2) Engineering Research Center of Tropical and Subtropical Aquatic Ecological Engineering, Ministry of Education, Guangzhou, 510632, China
Nitrogen (N) is an important nutrient element for the growth of algal cells. Although dissolved inorganic nitrogen (DIN) is considered the most important nutrient source for microalgae, dissolved organic nitrogen (DON) also contributes significantly to the total nutrient pool, and often exceed the concentration of the inorganic forms (Sipler and Bronk, 2015; Glibert, 2017). Many researchers have demonstrated that phytoplankton are capable of utilizing organic nutrients to sustain their growth when they meet the limitation of inorganic nutrients (Pustizzi et al., 2004; Cucchiari et al., 2008; Glibert and Burford, 2017). Actually, up to 70% of DON in the marine environment may be bioavailable (Berman and Bronk, 2003; Sipler and Bronk, 2015). Concentration and proportion of different forms of N can lead to changes in phytoplankton community composition and abundance (Glibert et al., 2016; Moschonas et al., 2017).
phytoplankton have been demonstrated in both natural assemblages and laboratory cultures (Kudela and Cochlan, 2000; Berman and Bronk, 2003; Glibert et al., 2006). Of particular interest has been the linkage between DON and the growth of harmful algal bloom (HAB) species, including the dinoflagellates Karenia brevis (Steidinger et al., 1998), Pfiesteria piscicida (Lewitus et al., 1999), Lingulodinium polyedrum (Kudela and Cochlan, 2000), the diatoms Pseudonitzschia spp. (Loureiro et al., 2009), the raphidophytes Heterosigma akashiwo (Herndon and Cochlan, 2007), Fibrocapsa japonica (Cucchiari et al., 2008), and the brown tide alga Aureococcus anophagefferens (Lomas and Glibert, 2000; Pustizzi et al., 2004; Herndon and Cochlan, 2007). The use of organic compounds may serve as an important ecological strategy for harmful flagellates in the specific competition especially in organic nutrients rich coastal waters (Glibert et al., 2006; Glibert and Burford, 2017).
In this study, we have examined the N growth capabilities of the three different marine phytoplankton taxa, Skeletonema costatum (diatom, Bacillariophyceae), Prorocentrum micans (dinoflagellate, Dinophyceae), and Chattonella marina (Raphidophyceae) in laboratory conditions. Effects of different N sources, concentrations and ratios between nitrate (NO3-N) and urea on the growth of algal cells were estimated. The Monod equation was applied to examine and compare the effect of ambient N concentrations on the growth of algal cells. The purpose of this study was to compare nitrogenous utilization of different microalgal species, and to understand their advantages in phytoplankton competition in the natural sea waters.
2 Materials and Methods 2.1 Algal CulturesUni-algal cultures of S. costatum, P. micans, and C. marina were isolated from Daya Bay, China. The stock cultures were maintained in autoclaved (121℃, 20 min) f/2 media (Guillard, 1975) at 20℃ ± 1℃ and salinity 30 and under 100 μmol photon m-2 s-1 of cool-white fluorescent illumination with a dark: light cycle of 12 h: 12 h. The cultures were treated with mixture antibiotics to destroy the external bacteria before the experiment. Firstly, 10 μg mL-1 penicillin (final concentration) was added in 100 mL cultures in exponential phase, and incubated for 24 h, then 10 mL of the penicillin-treated culture was inoculated to the fresh f/2 medium with 10 μg mL-1 streptomycin sulphate, and incubated for another 24 h, and followed by the treatment of 10 μg mL-1 kanamycin sulfate. The multi-antibiotics treated algal cultures were maintained in the sterile f/2 medium. Before the experiments, the cultures were transferred to the modified f/2 medium, which contained 100 μmol N L-1 (NaNO3), 7 μmol P L-1 (KH2PO4) and 70 μmol Si L-1 (Na2SiO3), approximating the maximum nutrient concentrations found in Daya Bay (Wang et al., 2009). Clones were maintained in the exponential growth phase by serial transfer in the modified f/2 medium. The cells for inoculation were pre-incubated for two days in the N-free medium.
2.2 Experiment Design 2.2.1 Growth under different nitrate concentrationsThe experiments were conducted in batch cultures. The growth of algal cells was examined at seven different concentrations of N (0, 5, 10, 50, 100, 500, 1000 μmol N L-1). The concentrations of P and Si were 7 μmol P L-1 and 70 μmol Si L-1, respectively. Silica was removed in the media for non-diatoms. Elements other than N, P and Si were the same as those in f/2 medium. To reduce the background N and P concentrations, the medium was made with artificial sea salt (Red Coral Sea, nutrient free formula) with salinity 30-31 and pH 7.9 ± 0.1. Algal cells were inoculated into triplicate 250 mL flasks with 150 mL of test medium. According to the cell sizes of the three taxa, the initial cell densities were set at about 5×104 cells mL-1 for S. costatum, and about 103 cell mL-1 for P. micans and C. marina. The experiment was run for 8 days. To minimize cell sedimentation, cultures were shaken by hand three times a day. The incubation condition was the same as that of algal culture in Section 2.1.
