Three new 14-noreudesmane-type sesquiterpenoids from the roots of Hippophae rhamnoides
Abstract
Hippophae rhamnoides L. (Elaeagnaceae), commonly known as sea buckthorn, is a medicinal plant valued for its diverse bioactive constituents and broad therapeutic potential. Phytochemical investigation of its roots led to the isolation of three new 14-noreudesmane-type sesquiterpenoids (1–3), together with sixteen known compounds (4–19). Their structures and absolute configurations were determined using comprehensive spectroscopic analyses and quantum chemical computations of electronic circular dichroism (ECD) spectra and 13C NMR shifts. Three new sesquiterpenoids (1–3) were tested for their antioxidant, anti-inflammatory, antibacterial, and α-glucosidase inhibitory activities. Unfortunately, none exhibited significant activity under the tested conditions. Among the isolated known compounds, all those displaying α-glucosidase inhibitory and antibacterial activities are pentacyclic triterpenoids. Hippophamide (17) showed significant DPPH and ABTS radical-scavenging activity. These results enriched the chemical profile of H. rhamnoides roots.Graphical Abstract

Keywords
Hippophae rhamnoides 14-noreudesmane sesquiterpenoids Bioactivity1 Introduction
Hippophae rhamnoides L., commonly known as sea buckthorn, is a member of the Elaeagnaceae family and is native to several regions across Asia and Europe [1, 2]. The plant is predominantly distributed in countries such as China, Turkey, Russia, Mongolia, Tajikistan, India, Greece, Afghanistan, and various other locations [3]. H. rhamnoides berries has long been utilized in Chinese traditional medicine to strengthen the spleen, promote digestion, relieve coughs, dispel phlegm, and invigorate blood circulation to dissipate blood stasis [4, 5]. H. rhamnoides is not only used in traditional Chinese medicine but also is an actinorhizal plant, which can enter a mutualistic symbiosis with Frankia. Extensive phytochemical analyses of H. rhamnoides covered various plant organs, such as the leaves [6], fruits [7, 8], branches [9], and seeds [10], and have identified a broad spectrum of secondary metabolites, including terpenoids, alkaloids, volatile oils, flavonoids and steroids. However, chemical studies on the roots remain scarce. So far, only five flavonoids have been documented from the roots [11]. In the ongoing phytochemical research on actinorhizal plant [12, 13], a detailed phytochemical studies of H. rhamnoides roots led to the discovery of three new 14-noreudesmane-type sesquiterpenoids (1–3), along with sixteen known compounds (4–19). This study presents the processes of extraction and isolation, followed by an in-depth structural analysis of these sesquiterpenoids. Additionally, their biological activities were evaluated, including antioxidant, anti-inflammatory, α-glucosidase inhibitory, and antibacterial properties.
2 Results and discussion
2.1 Structural elucidation
A total of nineteen compounds were successfully isolated and elucidated from the roots of H. rhamnoides (Fig. S1, supplementary file), including three newly identified 14-noreudesmane-type sesquiterpenoids (1–3) (Fig. 1), and sixteen known compounds (4–19) (Fig. S2, supplementary file).
The structures of compounds 1–3
Compound 1 was obtained as a brown oil with [α]D25=–14.1 (c 0.19, MeOH). Its molecular formula was established to be C15H22O2 from its molecular ion peak at m/z 234.1614 in the HR-EI-MS spectrum. The 1H-NMR spectrum (Table 1) displayed three methyl doublets at δH 1.27 (3H, d, J=7.0 Hz, H-15), 1.25 (3H, d, J=7.0 Hz, H-12) and 1.24 (3H, d, J=7.0 Hz, H-13), as well as a singlet at δH 3.78, indicative of an oxygen-substituted methyl group (–OCH3). The 13C-NMR data (Table 1) in combination with the HSQC spectrum revealed a total of fifteen carbon signals, which were classified as three methyls, one oxygenated methyl, three methylenes, three methines, and five quaternary carbons. The 1H-1H COSY spectrum of 1 showed two spin-coupled systems of H-1/H-2/H-3/H-4/H-15 and H-12/H-11/H-13 suggesting the presence of a connections of C-1/C-2/C-3/C-4/C-15 and an isopropyl group [14] (Fig. 2). The remaining carbon signals could be ascribed to a benzene ring and an oxygenated methyl. In the HMBC spectrum, δH 3.25 (H-11), 1.25 (H-12), and 1.24 (H-13) correlated with δC 132.5 (C-7), and δH 3.25 (H-11) correlated with δC 121.2 (C-6) and 143.8 (C-8), suggesting the isopropyl group linked with the benzene ring via C-7. The HMBCs from H-1 to C-5, C-9 and C-10, from H-15 to C-5, from H-6 to C-4, indicated that the moiety of C-1/C-2/C-3/C-4/C-15 were attached to the benzene ring through C-1 linking with C-10, and through C-4 linking with C-5. To meet the requirements of the molecular formula and the presence of five quaternary carbons, a hydroxyl group should be substituted on the benzene ring. The hydroxyl group (δH 5.60, s) was assigned to C-8 based on its HMBC correlation to C-7. Consequently, the oxygenated methyl group was positioned at C-9. By comparing the measured electronic circular dichroism (ECD) spectra of 1 with the calculated 4R-1 (Fig. 4), the absolute configuration of 1 was determined as 4R. Thus, compound 1 was elucidated as (4R)-9-methoxy-14-noreudesma-5, 7, 9-trien-8-ol.
