2) College of Marine Life Science, Ocean University of China, Qingdao 266003, China;
3) Department of Life Sciences, Natural History Museum, London SW7 5BD, UK
Gemcitabine is a chemotherapeutic drug that kills cells during their division process (Zhang et al., 2017). This drug is used to kill cancer cells that show rapid division rates. However, it also target several essential cells that can divide, e.g., the cells in the skin, scalp, and lining of stomach, testes, and bone marrow (Rachel, 2009). Therefore, gemcitabine has numerous adverse side effects, including suppression of bone marrow function; loss of white blood cells, platelets, and red blood cells; and harming the sperm (Siddall et al., 2017; Zhang et al., 2017). Nevertheless, although gemcitabine has been used clinically via intravenous delivery or direct perfusion, there are only a few reports regarding its cytotoxicity (Krown, 2011; Dubey et al., 2016; Birhanu et al., 2017; Dyawanapelly et al., 2017; Pishvaian and Brody, 2017).
Ciliated protozoa have been widely used as test organisms in bioassays of chemical drugs as they have several advantages such as ease of culture, short generation time, and simple developmental stages (Gray and Ventilla, 1973; Dive and Leclerc, 1975; Rogerson, 1983; Moreno-Garrido and Canavate, 2001; Xu et al., 2004). Furthermore, as they are separated from their environment only by a cell membrane, they respond rapidly to changes in their surroundings and are hence used as bioassay tools for assessing cytotoxicity (Dive and Leclerc, 1975; Bearden et al., 1999; Girling et al., 2000; Herllung-Larsen et al., 2000; Seward et al., 2001; Fuma et al., 2003; Xu et al., 2004).
In this study, the model ciliated protozoan Euplotes vannus was used as a test cell to determine the lethal and threshold concentrations of gemcitabine. The aims of this study were to provide experimental evidence regarding the cytotoxicity of gemcitabine and to evaluate the potential of E. vannus as a model organism in bioassays of chemical drugs.
2 Materials and Methods 2.1 Cultivation of E. vannus and Preparation of Gemcitabine SolutionsThe marine ciliate E. vannus was obtained from the Laboratory of Protozoology, Ocean University of China, Qingdao, China, and was identified based on a combination of morphological and molecular data (Chen and Song, 2002; Hong et al., 2017). Clonal and mass cultures of E. vannus were grown at 25℃ in sterilized artificial marine water comprising 28 g of NaCl, 0.8 g of KCl, 5 g of MgCl2·6H2O, and 1.2 g of CaCl2 per 1000 mL of distilled water (salinity 28).
Ammonia stock solutions (salinity 28, temperature 25℃) were prepared by adding gemcitabine to artificial marine water. Test solutions with different concentrations of gem- citabine were prepared by mixing the stock solution with artificial marine water in appropriate proportions.
2.2 Acute Toxicity TestFor the acute toxicity test, 1 mL of gemcitabine solution was added to each well of a 48-well cell culture plate. To each well, ten E. vannus cells were added. The acute toxicity test was carried out in the dark at 25℃. The 30-min experiments were designed as five treatments (including one control), with each treatment as three independent lines (replicates). The concentrations of gemcitabine for the five treatments were 0, 6, 12, 24, and 48 mg mL-1, respectively. For each line, the number of dead cells, including those unable to swim or creep on the bottom of the well, was recorded every 6 min in vivo under a stereomicroscope.
The median lethal concentrations (LC50) for 6, 12, 18, 24, and 30 min were calculated using the probit-regression routine of the program IBM SPSS Statistics version 22.0, International Business Corp, USA. The toxicity curve equation was determined using the program SigmaPlot 12.5, Systat Software Inc, USA.