2.2.2 Growth under different nitrogen compoundsSeven N compounds were used in this experiment as the sole sources of N supplied, including nitrate (NO3), urea, alanine (Ala), glycine (Gly), threonine (Thr), serine (Ser), and asparaginic acid (Asp). Nitrogen concentration of all experimental groups was 100 μmol N L-1, and P and Si concentrations were the same as those in Section 2.2.1. Test group N0P0 was set as both N- and P-free control, in which neither N nor P was added. N0Pi was the N-free control, in which no N compound was added and P and Si concentrations were the same as the other test groups. To avoid the decomposition of N and P compounds during autoclave treatment, all compounds were added individually to the autoclaved medium (N- and P-free f/2 medium) after filtration through a 0.1 μm pore size disposable syringe filter (Millipore Corporation, Bedford, MA, USA). The methods for pre-incubation and culture conditions were the same as the described in Section 2.2.1.
2.2.3 Growth under different ratios of urea to nitrateUsing urea and NO3-N as N sources, five experimental groups were set with the relative urea-N proportions of 0, 25%, 50%, 75% and 100%, respectively. The sum concentration of urea and NO3-N was 100 μmol N L-1, and P and Si concentrations were the same in Section 2.2.1.
2.3 Cell Counting and Relative GrowthThe cell number was counted every day during the incubation period. The cell counting was performed in a cell counting chamber by placing 0.05-0.1 mL of culture into the chamber, fixed with a drop of Lugol's fixative, and observed under an inverted microscope (Leica DM IRB) at a magnification of 200×. Each sample was counted more than three times until the differences in cell counts were less than 10%.
2.4 Specific Growth Rate (μ) and Half-Saturation Constant (Ks)Specific growth rate (μ, divisions d-1) was calculated using the following equation:
| $ \mu\left(\text { divisions } \mathrm{d}^{-1}\right)=\frac{\ln N 2-\ln N 1}{t 2-t 1}, $ | (1) |
where N2 and N1 are cell densities at times t2 and t1. N2 and N1 are cell densities at d2 and d0 in this study.
The growth rate at the exponential growth phase was used to calculate μmax and Ks using the Monod equation (1949):
| $ \mu = \frac{{{\mu _{\max }} \times S}}{{{K_{\rm{s}}} + S}}, $ | (2) |
where μ is the specific growth rate (divisions d-1) in the test groups, μmax is the maximum specific growth rate (divisions d-1), S is the nutrient concentration (μmol L-1), and Ks, the half-saturation constant for growth, is the nutrient concentration at μmax/2.
2.5 Data AnalysisThe mean and standard deviation were calculated for each treatment from three independent replicate cultures. The means and standard deviations (SD) of all data were calculated and graphed. One-way ANOVA was performed to compare the test groups with the controls and the significant difference among groups using SPSS 19.0 for Windows. Differences are termed significant when P < 0.05. The relationships between growth rate and nutrient concentration were fitted with the Monod equation using Originpro 2019.
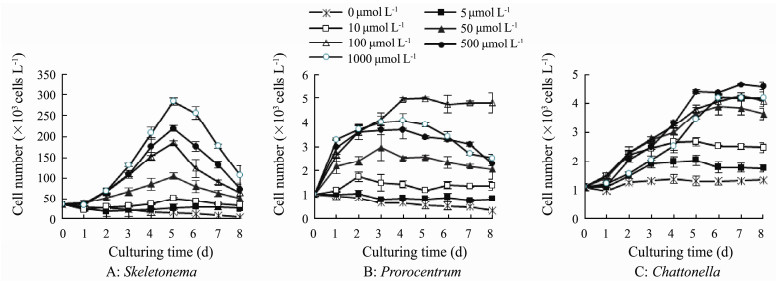
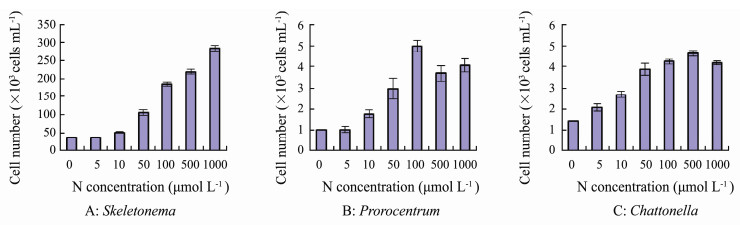
3 Results 3.1 Growth of the Three Phytoplankton Taxa Under Different NO3-N ConcentrationsThe growth curves of the three microalgae obtained under different nitrate concentrations (0-1000 μmol N L-1) are shown in Fig. 1. Growth of S. costatum was observed at concentrations ≥ 10 μmol L-1, with maximum yields increasing with increases in N concentrations from 10 to 1000 μmol L-1 (Figs. 1A, 2A). The maximum yields occurred in day 5 after incubation, and reached 2.84×105 cells mL-1 at the concentration of 1000 μmol L-1.
|
Fig. 1 Growth of the three phytoplankton taxa under different nitrate concentrations. |
|
Fig. 2 Maximum yields of the three phytoplankton taxa under different nitrate concentrations. |
P. micans could not grow at low N concentrations (0-5 μmol L-1) (Figs. 1B, 2B), and significant increases in growth were observed at concentrations more than 10 μmol L-1 (P < 0.01). Best growth occurred at concentration of 100 μmol L-1 with the maximum yield of 4.99×103 cells mL-1, which was significantly higher than those under other N concentrations (P < 0.01).