1H (500 MHz) and 13C (125 MHz) NMR data of compounds 1–3 in CDCl3
1H– 1H COSY and key HMBC correlations of 1–3
Compound 2 was obtained as a pale-brown oil with [α]D25=–20 (c 0.11, MeOH). It was found to have the molecular formula C14H20O according to the molecular ion peak at m/z 204.1511 in the HR-EI-MS spectrum. The 1H-NMR spectrum (Table 1) displayed three methyl doublets at δH 1.30 (3H, d, J=7.1 Hz, H-15), 1.23 (3H, d, J=7.0 Hz, H-13), and 1.22 (3H, d, J=7.0 Hz, H-12). The 13C-NMR data in combination with the HSQC spectrum revealed a total of fourteen carbon signals, which were classified into three methyls, three methylenes, four methines, and four quaternary carbons (Table 1). Its 1H and 13C NMR as well as 1H-1H COSY pattern was closely similar to that of compound 1, which indicated that compound 2 possessed the 14-noreudesmane skeleton. The key difference between them was the absence of the methoxy group and the presence of an additional aromatic proton signal at δH 6.52 (1H, d, J=1.7 Hz, H-8) in the 1H-NMR spectrum of 2. The coupling constant between two aromatic protons was 1.7 Hz, which indicated that they are meta-position. This is also confirmed by HMBCs from H-11 to C-6, C-7 and C-8. To meet the requirements of the molecular formula and the presence of four quaternary carbons, a hydroxyl group should be substituted on the benzene ring at C-9. The absolute configuration of 2 was further determined as 4R-2 by comparing the calculated ECD curve with its experimental values (Fig. 4). As a result, compound 2, was fully characterized as (4R)-14-noreudesma-5, 7, 9-trien-9-ol.
Compound 3 was obtained as a brown oil with [α]D25=–12.4 (c 0.15, MeOH). Its molecular formula was determined as C15H22O4 based on the pseudo-molecular ion at m/z 267.1597 [M+H]+ observed in positive HR-ESI–MS. The 1H-NMR spectrum (Table 1) revealed resonances for one aromatic proton at δH 6.79 (s, H-6), two methyls at δH 1.29 (d, 7.0 Hz, H-12) and 1.31 (d, 7.0 Hz, H-15), and a methoxy group at δH 3.79 (s). The 13C-NMR spectrum (Table 1) displayed fifteen carbon signals, five of which are quaternary carbons δC (145.0, 144.2, 131.5, 128.5, and 126.9), four methines, three methylenes, and three methyl carbons. After comparing the 1H and 13C NMR data with those of compound 1, it was easy to find 3 also possesses 14-noreudesmane skeleton. With the help of the HSQC spectrum, all protons were correlated to their respective carbons (Table 1). The 1H-1H COSY spectrum showed two proton spin-coupled systems of H-1/H-2/H-3/H-4/H-15 and H-12/H-11/H-13 (Fig. 2). C-3 was substituted by a hydroxy group based on the observation of δH 3.80 (m, H-3) and δC (72.3, C-3). C-13 was also linked with a hydroxy group, which was confirmed in the same way like C-3. In the low-field region of 13C NMR spectrum, 3 shared highly similar signal pattern of the benzene ring to that of compound 1. The substitutions of a methoxy group at C-9 and of a hydroxy group at C-8 on the benzene ring were confirmed based on the HMBC observations from H-1 and the methoxy protons to C-9, as well as from H-11 to C-8, respectively. Consequently, compound 3 was identified as 9-methoxy-14-noreudesma-5, 7, 9-triene-3, 8, 13-triol. In order to assign the relative configuration of 3, four stereoisomers, (3S, 4S, 11R)-, (3R, 4S, 11R)-, (3S, 4S, 11S)-, and (3R, 4S, 11S)-3 were subjected to theoretical simulations for 13C NMR shifts using the GIAO method [15–18]. The goodness of fit among the predicted 13C NMR data of four stereoisomers and the experimental data of 3 were evaluated by the modified DP4 probability (DP4+) values [19, 20]. As can be seen in Fig. 3, the calculated 13C NMR shifts of (3S, 4S, 11S)-isomer showed the best match with the measured data of 3 (DP4+probability: 99.35%), leading assignment of the relative configurations 3S*, 4S*, 11S*. Then, (3S, 4S, 11S)-3 was calculated for the ECD spectrum. As can be seen in Fig. 4, the calculated ECD curve was in excellent fit with the experimental spectrum of 3. Thus, 3 was tentatively assigned to have the absolute configurations 3S, 4S, 11S.
Linear regression analysis of the calculated 13C NMR shifts of four possible stereoisomers against the measured shifts of 3 and the goodness of fit test by DP4 + probability analysis
Comparison of the measured and calculated ECD spectra of 1–3 in methanol
Compounds 1–3 were 14-noreudesmane type sesquiterpenoids. Previously, four 14-sesquiterpenoids of this type, namely hipponorterpenes A and B, 6, 9-dihydroxy-1-oxo-14-noreudesma-5, 7, 9-triene and 6-hydroxy-2-isopropyl-5-methoxy-8-methylnaphthalene-1, 4-dione, were also reported from H. rhamnoides, but from its berries and juice [14, 21]. Interestingly, such sesquiterpenoids have been isolated from some other natural sources, including Nicotiana tabacum (Solanaceae) [22], Alpinia oxyphylla (Zingiberaceae) [23], the fungus Hypoxylon rickii [24], and the edible mushroom Flammulina velutipes [25].
