Methods A total of 2794 representative, Mycobacterium tuberculosis isolates from treatment-naive patients were subjected to drug susceptibility testing, and risk factors for drug-resistant TB were analyzed. We also analyzed MDR-TB strain sublineages, drug-resistance-conferring mutations, and risk factors associated with clustered primary MDR strains.
Results Among 2794 Mycobacterium tuberculosis isolates from treatment-naive patients, the prevalence of any resistance to first-line drugs was 33.2% and the prevalence of MDR-TB was 5.7%. We did not find any risk factors significantly associated with resistance to first-line drugs. The 93 primary MDR-TB isolates were classified into six sublineages, of which, 75 (80.6%) isolates were the RD105-deleted Beijing lineage. The largest sublineage included 65 (69.9%) isolates with concurrent deletions of RD105, RD207, and RD181. Twenty-nine (31.2%) primary MDR strains grouped in clusters; MDR isolates in clusters were more likely to have S531L rpoB mutation.
Conclusion This study indicates that primary drug-resistant TB and MDR-TB strains are prevalent in China, and multiple measures should be taken to address drug-resistant TB.
Drug-resistant tuberculosis (TB) is an increasing health problem worldwide,with China being one of the countries most burdened with drug-resistant TB. In 2007,it was estimated that in China there were 110,000 incident cases of multidrug-resistant tuberculosis (MDR-TB) and 8200 incidence cases of extensively drug-resistant (XDR) TB[1]. Generally,people consider the main causes of MDR-TB to be poor patient management,non-adherence to the prescribed regimen,a substandard national program,or some combination of these three. Successful treatment of TB has benefits for the patient and reduces the drug-resistant TB burden of a country. It is recommended that the treatment of drug-resistant TB should be guided by drug susceptibility test (DST) results[2]. In China,according to current guidelines,culture and DST for Mycobacterium tuberculosis (M. tuberculosis) are recommended only in specific patients considered to be at risk for drug-resistant TB[3]. Access to culture and DST for diagnosis of drug-resistant TB in new TB cases remains unavailable in many areas of China,and new patients with pulmonary TB are often treated empirically with standardized short-course chemotherapy regimen[4].
Previous studies covering part areas of China showed that some patients had primary drug-resistant TB and the prevalence of such resistance among the Chinese population has shown an upward trend[5, 6].Reports from previous studies indicated patients with initial drug resistance who were treated with only standardized regimens showed incidences of failure,relapse,and acquired drug resistance were substantially increased[7, 8]. According to Guidelines of World Health Organization[9],some Chinese provinces carried out surveys on drug resistance in tuberculosis patients- the frequency of drug resistance varies greatly across different regions. In addition,no studies using standardized methodology have been performed to evaluate primary drug resistance in new cases. Therefore,the objective of the present study was to investigate prevalence of primary drug-resistant TB and to contribute new data on the transmission of primary drug-resistant TB in China.
MATERIALS AND METHODS Study Objects and Data CollectionWe performed this study using representative M. tuberculosis strains isolated from a national survey of drug-resistant TB in China[1]. During the survey,data on social and demographic characteristics,clinical characteristics,exposure to TB,and TB treatment history were collected from each of the newly diagnosed,smear-positive TB patients through a standard questionnaire. The patient's treatment history was based on patient interview and validated by review of the patient's medical record. Patients who had no history of TB treatment or evidence of TB treatment on medical record were included into this study. Patients who had been treated for TB,or there was evidence of TB treatment on their medical records were excluded from this study.
Ethics StatementThe study protocol was approved by the Ethical Review Committees of Chinese Center for Disease Control and Prevention. Written informed consent was obtained from the study participants.
Drug Resistance Testing and Molecular FingerprintingThe smear-positive specimens were cultured and subjected to identification of M. tuberculosis by p-nitrobenzoic acid method. Susceptibility to rifampicin (RFP),isoniazid (INH),streptomycin (SM),ethambutol (EMB),kanamycin (KM) and ofloxacin (OFX) was performed on Löwenstein-Jensen (L-J) medium,according to WHO standard proportional method. DNA specimens were extracted from strains,and all isolates were characterized by spoligotyping according to standard methods[10].
Sequencing of Drug Resistance GenesDNA from primary MDR-TB isolates was amplified and sequenced for the drug resistance-determining regions of rpoB and katG using primers rpoB F (5’-tacggtcggcga gctgatcc-3’) and rpoB R (5’-tacggcgtt tcgatgaacc-3’),katG F (5’-aacgacgtcga aacagcggc-3’),and katG R (5’-gcgaactcgtcg gccaattc-3’). Sequences were compared with published sequences for M. tuberculosis H37Rv by using DNAstar and BioEdit software.
