肝纤维化是指肝脏内细胞外基质过度沉淀的病理过程[1]。氧化应激是肝纤维化的重要发病机制[2]。活化的肝星状细胞(hepatic stellate cell,HSC)被认为是产生细胞外基质的主要来源,在肝纤维化的发生发展中起着关键作用[3]。大麻素Ⅱ型受体(CB2)是机体内源性大麻素系统(endocannabinoid system,ECS)的一种G蛋白偶联受体,可表达于肝纤维化的肝组织中[4]。ECS在炎症反应、氧化应激损伤中发挥重要的调节作用,ESC失调和肝炎、酒精性肝病等许多肝病有关[5]。CB2受体激动剂可防止肺纤维化及减少全身成纤维细胞增殖和自身抗体发展[6]。大麻素CB2受体高表达于肝纤维化的细胞,有抗肝纤维化作用,而无成瘾性[7, 8]。因此,大麻素CB2受体激动剂可能成为抗肝纤维化药物研究的新亮点。研究证实,核转录相关因子(nuclear factor erythroid-2 related factor 2,Nrf2)是调节抗氧化应激反应的重要转录因子,在氧化应激等情况下被激活,进入细胞核并启动多种抗氧化酶及Ⅱ相酶基因表达,在细胞抵御氧化应激反应机制中起着重要作用[9-12]。本研究通过体外培养HSC-T6细胞,观察CB2受体激动剂AM1241对氧化应激作用下HSC-T6细胞的影响及其机制,结果报告如下。
1 材料与方法 1.1 主要试剂依氏低糖培养基(Dulbecco′s modification of Eagle′s medium,DMEM)、胎牛血清(美国HyClone公司),Matrigel胶、聚偏二氟乙烯膜(polyvinylidene fluoride membrane,PVND)、默克密理博发光液(美国Millipore公司),AM1241(美国Cayman公司)、葡萄糖氧化酶(glucose oxidase,GO)(美国Sigma公司),α-平滑肌肌动蛋白(alpha-smooth muscle actin,α-SMA)单克隆抗体、Nrf2单克隆抗体(美国abcam公司),I型胶原(type Col collagen I,Col I)酶联免疫试剂盒(美国CUSABIO公司),超氧化物歧化酶(superoxide dismutase,SOD)、丙二醛检测试剂盒(南京建成公司),HSC-T6细胞(上海中科院,货号:BRL 3A)。
1.2 分组与处理HSC-T6细胞在含10%胎牛血清的DMEM低糖培养液中,置于37 ℃、5%CO2培养箱中饱和湿度下培养,待细胞生长至对数期时进行实验。实验设对照组、氧化应激组、低、高剂量AM1241干预组,每组设3个复孔;对照组常规培养;氧化应激组用含100 mU/L GO完全培养液培养2 h,制备细胞氧化应激模型;低、高剂量AM1241干预组分别加入20、80 μmol/L AM1241作用3 h后,再分别加入100 mU/L GO作用2 h,收集细胞备测。
1.3 指标与方法 1.3.1 HSC-T6细胞中α-SMA表达检测采用细胞免疫化学染色法,将HSC-T6细胞以每孔4×104个/mL接种于预先放有载玻片的6孔培养板中,完全培养液培养24 h,取出载玻片,4%多聚甲醛中固定15 min,0.1%Triton x-100破膜5 min,3%双氧水作用5 min,羊血清封闭30 min,每步磷酸盐缓冲液(phosphate buffer saline,PBS)清洗 3次,每次5 min。加入兔抗鼠一抗α-SMA单克隆抗体(1:100)4 ℃孵育12 h后,PBS清洗 3次,每次5 min,用生物素标记的羊抗兔二抗37 ℃孵育30 min,二氨基联苯胺显色液显色(避光,镜下观察至棕色) 约 3~10 min,PBS洗 2次,每次 1 min,苏木素复染 0.5~1 min,自来水洗 30 min,98%酒精返蓝,中性树胶封片。用PBS代替一抗体作为阴性对照,阳性细胞为胞质染成棕黄色;200和400倍镜下观察。
1.3.2 HSC-T6细胞培养液上清中Col I含量检测采用酶联免疫法,收集各组细胞培养液上清2 000 r/min离心15 min,取上清用样本稀释液进行1:100倍稀释后,每孔分别加样100 μL,37 ℃温育30 min,弃去液体,甩干,不洗涤;加生物素标记抗体工作液,37 ℃温育1 h,弃去液体,洗涤3次,拍干;加辣根过氧化物酶标记亲和素工作液,37 ℃温育1 h,弃去液体,洗涤5次,拍干;加入显色剂37 ℃避光显色15 min。加终止液50 min,以空白管调零,5 min内450 nm波长测量各孔吸光度(A)值。
