2) Laboratory for Marine Ecology and Environmental Science, Qingdao Marine Science and Technology Center, Qingdao 266237, China;
3) Institute of Marine Chemistry, Ocean University of China, Qingdao 266100, China
Methyl halides, such as chloromethane (CH3Cl), bromomethane (CH3Br), and iodomethane (CH3I), are significant trace greenhouse gases that play a crucial role in global climate change. These compounds can absorb infrared radiation, contributing to global warming (Abrahamsson et al., 2018; WMO, 2018; Bidleman et al., 2020). Additionally, when exposed to ultraviolet light, methyl halides in the atmosphere can produce halogen free radicals (X·), which can influence the concentration of other greenhouse gases through atmospheric chemical reactions (Molina et al., 1974). More importantly, methyl halides are critical ozone-depleting substances (Hossaini et al., 2015; Carpenter et al., 2017; Iglesias-Suarez et al., 2020; Zhang et al., 2020).
Methyl halides originate from both biotic and abiotic processes, with abiotic processes in the ocean including chemical and photochemical reactions. Photochemical reactions are a significant abiotic source of methyl halides. Previous studies have shown that photochemical reactions can produce CH3Br (Keppler et al., 2000), CH3Cl (Moore, 2008; Yang et al., 2020), and CH3I (Moore and Zafiriou, 1994; Hu et al., 2016). Yang et al. (2010) found that these photochemical reactions predominantly occur in surface seawater. Colored dissolved organic matter (CDOM) in the ocean can undergo photochemical reactions in chlorine-containing solutions to form CH3Cl (Moore, 2008). Richter et al. (2004) demonstrated the photochemical generation of CH3I by exposing disinfected tropical Atlantic seawater to light. It has been shown that environmental pollutants with methoxy groups can act as methyl sources in the photochemical synthesis of methyl halides. The methyl group is derived from precursor substances containing the methoxy group (R-O-CH3), which then reacts photochemically with halogen ions (X−) to produce methyl halides. Additionally, Yang et al. (2020) indicated that organic matter with ethy-loxy or methylamino group structures, such as 2-ethoxy-phenol and carbofuran, can produce methyl halides in the presence of X−.
The photochemical production of methyl halides is influenced by environmental factors. The generation rate of CH3I and CH3Cl under light conditions is significantly higher compared to that under dark conditions (Moore and Zafirious, 1994; Moore, 2008). Ziska et al. (2013) attributed the high concentration of CH3I in subtropical circulation waters to photochemical production. Stemmler et al. (2014) simulated the global production of CH3I in seawater and suggested that photochemical generation and release are the primary sources of CH3I in surface seawater. These studies indicate that light promotes the generation of methyl halides in seawater; however, the effects of light type, light intensity, and light radiation band on the generation of methyl halides, as well as the optimal conditions, still need further exploration.
Dissolved organic matter (DOM) can be photochemically degraded to form methyl halides (Tai et al., 2012). It has been demonstrated in marine and estuarine regions that DOM provides a carbon source for the photochemical production of methyl halides (CH3I and CH3Cl) (Moore, 2008). Moore and Zafirious (1994) found that the photochemical production of CH3I in coastal seawater was higher than that in Labrador water, possibly due to the higher concentration of organic matter in coastal seawater. Moreover, Chen et al. (2020) found a significant positive correlation between CH3I production and dissolved organic carbon (DOC) content in the Yangtze River Basin, indicating that DOC provides the precursor for the photochemical reaction; high concentrations of organic matter would favor CH3I production. Liu et al. (2015) discovered that DOM can generate transient brominated substances in seawater and that different DOM types can affect the yield of these transient brominated substances. It is particularly important to note that the type of DOM also affects the yield of methyl halides. Previous studies have shown that large molecular weight organic matter exhibits higher biological and chemical activity (Amon and Benner, 1996), and that colloids and large molecular weight organic matter are more prone to photodegradation than small molecular weight organic matter (Zhu et al., 2017). While DOM promotes the photochemical production of methyl halides in seawater, it can also act as an inhibitor in some cases. This is mainly because the reaction sites on DOM can also react with free radicals, resulting in competition with the formation of methyl halides (Ge et al., 2009). Additionally, DOM may combine with organic matter (Burns et al., 1996), which also leads to inhibition of photochemical reactions.
In the photochemical production of methyl halides, the concentration of halogen ions (X−) is also an important influencing factor. The addition of I− significantly increased the concentration of CH3I in deoxidized water samples (Moore and Zafirious, 1994), and the increase of X− concentration had a promoting effect on the photochemical production of methyl halides. Other parameters, such as pH, DO, Fe3+, and salinity, can also affect the photochemical production of methyl halides in seawater (Chen et al., 2020). A high pH value promoted the photochemical production of CH3Cl (Moore, 2008). Similarly, Huang et al. (2002) found that the production of trichloromethane (CHCl3) under alkaline conditions was significantly higher than under neutral conditions during the chlorination disinfection of drinking water. However, Yang et al. (2020) drew a different conclusion; when CH3Cl was generated under UV254 irradiation using guaiacol as a precursor, they found that the CH3Cl yield decreased with increasing pH in the range of 5.5 – 10.5, with the lowest yield at pH 10.5. This suggests that the effect of pH on the photochemistry of methyl halides still needs further research for confirmation.
The content of DO in the ocean also affects the photochemical production of methyl halides. The presence of oxygen inhibits the production of CH3I, as some scholars believe that oxygen acts as a radical inhibitor; ·CH3 can react with oxygen to produce CH3OO·, thereby inhibiting the production of methyl halides (Zafirious et al., 1990). Yang et al. (2020) noted that the formation of CH3Cl remained largely unchanged regardless of the presence of oxygen. Fe3+ can induce and catalyze the oxidation of organic substrates in nature to produce alkanes, olefins, oxygenated hydrocarbons, and halogenated hydrocarbons (Comba et al., 2015). Fe3+ could oxidize DOM, affecting CH3I generation by altering photochemical processes. Fe3+ generates hydroxyl radicals (OH·) and Fe2+ under light conditions, which react with DOM in the environment. Light-induced OH· can oxidize most DOM to generate CH3·, which reacts with halogen ions to produce CH3I. Therefore, studying the effect of Fe3+ on the photochemical production of CH3I is necessary (Bai et al., 2023). Keppler et al. (2000) showed that Fe3+ can oxidize guaiacol to produce o-quinone, while it is reduced to ferrous ions (Fe2+), during which halogen ions combine with methyl groups to form methyl halides. Zhu et al. (2007) studied the role of Fe3+ in the chlorination disinfection of drinking water and found that when the pH was slightly acidic, Fe3+ had an inhibitory effect on the formation of trimethyl halides, but when the pH was slightly alkaline, Fe3+ promoted the formation of trimethyl halides.
