yin, H., Peng, H., Ye J. S., Qin, H. M., Zhang, N., et al., 2009. Characteristics of a biosurfactant produced by Pseudomonas aeruginosa S6. Acta Scientiae Circumstantiae, 29: 102-110. (  0) 0) |
Rajesh, P. P., Jadhav, D. A., and Ghangrekar, M. M., 2015. Improving performance of microbial fuel cell while controlling methanogenesis by Chaetoceros pretreatment of anodic inoculum. Bioresource Technology, 180: 66-71. DOI:10.1016/j.biortech.2014.12.095 (  0) 0) |
Read, S. T., Dutta, P., Bond, P. L., Keller, J., and Rabaey, K., 2010. Initial development and structure of biofilms on microbial fuel cell anodes. BMC Microbiology, 10: 98-107. DOI:10.1186/1471-2180-10-98 (  0) 0) |
Samelak, I., Balaban, M., Antic, M., Knudsen, T. S., and Jovancicevic, B., 2020. Geochromatographic migration of oil pollution from a heating plant to river sediments. Environmental Chemistry Letters, 18: 459-466. DOI:10.1007/s10311-019-00937-2 (  0) 0) |
Sathe, S. M., Chakraborty, I., and Ghangrekar, M. M., 2021. Wastewater treatment and concomitant bioelectricity production using microbial fuel cell: Present aspects, up-scaling and future Inventiveness. Transactions of the Indian National Academy of Engineering, 6: 633-651. DOI:10.1007/s41403-021-00245-8 (  0) 0) |
Sette, L. D., Simioni, K. C. M., Vasconcellos, S. P., Dussan, L. J., Neto, E. V. S., and Oliveira, V. M., 2007. Analysis of the composition of bacterial communities in oil reservoirs from a southern offshore Brazilian Basin. Antonie Van Leeuwenhoek International Journal of General and Molecular Microbiology, 91: 253-266. DOI:10.1007/s10482-006-9115-5 (  0) 0) |
Shi, Z. X., Li, Y. M., Dong, L. M., Guan, Y. H., and Bao, M. T., 2021. Deep remediation of oil spill based on the dispersion and photocatalytic degradation of biosurfactant-modified TiO2. Chemosphere, 281: 130744-130752. DOI:10.1016/j.chemosphere.2021.130744 (  0) 0) |
Sponza, D. T., and Gok, O., 2010. Effect of rhamnolipid on the aerobic removal of polyaromatic hydrocarbons (PAHs) and COD components from petrochemical wastewater. Bioresource Technology, 101: 914-924. DOI:10.1016/j.biortech.2009.09.022 (  0) 0) |
Tang, C. Y., Fu, Q. S., Criddle, C. S., and Leckie, J. O., 2007. Effect of flux (transmembrane pressure) and membrane properties on fouling and rejection of reverse osmosis and nanofiltration membranes treating perfluorooctane sulfonate containing wastewater. Environmental Science & Technology, 41: 2008-2014. (  0) 0) |
Wan, J. Z., Chai, L. N., Lu, X. H., Lin, Y. S., and Zhang, S. T., 2011. Remediation of hexachlorobenzene contaminated soils by rhamnolipid enhanced soil washing coupled with activated carbon selective adsorption. Journal of Hazardous Materials, 189: 458-464. DOI:10.1016/j.jhazmat.2011.02.055 (  0) 0) |
Wang, A. J., Cheng, H. Y., Ren, N. Q., Cui, D., Lin, N., and Wu, W. M., 2012. Sediment microbial fuel cell with floating biocathode for organic removal and energy recovery. Frontiers of Environmental Science & Engineering, 6: 569-574. (  0) 0) |
Wang, C. Y., Li, X., Guo, J., Lv, Y. C., and Li, Y. W., 2018. Biodegradation of marine oil spill residues using aboriginal bacterial consortium based on Penglai 19-3 oil spill accident, China. Ecotoxicology and Environmental Safety, 159: 20-27. DOI:10.1016/j.ecoenv.2018.04.059 (  0) 0) |
Whang, L. M., Liu, P. W., Ma, C. C., and Cheng, S. S., 2008. Application of biosurfactants, rhamnolipid, and surfactin, for enhanced biodegradation of diesel-contaminated water and soil. Journal of Hazardous Materials, 151: 155-163. DOI:10.1016/j.jhazmat.2007.05.063 (  0) 0) |
Zhang, H., Chao, B., Gao, X., Cao, X., and Li, X., 2022. Effect of starch-derived organic acids on the removal of polycyclic aromatic hydrocarbons in an aquaculture-sediment microbial fuel cell. Journal of Environmental Management, 311: 114783-114795. DOI:10.1016/j.jenvman.2022.114783 (  0) 0) |
Zhang, J., Li, R., and Ji, M., 2015. Influence of sediment characteristics on the power generation capability. Acta Scientiae Circumstantiae, 35: 797-804. (  0) 0) |
Zhou, C. Y., Fu, Y. B., Zhang, H. S., Chen, W., Liu, Z., Liu, Z. H., et al., 2018. Structure design and performance comparison of large-scale marine sediment microbial fuel cells in lab and real sea as power source to drive monitoring instruments for long-term work. Ionics, 24: 797-805. DOI:10.1007/s11581-017-2251-2 (  0) 0) |
 2025, Vol. 24
2025, Vol. 24