1.3.3 HSC-T6细胞中丙二醛含量、SOD活力检测收集HSC-T6细胞,弃去细胞培养上清,将细胞裂解后,取细胞裂解液,设空白管(无水乙醇)、标准管(10 nmol/mL标准品),测定管(测试样品),每管加100 μL样品,按试剂盒说明书操作,酶标仪测定吸光度(A)值,再分别根据公式计算丙二醛含量和SOD活力;每组设6个复孔。
1.3.4 HSC-T6细胞总蛋白和胞核中Nrf2蛋白表达检测采用Western blot法,收集细胞,分别提取细胞总蛋白和核蛋白。(1)总蛋白提取:分别加入5倍细胞含有蛋白酶抑制剂的细胞裂解液,将细胞吹悬,冰上裂解15~20 min,用1 mL注射器对裂解液反复抽吸20次,再继续冰上裂解15~20 min,4 ℃离心,12 000 r/min 20 min;将上清移入1.5 mL 微量离心管中,加入1/2体积上样缓冲液,沸水中煮5 min;4 ℃离心,12 000 r/min 5 min,得细胞总蛋白,-20 ℃冰箱中保存备用。(2)核蛋白提取:每20 μL细胞沉淀加入200 μL含苯甲基磺酰氟(phenylmethylsulfonyl fluoride,PMSF)的细胞浆蛋白抽提试剂A,剧烈震荡5 s,把细胞沉淀完全悬浮并分散开,冰浴10 min;加入细胞浆蛋白抽提试剂B 10 μL,剧烈震荡5 s,冰浴1 min。再剧烈震荡5 s,4 ℃离心,12 000 r/min 5 min;沉淀加入50 μL含PMSF的细胞核蛋白抽提试剂,剧烈震荡30 s,把细胞沉淀完全悬浮并分散开,然后放回冰中,每隔1 min再剧烈震荡30 s,共30 min;4 ℃离心,12 000 r/min 10 min,立即吸取上清至一预冷的微量离心管中,即细胞核蛋白,-20 ℃冰箱中保存备用。(3)聚氰基丙烯酸正丁酯(bicinchoninic acid,BCA)蛋白定量法计算各组蛋白浓度:每组取20 μg蛋白进行聚丙烯酰胺凝胶电泳(polyacrylamide gelelectrophoresis,PAGE)湿转于PVDF膜上,5%牛血清白蛋白室温封闭2 h,兔抗鼠一抗Nrf2单克隆抗体(1:2 000),4 ℃孵育12 h,辣根过氧化物酶标记羊抗兔二抗(1:20 000)室温孵育1 h,Tris-HCl缓冲盐溶液清洗后默克密理博发光液显色、曝光,Image J 软件分析灰度值;以目的条带和内参条带比值进行半定量分析。
1.4 统计分析实验数据采用
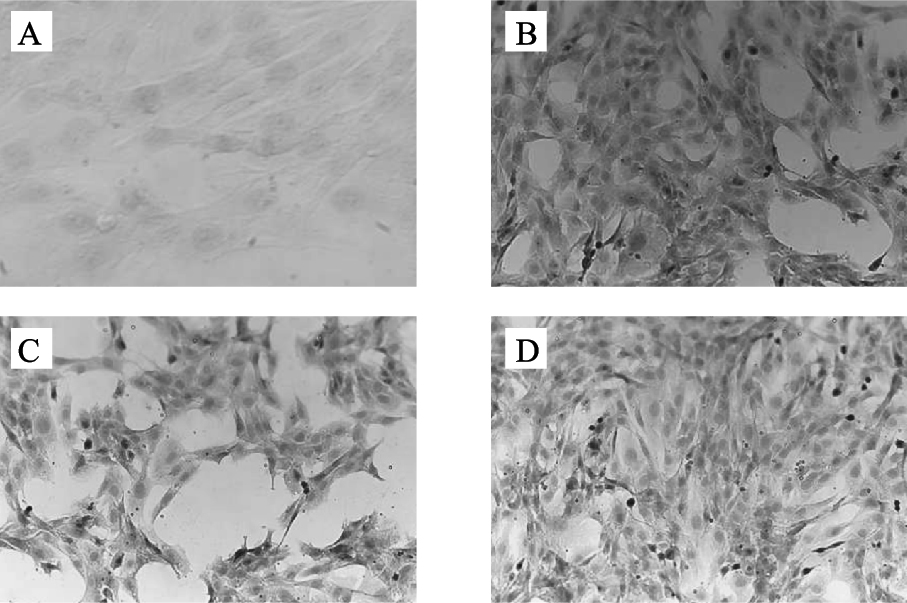
|
注:A:对照组;B:氧化应激组;C、D:20、80 μmol/L AM1241干预组。 图 1 AM1241对HSC-T6细胞α-SMA表达影响(细胞免疫化学染色,A:×400;B、C、D:×200) |
细胞免疫化学染色结果显示,α-SMA 主要表达于HSC-T6细胞胞质中,对照组HSC-T6细胞胞质中无α-SMA表达(图 1)。与氧化应激组(142±12)比较,20、80 μmol/L AM1241干预组HSC-T6细胞胞质α-SMA表达[分别为(109±13)、(89±14)]均下降(P < 0.05)。对照组、氧化应激组及20、80 μmol/L AM1241干预组HSC-T6细胞培养上清中Col I含量分别为(203.89±0.10)、(269.03±0.12)、(170.40±0.14)、(62.46±0.16)μg/mL;与对照组比较,氧化应激组HSC-T6细胞培养上清中Col I含量升高(P < 0.05);与氧化应激组比较,AM1241干预组HSC-T6细胞培养上清中Col I含量均下降,差异均有统计学意义(P < 0.05)。
2.2 AM1241 对HSC-T6细胞氧化应激指标影响(表 1)| 表 1 AM1241对HSC-T6细胞丙二醛含量及SOD活力影响(x±s, n=6) |