The sixteen known compounds were identified as arjunolic acid (4) [26], 18Hα, 3β, 20β-ursanediol (5) [27], 3β-hydroxyolean-12-en-28-al (6) [28], 2α, 3β, 19α, 23-tetrahydroxyolean-12-en-28-oic acid (7) [29], erythrodiol (8) [30], oleanolic acid (9) [31], ursolic acid (10) [32], maslinic acid (11) [33], ergosterol endoperoxide (12) [34], stigmast-4-en-3-one (13) [35], β-sitosterol (14) [36], daucosterol-6'-linoleate (15) [37], (+)-catechin (16) [38], hippophamide (17) [39], dehydrodiconiferyl alcohol (18) [40], 22-O-(4-hydroxy-3-methoxy-cinnamy1) docosanoic acid (19) [41], (Fig. S2, supplementary file), by comparing their spectroscopic data with previously documented values in literature.
2.2 Antioxidant activity
The antioxidant activity of all compounds was evaluated using two in vitro assays. As summarized in Table 2, compounds 16 and 17 showed notable activity in the DPPH radical scavenging assay, with IC50 values of 30.66 μM and 43.15 μM, respectively, compared to L-ascorbic acid (IC50=23.77 μM). In the ABTS cation radical scavenging assay, both compounds 16 and 17 displayed even stronger activity, with IC50 values of 13.17 μM and 20.31 μM, respectively, outperforming L-ascorbic acid (IC50=24.85 μM). Three new compounds 1–3 were tested in the cellular reactive oxygen species (ROS) scavenging assay at 50 μM. All of them showed inhibition rate below 10%. Compared to curcumin (IC50=16.84 μM), the intracellular antioxidant activity of these compounds was negligible.
Antioxidant activity of compounds 1–19
2.3 α-Glucosidase inhibitory activity
Compounds 1–19 were evaluated for their inhibitory activity against α-glucosidase, using corosolic acid as positive control. As shown in Table 3, compound 10 exhibited the most potent inhibitory effect, with an IC50 value of 4.81 μM, equivalent to that of corosolic acid (IC50=5.60 μM). Compounds 4, 5, 8, 9, and 11 showed moderate activity, with IC50 values ranging from 8.96 to 33.58 μM. All active compounds are triterpenoids, while the positive control corosolic acid is also a triterpenoid.
α-Glucosidase inhibitory activity of compounds 1–19
2.4 Antibacterial activity
All 19 isolated compounds were evaluated for their antibacterial activity against three bacterial strains: Staphylococcus aureus (MSSA, CMCC 26003), methicillin-resistant Staphylococcus aureus (MRSA, JCSC 3063), and Escherichia coli (EC, ATCC 8739) (Table 4). Minimum inhibitory concentrations (MICs) were determined and compared with positive controls, including kanamycin, vancomycin, and polymyxin. Among the tested compounds, compound 9 exhibited weak antibacterial activity against MSSA and MRSA, with an MIC of 100 μM. Compound 10 displayed weak antibacterial activity against both MSSA and MRSA, with an MIC value of 25 μM. None of the compounds demonstrated antibacterial activity against the Gram-negative strain E. coli.
Antibacterial activity of compounds 1–19 (MIC, μM)
2.5 Anti-inflammatory activity
To evaluate the anti-inflammatory potential of three new compounds (1–3), their ability to suppress nitric oxide (NO) production was tested using LPS-stimulated RAW 264.7 murine macrophage cells. Compounds 1–3 were tested at concentrations of 50, 25 and 10 μM. However, these compounds exhibited NO production inhibitory rates below 10% at all concentrations tested, compared to the positive control, indomethacin (IC50=36.29 µM).
3 Experimental procedures
3.1 General
Column chromatography (CC) was performed using silica gel (80–100 mesh and 200–300 mesh; Yantai Jiangyou Silica Gel Development Co., Ltd., Yantai, China), Sephadex LH-20 (Pharmacia Fine Chemicals Co., Ltd., Uppsala, Sweden), and reversed-phase C18 (YMC Co., Ltd., Japan) materials. Preparative high-performance liquid chromatography (HPLC) was carried out with an LC-6AD pump system equipped with an SPD UV/Vis detector (Shimadzu, Kyoto, Japan), using a Cosmosil column (250 mm × 10 mm i.d., 5 μm particle size, Kyoto, Japan) at a flow rate of 2 mL/min. Thin-layer chromatography (TLC) was performed on precoated silica gel plates (HSGF254; Yantai Jiangyou Silica Gel Development Co., Ltd., Yantai, China), with spot detection achieved by spraying with a 5% vanillin in sulfuric acid solution followed by heating. Proton (1H) and carbon (13C) nuclear magnetic resonance (NMR) spectra were recorded on a Bruker DRX-500 NMR spectrometer (Bruker Biospin GmbH, Rheinstetten, Germany), with chemical shifts referenced to residual solvent peaks. The spectrometer operated at 500 MHz for 1H NMR and 125 MHz for 13C NMR. High-resolution electron impact mass spectrometry (HR-EI-MS) and Electrospray ionization mass spectrometry (ESI–MS) data were obtained using a Thermo Scientific DFS magnetic mass spectrometer (Thermo Corporation). Optical rotation values (αD) were measured on a Perkin-Elmer 343 polarimeter using methanol (MeOH) as the solvent. Circular Dichroism (CD) spectrum was recorded on a Chirascan circular dichroism spectrometer (Applied Photophysics Ltd., Surrey, UK).