2.3 Chronic Toxicity TestThe inhibition effect of gemcitabine on the growth of E. vannus was also investigated in 48-well culture plates at 25℃. To each well, 1 mL of test solution and ten E. vannus cells were added. The concentration of bacteria in each well was maintained at 108 cells mL-1 by adding Vibrio sp. The tests were performed using five different concentrations of gemcitabine, 0, 0.7, 3.5, 7, and 14 mg mL-1, with three replicates of each concentration. The ciliate cells in each well were enumerated in vivo every 12 h under a stereomicroscope for up to 120 h.
The increase of the number of ciliate cells over the total experimental period was evaluated to ascertain whether it is fitted to the following logistic model:
| $ N_{t}=N_{\max } /\left[1+\mathrm{e}^{(a-r t)}\right], $ |
where Nt, the density of ciliate cells at time t; Nmax, the estimated maximum density; r, the growth rate; a, the coefficient constant of initial density; and T50, the time to 50% Nmax.
All parameters were estimated using the program SigmaPlot. Fitness tests were conducted to determine whether the growth curve fits to the logistic model at the 0.05 significance level (Zhang et al., 2012).
The median inhibition concentration (IC50) was computed using regression analysis of the relationship between the concentration of gemcitabine and the treatment/control ratios (r%) of growth rates using SigmaPlot 12.5 (Zhang et al., 2012).
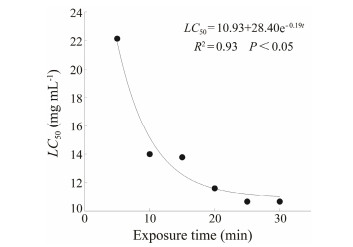
3 Results 3.1 Acute Toxicity and Toxicity CurvesBased on the probit-regression analysis of log-dose and mortality data at exposure times of 6, 12, 16, 24, and 30 min, the LC50 values were calculated and are shown in Fig. 1. The LC50 values decreased with increasing duration of exposure. The regression analysis demonstrated that the toxicity curve significantly fitted to the following equation:
| $ L{C_{50}} = 10.93{\rm{ }}28.4{{\rm{e}}^{{\rm{ - 0}}{\rm{.19}}t}}, $ |
|
Fig. 1 Toxicity curve of gemcitabine for Euplotes vannus up to 30 min of exposure. |
where R2 = 0.93; P < 0.05, t = exposure time (Fig. 1).
Using this model equation, it is possible to predict LC50 values of gemcitabine with different concentrations at a wide range of exposure times.
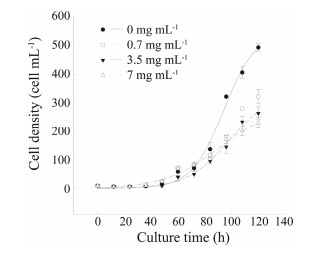
3.2 Inhibition Effect of Gemcitabine on Cell GrowthThe inhibition effect of gemcitabine on the population growth of E. vannus for up to 120 h is depicted in Fig. 2. Regression analyses revealed that the growth curves of the four treatments (except 14 mg mL-1) significantly fitted to the logistical model equation (R2 > 0.95; P < 0.05). The data pertaining to the treatment with 14 mg mL-1 of gemcitabine were omitted due to the absence of growth.
|
Fig. 2 Growth curves showing the inhibition effect of gemcitabine on individual growth of Euplotes vannus after an exposure time of 30 min. |
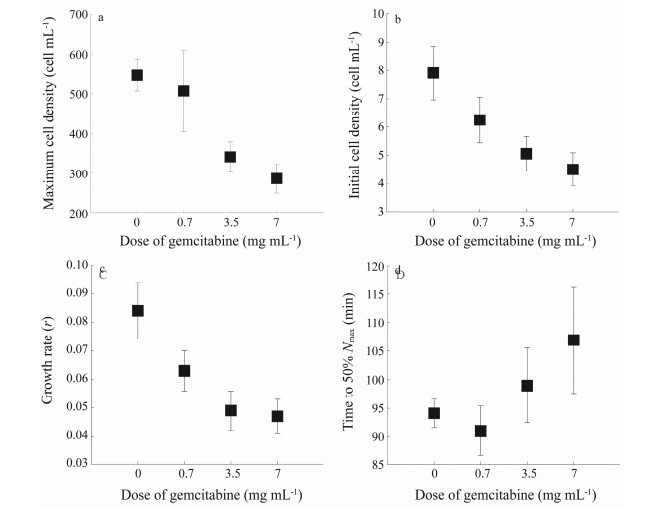
Based on the model equation, the estimated maximum density, the initial density, the growth rate, and the time to reach 50% of maximum density of the ciliate cells were obtained (Fig. 3). The maximum density values of gemcitabine with concentrations of 3.5 and 7.0 mg mL-1 were significantly lower than those of the control (P < 0.05) (Fig. 3a), whereas in terms of growth rate, the values at all gemcitabine treatments were significantly lower than those of the control (P < 0.05) (Figs. 3b and c).