Determination of Strain Lineages and ClustersMultiplex PCRs were performed to identify lineages of the primary MDR-TB strains as reported previously[11, 12],and the lineages were defined based on the presence or absence of the region of difference (RD) 105,RD181,RD207,RD150,and RD142. Isolates were considered to belong to the same cluster if the strains had the same phenotypic drug resistance profile,the same M. tuberculosis lineage,and the same drug resistance-conferring mutations. Isolates were considered unique if any of these criteria were not met. Cluster analysis was performed by BioNumerics Software (Version 5.1,Belgium).
Statistical AnalysisTo explore the relationship between drug resistance or strain cluster with demographic and bacterial variables,univariate analysis using Chi-square test or Fisher’s exact test,odds ratios (OR) were calculated with 95% confidence intervals (95% CI). Continuous variables were compared using the Student’s t-test or non-parametric test. Differences were considered as significant if P<0.05. Statistical analyses were performed using the SPSS statistical package (SPSS,Chicago,USA).
RESULTS Characteristics of Study Objects and Drug Resistance ProfilesDuring the TB baseline survey,3037 new smear-positive cases of tuberculosis were enrolled,of whom 240 patients had taken anti-tuberculosis drugs for less than one month and 2797 new cases had not reported treatment history for TB (3 cases were excluded as they did have treatment history for TB on their medical records). Ultimately,2794 new cases with no history of using anti-tuberculosis drugs were included into this study. There were 1937 males (69.3%) and 857 females (30.7%),with the age of the patients ranged from 14 to 93 years and a median age of 44.46 (±18.9) years.
Table 1 shows the profile of drug resistance in the patients. Among the 2794 new TB cases studied,1867 (66.8%) excreted bacilli that were susceptible to all four first-line drugs,and 927 (33.2%) were resistance to any first-line drugs. When each drug was considered separately,the highest rates of any resistance to first-line drugs were observed for Streptomycin (26.8%) and Isoniazid (15.7%),followed by Rifampin (6.6%) and Ethambutol (5.0%). Highest monoresistance was observed against Streptomycin (14.6%),followed by Isoniazid (3.7%). One hundred and sixty (5.7%) new cases were infected by MDR strains.
|
|
Table 1. Prevalence of Primary Drug Resistant Clinical Strains of M. tuberculosis Isolated in China from 2007-2008 |
Among strains tested in this study,2679 (95.9%) were susceptible to the second-line drugs Ofloxacin and Kanamycin. Any resistance to Ofloxacin,Kanamycin was 73 (2.6%) and 55 (2.0%),respectively,compared with 17 (0.6%) and 7 (0.3%) for monoresistance to Ofloxacin and Kanamycin,respectively. Prevalence of XDR TB was 0.4%.
Of 2744 strains with available spoligotyping results,1748 (63.7%) belonged to Beijing genotype,and for 158 spoligotyped MDR strains,93 (58.9%) were Beijing genotype and 65 (41.1%) were non-Beijing genotype strains.
Risk Factors for Primary Drug-Resistant TBTable 2 shows an analysis of the variables potentially associated with drug resistance in this study. None of the variables studied were found to be statistically associated with TB patients with any resistance to first-line drugs. Female patients were more likely to have primary MDR-TB (OR=1.64,95% CI 1.18-2.27).
|
|
Table 2. Univariable Analysis for Risk Factors for Primary Drug Resistant TB and MDR-TB |
By screening lineage-specific genetic markers,93 primary MDR-TB strains were classified into six sublineages. 75 (80.6%) of the strains were RD105-deleted East Asian or Beijing lineage,of which,65 (86.7%) isolates were in sublineage 1 with concurrent deletions of RD105,RD207,and RD181. No significant difference was observed between lineages with respect to the associations with S315T KatG and S531L rpoB mutations,(Fisher’s 2-tailed exact test: P=0.802,P=1.000,respectively),and there was no significant difference between lineages and age (F=0.678,P=0.642) or sex (Fisher’s 2-tailed exact test: P=0.306). (Table 3).
|
|
Table 3. Characteristics of Sublineages and Resistance Mutations Found in 93 Primary MDR-TB Patients in China |
As seen in Table 4 and Figure 1,clustering analysis revealed the presence of ten clusters (5,5,4,3,2,2,2,2,2,2) in 93 MDR strains,where a total of 29 (31.2%) patients were included over the ten clusters. In sublineage 1,there were three larger clusters with S315T KatG and S531L rpoB mutations and similar drug resistance profiles,except for ethambutol or streptomycin resistance. Risk factors associated with patients whether or not they were grouped in clusters revealed that those patients in clusters were more likely to have the S531L rpoB mutation. We did not find any other factors such as age,sex,ethnic,contact history or S315T KatG associated with clustering.