与对照组比较,氧化应激组HSC-T6细胞中丙二醛含量上升,SOD活性下降(P < 0.05);与氧化应激组比较,AM1241干预组HSC-T6细胞中丙二醛含量均下降,SOD活性均上升(P < 0.05)。
2.3 AM1214对HSC-T6细胞Nrf2总蛋白与核蛋白表达影响(图 2)
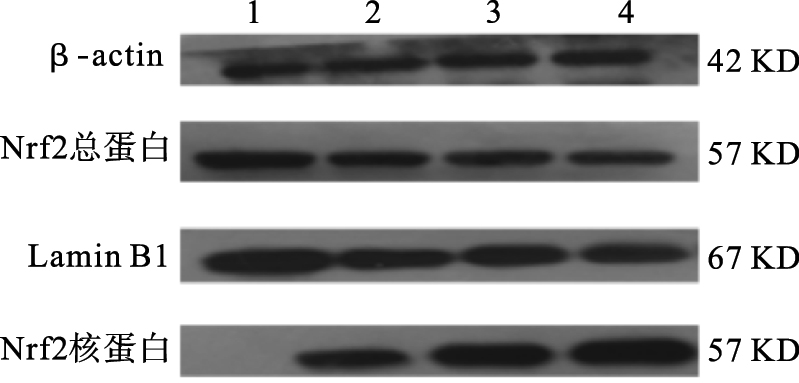
|
注:1.对照组;2.氧化应激组;3、4:20、80 μmol/L AM1241干预组。 图 2 AM1241对HSC-T6细胞Nrf2总蛋白与核蛋白表达影响 |
对照组、氧化应激组及20、80 μmol/L AM1241干预组HSC-T6细胞中Nrf2总蛋白表达量分别为(0.62±0.021)、(0.60±0.015)、(0.59±0.020)、(0.61±0.032),差异无统计学意义(P>0.05)。对照组、氧化应激组及20、80 μmol/L AM1241干预组HSC-T6细胞中Nrf2核蛋白表达量分别为(0.01±0.021)、(0.41±0.014)、(0.68±0.014)、(0.79±0.020);与对照组比较,氧化应激组HSC-T6细胞中Nrf2核蛋白表达量增加(P < 0.05);与氧化应激组比较,AM1241干预组HSC-T6中Nrf2核蛋白表达量均增加,差异均有统计学意义(P < 0.05)。
3 讨 论肝纤维化是以细胞外基质过度沉淀为形态学基础的复杂病理过程,肝星状细胞在肝纤维化过程中起核心作用[13]。活化的肝星状细胞产生大量以胶原为主的细胞外基质成分和细胞因子,是肝纤维化形成的中心环节[14]。正常情况下,肝星状细胞通常处于静息状态,储存脂肪和维生素A,合成和分泌多种胶原酶及少量细胞外基质[15]。氧化应激作用使肝星状细胞活化并分泌出大量细胞外基质导致肝纤维化,并表达α-SMA,目前被公认为是预防和治疗慢性肝病的重要靶细胞[16, 17]。肝星状细胞活化的标志是表达α-SMA[18]。研究表明,HSC-T6激活的各阶段均有氧化应激发挥重要作用,氧化应激中产生的大量活性氧超氧阴离子可氧化不饱和脂质导致脂质过氧化,产生脂质过氧化物丙二醛和4-壬烯等,脂质过氧化物可直接激活肝星状细胞,介导肝星状细胞的分化增殖和胶原合成[19, 20]。丙二醛作为脂质过氧化产物之一,可以反映脂质过氧化的程度,也间接反映细胞氧化损伤程度,丙二醛含量测定常与SOD活力测定相互配合,SOD活力的高低间接反映机体清除氧自由基能力,而丙二醛含量可间接反应细胞受氧自由基攻击的程度[21]。本研究结果显示,对照组HSC-T6细胞中α-SMA表达呈阴性,氧化应激组、AM1241干预组HSC-T6细胞中α-SMA 均呈阳性表达,表明HSC-T6细胞已经活化。
大麻素CB2受体是机体内源性大麻素系统的一种G蛋白偶联受体[22]。在慢性肝病中大麻素CB2主要表达在肝脏非实质细胞,在体外激活的肝星状细胞中也有表达,其表达可能与抑制肝纤维化作用有关[23]。Nrf2属于亮氨酸拉链家族的调节抗氧化应激反应的重要转录因子,生理情况下Nrf2作用被抑制,在氧化应激等情况下被激活,启动抗氧化反应元件(antioxidantresponseelement,ARE)调控的第Ⅱ相解毒酶及抗氧化酶基因表达,增加细胞对氧化应激和亲电子化学物质的抗性[24-26]。葡萄糖氧化酶与培养基中的葡萄糖反应生成葡萄糖醛酸和双氧水等自由基活化HSC-T6细胞,合成大量ECM,如Col I;活性氧超氧阴离子产生的自由基通过脂质过氧化作用而损伤HSC-T6细胞,产生脂质过氧化物丙二醛,SOD 作为超氧阴离子自由基的清除剂可抑制自由基启动的脂质过氧化反应。本研究结果显示,在氧化应激作用下,HSC-T6细胞Col I含量增高,丙二醛浓度随大麻素CB2受体激动剂AM1241浓度增加而明显降低,SOD活力升高。提示,大麻素CB2受体激动剂AM1241 可以减轻HSC-T6细胞的氧化应激损伤。
本研究结果还显示,在氧化应激作用下Nrf2由胞浆进入胞核,而且AM2141高剂量组HSC-T6细胞胞核中Nrf2含量高于低剂量组。提示,在氧化应激作用下,大麻素CB2受体激动剂可促使Nrf2由HSC-T6细胞胞浆进入胞核,Nrf2进入胞核启动Ⅱ相解毒酶及抗氧化酶基因表达,增加细胞对氧化应激和亲电子化学物质的抗性,起到抗氧化作用。