In this study, guaiacol, a natural monomer of humic acid containing a methoxy group (R-O-CH3), was used as the research object to study the photochemical production of methyl halides in a simulated artificial seawater system. The effects of various marine environmental factors, such as illumination time, type, intensity, precursor substance concentration, pH, Fe3+ concentration, DO, and halogen ion (X−) concentration, on the photo induced production of methyl halides were evaluated. Seawater from different locations offshore Qingdao was selected for photochemical experiments to study the influence of varying contents and sources of dissolved organic matter (DOM) on the photochemical production of halogenated hydrocarbons. This research aims to enrich the data of photochemical production of halogenated hydrocarbons in the ocean in China, provide essential parameters to fill relevant gaps, and offer a more scientific basis for understanding their formation mechanisms.
2 Methods 2.1 Preparation of SolutionsTo study the photochemical production of methyl halides in natural seawater, a sampling survey was conducted in the coastal sea area of Qingdao, China, on April 19, 2021. The sampling stations are shown in Fig.1. Samples from surface seawater (0 – 5 m) were collected and filtered on-site using 0.45 μm and 0.2 μm glass fiber filter membranes to remove phytoplankton and bacteria. These samples were then brought to the laboratory for immediate treatment and analysis. According to Martin's (1972) method, the composition of artificial seawater includes 24.7 g sodium chloride (NaCl), 13.0 g magnesium chloride hexahydrate (MgCl2·6H2O), and 9.0 g sodium sulfate decahydrate (Na2SO4·10H2O) dissolved in 954 g H2O to obtain 1 L of artificial seawater. Guaiacol (20 μmol L−1) and halide ions (X−: Cl−, I−) served as precursors for methyl halides. Seawater samples were extracted with a 100 mL air-tight syringe and placed in sealed quartz bottles. The samples were exposed to a solar lighting simulator in a constant temperature water bath at 20℃ with a light intensity of 550 W m−2 for 5 h. Solutions were prepared by step-by-step dilution to achieve concentrations of 6, 0.6, and 0.06 ppm KI solution, and 100, 10, 1, and 0.1 ppm KBr solution.
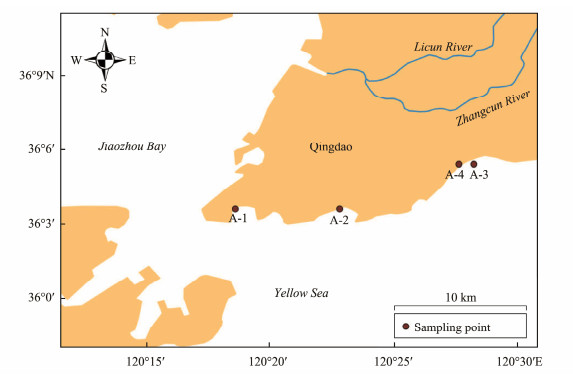
|
Fig. 1 Locations of sampling sations. |
Potassium iodide and potassium bromide of analytical grade were purchased from Aladdin Company. Sodium chloride (analytical grade), magnesium chloride hexahydrate (analytical grade), sodium sulfate decahydrate (analytical grade), guaiacol (chemically pure), and iron (III) chloride hexahydrate (analytical grade) were purchased from Sinopharm Group Chemical Reagent Co., Ltd.. Standard gases of VHCs were obtained from Spectra Gases (America).
2.2 Irradiation ExperimentsSeawater was cleaned with high-purity nitrogen to remove background methyl halides, ensuring blank levels before the irradiation experiment. The quartz tube in the irradiation experiment reactor was tested for gas tightness. Prepared solutions were extracted with a 100 mL air-tight syringe and placed into sealed 50 mL quartz bottles without headspace. The quartz bottles were irradiated in a Sun-test CPU solar simulator, maintained in a constant temperature water bath at 20℃. The xenon arc lamp in the solar simulator was adjusted for illumination intensity, ranging from 400 to 765 W m−2, depending on the experimental requirements.
For the light radiation experiments, the reactor was kept in a solar simulator at 20℃ and 550 W m−2 for 5 h. Quartz bottles were wrapped in various materials: full light, Mylar-D film (transmitting 80% UV-A and small amounts of UV-B), resin UF3 Plexiglas film (attenuating all UV-A and UV-B while allowing visible light), and double-layer aluminum foil (lightproof, as a dark control) to investigate photochemical production of methyl halides under different light radiation bands of UV-B and UV-A. After exposure, 40 mL of sample was extracted with an air-tight syringe and injected into the purge-and-trap device for pretreatment, followed by gas chromatograph analysis.
2.3 Analytical ProcedureThe concentrations of CH3Cl and CH3I in all samples were determined using a lab-established purge-and-trap pretreatment system coupled with a gas chromatograph (Agilent 6890N) equipped with a microelectron capture detector (μECD) (Yang et al., 2007; Lu et al., 2010). Samples were carefully extracted with minimal disturbance to avoid air contamination and transferred to the purge-and-trap pretreatment system. The samples were purged with high-purity nitrogen (N2) at a flow rate of 60 mL min−1 for 14 min. The purged substances passed through a drying tube containing magnesium perchlorate (Mg(ClO4)2) and sodium hydroxide (NaOH) to remove water (H2O) and carbon dioxide (CO2), respectively. The gas was trapped in a stainless steel tube with liquid nitrogen at −150℃ for 14 min, then desorbed with boiling hot water for 2 min and analyzed in a gas chromatographic column (DB-624 elastic quartz capillary column; Agilent; 60 m; 0.53 mm; 3 μm) (Li et al., 2018). Each experimental group had three parallel samples. Methyl halides were quantified using external standard calibration. The detection limits for CH3Cl and CH3I were 0.03 – 0.5 pmol L−1, with precisions of 3% – 6% and recoveries of 93.8% – 109.7%, confirming the method's accuracy and feasibility.
Dissolved organic carbon (DOC) concentration was determined by catalytic high-temperature oxidation using a TOC-VCPH (SHIMADZU) total organic carbon analyzer. An 80 μL sample was combusted, and the CO2 produced was detected by a gas analyzer (NDIR). The DOC concentration was calculated using the calibration curve of a potassium hydrogen phthalate standard solution.
3 Results and Discussion 3.1 Influencing Factors 3.1.1 Illumination time and radiation wavebandsWith the increase in illumination time, the production of CH3I and CH3Cl in different optical radiation bands in seawater samples showed an increasing trend, as shown in Fig.2. Initially, the amount of methyl halides produced under dark conditions was slightly higher than under light conditions. This could be due to the activity of microorganisms in the seawater related to methyl halide production, consistent with the hypothesis that halides can be produced by microbial release under dark conditions (Fuse et al., 2003). However, in the later stages, the concentration of methyl halides (CH3I and CH3Cl) under light conditions was significantly higher than under dark conditions. Moore and Zafiriou (1994) found that the continuous production of CH3I in oxygenated water under sunlight far exceeded the amount produced under dark conditions, concluding that CH3I was produced by the interaction between sunlight and seawater. Yang et al. (2020) also determined that light plays a crucial role in the formation of CH3Cl, with the concentration of CH3Cl in a dark environment remaining relatively unchanged.