|
Fig. 3 Effects of gemcitabine on the maximum cell density (a), initial cell density (b), growth rate (c), and the time to 50% maximum cell density (d) of Eulpotes vannus. |
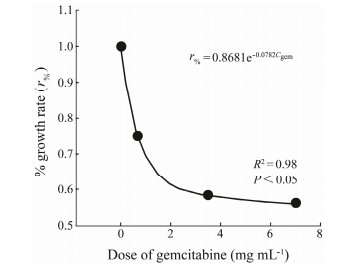
The relationship between gemcitabine concentration and the treatment/control ratios (r%) of growth rates is depicted in Fig. 4. The regression analysis showed that the dose-r% significantly fitted to the following equation:
| $ r_{\%}=0.8681 \mathrm{e}^{-0.0782 C_{\mathrm{gem}}}, $ |
|
Fig. 4 Relationships between dose of gemcitabine and growth rates of Eulpotes vannus. |
where r% is the growth rate with inhibition of gemcitabine, and Cgem is the concentration of gemcitabine; R2 = 0.99 and P < 0.05.
Based on this equation, the IC50 value was obtained, i.e., when r% = 50%, Cgem = 7.05 mg mL-1.
4 DiscussionE. vannus is a well-known model ciliate that has traditionally been used as a bioassay organism in ecotoxicological studies (Coppellotti, 1998; Fernandez-Leborans, 2000; Xu et al., 2004; Zhou et al., 2011; Li et al., 2014; Hong et al., 2015). In the present study, E. vannus exhibited a measurable dose-response to gemcitabine and hence may be applied as a useful model organism in bioassays of cytotoxicity of this chemotherapeutic drug.
Owing to its hydrophilic properties, gemcitabine is transported into cells via molecular transporters for nucleosides and is pharmacologically active as gemcitabine triphosphate after the attachment of a phosphate ion to the gemcitabine molecule (Alvarellos et al., 2014). The thrice- phosphorylated gemcitabine molecule can masquerade as cytidine and thus be incorporated into DNA. This allows a native (or normal) nucleoside base to be added next to it and leads to 'masked chain termination' by creating an irreparable error that leads to inhibition of further DNA synthesis, thereby resulting in cell death (Alvarellos et al., 2014). Furthermore, the twice-phosphorylated gemcitabine can inhibit the enzyme ribonucleotide reductase and thus can drive the cell to uptake more gemcitabine from outside the cell (Cerqueira et al., 2007; Alvarellos et al., 2014).
Gemcitabine is clinically used to treat bladder cancer by irrigation of bladder with a dose of 20 mg mL-1 for 30 min (Alvarellos et al., 2014). In the present study, the 30-min LC50 value was 10.66 mg mL-1 (temperature, 25℃), and the LC50 values decreased with increasing exposure time following the toxicity curve equation
| $ L C_{50}=10.93+28.4 \mathrm{e}^{-0.19 t}, $ |
where R2 = 0.93, P < 0.05, and t is exposure time. Therefore, from this equation, we predict that the clinical dose of gemcitabine (20 mg mL-1) is higher than the 30-min LC50 value and almost the same as the 6-min LC50 value (19.88 mg mL-1) for E. vannus cells. This implies that 20 mg mL-1 gemcitabine kills 50% of E. vannus cells within 6 min. Based on these results, the IC50 value for inhibiting the growth of E. vannus cells was 7.05 mg mL-1. This finding suggests that the model ciliate is sensitive to gemcitabine.