| [1] | Neeraj KS, Frank AA. Adipocytokine hepatic fibrosis[J]. Trends in Endocrinology and Metabolism, 2015, 26 :153–161 . |
| [2] | Wang CY, Liu Q, Huang QX, et al. Activation of PPARγ is required for hydroxysafflor yellow A of Carthamus tinctorius to attenuate hepatic fibrosis induced by oxidative stress[J]. Phytomedicine, 2013, 20 :592–599 . |
| [3] | Lu L, Wang JL, Luet HJ, et al. MicroRNA-130a and-130b enhance activation of hepatic stellate cells by suppressing PPARγ expression:a rat fibrosis model study[J]. Biochemical and Biophysical Research Communications, 2015, 465 :387–393 . |
| [4] | Julien B, Grenard P, Teixeira-Clerc F, et al. Antifibrogenic role of the cannabinoid receptor CB2 in the liver[J]. Gastroenterology, 2006, 128 :742–755 . |
| [5] | Caraceni P, Domenicali M, Giannone F, et al. The role of the endocannabinoid system in liver diseases[J]. Best Practice and Research Clinical Endocrinology and Metabolism, 2009, 23 :65–77 . |
| [6] | Antoniades CG, Berry PA, Wendon JA, et al. The importance of immune dysfunction in determining outcome in acute liver failure[J]. Hepatol, 2008, 49 :845–861 . |
| [7] | Mallat A, Teixeira-Clerc F, Lotersztajn S. Cannabinoid signaling and liver therapeutics[J]. Journal of Hepatology, 2013, 59 :891–896 . |
| [8] | Kathline K, Dan HM, Alexandros M, et al. AM1241,a cannabinoid CB2receptor selective compound,delays diseaseprogression in a mouse model of amyotrophic lateral sclerosis[J]. European Journal of Pharmacology, 2006, 542 :100–105 . |
| [9] | Park JS, Kang DH, Lee DH, et al. Fenofibrate activates Nrf2 through p62-dependent Keap1 degradation[J]. Biochemical and Biophysical Research Communications, 2015, 465 :542–547 . |