3.2 Plant material
Roots of Hippophae rhamnoides were collected in April 2023 from Diqing, Yunnan province, China, by Kunming Zhifen Biotechnology Company, and were botanically authenticated by this company. A voucher specimen (No. Zzy20230414) was deposited at the phytochemistry laboratory, South China Botanical Garden, Chinese Academy of Sciences.
3.3 Extraction and isolation
The air-dried roots were pulverized into a fine powder (14 kg) and extracted three times with 95% aqueous ethanol. The combined filtrates were concentrated under reduced pressure to obtain a crude extract weighing 1007.6 g. The extract was successively partitioned with petroleum ether (4.5 L × 3), ethyl acetate (4.5 L × 3), and n-butanol (4.5 L × 3). The resulting fractions were concentrated to dryness under vacuum yielding petroleum ether-soluble (38.0 g), ethyl acetate-soluble (75.2 g), and n-butanol-soluble (418.8 g) fractions. The ethyl acetate fraction (75.2 g) underwent purification through silica gel column chromatography (CC), Sephadex LH-20 CC, preparative high-performance liquid chromatography (HPLC), and reversed-phase C18 (RP-C18) CC to isolate individual compounds. A comprehensive flowchart detailing the phytochemical extraction and isolation procedure for compounds 1–19 is provided in Fig. S2, supplementary file.
3.4 Spectroscopic data new compounds
3.4.1 (4R)-9-Methoxy-14-noreudesma-5, 7, 9-trien-8-ol (1)
Brown oil; [α]D25 = –14.1 (c 0.19, MeOH); UV (MeOH) λmax nm (log ε): 202 (3.60), 280 (2.44); HR-EI-MS m/z 234.1614 (calculated for C15H22O2, 234.1614); 1H (500 MHz) and 13C (125 MHz) NMR data, see Table 1.
3.4.2 (4R)-14-Noreudesma-5, 7, 9-trien-9-ol (2)
Pale brown oil; [α]D25 = –20 (c 0.11, MeOH); UV (MeOH) λmax nm (log ε): 203 (3.63), 281 (2.35); HR-EI-MS m/z 204.1511 (calculated for C14H20O, 204.1509); 1H (500 MHz) and 13C (125 MHz) NMR data, see Table 1.
3.4.3 9-Methoxy-14-noreudesma-5, 7, 9-triene-3, 8, 13-triol (3)
Brown oil; [α]D25 = –12.4 (c 0.15, MeOH); UV (MeOH) λmax nm (log ε): 203 (3.61), 281 (2.43); HR-ESI–MS (positive) m/z 267.1597 (calculated for C15H23O4, 267.1591); ESI–MS (positive) m/z 267 [M+H]+ and 289 [M+Na]+, C15H22O4; 1H (500 MHz) and 13C (125 MHz) NMR data, see Table 1.
3.5 Computational details
Conformational search was done using Molecular Merck force field (MMFF) embedded in Spartan'14 software (Wavefunction Inc., Irvine, CA, USA). Density functional theory (DFT) and time-dependent density functional theory (TDDFT) calculations were performed with Gaussian09 RevD.01 [42]. Double-hybrid (DH) DFT calculations were conducted with ORCA 5.0.4 program package using RIJCOSX approximation, tight SCF criteria [43]. For conformational analysis, conformers of compounds 1, 2 and 3 each within an 8 kcal/mol energy window from MMFF conformational search were subjected to geometry optimizations followed by frequency calculations using DFT method at the B3LYP [44, 45] -GD3BJ [46, 47] /6-31G(d) level of theory. To simulate the 13C NMR shifts of 3, the optimized low-energy conformers (relative electronic energies < 4.0 kcal/mol) were subjected to NMR calculations using the gauge including atomic orbitals (GIAO) [15, 17, 18] method at the mPW1PW91/6–311+G (d, p) level [16] of theory with the solvent model PCM for chloroform. The unscaled chemical shifts (δu) were computed using TMS as reference standard according to δu=σ0−σx (where σx is the Boltzmann averaged shielding tensor and σ0 is the shielding tensor of TMS computed at the same level employed for σx). The Boltzmann averaging was done using the relative energies obtained from the single-point NMR calculations [19, 20] The goodness of fit test between the simulated 13C NMR data of the four stereoisomers and the experimental shifts of 3 were evaluated by the improved DP4 probability (DP4+) [19, 20]. For calculations of the ECD spectra, the optimized geometries were subjected to single point calculations using the DH-DFT method at the (PWPB95 [48]-GD3BJ/def2-QZVPP [49] level of theory with the SMD [50] solvent model for MeOH to obtain more accurate electronic energies. The TDDFT calculations were carried out using M06-2X [51], PBE1PBE (PBE0) [52], M11-L [53], and revTPSS [54] functionals in combination with the TZVP [51] basis set and the PCM solvation model for MeOH. The number of excited states was 30 for all conformers. The results were visualized and exported using the SpecDis program [55]. The calculated ECD spectra was generated as a sum of Gaussian curve using rotatory strengths computed in the dipole-velocity gauge from ECD data of the individual conformers. Boltzmann distributions of the conformers in equilibrium population were estimated from the relative Gibbs free energies (ΔG) at 298.15K.