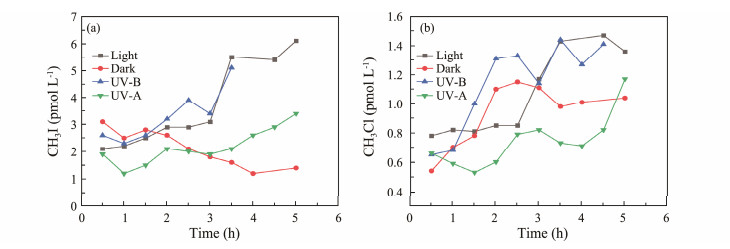
|
Fig. 2 Effect of illumination time and radiation wavebands on CH3I (a) and CH3Cl (b) photochemical production. |
Chen et al. (2020) and Liu et al. (2020) studied the photochemical production of CH3I and CH3Cl using humic acid as a precursor. They found that the production of CH3I and CH3Cl increased with radiation time, suggesting that CH3I and CH3Cl may have similar photochemical production mechanisms. Furthermore, Stemmler et al. (2014) used models to explain the global observations of photochemical production of CH3I surface concentrations, emphasizing the correlation between CH3I concentration and light. These studies fully demonstrate that light is a crucial factor in the production of methyl halides, a finding also confirmed in this experiment.
The amount of CH3I and CH3Cl produced under UV-B radiation was roughly the same as, or even slightly higher than, that under light for some periods, while the amount of methyl halides produced under UV-A radiation was significantly less. This suggests that light promotes the formation of CH3I and CH3Cl, with UV-B radiation having a greater impact on the photochemistry of methyl halides (Fig.2). Li et al. (2021) also concluded that UV-B promotes CH3I production through photochemical culture experiments. This result may be due to CDOM in seawater being easily excited by ultraviolet bands to form excited state CDOM (3CDOM*) (Zepp et al., 1985). Additionally, NO3-N and NO2-N in seawater primarily absorb UV-B sunlight to produce hydroxyl radicals (OH·) (Goldstein and Rabani, 2007; Carena and Vione, 2016). Both 3CDOM* and OH· can oxidize X− to generate X·, which then participates in the photochemical production of methyl halides (Jammoul et al., 2009; Brigante et al., 2014; Dong et al., 2021).
3.1.2 Illumination intensityThe production of CH3I and CH3Cl in samples under five different illumination intensities after 5 h of exposure is shown in Fig.3. At the same illumination time, the production of CH3I and CH3Cl increased with the increasing illumination intensity. For CH3Cl, the production was directly proportional to the light intensity. In the case of CH3I, production did not change significantly when the illumination intensity increased from 450 W m−2 to 650 W m−2, but it significantly increased when the illumination intensity further increased from 650 W m−2 to 765 W m−2. This could be because CH3I might also be produced under dark conditions. Goodwin et al. (1997) found that CH3I could be produced under dark conditions through contrast experiments conducted in both dark and light conditions.
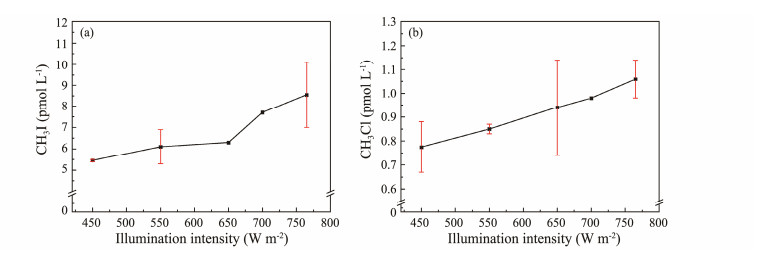
|
Fig. 3 Effect of illumination intensity on CH3I (a) and CH3Cl (b) photochemical production. |
High radiation intensity favors the photochemical generation of CH3I in seawater (Shi et al., 2014). Archer et al. (2007) showed that the concentration of CH3I was related to seasonal variations in light; higher illumination intensity in summer correlated with increased CH3I concentrations in seawater. Chen et al. (2020) also suggested that higher illumination intensity promoted the photochemical production of CH3I in artificial seawater. Increased illumination intensity provides more energy, facilitating the excitation of Cl−, as well as the sensitized photolysis and self-photolysis of the precursor. Studies have shown that the photochemical formation mechanisms of CH3Cl and CH3I are similar (Moore, 2008; Ooki et al., 2010; Yang et al., 2020). Both Cl− and I− can break the carbon-oxygen bond (-O-CH3) and mediate the reaction of the methyl group (-CH3) to produce CH3I and CH3Cl. Therefore, it is speculated that increased light intensity promotes the production of CH3I and CH3Cl in seawater.
3.1.3 Guaiacol concentrationAs guaiacol concentration increased, the production of CH3I and CH3Cl also showed an upward trend, as shown in Fig.4. However, the rate of increase was not directly proportional to the guaiacol concentration. At higher guaiacol concentrations, the increase in CH3I concentration gradually leveled off, while CH3Cl production either plateaued or declined. The highest CH3Cl production occurred at a guaiacol concentration of 96 μmol L−1.
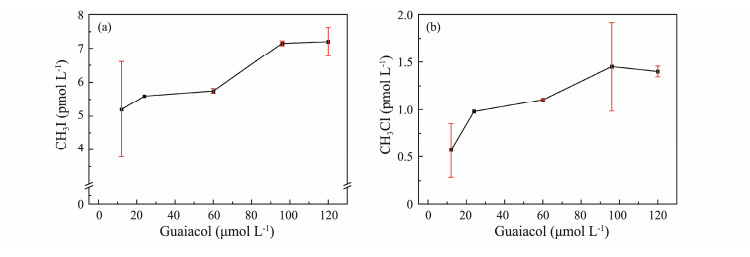
|
Fig. 4 Effect of guaiacol concentration on CH3I (a) and CH3Cl (b) photochemical production. |
High concentrations of organic matter can promote the production of methyl halides, likely because organic matter acts as both a photoadsorbent and a precursor of ·CH3 (Moore, 1994). Chen et al. (2020) found that increasing humic acid concentrations promoted the photochemical production of CH3I in artificial seawater. Research has indicated that methoxy-containing simple lignin molecules may be the methyl source for methyl halides (Moore, 2008). Reckhow et al. (1990) demonstrated that the methyl group in methyl halides originates from precursor substances containing methoxy (R-O-CH3). Guaiacol, a natural humic acid monomer containing the R-O-CH3 structure, is considered a good methyl source. Thus, higher guaiacol concentrations result in higher photochemical yields of CH3I and CH3Cl. However, the photochemical production mechanism of CH3I appears to be a second-order reaction, where the concentrations of ·I and ·CH3 influence CH3I production. Therefore, at high guaiacol concentrations, the reaction may become I− limited, with the limiting I- affecting the reaction rate, resulting in CH3I production that is not directly proportional to guaiacol concentration. Yang et al. (2020) found no linear relationship between CH3Cl yield and guaiacol concentration under UV254 irradiation, possibly due to the optical shielding effect of high guaiacol concentrations limiting the photochemical reaction. Liu (2020) observed that the formation rate constant of CH3Cl, corrected for light shielding, was almost linearly related to humic acid concentration.