It should be noted that to test the cytotoxicity of gemcitabine to the human bladder, mammalian (preferably human) bladder cell lines would be expected to better reflect the cytotoxic response. However, till date, there have been only a few reports on the successful cultivation of bladder cells in vitro (Burrows et al., 2017). In comparison, the ciliate E. vannus has several advantages in bioassays. For example, it can be maintained more easily and at less cost than human body cell lines. Furthermore, the preparation of test solutions for cytotoxicity tests is simpler as E. vannus lives in water rather than in a complex physiological solution (Dayeh et al., 2005). On the other hand, E. vannus is a free-living marine ciliated protozoan and exhibits a higher degree of complexity of cell structure and function than mammalian tissue cells (Zhang et al., 2015). Additional tests are therefore needed to determine whether E. vannus cells have the same sensitivity to gemcitabine as mammalian cells in general, and bladder cells in particular. Nevertheless, the present findings suggest that E. vannus is a useful test organism in bioassays of the cytotoxicity of gemcitabine.
5 ConclusionsThe model ciliate species E. vannus exhibited a sensitive dose-dependent response to gemcitabine and can be used as a robust test organism in toxicology bioassays for short duration of exposure. The LC50 values decreased with increasing exposure time and well fitted to the toxicity curve equation. These results show that the clinical dose of gemcitabine (20 mg mL-1) is higher than the 30- min LC50 value and is almost the same as the 6-min LC50 value for E. vannus cells.
AcknowledgementsThis work was supported by the National Natural Science Foundation of China (Nos. 31672308 and 40206021).
Alvarellos, M. L., Lamba, J., Sangkuhl, K., Thorn, C. F., Wang, L., Klein, D. J., Altman, R. B. and Klein, T. E., 2014. PharmGKB summary: Gemcitabine pathway. Pharmacogenetics and Genomics, 24: 564-574. DOI:10.1097/FPC.0000000000000086 (  0) 0) |
Bearden, A. P., Sinks, G. D. and Schultz, T. W., 1999. Acclimation to sublethal exposures to a model nonpolar narcotic: Population growth kinetics and membrane lipid alterations in Tetrahymena pyriformis. Aquatic Toxicology, 46: 11-21. DOI:10.1016/S0166-445X(98)00110-6 (  0) 0) |
Birhanu, G., Javar, H. A., Seyedjafari, E. and Zandi-Karimi, A., 2017. Nanotechnology for delivery of gemcitabine to treat pancreatic cancer. Biomedicine and Pharmacotherapy, 88: 635-643. DOI:10.1016/j.biopha.2017.01.071 (  0) 0) |
Burrows, M. T., Burns, J. E. and Suzuki, Y., 2017. Studies on the growth of cells: The cultivation of bladder and prostatic tumors outside the body. The Journal of Urology, 197: s2-s14. (  0) 0) |
Cerqueira, N. M., Fernandes, P. A. and Ramos, M. J., 2007. Understanding ribonucleotide reductase inactivation by gemcitabine. Chemistry: A European Journal, 13: 8507-8515. DOI:10.1002/chem.200700260 (  0) 0) |
Chen, Z. and Song, W., 2002. Phylogenetic postions of Aspidisca steini and Euplotes vannus within the order Euplotida (Hypotrichia: Ciliophora) inferred from complete small subunit ribosomal RNA gene sequences. Acta Protozoologica, 41: 1-9. (  0) 0) |