3.6 Antioxidant activity
Antioxidant activity was evaluated by in vitro DPPH radical scavenging assay, ABTS radical cation scavenging assay, and cellular reactive oxygen species (ROS) scavenging assay following our lab previous methods [56, 57].
3.7 α-Glucosidase activity
The α-glucosidase inhibitory activity of compounds 1–19 was assessed based on a previously reported method with slight modifications [58]. In a 96-well microtiter plate, 70 µL of phosphate buffer, 10 µL of α-glucosidase enzyme solution (0.5 U/mL), and 10 µL of the test compound at varying concentrations (50, 25, 12.5, 6.25, and 3.12 μM) were added to each well. Corosolic acid (10 µL) was used as a positive control. For the negative control, the test compound was replaced with phosphate buffer, while the blank control was prepared by omitting the enzyme and substituting it with buffer. The reaction mixtures were incubated at 37 ℃ for 10 min, followed by the addition of 10 µL of PNPG solution per well. After a further 20 min incubation at 37 ℃, the reaction was terminated by adding 100 µL of Na2CO3 solution. The absorbance was measured at 405 nm using a microplate reader. All experiments were conducted in triplicate, and results are expressed as mean values±standard deviation (SD).
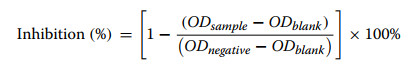
|
3.8 Antibacterial activity
The antibacterial activity of compounds 1–19 was evaluated against Staphylococcus aureus (MSSA, CMCC 26003), methicillin-resistant Staphylococcus aureus (MRSA, JCSC 3063), and Escherichia coli (EC, ATCC 8739) using a resazurin-based microdilution assay. Bacterial suspensions were prepared by adding 30 µL of the inoculum to 5 mL of Brain Heart Infusion (BHI) medium followed by incubation at 37 ℃ in a shaking incubator set at 200 rpm for 12 h. The bacterial suspension was then diluted to an optical density (OD) of 0.07 ± 0.002, corresponding to 1.25 × 106 CFU/mL. A resazurin stock solution (1 mg/mL) was prepared in sterile water and diluted to 100 µg/mL for use. Stock solutions of compounds were prepared in biological-grade DMSO at 1 mM, and used as test samples. The assay was performed on 96-well plates. First, 90 µL of a resazurin-bacterial mixed solution (in a 3:2 volume ratio) and 10 µL of the test samples were added to the first row (Row A) and thoroughly mixed using a pipette. Then, 50 µL of the resazurin-bacterial solution was added to rows B–H. Next, 50 µL mixture from Row A was transferred to Row B and mixed well; this process was repeated for Rows C–H. Two-fold serial dilutions were performed across the rows. The plate was incubated at 37℃ for 6–8 h. The MIC was determined by the lowest concentration at which the bacterial solution changed from purple to pink, indicating inhibition of growth. Each test was performed in triplicate. All bacterial strains were obtained from the Guangdong Institute of Microbiology (Guangzhou, China). Vancomycin and kanamycin were used as positive controls for MSSA and MRSA, and polymyxin for EC.
3.9 Anti-inflammatory activity
The anti-inflammatory potential of compounds 1–3 was evaluated by measuring their ability to inhibit nitric oxide (NO) production in lipopolysaccharide (LPS)-stimulated RAW 264.7 macrophages, following our previously established protocols [57].
4 Conclusion
In conclusion, the phytochemical exploration of H. rhamnoides roots yielded three new 14-noreudesmane-type sesquiterpenoids and a suite of known compounds. The main constituents were oleanane-type triterpenoids. While the newly identified sesquiterpenoids exhibited limited biological activity, several triterpenoids demonstrated promising α-glucosidase inhibitory, and antibacterial properties. Hippophamide (17) showed significant antioxidant activity with DPPH and ABTS radicals scavenging potential. These results enriched the chemical profile of H. rhamnoides roots.
Notes
Acknowledgements
We would like to thank Mr. Yunfei Yuan, South China Botanical Garden, Chinese Academy of Sciences (CAS), for NMR spectroscopic measurements; Dr. Hanxiang Li, South China Botanical Garden, CAS, for GC-EI-MS experiment; Prof. Ping Wu, South China Botanical Garden, Chinese Academy of Sciences, for ESI-MS experiment; Prof. Changan Geng, Kunming Institute of Botany, CAS, for EI- and HR-EI-MS experiment; Senior engineer Aijun Sun, Institute of South China Sea Oceanology, CAS, for ECD experiment. The photo of Hippophae rhamnoides fruits used in Graphical Abstract is courtesy of Prof. Caiyun He, Research Institute of Forestry, Chinese Academy of Forestry.