Moore (2008) speculated that an intermediate plays a direct or indirect role in methyl halide formation, possibly serving two functions: 1) acting as a direct precursor by reacting with X−, and 2) catalyzing methyl halide production. Yang et al. (2020) showed that precursor photodegradation produces the arenium ion intermediate, leading to Cl− nucleophilic attack, breaking the C-O bond on the methoxy group, and ultimately resulting in CH3Cl formation. The second potential role is that the energy transfer of active intermediates in the excited state induces free radical reactions in other compounds, influencing the photochemical reactions of organic compounds (Bi, 2017). Dallin et al. (2009) indicated that protonation of the aromatic ring is a key step in the photochemical reaction of the precursor. Thus, guaiacol concentration affects the amount of the intermediate, thereby influencing the photochemical yields of CH3I and CH3Cl.
3.1.4 Halogen ion (X−) concentrationFig.5 shows the changes in CH3I production with increasing concentrations of I− and Br−. The production of CH3I significantly increased with higher I− concentrations, while it decreased as Br- concentration increased. Moore and Zafiriou (1994) reported a possible photochemical production pathway of CH3I in seawater. As a source of iodine free radicals, an increased I− concentration would naturally affect CH3I production. Simulation experiments have shown that CH3I can be produced in filtered seawater, with production increasing alongside I− concentration (Lu et al., 2008). Chen et al. (2020) demonstrated that a higher I− concentration led to a more thorough reaction with humic acid, thereby increasing CH3I production. These findings align with our experimental results, indicating that an increase in I− concentration significantly promotes the photochemical production of CH3I. The higher the I− concentration, the more pronounced the effect. A turning point in the curve (Fig.5) may suggest that the reaction is a second-order reaction, where the limiting factor becomes the concentration of the precursor guaiacol, thereby reducing the influence of I− concentration on the reaction.
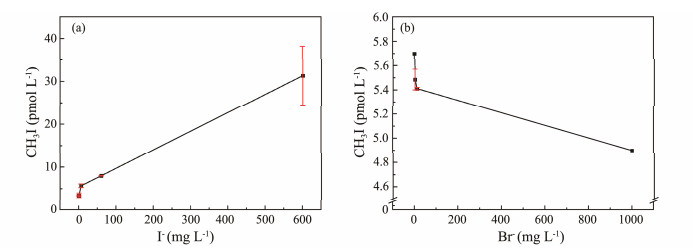
|
Fig. 5 Effect of I− (a) and Br− (b) concentration on CH3I photochemical production. |
Similarly, Br−, as a type of X−, has been extensively studied for its role in producing CH3Br under photochemical conditions. With increasing Br− concentration, the yield of CH3Br from photochemical reactions also increases (Bi et al., 2017). However, when the precursor concentration is fixed, the amount of ·CH3 produced by photolysis is also fixed. Different X− types will compete for ·CH3. Therefore, with higher Br− concentration, the decreased CH3I production likely results from I− and Br− competing for the ·CH3 produced by photolysis of the precursor material.
Regarding CH3Cl production, changes in its quantity with increasing I− and Br− concentrations are shown in Fig.6. CH3Cl production shows a clear downward trend as X− concentration increases. However, when X− concentration reaches a certain level, further increases have no significant impact on CH3Cl photochemical production. DOM induces electron transfer after UV radiation absorption, converting Cl− to Cl·, which then attacks the methyl group of guaiacol to form CH3Cl (Gjessing and Kallqvist, 1991). In seawater, Br− and I− primarily compete with Cl− at the reaction site of precursor substances, causing CH3Cl production to decrease as Br− and I− concentrations increase. Compared to Br− and I−, Cl− is not a good leaving group. The SN2 reaction of Br− and I− to CH3Cl is difficult under seawater conditions (Liu et al., 2015). Thus, CH3Cl concentration remains essentially constant once Br− and I− concentrations reach a certain level.
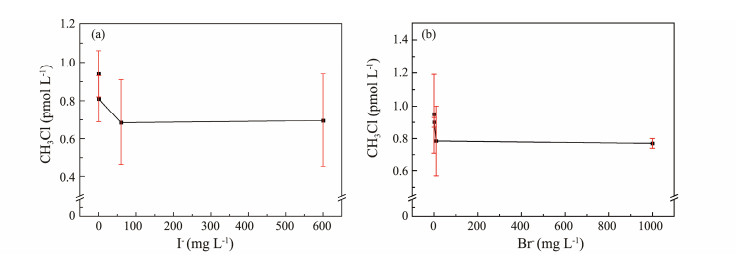
|
Fig. 6 Effect of I− (a) and Br− (b) concentration on CH3Cl photochemical production. |
Fig.7 illustrates the concentrations of CH3I and CH3Cl produced under different pH conditions after irradiation. The maximum yield of CH3I occurred at pH 3.58, accompanied by a significant increase in CH3Cl production. Conversely, near-neutral pH inhibited both photochemical reactions. At pH 9.04 (alkaline), the yield of CH3Cl peaked, but CH3I production decreased significantly.
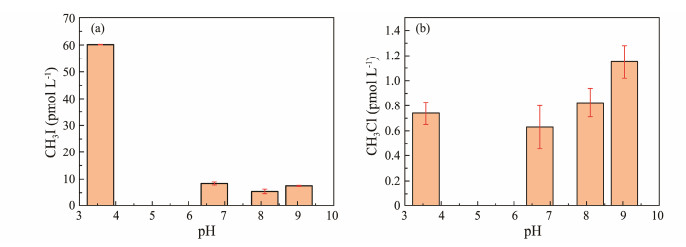
|
Fig. 7 Effect of pH on CH3I (a) and CH3Cl (b) photochemical production. |
These results suggest that the photochemical reaction of guaiacol promoting CH3I and CH3Cl production is enhanced under acidic conditions. Yang et al. (2020) proposed that aryl ion intermediates formed during guaiacol's photochemical reaction can degrade via two pathways: demethylation to CH3Cl and demethoxylation to methanol (CH3OH). As pH increases, the concentration of OH− in the solution rises, favoring CH3OH as the dominant kinetic product (Bi, 2017). This explains why CH3I and CH3Cl production is significantly higher under acidic conditions compared to alkaline conditions. Additionally, the presence of numerous molecular association states of guaiacol, which exhibit stronger photochemical activity under acidic conditions, further promotes CH3I and CH3Cl production (Yang et al., 2020).
In alkaline environments, the photochemistry of CH3I is inhibited while that of CH3Cl is promoted. Due to the poor leaving ability of Cl− compared to I−, CH3I is prone to bimolecular nucleophilic substitution reactions with OH− under alkaline conditions, yielding CH3OH (Liu et al., 2015). However, the replacement of Cl− by OH− is challenging, thus hindering CH3Cl production. Moore (2008) observed a pH-dependent formation of CH3Cl, with significantly enhanced photochemical production at higher pH levels. The highest demethylation efficiency of the precursor occurs at pH > 8, contributing to increased photochemical CH3Cl production (Dallin et al., 2009).