|
|
Table 4. Univariable Analysis for Risk Factors for Grouping in Cluster of Primary MDR-TB |
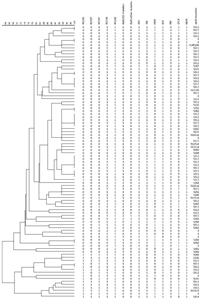
|
Download: |
| Figure 1. Clustering map of 93 cases of primary MDR-TB patients in China. | |
We found that the prevalence of primary drug-resistant TB was high in China and 5.7% of the patients were primary MDR-TB. Previous studies from Jiangsu,Shanghai China also indicated a high prevalence of primary MDR-TB[13, 14]. Additional findings show transmission of drug-resistant TB strains among the general population in China[15]. The rate of primary MDR-TB from this study was comparable to reports from Peru[16] and Vietnam[17],lower than some reports from India,Russian Federation and Saudi Arabia[18, 19, 20, 21],and higher than that of Brazil and Ethiopia[22, 23, 24].
At present,the China National TB control program implement DOTS (directly observed therapy,short course) strategy,and standardized,short-course chemotherapy is recommended for patients with previously untreated tuberculosis[4]. Considering the high prevalence of primary drug-resistant TB among new smear-positive cases,standardized short-course chemotherapy may result in inadequate treatment,generating MDR-TB,which in turn increases the transmission of drug resistant TB strains[25, 26].
According to data from national drug resistant TB baseline survey[1],the detected total number of cases who are resistant to first-line drug and MDR were 1503 and 401,respectively,and among new smear-positive,untreated cases,the counterpart number of cases was 927 and 160,respectively. If these new cases cannot access DST test,more than 60% of drug-resistant TB and 40% of MDR-TB patients will not be correctly diagnosed. The results here are in accordance with the results of the previous study[27]. Therefore,in conclusion to resulting data of this study,DST tests should be used to screen all new smear-positive TB cases.
In this study,31.2% of the primary MDR-TB grouped in clusters. None of the factors showed statistically significant association with clustering patients except the S531L rpoB mutation. The S531L rpoB mutation is the most prevalent rifampin resistance-conferring mutation in clinical strains. For these clinical isolates,it was suggested that the rpoB S531L mutation incurred low or no fitness cost in these particular strains’ genetic backgrounds[28]. Another study indicated that secondary mutations were acquired that mitigate the initial low fitness cost of the rpoB S531L mutation and contributed to the spread of drug resistance strains[29].
The study has some limitation: only patients attending clinics of tuberculosis prevention and control system were included,although most of the TB patients were treated in this system in the survey period,TB cases that were treated in hospital were not included in this study,and this study may promote a selection bias. Despite this limitation,this study investigated a large number of TB isolates representative of the tuberculosis prevention and control system of the country,and developed profile of the status of primary drug-resistance in China.
In summary,this study provides evidence that prevalence of primary drug-resistant TB and MDR-TB was high in China,and drug-resistant M.tuberculosis strains had disseminated across general populations. These findings emphasize that all new smear-positive TB cases should be screened with DST to identify drug-resistant TB and MDR-TB,and,if possible,rapid DST diagnostics should be implemented to reduce transmission of drug-resistant TB. The findings of this study consolidate that infection control should be considered as priority strategy to address drug-resistant TB in China.
ACKNOWLEDGEMENTSConflicts of interest: The authors have declared that no competing interests exist.
| [1] | Zhao Y, Xu S, Wang L, et al. National survey of drug-resistant tuberculosis in China. N Engl J Med, 2012; 366, 2161-70. |
| [2] | Blumberg HM, Burman WJ, Chaisson RE, et al. American Thoracic society/Centers for Disease Control and Prevention/Infectious Diseases Society of America: treatment of tuberculosis. Am J Respir Crit Care Med, 2003; 167, 603-62. |
| [3] | World Health Organization. Guidelines for the programmatic management of drug-resistant tuberculosis. WHO Report 2011. WHO/HTM/TB/2011.6. Geneva, Switzerland: WHO; 2011. |
| [4] | World Health Organization. Treatment of tuberculosis: guidelines for national programmes. WHO/CDS/TB/2000.313. 3rd edition. Geneva, Switzerland. |