Author contributions
FAG carried out the methodology, investigation, formal analysis, visualization, data curation, writing—original draft, and writing—review & editing; PL contributed to biological investigation; SB contributed to isolation; GD contributed to isolation and formal analysis; ZZ was responsible for conceptualization, supervision, validation, resources, funding acquisition, project administration, and writing—review & editing; HX and XW contributed to ECD calculations, writing—review & editing, and project supporting. All authors read and approved the final manuscript.
Funding
This research was supported by the Natural Science Foundation of Guangdong Province (grant number 2025A1515010367), National Natural Science Foundation of China (grant number 31970376), and Guangdong Science and Technology Plan Project (grant number 2023B1212060046).
Data availability
The experimental data supporting this work are accessible within the article and its Additional file 1.
Declarations
Competing interests
The authors declare that they have no known financial or personal competing interests that could have influenced the research reported in this manuscript.
References
-
1.Singh V. Global distribution of sea buckthorn (Hippophae sp) resources and their utilization. In: The Sea Buckthorn Genome. UK: Springer International Publishing; 2022. PubMed Google Scholar
-
2.Jubayer MF, Mazumder MAR, Nayik GA, Ansari MJ, Ranganathan TV. Hippophae rhamnoides L.: Sea buckthorn. In: Immunity Boosting Medicinal Plants of the Western Himalayas. UK: Springer; 2023. PubMed Google Scholar
-
3.Ma QG, He NX, Huang HL, Fu XM, Zhang ZL, Shu JC, et al. Hippophae rhamnoides L.: a comprehensive review on the botany, traditional uses, phytonutrients, health benefits, quality markers, and applications. J Agric Food Chem. 2023;71: 4769-88. CrossRef PubMed Google Scholar
-
4.Dong W, Tang Y, Qiao J, Dong Z, Cheng J. Sea buckthorn bioactive metabolites and their pharmacological potential in digestive diseases. Front Pharmacol. 2025;16: 1637676. CrossRef PubMed Google Scholar
-
5.He N, Wang Q, Huang H, Chen J, Wu G, Zhu M, et al. A comprehensive review on extraction, structure, detection, bioactivity, and metabolism of flavonoids from sea buckthorn (Hippophae rhamnoides L.). J Food Biochem. 2023;2023: 4839124. CrossRef PubMed Google Scholar
-
6.Kubczak M, Khassenova AB, Skalski B, Michlewska S, Wielanek M, Sklodowska M, et al. Hippophae rhamnoides L. leaf and twig extracts as rich sources of nutrients and bioactive compounds with antioxidant activity. Sci Rep. 2022;12: 1095. CrossRef PubMed Google Scholar
-
7.Ma Q, Guan Y, Sang Z, Dong J, Wei R. Isolation and characterization of auronlignan derivatives with hepatoprotective and hypolipidemic activities from the fruits of Hippophae rhamnoides L. Food Funct. 2022;13: 7750-61. CrossRef PubMed Google Scholar
-
8.Lee DE, Park KH, Hong JH, Kim SH, Park KM, Kim KH. Anti-osteoporosis effects of triterpenoids from the fruit of sea buckthorn (Hippophae rhamnoides) through the promotion of osteoblast differentiation in mesenchymal stem cells, C3H10T1/2. Arch Pharm Res. 2023;46: 771-81. CrossRef PubMed Google Scholar
-
9.Tkacz K, Wojdyło A, Turkiewicz IP, Nowicka P. Triterpenoids, phenolic compounds, macro-and microelements in anatomical parts of sea buckthorn (Hippophae rhamnoides L.) berries, branches and leaves. J Food Compos Anal. 2021;103: 104107. CrossRef PubMed Google Scholar
-
10.Ding J, Hu N, Li G, Wang H-L. Megastigmanes and flavonoids from seeds of Hippophae rhamnoides. Chem Nat Compd. 2024;60: 21-5. CrossRef PubMed Google Scholar
-
11.Hibasami H, Mitani A, Katsuzaki H, Imai K, Yoshioka K, Komiya T. Isolation of five types of flavonol from sea buckthorn (Hippophae rhamnoides) and induction of apoptosis by some of the flavonols in human promyelotic leukemia HL-60 cells. Int J Mol Med. 2005;15: 805-9. PubMed Google Scholar
-
12.Xu Y, Xu Y, Huang Z, Luo Y, Gao R, Xue J, et al. 3-pentanol glycosides from root nodules of the actinorhizal plant Alnus cremastogyne. Phytochemistry. 2023;207: 113582. CrossRef PubMed Google Scholar
-
13.Jin Y, Xu Y, Huang Z, Zhou Z, Wei X. Metabolite pattern in root nodules of the actinorhizal plant Casuarina equisetifolia. Phytochemistry. 2021;186: 112724. CrossRef PubMed Google Scholar
-
14.Rédei D, Kúsz N, Rafai T, Bogdanov A, Burián K, Csorba A, et al. 14-noreudesmanes and a phenylpropane heterodimer from sea buckthorn berry inhibit Herpes simplex type 2 virus replication. Tetrahedron. 2019;75: 1364-70. CrossRef PubMed Google Scholar