3.1.6 Fe3+ concentrationStudies on the impact of Fe3+ concentration on the photochemical production of methyl halides in the ocean are currently limited. Increasing Fe3+ concentration greatly enhances CH3I production, indicating Fe3+ promotes CH3I production, with higher concentrations exerting stronger effects on its photochemistry. Conversely, lower Fe3+ concentrations significantly increase CH3Cl production, while higher concentrations have minimal impact on CH3Cl photochemical production (Fig.8).
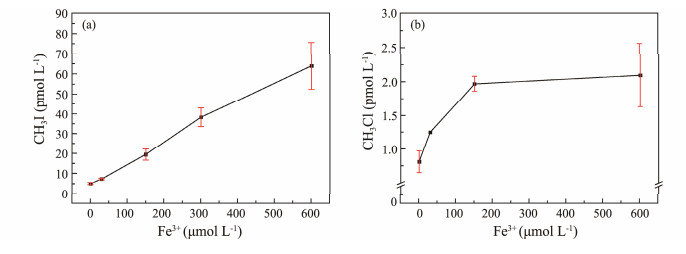
|
Fig. 8 Effect of Fe3+ concentration on CH3I (a) and CH3Cl (b) photochemical production. |
Bai et al. (2023) observed that under xenon lamp irradiation, the addition of Fe3+ significantly promotes methyl halide formation. They proposed a mechanism where [Fe(OH)]2+ (the main form of Fe3+) undergoes photolysis to produce Fe2+ and OH·. OH· oxidizes halides to form intermediate XHO−, generating X· that promotes methyl halide production. Light-induced OH· also oxidizes most dissolved organic matter (DOM) to CH3·, which reacts with X− to produce methyl halides. Furthermore, Fe3+ can complex with alcohols, phenols, and other structures in guaiacol to form complexes, enhancing precursor material activity and subsequently increasing photochemical reaction rates (Porasso et al., 2002; Bi et al., 2017).
Additionally, Fe3+ in solution can influence system pH. Zhao et al. (2004) noted that the photoreaction rate of organic acid-Fe3+ complexes increases under acidic conditions. This suggests Fe3+ affects methyl halide photochemistry by altering system pH. However, Fe3+ precipitates fully at pH 3.2, reducing its availability for photoreactions and limiting its ability to promote photochemical reactions. Excess Fe3+ also leads to the production of polyhalomethanes during photochemical reactions (Bi et al., 2017). At high Fe3+ concentrations, competition between polyhalomethane and CH3Cl production occurs, with CH3Cl production plateauing despite increasing Fe3+ concentration.
3.1.7 Dissolved oxygen (DO)Fig.9 illustrates the effects of dissolved oxygen (DO) on the photochemistry of CH3I and CH3Cl. The CH3I yield in water samples without deoxygenation was 6.1 pmol L−1, whereas in deoxygenated water samples, it increased to 7.7 pmol L−1, a 25% increase. Conversely, CH3Cl production in water samples without deoxygenation was 1.4 pmol L−1, but it was not detected in deoxygenated water samples. These results indicate that dissolved oxygen significantly inhibits CH3I photochemical production while promoting CH3Cl photochemical production.
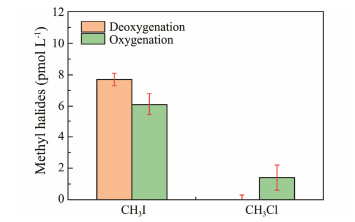
|
Fig. 9 Effect of DO on CH3I and CH3Cl photochemical production. |
Moore (1994) first observed that oxygen inhibits CH3I photochemical production, likely due to oxygen reacting with ·CH3, a major sink for ·CH3. In alkane free radical substitutions, oxygen addition markedly reduces reaction rates as ·CH3 preferentially reacts with oxygen to form CH3OO· radicals, which then react with halogens to produce methyl halides (Yang et al., 2010). Studies have shown that under deoxygenated conditions, the degradation rate of guaiacol decreases by approximately half, with no significant increase in methyl halide production (Lu et al., 2010). Guaiacol photodegradation pathways involve oxygen-independent production of methyl halides and oxygen-dependent production of substances like methanol (CH3OH) (Yang et al., 2020). Thus, dissolved oxygen inhibits CH3I production but promotes CH3Cl production.
Bai et al. (2023) observed decreased CH3Cl production after deoxygenation, highlighting oxygen's role in organic matter mineralization and suggesting that oxygen deficiency stalls CH3Cl photochemistry. Ultraviolet radiation generates transient reactive oxygen species (ROS) in aquatic environments, which degrade humic substances, thereby promoting CH3Cl production (Paul et al., 2012). Additionally, Cl· reacts with ROS to produce reactive chlorine species (RCS), which also influence CH3Cl production.
3.1.8 SalinityFig.10 illustrates the variation in CH3I and CH3Cl production with seawater salinity after irradiation. CH3I yield decreases with increasing salinity, while CH3Cl yield increases. Salinity's most direct indicator is the change in Cl− concentration, which indirectly affects the photochemical production of CH3I and CH3Cl (Chiron et al., 2006). Liu (2020) demonstrated that higher Cl− concentrations significantly promote CH3Cl photochemical production. Similarly, Bi (2017) observed that Cl− enhances CH3Cl production, with a more pronounced effect at higher concentrations. Their findings support that increased salinity promotes CH3Cl photochemical production.
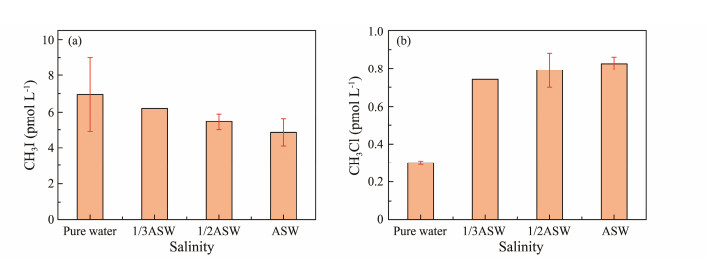
|
Fig. 10 Effect of salinity on CH3I (a) and CH3Cl (b) photochemical production. |
Hu et al. (2016) observed a significant negative correlation between CH3I concentration and seawater salinity. They concluded that higher Cl− concentrations in seawater lead to lower CH3I concentrations, consistent with our experimental results. They proposed that this decrease in CH3I concentration is primarily due to chemical losses. Increased Cl− concentrations compete with methyl groups from precursors via ·Cl radicals, thereby reducing CH3I production. Additionally, iodine, being a good leaving group, undergoes bimolecular nucleophilic substitution (SN2) reactions with Cl− under high salinity conditions, leading to the production of CH3Cl and reducing macroscopic CH3I concentrations.