Coppellotti, O., 1998. Sensitivity to copper in a ciliate as a possible component of biological monitoring in the Lagoon of Venice. Archives of Environmental Contamination and Toxicology, 35: 417-425. DOI:10.1007/s002449900397 (  0) 0) |
Dayeh, V. R., Lynn, D. H. and Bols, N. C., 2005. Cytotoxicity of metals common in mining effluent to rainbow trout cell lines and to the ciliated protozoan Tetrahymena thermophila. Toxicology in Vitro, 19: 399-410. DOI:10.1016/j.tiv.2004.12.001 (  0) 0) |
Dive, D. and Leclerc, H., 1975. Standardized test method using protozoa for measuring water pollutant toxicity. Progress in Water Technology, 7: 67-72. (  0) 0) |
Dubey, R. D., Saneja, A., Gupta, P. K. and Gupta, P. N., 2016. Recent advances in drug delivery strategies for improved therapeutic efficacy of gemcitabine. European Journal of Pharmaceutical Sciences, 93: 147-162. DOI:10.1016/j.ejps.2016.08.021 (  0) 0) |
Dyawanapelly, S., Kumar, A. and Chourasia, M. K., 2017. Lessons learned from gemcitabine: Impact of therapeutic carrier systems and gemcitabine's drug conjugates on cancer therapy. Critical Reviews in Therapeutic Drug Carrier Systems, 34: 63-96. DOI:10.1615/CritRevTherDrugCarrierSyst.2017017912 (  0) 0) |
Fernandez-Leborans, A., 2000. Protozoan species in three epibenthic areas of the Cantabrian Sea: Relations with environmental factors. Journal of the Marine Biological Association of the United Kingdom, 80: 407-418. DOI:10.1017/S0025315400002101 (  0) 0) |
Fuma, S., Ishii, N., Takeda, H., Miyamoto, K., Yanagisawa, K., Ichimasa, Y., Saito, M., Kawabata, Z. and Polikarpov, G., 2003. Ecological effects of various toxic agents on the aquatic microcosm in comparison with acute ionizing radiation. Journal of Environmental Radioactivity, 67: 1-14. DOI:10.1016/S0265-931X(02)00143-1 (  0) 0) |
Girling, A. E., Pascoe, D., Janssen, C. R., Peither, A., Wenzel, A., Schafer, H., Neumeier, B., Mitchell, G. C., Taylor, E. J., Maund, S. J., Lay, J. P., Juttner, I., Crossland, N. O., Stephenson, R. R. and Persoone, G., 2000. Development of methods for evaluating toxicity to freshwater ecosystems. Ecotoxicology and Environemental Safety, 45: 148-176. DOI:10.1006/eesa.1999.1847 (  0) 0) |
Gray, J. S. and Ventilla, R. J., 1973. Growth rates of sediment living marine protozoa as a toxicity indicator for heavy metals. AMBIO, 2: 118-121. (  0) 0) |
Herllung-Larsen, P., Assaad, F., Pankratova, S. B., Saietz, L. and Skovgaard, L. T., 2000. Effects of pluronic F-68 on Tetrahymena cells: Protection against chemical and physical stress and prolongation of survival under toxic conditions. Biotechnology, 76: 185-195. (  0) 0) |
Hong, Y., Tan, Y., Meng, Y., Yang, H., Zhang, Y., Warren, A., Li, J. and Lin, X., 2017. Evaluation of biomarkers for ecotoxicity assessment by dose-response dynamic models: Effects of nitrofurazone on antioxidant enzymes in the model ciliated protozoan Euplotes vannus. Ecotoxicology and Environmental Safety, 144: 552-559. DOI:10.1016/j.ecoenv.2017.06.069 (  0) 0) |
