2. 广东环境保护工程职业学院 机电工程系,广东 佛山 528216
2. Department of Mechanical and Electrical Engineering, Guangdong Polytechnic of Environmental Protection Engineering, Foshan 528216, China
氢能作为一种储量丰富、来源广泛、能量密度高的绿色清洁能源,已受到人们的广泛重视,被视为一种潜在的理想能源[1]. 氢能的开发和利用需要解决氢的制取、储运和应用3个问题[2]. 固态储氢因具有储氢密度大、安全性高的特点而受到科研人员的广泛关注[3]. 传统储氢合金的储氢密度低而难以满足车载燃料电池的需求,轻质储氢材料由于其储氢密度高而成为近年来的研究热点,如MgH2、硼氢化物、铝氢化物和氨基氢化物等[4-6].
硼氢化锂(LiBH4)的储氢质量分数和体积分数分别高达18.4%和121 kg/m3. 由于组成元素之间的强化学键作用,LiBH4的吸放氢反应存在热力学稳定性高和动力学缓慢等问题,通常需要在400 ℃以上才能够释放大量氢气[7]. 为此,研究人员对其开展了广泛的研究,采取了去稳定化、纳米限域和元素替代等方法,以期降低LiBH4的放氢温度和提高放氢动力学性能[8-10].
去稳定化是指通过添加合适的去稳定化剂,构建复合储氢体系,使放氢反应焓变降低,从而有效地降低放氢温度和提高放氢动力学性能. Vajo等[11-12]首先报道了LiBH4-MgH2复合储氢体系,通过改变LiBH4的放氢反应路径有效地降低了LiBH4的稳定性. 此外,研究人员还开展了大量的工作,陆续报道了金属/非金属单质、金属氢化物和金属氧化物等对改善LiBH4放氢性能的作用[13-21]. 纳米限域是采用熔融渗入或溶剂浸渍的方法将LiBH4装载于多孔材料中,目前已研究的多孔材料包括碳基材料、金属有机框架和介孔二氧化硅等[22-27]. Liu等[28-29]将LiBH4限域于孔径为2 nm的有序多孔碳中,显著地降低了放氢温度并抑制了副产物B2H6气体的释放;并且发现通过调控孔径大小可以改变LiBH4的物化性能. 元素替代是基于Li+对[BH4]-基团的电子补偿导致LiBH4热力学稳定性高而提出的方法. 该方法通过采用电负性更高的金属阳离子替代部分Li+,抑制电子向[BH4]-基团转移,从而降低LiBH4的结构稳定性[30]. 此外,研究人员还提出阴离子替代的方法,采用具有不同体积大小的阴离子实现对H-或[BH4]-基团的替代. 目前,研究人员开发了多种元素替代硼氢化合物,如LiK(BH4)2、Li1-xZrx(BH4)n、Li4Al3(BH4)13、Li8B8H32-xFx和Li(BH4)1-xClx等[31-36].
本文通过高能球磨法构建LiBH4-NdCl3储氢体系,系统考察了预球磨、球磨时间、球粉比和掺杂量等球磨工艺参数对体系放氢性能的影响规律,从而阐明表面能和界面接触面积的作用,并且探索了LiBH4-NdCl3的去稳定化机理.
1 实验部分 1.1 样品的制备LiBH4(纯度
| 表 1 不同球磨工艺下的LiBH4-NdCl3体系 Table 1 LiBH4-NdCl3 samples prepared under different ball milling conditions |
采用Hiden Qic20质谱仪进行程序升温脱附–质谱(TPD-MS)测试变温放氢特性,同时检测气体组分;加热温度以2 ℃/min的升温速率从室温加热至500 ℃,同时采用60 mL/min的高纯氩作为载气. 采用美国Setaram的PCTPro2000测试等温放氢动力学特性,蓄气池初始压力设为零,放氢量相对于LiBH4计算. 采用Philips X’Pert XRD对样品物相结构进行表征,测试过程采用3M膜保护样品,该膜在XRD图谱的2θ=18°处产生漫散衍射峰. 采用Zeiss Supra 40 SEM对材料微观组织结构进行表征. 为避免样品与空气中的水、氧等物质发生化学反应,所有样品准备过程都在手套箱中进行. 手套箱内为高纯氩,水氧质量分数含量低于3×10–6.