Growth of C. marina was observed under nitrate concentrations of 5-1000 μmol L-1 (Figs. 1C, 2C), and cell numbers in these treatments were significantly higher than that in N-free control (P < 0.01). The maximum yields increased with increasing concentration of NO3-N to 500 μmol L-1, which was 4.64×103 cells mL-1 (Fig. 2C).
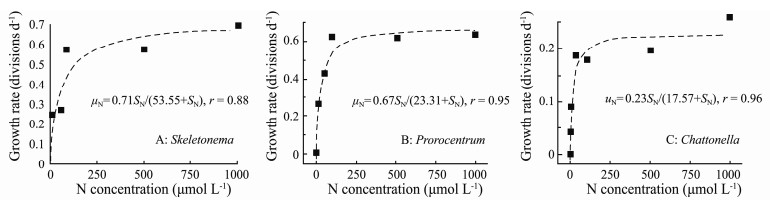
The relationships between specific growth rates (μ) of the three microalgae and the concentrations of NO3-N were determined using the Monod equation (Fig. 3). From the equations, the values of μmax and Ks for NaNO3 were 0.71 divisions d-1 and 53.55 μmol L-1 for S. costatum (r = 0.88), 0.67 divisions d-1 and 23.31 μmol L-1 for P. micans (r = 0.95), and 0.23 divisions d-1 and 17.57 μmol L-1 for C. marina (r = 0.96), respectively.
|
Fig. 3 Relationships between growth rates (μ) and NO3-N concentrations of the three microalgal taxa. The curves were fitted to the Monod equation. |
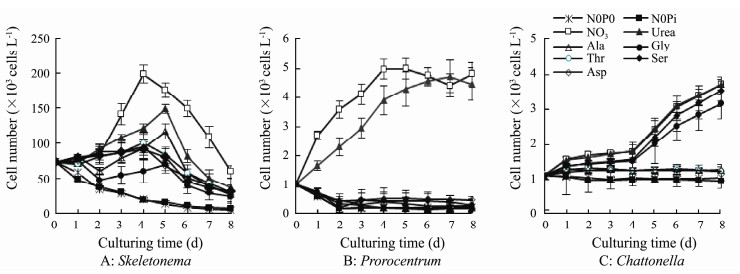
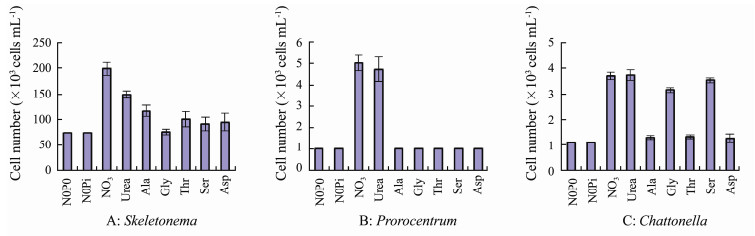
Of the seven compounds provided as N sources, S. costatum grew in both inorganic and the six organic N sources (Figs. 4A, 5A), however the maximum yields in all organic N sources were significantly lower than that obtained in treatment with NO3-N (P < 0.05, or P < 0.01). P. micans grew only in treatments with NO3-N and urea, and cell numbers in the other five amino acids (Ala, Gly, Thr, Ser, and Asp) and the N-free controls (N0P0 and N0Pi) never exceeded the initial inoculation numbers (Figs. 4B, 5B). C. marina grew well in inorganic and three organic N compounds (urea, Gly, and Ser, Figs. 4C, 5C), and cell numbers in the other three amino acids (Ala, Thr, and Asp) increased but showed no significant differences with the N-free controls and the initial inoculation numbers (P > 0.05). The highest yields were obtained in the inorganic NO3-N treatment for S. costatum and P. micans, and were recorded in both NO3-N and urea for C. marina (Fig. 5).