3.2 Formation of CH3I and CH3Cl in Natural SeawaterFig.11 compares the changes in CH3Cl and CH3I concentrations in natural seawater from different stations under varying conditions. The production of methyl halides in both light and dark conditions was significantly higher than the initial concentration of halomethanes in seawater, indicating their continuous production under these conditions. Light exposure resulted in significantly greater methyl halide production compared to darkness, highlighting light's role in promoting methyl halide production in natural seawater. Studies have shown that methyl halides in the ocean can be produced by microorganisms such as bacteria (Fuse et al., 2003) and cyanobacteria (Hughes et al., 2011). It is speculated that microbial activities may also release methyl halides in darkness, a hypothesis that warrants further investigation.
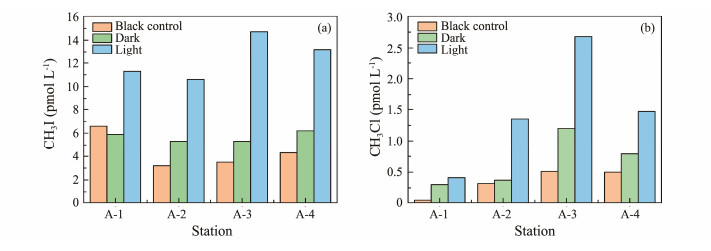
|
Fig. 11 Changes in CH3I (a) and CH3Cl (b) concentration in natural seawater under light and dark conditions. |
The concentration of CH3I was consistently higher than that of CH3Cl at different stations. Daily accumulation rates of CH3I and CH3Cl were reported to be 73% and 41% higher in light conditions than in darkness, respectively (Liu et al., 2020). In terms of photochemical reactions, nucleophilic substitution of CH3Br and CH3I with Cl− generates CH3Cl (Elliot and Rowland, 1993). Chloride-containing halocarbons produced via nucleophilic reactions are predominant in deep-sea areas, whereas CH3Cl in coastal areas mainly stems from bioproduction (Carpenter and Liss, 2000).
CH3I and CH3Cl concentrations were higher at stations A-3 and A-4 compared to A-2 and A-1 (Fig.1). Generally, water temperatures near Qingdao decrease gradually from offshore to coastal areas, mirroring salinity trends (StemmLer et al., 2014). Optimal temperatures and nutrientrich conditions accelerate phytoplankton growth, thereby enhancing CH3I and CH3Cl production. Stations A-3 and A-4, located near suspected sewage outlets, exhibit increased anthropogenic sources of CH3I and CH3Cl. Higher dissolved organic carbon (DOC) content provides ample reactants for photochemical reactions that produce methyl halides in seawater (Yuan et al., 2019). To explore DOC's impact on methyl halide production further, DOC values in natural seawater samples from different stations were measured, and correlation graphs were plotted for CH3I and CH3Cl production (Fig.12). Linear fitting yielded R2 values of 0.895 and 0.958, respectively. The production of CH3I and CH3Cl correlates positively and significantly with DOC content. This supports the hypo-thesis that methyl halide production is influenced by DOC content, consistent with findings by Shi et al. (2014). DOM in seawater under light irradiation can enter an excited state, oxidizing X− to generate X· (Dong et al., 2021). Additionally, DOM acts as a precursor for methyl halides, underscoring the importance of DOC in the photochemical generation of CH3I and CH3Cl in natural seawater.
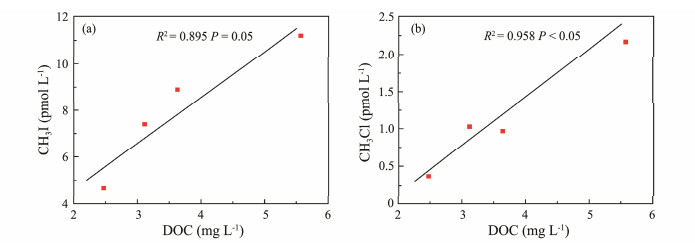
|
Fig. 12 Relationships between concentration of CH3I (a) and CH3Cl (b) and DOC. |
This study investigated the photochemical production of methyl halides in artificial seawater and natural seawater from the Qingdao coastal area. Guaiacol was identified as a crucial precursor for the photochemical formation of methyl halides. The concentration of methyl halides from photochemical processes was enhanced by increased concentrations of guaiacol, X−, and Fe3+, as well as by higher illumination intensity and duration. A notable inhibitory effect on the photochemical production of CH3I was observed at high pH values and in the presence of dissolved oxygen, attributed to reactions between OH− and ·CH3 radicals. The study demonstrated that methyl halide production in natural seawater was significantly higher under light conditions compared to darkness, highlighting light as a critical factor in promoting seawater photochemical reactions. Furthermore, a positive correlation between methyl halide concentrations and dissolved organic carbon (DOC) in natural seawater was observed. This correlation suggests that abundant DOC in seawater provides ample substrate for the photochemical production of methyl halides. These findings underscore the complex interplay of environmental factors influencing methyl halide dynamics in seawater, with implications for understanding their production mechanisms and environmental impact. Further research is warranted to explore these interactions comprehensively, especially in the context of varying environmental conditions and microbial activities in marine ecosystems.
AcknowledgementsThis work was funded by the Natural Science Foundation of Shandong Province, China (No. ZR2021MD034), and the National Natural Science Foundation of China (No. 42276039).
Amon, R. M., and Benner, R., 1996. Bacterial utilization of different size classes of dissolved organic matter. Limnology and Oceanography, 41(1): 41-51. DOI:10.4319/lo.1996.41.1.0041 (  0) 0) |
Archer, S. D., Goldson, L. E., Liddicoa, M. I., Cummings, D. G., and Nightingale, P. D., 2007. Marked seasonality in the concentrations and sea-to-air flux of volatile iodocarbon compounds in the western English Channel. Journal of Geophysical Research, 112(C8): C8009. (  0) 0) |
Bai, X. L., Yang, Q., Yang, G., Hao, B. Q., Zhang, R. Y., Duan, R., et al., 2023. Alkyl halide formation from degradation of carboxylic acids in the presence of Fe(III) and halides under light irradiation. Water Research, 235: 119842. DOI:10.1016/j.watres.2023.119842 (  0) 0) |
Bi, X. W., 2017. Photochemical formation of volatile halogenated hydrocarbons in seawater. Master thesis. Dalian Maritime University (in Chinese with English abstract).