-
15.Wolinski K, Hinton JF, Pulay P. Efficient implementation of the gauge-independent atomic orbital method for NMR chemical shift calculations. J Am Chem Soc. 1990;112: 8251-60. CrossRef PubMed Google Scholar
-
16.Lodewyk MW, Soldi C, Jones PB, Olmstead MM, Rita J, Shaw JT, et al. The correct structure of aquatolide-experimental validation of a theoretically predicted structural revision. J Am Chem Soc. 2012;134: 18550-3. CrossRef PubMed Google Scholar
-
17.Ditchfield R. Self-consistent perturbation theory of diamagnetism: Ⅰ. a gauge-invariant LCAO method for NMR chemical shifts. Mol Phys. 1974;27: 789-807. CrossRef PubMed Google Scholar
-
18.Ditchfield R. Molecular orbital theory of magnetic shielding and magnetic susceptibility. J Chem Phys. 1972;56: 5688-91. CrossRef PubMed Google Scholar
-
19.Smith SG, Goodman JM. Assigning stereochemistry to single diastereoisomers by GIAO NMR calculation: the DP4 probability. J Am Chem Soc. 2010;132: 12946-59. CrossRef PubMed Google Scholar
-
20.Grimblat N, Zanardi MM, Sarotti AM. Beyond DP4: an improved probability for the stereochemical assignment of isomeric compounds using quantum chemical calculations of NMR shifts. J Org Chem. 2015;80: 12526-34. CrossRef PubMed Google Scholar
-
21.Zhang XL, Na HY, Li PS, Chen YX, Li ZL, Liang GD, et al. Hipponorterpenes A and B, two new 14-noreudesmane-type sesquiterpenoids from the juice of Hippophae rhamnoides. Phytochem Lett. 2022;52: 82-6. CrossRef PubMed Google Scholar
-
22.Yang CY, Geng CA, Huang XY, Wang H, Xu HB, Liang WJ, et al. Noreudesmane sesquiterpenoids from the leaves of Nicotiana tabacum. Fitoterapia. 2014;96: 81-7. CrossRef PubMed Google Scholar
-
23.Park DH, Lee JW, Jin Q, Jeon WK, Lee MK, Hwang BY. A new noreudesmane-type sesquiterpenoid from Alpinia oxyphylla. Bull Korean Chem Soc. 2014;35: 1565-7. CrossRef PubMed Google Scholar
-
24.Kuhnert E, Surup F, Wiebach V, Bernecker S, Stadler M. Botryane, noreudesmane and abietane terpenoids from the ascomycete Hypoxylon rickii. Phytochemistry. 2015;117: 116-22. CrossRef PubMed Google Scholar
-
25.Tao Q, Ma K, Yang Y, Wang K, Chen B, Huang Y, et al. Bioactive sesquiterpenes from the edible mushroom Flammulina velutipes and their biosynthetic pathway confirmed by genome analysis and chemical evidence. J Org Chem. 2016;81: 9867-77. CrossRef PubMed Google Scholar
-
26.Ramesh AS, Christopher JG, Radhika R, Setty CR, Thankamani V. Isolation, characterisation and cytotoxicity study of arjunolic acid from Terminalia arjuna. Nat Prod Res. 2012;26: 1549-52. CrossRef PubMed Google Scholar
-
27.Wang F, Li ZL, Cui HH, Hua HM, Jing YK, Liang SW. Two new triterpenoids from the resin of Boswellia carterii. J Asian Nat Prod Res. 2011;13: 193-7. CrossRef PubMed Google Scholar
-
28.Kamiya K, Yoshioka K, Saiki Y, Ikutat A, Satak T. Triterpenoids and flavonoids from Paeonia lactiflora. Phytochemistry. 1997;44: 141-4. CrossRef PubMed Google Scholar
-
29.Xia M, Tan J, Yang L, Shang Z, Zhao Q, Shi G. Studies on chemical constituents in Patrinia scabiosaefolia. Chin Tradit Herbal Drugs. 2010;41: 1612-5. PubMed Google Scholar
-
30.Osman W, Ibrahim M, Adam M, Mothana R, Mohammed M, Abdoon I, et al. Isolation and characterization of four terpenoidal compounds with potential antimicrobial activity from Tarconanthus camphorantus L. (Asteraceae). J Pharm Bioallied Sci. 2019;11: 373-9. CrossRef PubMed Google Scholar
-
31.Castellano JM, Sara RR, Perona JS. Oleanolic acid: extraction, characterization and biological activity. Nutrients. 2022;14: 623. CrossRef PubMed Google Scholar
-
32.Gnoatto SC, Klimpt AD, Nascimento SD, Galera P, Boumediene K, Gosmann G, et al. Evaluation of ursolic acid isolated from Ilex paraguariensis and derivatives on aromatase inhibition. Eur J Med Chem. 2008;43: 1865-77. CrossRef PubMed Google Scholar
-
33.Murakami C, Myoga K, Kasai R, Ohtani K, Kurokawa T, Ishibashi S, et al. Screening of plant constituents for effect on glucose transport activity in Ehrlich ascites tumour cells. Chem Pharm Bull. 1993;41: 2129-31. CrossRef PubMed Google Scholar
-
34.Kwon HC, Zee SD, Cho SY, Chop SU, Lee KR. Cytotoxic ergosterols from Paecilomyces sp. J300. Arch Pharm Res. 2002;25: 851-5. CrossRef PubMed Google Scholar
-