|
Fig. 4 Growth of the three phytoplankton taxa under different nitrogen compounds. |
|
Fig. 5 Maximum yields of the three phytoplankton taxa under nitrogen compounds. |
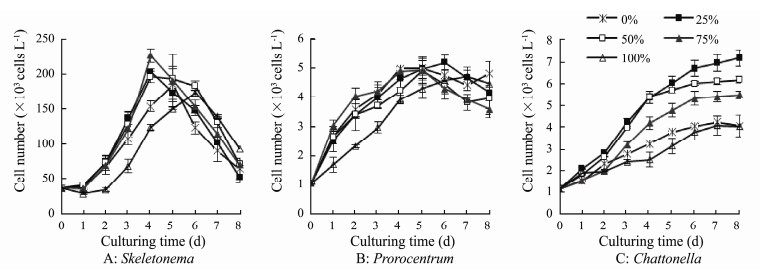
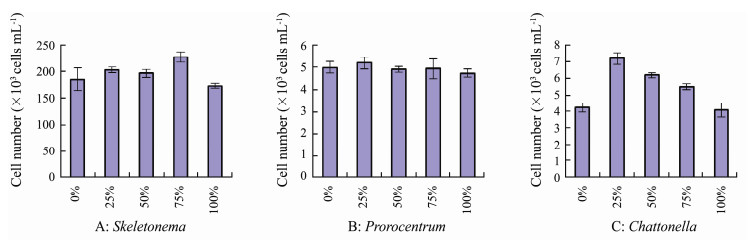
In the experiments with various urea: nitrate ratios, algal cells grew well in all cultures (Figs. 6, 7). Cell numbers of S. costatum increased significantly after experiencing one day lag period, and reached to the maximum numbers of (1.73-2.26)×105 cells mL-1 at day 4-6 (Fig. 6A). The highest yield of S. costatum was recorded in treatment with 75% urea-N (Fig. 7A), which was significantly higher than those in the other treatments (P < 0.05). The growth of P. micans showed similar patterns under different treatments, and cell number increased rapidly at day 1 after incubation, and then dropped at day 5-6 (Fig. 6B). There were no significant differences in maximum yields of P. micans among all treatments (P > 0.05, Fig. 7B). C. marina grew better in the mixture N sources than using NO3-N or urea as the sole N source (P < 0.05, or < 0.01). Best growth of C. marina occurred in treatment with 25% urea-N, and growth decreased as increasing proportions of urea-N (Figs. 6C, 7C).
|
Fig. 6 Growth of the three phytoplankton taxa under different proportions (%) of urea-N. |
|
Fig. 7 Maximum yields of the three phytoplankton taxa under different proportions (%) of urea-N. |
The ability to use various nutrient components for growth differs among phytoplankton species, which may be one of the major factors accounting for species succession in situ (Pustizzi et al., 2004). Organic N compounds are considered as valuable N sources for the growth of many HAB species, such as dinoflagellates K. brevis (Steidinger et al., 1998; Killberg-Thoreson et al., 2014), P. piscicida (Lewitus et al., 1999), L. polyedrum (Kudela and Cochlan, 2000), the raphidophytes H. akashiwo (Herndon and Cochlan, 2007), F. japonica (Cucchiari et al., 2008), and the brown tide alga A. anophagefferens (Pustizzi et al., 2004). Our results demonstrated that NO3-N and urea served as good N sources for all of the three phytoplankton taxa (Figs. 4, 5). P. micans cannot grow in medium of the five free amino acids. C. marina can effectively use the two amino acids Gly and Ser, however cannot utilize the other three amino acids. Though amino acids are small organic N sources that would be easy to be used by phytoplankton for growth, amino acids are the least prioritized N substrates to K. brevis including NH4-N, NO3-N, urea, and humic-N (Killberg-Thoreson et al., 2014). Some phytoplankton taxa cannot even grow on organic N compounds including urea, uric acid and various amino acids, for example Levanderina fissa (formerly Gyrodinium instriatium, Nagasoe et al., 2010; Wang et al., 2017) and Chattonella ovata (Yamaguchi et al., 2008). S. costatum grew well on a variety of N sources including NO3-N, urea and all the five free amino acids. The results suggested that S. costatum had the ability to use a diverse array of N compounds, while P. micans and C. marina can only utilize selective organic N sources. Other reports also demonstrated that Skeletonema had high affinity for DON (Kwon and Oh, 2014; Li et al., 2017).
In natural aquatic environments, urea represents the majority of the organic N, and is important for phytoplankton as the N source (Moschonas et al., 2017). Dissolved amino acids are also utilized by many phytoplankton species (Hu et al., 2014; Moschonas et al., 2017). Shilova et al. (2017) found that urea addition resulted in the greatest increases in chlorophyll a and productivity compared to those of nitrate and ammonium in the oligotrophic waters of the North Pacific Ocean. Results from an annual survey in Loch Creran of Scotland indicated that the abundances of dominant phytoplankton species, mostly small diatoms such as Skeletonema spp., Thalassiosira spp., Pseudo-nitzschia spp., and Chaetoceros spp., showed significant correlations with urea and dissolved free amino acids (Moschonas et al., 2017). Therefore, the ability of S. costatum to use wide kinds of organic N compounds provide this species an advantage to predominate in DON rich coastal and estuarine waters. Actually, S. costatum has been reported as a common dominant diatom in the worldwide coastal sea areas (Wang et al., 2009; Moschonas et al., 2017; Zhou et al., 2017).
High ratios of DON to DIN have been suggested to favor the growth of the brown tide alga A. anophagefferens (Pustizzi et al., 2004), and increase loadings of urea from coastal runoff may be a determining factor in the blooms of Pseudo-nitzschia (Howard et al., 2007). All of the three phytoplankton taxa grew well under different proportions of urea-N in our study, and C. marina grew significantly better in cultures with both NO3- and urea-N (Figs. 6, 7). The better growth in mixture N sources of C. marina thus make this species an advantage in interspecific competition in natural sea waters.