(  0) 0) |
Bidleman, T. F., Andersson, A., Haglund, P., and Tysklind, M., 2020. Will climate change influence production and environmental pathways of halogenated natural products. Environmental Science and Technology, 54(11): 6468-6485. DOI:10.1021/acs.est.9b07709 (  0) 0) |
Brahamsson, K., Granfors, A., Ahnoff, M., Cuevas, C. A., and Saiz-Lopez, A., 2018. Organic bromine compounds produced in sea ice in Antarctic winter. Nature Communications, 9(1): 5291. DOI:10.1038/s41467-018-07062-8 (  0) 0) |
Brigante, M., Minella, M., Mailhot, G., Maurino, V., Minero, C., and Vione., D., 2014. Formation and reactivity of the dichloride radical (Cl2−) in surface waters: A modelling approach. Chemosphere, 95: 464-469. DOI:10.1016/j.chemosphere.2013.09.098 (  0) 0) |
Burns, S. E., Hassett, J. P., and Rossi, M. V., 1996. Binding effects on humic-mediated photoreaction: Intrahumic dechlorination of mirex in water. Environmental Science and Technology, 30(10): 2934-2941. DOI:10.1021/es950906i (  0) 0) |
Carena, L., and Vione, D., 2016. Photochemical reaction of peroxynitrite and carbon dioxide could account for up to 15% of carbonate radicals generation in surface waters. Environmental Chemistry Letters, 14: 183-187. DOI:10.1007/s10311-016-0549-3 (  0) 0) |
Carpenter, L. J., Yokouchi, Y., and Atlas, E. L., 2017. Introduction to special issue on natural halocarbons in the atmosphere. Journal of Atmospheric Chemistry, 74(2): 141-143. DOI:10.1007/s10874-017-9364-x (  0) 0) |
Chen, Y., Liu, S. S., Yang, G. P., and Zhen, H., 2020. Influence factors on photochemical production of methyl iodide in seawater. Journal of Ocean University of China, 19: 1353-1361. DOI:10.1007/s11802-020-4463-8 (  0) 0) |
Chiron, S., Minero, C., and Vione, D., 2006. Photodegradation processes of the antiepileptic drug carbamazepine, relevant to estuarine waters. Environmental Science and Technology, 40(19): 5977-5983. DOI:10.1021/es060502y (  0) 0) |
Comba, P., Kerscher, M., Krause, T., and Schaler, H. F., 2015. Iron-catalysed oxidation and halogenation of organic matter in nature. Environmental Chemistry, 12(4): 381-395. DOI:10.1071/EN14240 (  0) 0) |
Dallin, E., Wan, P., Krogh, E., Gill, C., and Moore, R. M., 2009. New pH-dependent photosubstitution pathways of syringic acid in aqueous solution: Relevance in environmental photochemistry. Journal of Photochemistry and Photobiology A, 207(2-3): 297-305. DOI:10.1016/j.jphotochem.2009.07.023 (  0) 0) |
Dong, Y., Peng, W., and Liu, Y., 2021. Photochemical origin of reactive radicals and halogenated organic substances in natural waters: A review. Journal of Hazardous Materials, 401: 123884. DOI:10.1016/j.jhazmat.2020.123884 (  0) 0) |
Elliott, S., and Rowland, F. S., 1993. Nucleophilic substitution rates and solubilities for methyl halides in seawater. Geophysical Research Letters, 20(11): 1043-1046. DOI:10.1029/93GL01081 (  0) 0) |
Fuse, H., Inoue, H., Murakami, K., Takimura, O., and Yamaoka, Y., 2003. Production of free and organic iodine by Roseovarius spp. FEMS Microbiology Letters, 229(2): 189-194. DOI:10.1016/S0378-1097(03)00839-5 (  0) 0) |
Ge, L., Chen, J. W., Qiao, X. L., Li, L, J., and Cai, X. Y., 2009. Light-source-dependent effects of main water constituents on photodegradation of phenicol antibiotics: Mechanism and kinetics. Environmental Science and Technology, 43(9): 3101-3107. DOI:10.1021/es8031727 (  0) 0) |
Gjessing, E. T., and Källqvist, T., 1991. Algicidal and chemical effect of UV-radiation of water containing humic substances. Water Research, 25(4): 491-494. DOI:10.1016/0043-1354(91)90087-7 (  0) 0) |
Goldstein, S., and Rabani, J., 2007. Mechanism of nitrite formation by nitrate photolysis in aqueous solutions: The role of peroxynitrite, nitrogen dioxide, and hydroxyl radical. Journal of the American Chemical Society, 129(34): 10597-10601. DOI:10.1021/ja073609+ (  0) 0) |
Goodwin, K. D., North, W. J., and Lidstrom, M. E., 1997. Production of bromoform and dibromomethane by giant kelp: Factors affecting release and comparison to anthropogenic bromine sources. Limnology and Oceanography, 42(8): 1725-1734. DOI:10.4319/lo.1997.42.8.1725 (  0) 0) |
Hossaini, R., Chipperfield, M. P., Montzka, S. A., Rap, A., Dhomse, S., and Feng, W., 2015. Efficiency of short-lived halogens at influencing climate through depletion of stratospheric ozone. Nature Geoscience, 8(3): 186-190. DOI:10.1038/ngeo2363 (  0) 0) |
Hu, Q., Xie, Z., Wang, X., Yu, J., and Zhang, Y., 2016. Methyl iodine over oceans from the Arctic Ocean to the maritime Antarctic. Scientific Reports, 6(1): 26007. DOI:10.1038/srep26007 (  0) 0) |
Huang, X. D., and Wang, Z. S., 2002. Effect of the chlorination reaction conditions on the amount of trichloromethane produced. Water Supply and Drainage in China, 6: 14-17. (  0) 0) |
Hughes, C., Franklin, D. J., and Malin, G., 2011. Iodomethane production by two important marine cyanobacteria: Prochlorococcus marinus (CCMP 2389) and Synechococcus sp. (CCMP 2370). Marine Chemistry, 125(1): 19-25. (  0) 0) |
Iglesias-Suarez, F., Badia, A., Fernandez, R. P., Cuevas, C. A., Kinnison, D. E., Tilmes, S., et al., 2020. Natural halogens buffer tropospheric ozone in a changing climate. Nature Climate Change, 10: 147-154. DOI:10.1038/s41558-019-0675-6 (  0) 0) |
Jammoul, A., Dumas, S., D'anna, B., and George, C., 2009. Photoinduced oxidation of sea salt halides by aromatic ketones: A source of halogenated radicals. Atmospheric Chemistry and Physics, 9(13): 4229-4237. DOI:10.5194/acp-9-4229-2009 (  0) 0) |
Keppler, F., Eiden, R., Niedan, V., Pracht, J., and Schöler, H. F., 2000. Halocarbons produced by natural oxidation processes during degradation of organic matter. Nature, 403: 298-301. DOI:10.1038/35002055 (  0) 0) |
Li, C. S., He, Z., Zhang, J., and Yang, G. P., 2018. Study on the distribution and sea-gas flux of volatile halogenated hydrocarbons in the Yangtze Estuary and its adjacent waters in summer. Journal of Environmental Science, 38(2): 761-771. (  0) 0) |
Li, Y., He, Z., Yang, G. P., and Zou, Y., 2021. Spatial distribution and biogeochemical cycling of methyl iodide in the Yellow Sea and the East China Sea during summer. Environmental Pollution, 276(15): 116749. (  0) 0) |
Liu, H., Pu, Y. Y., Tong, T., Zhu, X. M., Sun, B., and Zhang, X. X., 2020. Photochemical generation of methyl chloride from humic aicd: Impacts of precursor concentration, solution pH, solution salinity and ferric ion. International Journal of Environmental Research and Public Health, 17(2): 503-516. DOI:10.3390/ijerph17020503 (  0) 0) |
Liu, Y., Thornton, D., Bianchi, T. S., Arnold, W. A., Shields, M. R., Chen, J., et al., 2015. Dissolved organic matter composition drives the marine production of brominated very short-lived substances. Environmental Science and Technology, 49(6): 3366-3374. DOI:10.1021/es505464k (  0) 0) |
Lu, X. L., Yang, G. P., Song, G. S., and Zhang, L., 2008. Advances in the studies on the marine chemistry of methyl iodine. Marine Science Bulletin, 27(2): 103-110 (in Chinese with English abstract). DOI:10.3969/j.issn.1001-6392.2008.02.016 (  0) 0) |
Lu, X. L., Yang, G. P., Song, G. S., and Zhang, L., 2010. Distributions and fluxes of methyl chloride and methyl bromide in the East China Sea and the Southern Yellow Sea in autumn. Marine Chemistry, 118(1): 75-84. (  0) 0) |
Martin, D. F., 1972. Marine Chemistry, vol. 1: Analytical Methods. 2nd edition. Marcel Dekker, Inc, New York, 143-158.