| [10] | Ji LL, Sheng YC, Zheng ZY, et al. The involvement of p62-Keap1-Nrf2 antioxidative signaling pathway and JNK in the protection of natural flavonoid quercetin against hepatotoxicity[J]. Free Radical Biology and Medicine, 2015, 85 :12–23 . |
| [11] | Steib CJ, Gmelin L, Pfeiler S, et al. Functional relevance of the cannabinoid receptor 2-heme oxygenase pathway:a novel target for the attenuation of portal hypertension[J]. Life Sciences, 2013, 93 :543–551 . |
| [12] | Li W, Kong AN. Molecular mechanisms of Nrf2-mediated antioxidant response[J]. Mol Carcinog, 2009, 48 :94–104 . |
| [13] | Wu LY, Liu SH, Liu Y, et al. Up-regulation of interleukin-22 mediates liver fibrosis via activating hepatic stellate cells in patients with hepatitis C[J]. Clinical Immunology, 2015, 158 :77–87 . |
| [14] | Wang Q, Dai XF, Yang WZ, et al. Caffeine protects against alcohol-induced liver fibrosis by dampening the cAMP/PKA/CREB pathway in rat hepatic stellate cells[J]. International Immunopharmacology, 2015, 25 :340–352 . |
| [15] | Raicevic G, Najar M, Najimi M, et al. Influence of inflammation on the immunological profile of adult-derived human liver mesenchymal stromal cells and stellatecells[J]. Cytotherapy, 2015, 17 :174–185 . |
| [16] | Yu FX, Ji XQ, Su LF, et al. Adipose-derived mesenchymal stem cells inhibit activation of hepatic stellate cells in vitro and ameliorate rat liver fibrosis in vivo[J]. Journal of the Formosan Medical Association, 2015, 114 :130–138 . |
| [17] | Zhang F, Ni CY, Kong DS, et al. Ligustrazine attenuates oxidative stress-induced activation of hepatic stellate cells by interrupting platelet-derived growth factor-β receptor-mediated ERK and p38 pathways[J]. Toxicology and Applied Pharmacology, 2012, 265 :51–60 . |
| [18] | Sophie C, Stéphanie P, Stéphanie C, et al. The hepatitis C virus core protein indirectly induces alpha-smooth muscle actin expression in hepatic stellate cells via interleukin-8[J]. Journal of Hepatology, 2010, 52 :635–643 . |