The Monod equation has been used to describe saturation kinetics in many field and laboratory studies (Taylor et al., 2006; Gobler et al., 2012). Maximum specific growth rates (μmax) and half saturation constants (Ks) are two important kinetic parameters derived from this model that quantify the growth responses to environmental nutrient concentrations. The maximum specific growth rates (μmax) and half-saturation constant (Ks) have been reported for the growth of a lot of phytoplankton taxa, however most Ks values are assessed for N uptake rates fitted to the Michealis-Menten equation (Table 1). Ks values for growth were generally < 10 μmol L-1, for example, 1.1 μmol L-1 for Prorocentrum minimum (Taylor et al., 2006), 2.06 μmol L-1 for Margalefidinium polykrikoides (formerly Cochlodinium polykrikoides, Gobler et al., 2012), 5.36 for C. marina (Oh et al., 2009), 8.98 μmol L-1 for Chattonella subsalsa, and 0.3 μmol L-1 for H. akashiwo (Zhang et al., 2006). In comparison with these species, Ks values obtained in this study were quite high, which were 53.55, 23.31, and 17.57 μmol L-1 for S. costatum, P. micans, and C. marina, respectively. These values were even 2.45-3.36 times greater than those obtained for the same species reported (Table 1). Differences in kinetic parameters are often observed in cells growing under different N sources and concentrations (Lomas and Glibert, 2000). Phytoplankton are able to take up more nutrients at higher nutrient concentrations and reach another maximum Ks value (Tantanasarit et al., 2013). The NO3-N uptake rate by Chaetoceros sp., as reported by Lomas and Glibert (2000), demonstrates that at N concentrations between 0 and 40 μmol L-1, the uptake rate reached a maximum plateau of 0.024 pmol cell-1h-1. However, at higher N concentrations, 50 to 260 μmol L-1, the uptake rate increased in a linear pattern with increasing N concentration in solution. Anyway, the Ks values obtained in our study were comparable with Ks in L. fissa at the same N levels, 0 to 1000 μmol L-1 (Nagasoe et al., 2010), and much lower than Ks in Chaetoceros calcitrans at higher N concentrations, 0 to 2110 μmol L-1 (Tantanasarit et al., 2013).
|
|
Table 1 Maximum specific growth rates (μmax) and half-saturation constants (Ks) of nitrogen for different phytoplankton taxa |
Meanwhile, cell size and shape are known to define species-specific differences in N uptake rates and Ks values, and larger cells are suggested to have a lower Ks values than smaller cells (Malone, 1980). The maximum growth rates decreased with increasing cell size in this study, and Ks value was significantly higher for S. costatum than those for the other two species (Table 1). The result suggested that S. costatum had a high N demand for growth. As a small diatom, S. costatum is characterized with high growth rate and short growth circle (Li et al., 2017). The high ambient N concentration stimulated rapid growth of diatoms, and spring diatom blooms in coastal systems typically develop under conditions of high NO3-N concentrations and a well-mixed water column (Lomas and Glibert, 2000). Nitrogen enrichment has been increasing constantly in the worldwide coastal sea areas due to the high anthropogenic loadings, especially for urea, which is widely used in nitrogenous fertilisers (Glibert et al., 2006, 2014; Glibert, 2017). Results of this study showed that S. costatum is capable to utilize wide kinds of organic N compounds. Therefore, the strategy of N utilization for S. costatum make this species an advantage in N enriched sea areas especially the DON-rich coastal waters. However, diatoms require silicon for their growth, and the rapid growth of diatoms may exhaust dissolved silicate in the water column, and thus lead to the collapse of diatom blooms (Wang et al., 2009). By this time, the flagellate species might predominate in the phytoplankton community due to their multiple ecological strategies, such as utilization of organic nutrients (Heisler et al., 2008), mixotrophy (Burkholder et al., 2008), allelopathy (Graneli et al., 2008; Li et al., 2019), and the release of toxic compounds (Sole et al., 2006). Therefore, a proportional shift away from a diatom-dominated community toward flagellates might be expected as the increase of anthropogenic nutrient loadings (Heisler et al., 2008).
AcknowledgementsThis study was supported by the Science & Technology Basic Resources Investigation Program of China (No. 2018FY100200), and the National Natural Science Foundation of China (No. 42076141).