(  0) 0) |
Molina, M. J., and Rowland, F. S., 1974. Stratospheric sink for chlorofluoromethanes: Chlorine atom-catalysed destruction of ozone. Nature, 249(5460): 810-812. DOI:10.1038/249810a0 (  0) 0) |
Moore, R. M., 2008. A photochemical source of methyl chloride in saline waters. Environmental Science & Technology, 42(6): 1933-1937. (  0) 0) |
Moore, R. M., and Zafiriou, O. C., 1994. Photochemical production of methyl iodide in seawater. Journal of Geophysical Research: Atmospheres, 99(D8): 16415-16420. DOI:10.1029/94JD00786 (  0) 0) |
Ooki, A., Tsuda, A., Kameyama, S., Takeda, S., Itoh, S., Suga, T., et al., 2010. Methylhalides in surface seawater and marine boundary layer of the Northwest Pacific. Journal of Geophysical Research, 115: C10013. (  0) 0) |
Paul, A., Dziallas, C., Zwirnmann, E., Gjessing, E. T., and Grossart, H. P., 2012. UV irradiation of natural organic matter (NOM): Impact on organic carbon and bacteria. Aquatic Sciences, 74(3): 443-454. DOI:10.1007/s00027-011-0239-y (  0) 0) |
Porasso, R. D., Benegas, J. C., Van Den, H., and Paoletti, S., 2002. Analysis of trace metal humic acid interactions using counterion condensation theory. Environmental Science and Technology, 236: 3815-3821. (  0) 0) |
Reckhow, D. A., Singer, P. C., and Malcolm, R. L., 1990. Chlorination of humic materials-by-product formation and chemical interpretations. Environmental Science and Technology, 24: 1655-1664. DOI:10.1021/es00081a005 (  0) 0) |
Richter, U., and Wallace, D. W. R., 2004. Production of methyl iodide in the tropical Atlantic Ocean. Geophysical Research Letters, 312(23): 203-218. (  0) 0) |
Shi, Q., Marandino, C., Petrick, G., Quack, B., and Wallace, D., 2014a. A time series of incubation experiments to examine the production and loss of CH3I in surface seawater. Journal of Geophysical Research: Oceans, 119: 8242-8254. DOI:10.1002/2014JC010223 (  0) 0) |
Shi, Q., Petrick, G., Quack, B., Marandino, C., and Wallace, D., 2014b. Seasonal variability of methyl iodide in the Kiel Fjord. Journal of Geophysical Research: Oceans, 119(3): 1609-1620. DOI:10.1002/2013JC009328 (  0) 0) |
Stemmler, I., Hense, I., Quack, B., and Maier-Reimer, E., 2014. Methyl iodide production in the open ocean. Biogeosciences, 11: 4459-4476. DOI:10.5194/bg-11-4459-2014 (  0) 0) |
Tai, C., Li, Y. B., Yin, Y. G., Cai, Y., and Jiang, G. B., 2012. Radical photochemical behavior of soluble organic matter in natural water bodies. Chemical Advances, 24(7): 1388-1397. (  0) 0) |
WMO (World Meteorological Organization), 2018. Scientific Assessment of Ozone Depletion: 2018, Global Ozone Research and Monitoring Project-Report No. 58, Geneva, Switzerland, 588pp.
(  0) 0) |
Yang, G. P., Lu, X. L., Song, G. S., and Wang, X. M., 2010. Purge-and-trap gas chromatography method for analysis of methyl chloride and methyl bromide in seawater. Chinese Journal of Analytical Chemistry, 38(5): 719-722. DOI:10.1016/S1872-2040(09)60046-3 (  0) 0) |
Yang, G. P., Yin, S. X., Lu, X. L., and Song, G. S., 2007. Determination of volatile halocardic hydrocarbons in seawater by purge-trap gas chromatography. Journal of Ocean University of China (Natural Science Edition), 37(2): 299-304. (  0) 0) |
Yuan, D., He, Z., and Yang, G. P., 2019. Spatiotemporal distributions of halocarbons in the marine boundary air and surface seawater of the Changjiang Estuary and its adjacent East China Sea. Marine Pollution Bulletin, 140: 227-240. DOI:10.1016/j.marpolbul.2019.01.040 (  0) 0) |
Zafirious, O. C., Blough, N. V., Micinski, E., Dister, B., Kieber, D., and Moffett, J., 1990. Molecular probe systems for reactive transients in natural waters. Marine Chemistry, 30: 45-70. DOI:10.1016/0304-4203(90)90061-G (  0) 0) |
Zepp, R. G., Schlotzhauer, P. F., and Sink, R. M., 1985. Photosensitized transformations involving electronic energy transfer in natural waters: Role of humic substances. Environmental Science and Technology, 19(1): 74-81. DOI:10.1021/es00131a008 (  0) 0) |
Zhang, W., Jiao, Y., Zhu, R., and Rhew, R. C., 2020. Methyl chloride and methyl bromide production and consumption in coastal Antarctic tundra soils subject to sea animal activities. Environmental Science and Technology, 54: 13354-13363. DOI:10.1021/acs.est.0c04257 (  0) 0) |
Zhao, R. D., 2004. Photochemical study of iron in seawater. Master thesis. Ocean University of China (in Chinese with English abstract).
(  0) 0) |
Zhu, Z. L., Wang, J., Ge, Y. X., Ma, H. M., and Zhao, J. F., 2007. Effect of Fe3+ and Br− on the generation of volatile halocarbons during the disinfection of drinking water chlorine. Environmental Science, 28(6): 1264-1267. DOI:10.3321/j.issn:0250-3301.2007.06.017 (  0) 0) |
Ziska, F., Quack, B., Abrahamsson, K., Archer, S. D., Atlas, E., Bell, T., et al., 2013. Global sea-to-air flux climatology for bromoform, dibromomethane and methyl iodide. Atmospheric Chemistry and Physics, 13(17): 8915-8934. DOI:10.5194/acp-13-8915-2013 (  0) 0) |
 2024, Vol. 23
2024, Vol. 23