| [19] | Dharmendra S, Arya PV. Modulatory potential of α-amyrin against hepatic oxidative stress through antioxidant status in wistar albino rats[J]. Journal of Ethnopharmacology, 2015, 161 :186–193 . |
| [20] | Lina AT, Sarit D, Mahmud M, et al. Leptin modulates lymphocytes' adherence to hepatic stellate cells is associated with oxidative status alterations[J]. Mitochondrion, 2013, 13 :473–480 . |
| [21] | Manas RB, Chulbul MA. Systemic treatment with a 5HT1a agonist induces anti-oxidant protection and preserves the retina from mitochondrial oxidative stress[J]. Experimental Eye Research, 2015, 140 :94–105 . |
| [22] | Mona F, Mahmoud M, Sahar ES, et al. Role of cannabinoid receptors in hepatic fibrosis and apoptosis associated with bile duct ligation in rats[J]. European Journal of Pharmacology, 2014, 742 :118–124 . |
| [23] | Mallat A, Lotersztajn S. Endocannabinoids as novel mediators of liver diseases[J]. Endocrinol Invest, 2006, 29 :58–65 . |
| [24] | Nazimabashir, Vaihundam M, Selvaraj M. Cadmium induced cardiac oxidative stress in rats and its attenuation by GSP through the activation of Nrf2 signaling pathway[J]. Chemico-Biological Interactions, 2015, 242 :179–193 . |
| [25] | Sandra S, Ana, eljka M, et al. The role of 17β-estradiol in the regulation of antioxidant enzymes via the Nrf2-Keap1 pathway in the livers of CBA/H mice[J]. Life Sciences, 2015, 130 :57–65 . |
| [26] | Lee BH, Hsu WH, Hsu YW, et al. Suppression of dimerumic acid on hepatic fibrosis caused from carboxymethyl-lysine(CML) by attenuating oxidativ stress depends on Nrf2 activation in hepatic stellate cells(HSCs)[J]. Food and Chemical Toxicology, 2013, 62 :413–419 . |
 2016, Vol. 32
2016, Vol. 32