Baek, S. H., Shimode, S., Han, M. S. and Kikuchi, T., 2008. Growth of dinoflagellates, Ceratium furca and Ceratium fusus in Sagami Bay, Japan: The role of nutrients. Harmful Algae, 7: 729-739. DOI:10.1016/j.hal.2008.02.007 (  0) 0) |
Berman, T. and Bronk, D. A., 2003. Dissolved organic nitrogen: A dynamic participant in aquatic ecosystems. Aquatic Microbial Ecology, 31: 273-305. (  0) 0) |
Burkholder, J. M., Glibert, P. M. and Skelton, H. M., 2008. Mixotrophy, a major mode of nutrition for harmful algal species in eutrophic waters. Harmful Algae, 8: 77-93. DOI:10.1016/j.hal.2008.08.010 (  0) 0) |
Collos, Y., Gagne, C., Laabir, M., Vaquer, A., Cecchi, P. and Souchu, P., 2004. Nitrogenous nutrition of Alexandrium catenella (Dinophyceae) in cultures and in Thau laggon, Southern France. Journal of Phycology, 40: 96-103. DOI:10.1046/j.1529-8817.2004.03034.x (  0) 0) |
Cucchiari, E., Guerrini, F., Penna, A., Totti, C. and Pistocchi, R., 2008. Effect of salinity, temperature, organic and inorganic nutrients on growth of cultured Fibrocapsa japonica (Raphidophyceae) from the northern Adriatic Sea. Harmful Algae, 7: 405-414. DOI:10.1016/j.hal.2007.09.002 (  0) 0) |
Gilbert, P. M., 2017. Eutrophication, harmful algae and biodiversity - Challenging paradigms in a world of complex nutrient changes. Marine Pollution Bulletin, 124: 591-606. DOI:10.1016/j.marpolbul.2017.04.027 (  0) 0) |
Glibert, P. M. and Burford, M. A., 2017. Globally changing nutrient loads and harmful algal blooms recent advances, new paradigms, and continuing challenges. Oceanography, 30: 58-69. DOI:10.5670/oceanog.2017.110 (  0) 0) |
Glibert, P. M., Harrison, J., Heil, C. and Seitzinger, S., 2006. Escalating worldwide use of urea - A global change contributing to coastal eutrophication. Biogeochemistry, 77: 441-463. DOI:10.1007/s10533-005-3070-5 (  0) 0) |
Glibert, P. M., Wilkerson, F., Dugdale, R. C., Parker, A. E., Alexander, J. A., Blaser, S. and Murasko, S., 2014. Microbial communities from San Francisco Bay Delta respond differently to oxidized and reduced nitrogen substrates: Even under conditions that would otherwise suggest nitrogen sufficiency. Frontiers in Marine Science, 1: article 17. (  0) 0) |
Glibert, P. M., Wilkerson, F. P., Dugdale, R. C., Raven, J. A., Dupont, C. L., Leavitt, P. R., Parker, A. E., Burkholder, J. M. and Kana, T. M., 2016. Pluses and minuses of ammonium and nitrate uptake and assimilation by phytoplankton and implications for productivity and community composition, with emphasis on nitrogen-enriched conditions. Limnology and Oceanography, 61: 165-197. DOI:10.1002/lno.10203 (  0) 0) |
Gobler, C. J., Burson, A., Koch, F., Tang, Y. and Mulholland, M. R., 2012. The role of nitrogenous nutrients in the occurrence of harmful algal blooms caused by Cochlodinium polykrikoides in New York estuaries (USA). Harmful Algae, 17: 64-74. DOI:10.1016/j.hal.2012.03.001 (  0) 0) |
Graneli, E., Weberg, M. and Salomon, P. S., 2008. Harmful algal blooms of allelopathic microalgal species: The role of eutrophication. Harmful Algae, 8: 94-102. DOI:10.1016/j.hal.2008.08.011 (  0) 0) |
Guillard, R. R. L., 1975. Culture of phytoplankton for feeding marine invertebrates. In: Culture of Marine Invertebrates Animals. Smith, W. L., and Chanley, M. H., eds., Plenum Press, New York, 26-60.
(  0) 0) |
Heisler, J., Glibert, P. M., Burkholder, J. M., Anderson, D. M., Cochlan, W. and Dennison, W. C., 2008. Eutrophication and harmful algal blooms: A scientific consensus. Harmful Algae, 8: 3-13. DOI:10.1016/j.hal.2008.08.006 (  0) 0) |
Herndon, J. and Cochlan, W. P., 2007. Nitrogen utilization by the raphidophyte Heterosigma akashiwo: Growth and uptake kinetics in laboratory cultures. Harmful Algae, 6: 260-270. DOI:10.1016/j.hal.2006.08.006 (  0) 0) |
Howard, M. D. A., Cochlan, W. P., Ladizinsky, N. and Kudela, R. M., 2007. Nitrogenous preference of toxigenic Pseudonitzschia australis (Bacillariophyceae) from field and laboratory experiments. Harmful Algae, 6: 206-217. DOI:10.1016/j.hal.2006.06.003 (  0) 0) |
Hu, Z., Duan, S., Xu, N. and Mulholland, M. R., 2014. Growth and nitrogen uptake kinetics in cultured Prorocentrum donghaiense. PLoS One, 9: e94030. DOI:10.1371/journal.pone.0094030 (  0) 0) |
Hu, Z., Xu, N., Duan, X., Li, A. and Zhang, C., 2010. Effects of urea on the growth of Phaeocystis globose, Scrippsiella trochoidea, Skeletonema costatum. Acta Scientiae Circumstantiae, 30: 1265-1270 (in Chinese with English abstract). (  0) 0) |
Killberg-Thoreson, L., Mulholland, M. R., Heil, C. A., Sanderson, M. P., O'Neil, J. M. and Bronk, D. A., 2014. Nitrogen uptake kinetics in field populations and cultured strains of Karenia brevis. Harmful Algae, 38: 73-85. DOI:10.1016/j.hal.2014.04.008 (  0) 0) |