Hong, Y., Lin, X., Cui, X., Zhou, L., Al-Rasheid, K. A. S. and Li, J., 2015. Comparative evaluatation of genotoxicity induced by nitrofurazone in two ciliated protozoa by detecting DNA strand breaks and DNA-protein crosslinks. Ecological Indicators, 54: 153-160. DOI:10.1016/j.ecolind.2015.02.030 (  0) 0) |
Krown, S. E., 2011. Treatment strategies for Kaposi sarcoma in sub-Saharan Africa: Challenges and opportunities. Current Opinion in Oncology, 23: 463-468. DOI:10.1097/CCO.0b013e328349428d (  0) 0) |
Li, J., Zhou, L., Lin, X., Yi, Z. and Al-Rasheid, K. A. S., 2014. Characterizing dose-reponse of catalase to nitrofurazone exposure in model ciliated protozoan Euplotes vannus for ecotoxicity assessment: Enzyme activity and mRNA expression. Ecotoxicology and Environemental Safety, 100: 294-302. DOI:10.1016/j.ecoenv.2013.08.021 (  0) 0) |
Moreno-Garrido, I. and Canavate, J. P., 2001. Assessing chemical compounds for controlling predator ciliates in outdoor mass cultures of the green algae Dunaliella salina. Aquacultural Engineer, 24: 107-114. DOI:10.1016/S0144-8609(00)00067-4 (  0) 0) |
Pishvaian, M. J. and Brody, J. R., 2017. Therapeutic implications of molecular subtyping for pancreatic cancer. Oncology (Williston Park N.Y.), 31: 159-166, 168. (  0) 0) |
Rachel, A., 2009. Cancer Chemotherapy. Wiley-Blackwell, Oxford, 369pp.
(  0) 0) |
Rogerson, A., 1983. Determination and interpretation of hydrocarbon toxicity to ciliate protozoa. Aquatic Toxicology, 3: 215-228. DOI:10.1016/0166-445X(83)90042-5 (  0) 0) |
Seward, J. R., Sinks, G. D. and Schultz, T. W., 2001. Reproducibility of toxicity across mode of toxic action in the Tetrahymena population growth impairment assay. Aquatic Toxicology, 53: 33-47. DOI:10.1016/S0166-445X(00)00158-2 (  0) 0) |
Siddall, E., Khatri, M. and Radhakrishnan, J., 2017. Capillary leak syndrome: Etiologies, pathophysiology, and management. Kidney International, 92(1): 37-46. DOI:10.1016/j.kint.2016.11.029 (  0) 0) |
Xu, H., Song, W. and Warren, A., 2004. An investigation of the tolerance to ammonia of the marine ciliate Euplotes vannus (Protozoa, Ciliophora). Hydrobiologia, 519: 189-195. DOI:10.1023/B:HYDR.0000026505.91684.ab (  0) 0) |
Zhang, W., Xu, F., Han, J., Sun, Q. and Yang, K., 2015. Comparative cytotoxicity and accumulation of Roxarsone and its photodegradates in freshwater protozoan Tetrahymena thermophila. Journal of Hazardous Materials, 286: 171-178. DOI:10.1016/j.jhazmat.2015.01.001 (  0) 0) |
Zhang, W., Xu, H., Jiang, J., Zhu, M. and Al-Reshaid, K. A. S., 2012. Colonization dynamics in trophic-functional structure of periphytic protest communities in coastal waters. Marine Biology, 159: 735-748. DOI:10.1007/s00227-011-1850-0 (  0) 0) |
Zhang, X. W., Ma, Y. X., Sun, Y., Cao, Y. B., Li, Q. and Xu, C. A., 2017. Gemcitabine in combination with a second cytotoxic agent in the first-line treatment of locally advanced or metastatic pancreatic cancer: A systematic review and meta-analysis. Targeted Oncology, 12(3): 309-321. DOI:10.1007/s11523-017-0486-5 (  0) 0) |
Zhou, L., Li, J., Lin, X. and Al-Rasheid, K. A. S., 2011. Use of RAPD to detect DNA damage induced by nitrofurazone in marine ciliate, Euplotes vannus (Protozoa, Ciliophora). Aquatic Toxicology, 103: 225-232. DOI:10.1016/j.aquatox.2011.03.002 (  0) 0) |
 2019, Vol. 18
2019, Vol. 18