35.Jibril S, Sirat HM, Zakari A, Sani IM, Kendeson CA, Abdullahi Z, et al. Isolation of chemical constituents from n-hexane leaf extract of Cassia singueana del. (Fabaceae). Chem Search J. 2019;10: 14-20. PubMed Google Scholar
-
36.Ododo MM, Choudhury MK, Dekebo AH. Structure elucidation of β-sitosterol with antibacterial activity from the root bark of Malva parviflora. Springerplus. 2016;5: 1-11. CrossRef PubMed Google Scholar
-
37.Zhimin L, Lijun W, Bingya J, Bohang S, Huang J, Huiyuan G. Chemical constituents of chestnut kernel (Ⅲ). J Shenyang Pharm Univ. 2008;25: 856. PubMed Google Scholar
-
38.Zhu XD, Xu B, Wang F. Studies on the chemical components of Osyris wightiana. Nat Prod Res Dev. 2009;21: 956-9. PubMed Google Scholar
-
39.OuYang J, Zhou WN, Li G, Wang XY, Ding CX, Suo YR, et al. Three new alkaloids from Hippophae rhamnoides Linn. subsp. sinensis Rousi. Helv Chim Acta. 2015;98: 1287-91. CrossRef PubMed Google Scholar
-
40.Guangshu W, Muxin Z, Xiaohong Y, Jingda X. Studies on the non-alkaloid constituents of Hippeastrun vittatum in Amaryllidaceae. Chin Pharm J. 2005;40: 498-9. PubMed Google Scholar
-
41.Wang Yan WY, Su BingHe SB, Zhou XiaoYu ZX, Wang TianMin WT, Zhai YanJun ZY. Study on chemical constituents of processed fructus Polygoni orientalis. Liaoning J Tradit Chin Med. 2012;39: 505-8. PubMed Google Scholar
-
42.Gaussian 09, revision D.01. 2013, Gaussian, Inc.: Wallingford CT. PubMed Google Scholar
-
43.Neese F. Software update: the ORCA program system, version 4.0. WIRES Comput Mol Sci. 2018;8: e1327. CrossRef PubMed Google Scholar
-
44.Becke AD. Density-functional thermochemistry Ⅲ. The role of exact exchange. J Chem Phys. 1993;98: 5648-52. CrossRef PubMed Google Scholar
-
45.Lee CT, Yang WT, Parr RG. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B. 1988;37: 785-9. CrossRef PubMed Google Scholar
-
46.Grimme S, Antony J, Ehrlich S, Krieg H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J Chem Phys. 2010;132: 154104. CrossRef PubMed Google Scholar
-
47.Grimme S, Ehrlich S, Goerigk L. Effect of the damping function in dispersion corrected density functional theory. J Comput Chem. 2011;32: 1456-65. CrossRef PubMed Google Scholar
-
48.Goerigk L, Grimme S. Efficient and accurate Double-Hybrid-Meta-GGA density functionals evaluation with the extended GMTKN30 database for general main group thermochemistry, kinetics, and noncovalent interactions. J Chem Theory Comput. 2011;7: 291-309. CrossRef PubMed Google Scholar
-
49.Weigend F, Ahlrichs R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys Chem Chem Phys. 2005;7: 3297-305. CrossRef PubMed Google Scholar
-
50.Marenich AV, Cramer CJ, Truhlar DG. Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J Phys Chem B. 2009;113: 6378-96. CrossRef PubMed Google Scholar
-
51.Chen G, Wang Y, Zhao C, Korpelainen H, Li C. Genetic diversity of Hippophae rhamnoides populations at varying altitudes in the Wolong natural reserve of China as revealed by ISSR markers. Silvae Genet. 2008;57: 29. CrossRef PubMed Google Scholar
-
52.Perdew JP, Burke K, Ernzerhof M. Generalized gradient approximation made simple. Phys Rev Lett. 1996;77: 3865-8. CrossRef PubMed Google Scholar
-
53.Peverati R, Truhlar DG. M11-L: a local density functional that provides improved accuracy for electronic structure calculations in chemistry and physics. J Phys Chem Lett. 2012;3: 117-24. CrossRef PubMed Google Scholar
-
54.Perdew JP, Ruzsinszky A, Csonka GI, Constantin LA, Sun JW. Workhorse semilocal density functional for condensed matter physics and quantum chemistry. Phys Rev Lett. 2009;103: 026403. CrossRef PubMed Google Scholar
-
55.Bruhn T, Schaumloeffel A, Hemberger Y, Bringmann G. SpecDis: quantifying the comparison of calculated and experimental electronic circular dichroism spectra. Chirality. 2013;25: 243-9. CrossRef PubMed Google Scholar
-
56.Ma Q, Xie H, Li S, Zhang R, Zhang M, Wei X. Flavonoids from the pericarps of Litchi chinensis. J Agric Food Chem. 2014;62: 1073-8. CrossRef PubMed Google Scholar
-
57.Cheng XL, Li HX, Chen J, Wu P, Xue JH, Zhou ZY, et al. Bioactive diarylheptanoids from Alpinia coriandriodora. Nat Prod Bioprospect. 2021;11: 63-72. CrossRef PubMed Google Scholar
-
58.Liu SB, Zeng L, Xu QL, Chen YL, Lou T, Zhang SX, et al. Polycyclic phenol derivatives from the leaves of Spermacoce latifolia and their antibacterial and α-glucosidase inhibitory activity. Molecules. 2022;27: 3334. CrossRef PubMed Google Scholar
Copyright information
© The Author(s) 2026
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.