Kudela, R. M. and Cochlan, W. P., 2000. Nitrogen and carbon uptake kinetics and influence of irradiance for a red tide bloom off southern California. Aquatic Microbial Ecology, 21: 31-47. DOI:10.3354/ame021031 (  0) 0) |
Kwon, H. K. and Oh, S. J., 2014. The importance of dissolved organic nutrients on the interspecific competition between the harmful dinoflagellate Cochlodinium polykrikoides and the diatom Skeletonema sp. Journal of the Korean Society of Oceanography, 19: 232-242. (  0) 0) |
Lee, K. H., Jeong, H. J., Kim, H. J. and Lim, A. S., 2017. Nitrate uptake of the red tide dinoflagellate Prorocentrum micans measured using a nutrient repletion method: Effect of light intensity. Algae, 32(2): 139-153. DOI:10.4490/algae.2017.32.5.20 (  0) 0) |
Lewitus, A. J., Willis, B. M., Hayes, K. C., Burkholder, J. M., Glasgow, H. B., Glibert, P. M. and Burke, M. K., 1999. Mixotrophy and nitrogen uptake by Pfiesteria piscicida (Dinophyceae). Journal of Phycology, 35: 1430-1437. DOI:10.1046/j.1529-8817.1999.3561430.x (  0) 0) |
Li, B., Ou, L. and Lu, S., 2009. Effects of different kinds of nitrogen on growth and nitrate reductase activity of Chattonella marina (Raphidophyceae). Marine Environmental Science, 28(3): 264-267 (in Chinese with English abstract). (  0) 0) |
Li, X., Yan, T., Yu, R. and Zhou, M. J., 2019. A review of Karenia mikimotoi: Bloom events, physiology, toxicity and toxic mechanism. Harmful Algae, 90: 101702. DOI:10.1016/j.hal.2019.101702 (  0) 0) |
Li, Z., Zhou, Y., Wang, X., Shi, X. and Zhang, C., 2017. Effect of urea on the growth of Skeletonema costatum and Karenia mikimotoi. Acta Ecologica Sinica, 37: 3193-3200 (in Chinese with English abstract). (  0) 0) |
Lomas, M. W. and Glibert, P. M., 2000. Comparisons of nitrate uptake, storage, and reduction in marine diatoms and flagellates. Journal of Phycology, 36: 903-913. DOI:10.1046/j.1529-8817.2000.99029.x (  0) 0) |
Loureiro, S., Jauzein, C., Garcés,, E, ., Collos, Y., Camp, J., Vaqu, é and D, ., 2009. The significance of organic nutrients in the nutrition of Pseudo-nitzschia delicatissima (Bacillariophyceae). Journal of Plankton Research, 31: 399-410. DOI:10.1093/plankt/fbn122 (  0) 0) |
Malone, T. C., 1980. Algal size. In: The Physiological Ecology of Phytoplankton. Morris, I., ed., Blackwell Scientific Publications, Oxford, 433-463.
(  0) 0) |
Monod, J., 1949. The growth of bacterial cultures. Annual Review of Microbiology, 3: 371-394. DOI:10.1146/annurev.mi.03.100149.002103 (  0) 0) |
Moschonas, G., Gowen, R. J., Paterson, R. F., Mitchell, E., Stewart, B. M., McNeill, S., Glibert, P. M. and Davidson, K., 2017. Nitrogen dynamics and phytoplankton community structure: The role of organic nutrients. Biogeochemistry, 134: 125-145. DOI:10.1007/s10533-017-0351-8 (  0) 0) |
Nagasoe, S., Shikata, T., Yamasaki, Y., Matsubara, T., Shimasaki, Y., Oshima, Y. and Honjo, T., 2010. Effects of nutrients on growth of the red-tide dinoflagellate Gyrodinium instriatum Freudenthal et Lee and a possible link to blooms of this species. Hydrobiologia, 651: 225-238. DOI:10.1007/s10750-010-0301-0 (  0) 0) |
Nishikawa, T., Tarutani, K. and Yamamoto, T., 2009. Nitrate and phosphate uptake kinetics of the harmful diatom Eucampia zodiacus Ehrenberg, a causative organism in the bleaching of aquacultured Porphyra thalli. Harmful Algae, 8: 513-517. DOI:10.1016/j.hal.2008.10.006 (  0) 0) |
Nishikawa, T., Tarutani, K. and Yamamoto, T., 2010. Nitrate and phosphate uptake kinetics of the harmful diatom Coscinodiscus wailesii, a causative organism in the bleaching of aquacultured Porphyra thalli. Harmful Algae, 9: 563-567. DOI:10.1016/j.hal.2010.04.007 (  0) 0) |
Oh, S., Park, J. S. and Yoon, Y. H., 2009. Growth kinetics on the nutrient of the harmful algae Chattonella marina and C. ovata (Raphidophyceae) isolated from the South Sea of Korea. Korean Journal of Fisheries and Aquatic Sciences, 42: 674-682. DOI:10.5657/kfas.2009.42.6.674 (  0) 0) |
Pustizzi, F., MacIntyre, H., Warner, M. E. and Hutchins, D. A., 2004. Interaction of nitrogen source and light intensity on the growth and photosynthesis of the brown tide alga Aureococcus anophagefferens. Harmful Algae, 3: 343-360. DOI:10.1016/j.hal.2004.06.006 (  0) 0) |
Shilova, I. N., Mills, M. M., Robidart, J. C., Turk-Kubo, K. A., Bjorkman, K. M. and Kolber, Z., 2017. Differential effects of nitrate, ammonium, and urea as N sources for microbial communities in the North Pacific Ocean. Limnology and Oceanography, 62: 2550-2574. DOI:10.1002/lno.10590 (  0) 0) |
Sipler, R. E., and Bronk, D. A., 2015. Chapter 4: Dynamics of dissolved organic nitrogen. In: Biogeochemistry of Marine Dissolved Organic Matter. 2nd edition. Carlson, D., ed., Academic Press, Boston, 127-232.