| [5] | Li X, Zhang Y, Shen X, et al. Transmission of drug-resistant tuberculosis among treated patients in Shanghai, China. J Infect Dis, 2007; 195, 864-9. |
| [6] | Yang XY, Li YP, Mei YW, et al. Time and spatial distribution of multidrug-resistant tuberculosis among Chinese people, 1981-2006: a systematic review. Int J Infect Dis, 2010; 14, e828-37. |
| [7] | Lew W, Pai M, Oxlade O, et al. Initial drug resistance and tuberculosis treatment outcomes: systematic review and meta-analysis. Ann Intern Med, 2008; 149, 123-34. |
| [8] | Seung KJ, Gelmanova IE, Peremitin GG, et al. The Effect of Initial Drug Resistance on Treatment Response and Acquired Drug Resistance during Standardized Short-Course Chemotherapy for Tuberculosis. Clin Infect Dis, 2004; 39, 1321-8. |
| [9] | World Health Organization. Guidelines for surveillance of drug resistance in tuberculosis. WHO/CDS/TB/2003.320. Geneva, Switzerland. |
| [10] | Kamerbeek J, Schouls L, Kolk A, et al. Simultaneous detection and strain differentiation of Mycobacterium tuberculosis for diagnosis and epidemiology. J Clin Microbiol, 1997; 35, 907-14. |
| [11] | Reed MB, Pichler VK, McIntosh F, et al. Major Mycobacterium tuberculosis Lineages Associate with Patient County of Origin. J Clin Microbiol, 2009; 1119-28. |
| [12] | Gagneux S, DeRiemer K, Van T, et al. Variable host-pathogen compatibility in Mycobacterium tuberculosis. Proc Natl Acad Sci USA, 2006; 103, 2869-73. |
| [13] | Liu Q, Zhu L, Shao Y, et al. Rates and risk factors for drug resistance tuberculosis in Northeastern China. BMC Public Health, 2013; 13, 1171. |
| [14] | Zhao M, Li X, Xu P, et al. Transmission of MDR and XDR tuberculosis in Shanghai, China. PLoS One, 2009; 4, e4370. |
| [15] | Hu Yi, Mathema Barun, Jiang Weili, et al. Transmission Pattern of Drug-Resistant Tuberculosis and Its Implication for Tuberculosis Control in Eastern Rural China. PLoS One, 2011; 6, e19548. |
| [16] | Otero L, Krapp F, Tomatis C, et al. High prevalence of primary multidrug resistant tuberculosis in persons with no known risk factors. PLoS One, 2011; 6, e26276. |
| [17] | Hang NT, Maeda S, Lien LT, et al. Primary drug-resistant tuberculosis in Hanoi, Viet Nam: present status and risk factors. PLoS One, 2013; 8, e71867. |
| [18] | Gupta H, Kant S, Jain A, et al. Initial drug resistance pattern among pulmonary tuberculosis patients. Indian J Tuberc, 2013; 60, 154-61. |
| [19] | Mathuria JP, Samaria JK, Srivastava GN, et al. Primary and acquired drug resistance patterns of Mycobacterium tuberculosis isolates in India: a multicenter study. J Infect Public Health, 2013; 6, 456-64. |
| [20] | Zhdanova S, Heysell SK, Ogarkov O, et al. Primary multidrug-resistant Mycobacterium tuberculosis in 2 regions, Eastern Siberia, Russian Federation. Emerg Infect Dis, 2013; 19, 1649-52. |
| [21] | Asaad AM, Alqahtani JM. Primary anti-tuberculosis drugs resistance of pulmonary tuberculosis in Southwestern Saudi Arabia. J Infect Public Health, 2012; 5, 281-5. |
| [22] | Bastos GM, Cezar MC, Mello EC, et al. Prevalence of primary drug resistance in pulmonary tuberculosis patients with no known risk factors for such. J Bras Pneumol, 2012; 38, 733-9. |
| [23] | da Silva Garrido M, Ramasawmy R, Perez-Porcuna TM, et al. Primary drug resistance among pulmonary treatment-naïve tuberculosis patients in Amazonas State, Brazil. Int J Tuberc Lung Dis, 2014; 18, 559-63. |
| [24] | Yimer SA, Agonafir M, Derese Y, et al. Primary drug resistance to anti-tuberculosis drugs in major towns of Amhara region, Ethiopia. APMIS, 2012; 120, 503-9. |
| [25] | Chan ED, Iseman MD. Multidrug-resistant and extensively drug-resistant tuberculosis: a review. Curr Opin Infect Dis, 2008; 21, 587-95. |
| [26] | Faustini A, Hall AJ, Perucci CA. Risk factors for multidrug resistant tuberculosis in Europe: a systematic review. Thorax, 2006; 61, 158-63. |
| [27] | Brewer TF, Choi HW, Seas C, et al. Self-reported risks for multiple-drug resistance among new tuberculosis cases: implications for drug susceptibility screening and treatment. PLoS One, 2011; 6, e25861. |
| [28] | Gagneux S, Long CD, Small PM, et al. The competitive cost of antibiotic resistance in Mycobacterium tuberculosis. Science, 2006; 312, 1944-6. |
| [29] | de Vos M, Müller B, Borrell S, et al. Putative compensatory mutations in the rpoC gene of rifampin-resistant Mycobacterium tuberculosis are associated with ongoing transmission. Antimicrob Agents Chemother, 2013; 57, 827-32. |
 2016, Vol.29
2016, Vol.29 