(  0) 0) |
Sole, J., Garcia-Ladona, E. and Estrada, M., 2006. The role of selective predation in harmful algal blooms. Journal of Marine Systems, 62: 46-54. DOI:10.1016/j.jmarsys.2006.04.002 (  0) 0) |
Steidinger, K. A., Vargo, G. A., Tester, P. A., and Tomas, C. R., 1998. Bloom dynamics and physiology of Gymnodinium breve with emphasis on the Gulf of Mexico. In: Physiological Ecology of Harmful Algal Blooms. Anderson, D. M., Cembella, A. D., and Hallegraeff, G. M., eds., Springer-Verlag, Berlin, 133-153.
(  0) 0) |
Tantanasarit, C., Englande, A. J. and Babel, S., 2013. Nitrogen, phosphorus and silicon uptake kinetics by marine diatom Chaetoceros calcitrans under high nutrient concentrations. Journal of Experimental Marine Biology and Ecology, 446: 67-75. DOI:10.1016/j.jembe.2013.05.004 (  0) 0) |
Taylor, G. T., Gobler, C. J. and Sañudo-Wilhelmy, S. A., 2006. Speciation and concentrations of dissolved nitrogen as determinants of brown tide Aureococcus anophagefferens bloom initiation. Marine Ecology Progress Series, 312: 67-83. DOI:10.3354/meps312067 (  0) 0) |
Wang, Z. H., Guo, X., Ou, L. J. and Lin, L. C., 2017. Effects of nitrogen and phosphorus on the growth of Levanderina fissa: How it blooms in Pearl River Estuary. Journal of Ocean University of China, 16: 114-120. DOI:10.1007/s11802-017-3080-7 (  0) 0) |
Wang, Z. H., Zhao, J. G., Zhang, Y. J. and Cao, Y., 2009. Phytoplankton community structure and environmental parameters in aquaculture areas of Daya Bay, South China Sea. Journal of Environmental Science, 21: 1268-1275. DOI:10.1016/S1001-0742(08)62414-6 (  0) 0) |
Yamaguchi, H., Sakamoto, S. and Yamaguchi, M., 2008. Nutrition and growth kinetics in nitrogen and phosphorus-limited cultures of the novel red tide flagellate Chattonella ovata (Raphidophyceae). Harmful Algae, 7: 26-32. DOI:10.1016/j.hal.2007.05.011 (  0) 0) |
Yamamoto, T., Oh, S. J. and Kataoka, Y., 2004. Growth and uptake kinetics for nitrate, ammonium and phosphate by the toxic dinoflagellate Gymnodinium catenatum isolated from Hiroshima Bay, Japan. Fisheries Science, 70: 108-115. DOI:10.1111/j.1444-2906.2003.00778.x (  0) 0) |
Zhang, C. and Zhou, J., 1997. Nutrient uptake kinetics and growth under nutrient limitation of Pseudonitzschia. Oceanologia et Limnology Sinica, 28: 599-603 (in Chinese with English abstract). (  0) 0) |
Zhang, Y. H., Fu, F. X., Whereat, E., Coyne, K. J. and Hutchins, D. A., 2006. Bottom-up controls on a mixed-species HAB assemblage: A comparison of sympatric Chattonella subsalsa and Heterosigma akashiwo (Raphidophyceae) isolates from the Delaware Inland Bays, USA. Harmful Algae, 5: 310-320. DOI:10.1016/j.hal.2005.09.001 (  0) 0) |
Zhou, Y. P., Zhang, Y. M., Li, F. F., Tan, L. J. and Wang, J. T., 2017. Nutrients structure changes impact the competition and succession between diatom and dinoflagellate in the East China Sea. Science of the Total Environment, 574: 499-508. DOI:10.1016/j.scitotenv.2016.09.092 (  0) 0) |
 2021, Vol. 20
2021, Vol. 20