2 结果与讨论 2.1 放氢性能表征
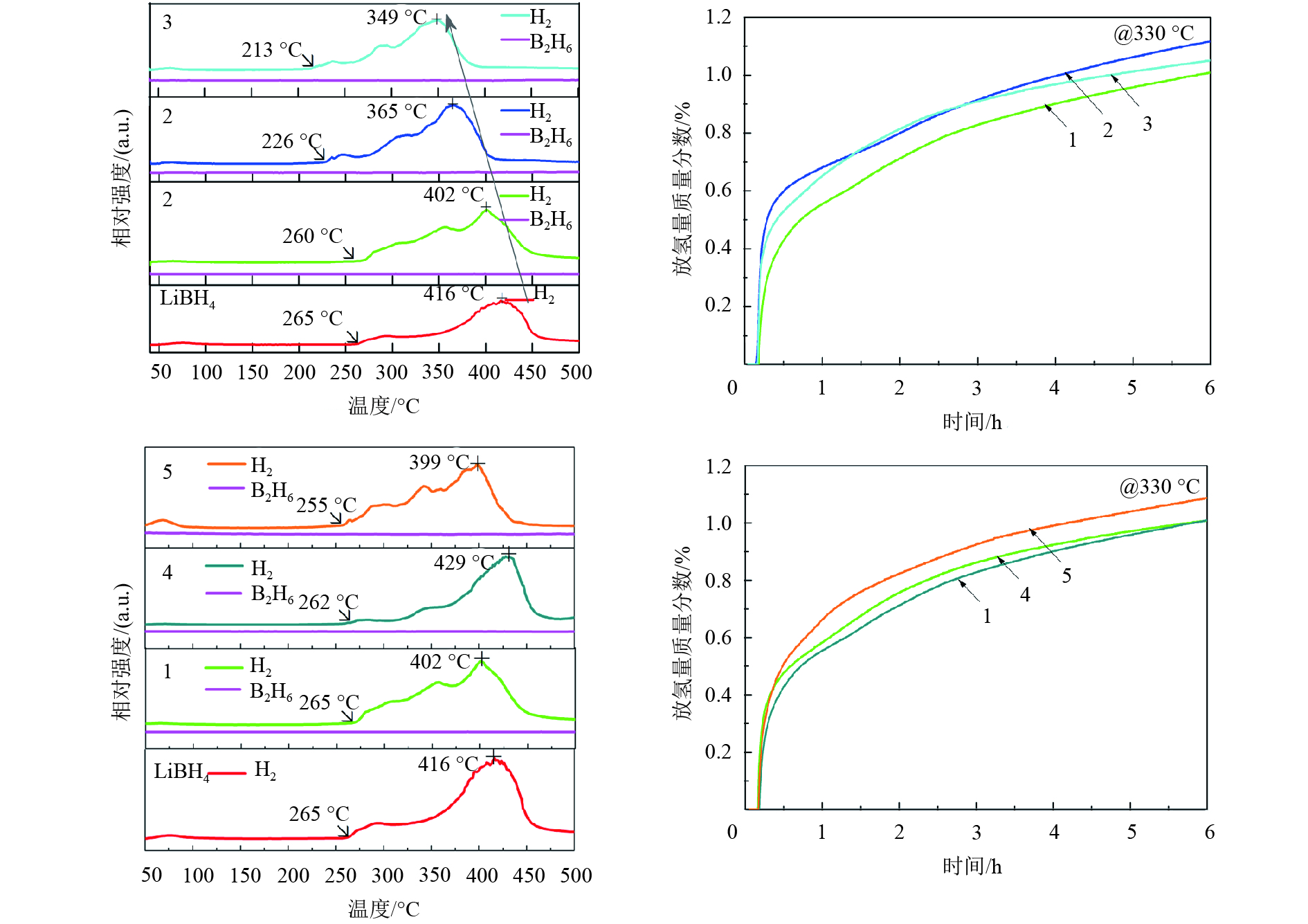
|
图 1 不同制备工艺条件下的LiBH4-NdCl3体系的变温放氢和等温放氢动力学曲线(其中NdCl3未预球磨) Figure 1 TPD-MS and isothermal dehydrogenation kinetics of LiBH4 doped with un-milled NdCl3 |
图1是掺杂未预球磨NdCl3的LiBH4-NdCl3体系在不同制备工艺条件下的变温放氢和等温放氢动力学特性曲线. 由图1可知,体系释放纯H2,并没有B2H6杂质气体的产生. 对于变温放氢性能而言,随着球磨时间的延长,LiBH4的初始放氢温度由265 ℃逐步降低至213 ℃,放氢峰值温度由416 ℃降至349 ℃. 这表明在低于LiBH4熔点(265 ℃)时,NdCl3能够促进LiBH4在固态下的放氢过程,同时有效地降低峰值温度[37]. 然而,随着球粉比的增大,LiBH4的初始放氢温度和峰值温度并没有得到明显的改善. 对于在330 ℃下的等温放氢动力学性能,当球磨时间由1 h增加至3 h时,可以有效提高LiBH4的放氢动力学性能;当球磨时间为5 h时,放氢动力学反而有所降低. 这应该与放氢过程中NdCl3与LiBH4的接触状态变化有关. 随着球粉比的增大,LiBH4的放氢动力学逐渐提高.
图2是掺杂预球磨NdCl3的LiBH4-NdCl3体系在不同制备工艺条件下的变温放氢和等温放氢动力学特性曲线. 由图2可知,球磨1 h和3 h的样品表现出最低的初始放氢温度和峰值温度,然而球磨5 h样品的放氢行为和纯LiBH4的相近. 当球粉比为40∶1时,样品具有最低的初始放氢温度和峰值温度,分别为172 ℃和311 ℃;当球粉比为120∶1时,样品的放氢行为与纯LiBH4的相似. 当调控NdCl3掺杂量时,发现掺杂质量分数5%和10% NdCl3的样品表现出相似的放氢行为;掺杂质量分数15%NdCl3的样品具有最低的初始放氢温度和峰值温度,分别为174 ℃和265 ℃,并且在330 ℃下的放氢量质量分数达到2.4%;掺杂质量分数20%NdCl3的样品则表现出多个放氢峰值温度. 增加掺杂量能够改善放氢性能主要得益于NdCl3与LiBH4的接触面积增加的结果. 由此可见,与掺杂未预球磨NdCl3的样品相比,掺杂预球磨NdCl3的样品具有更佳的放氢性能.
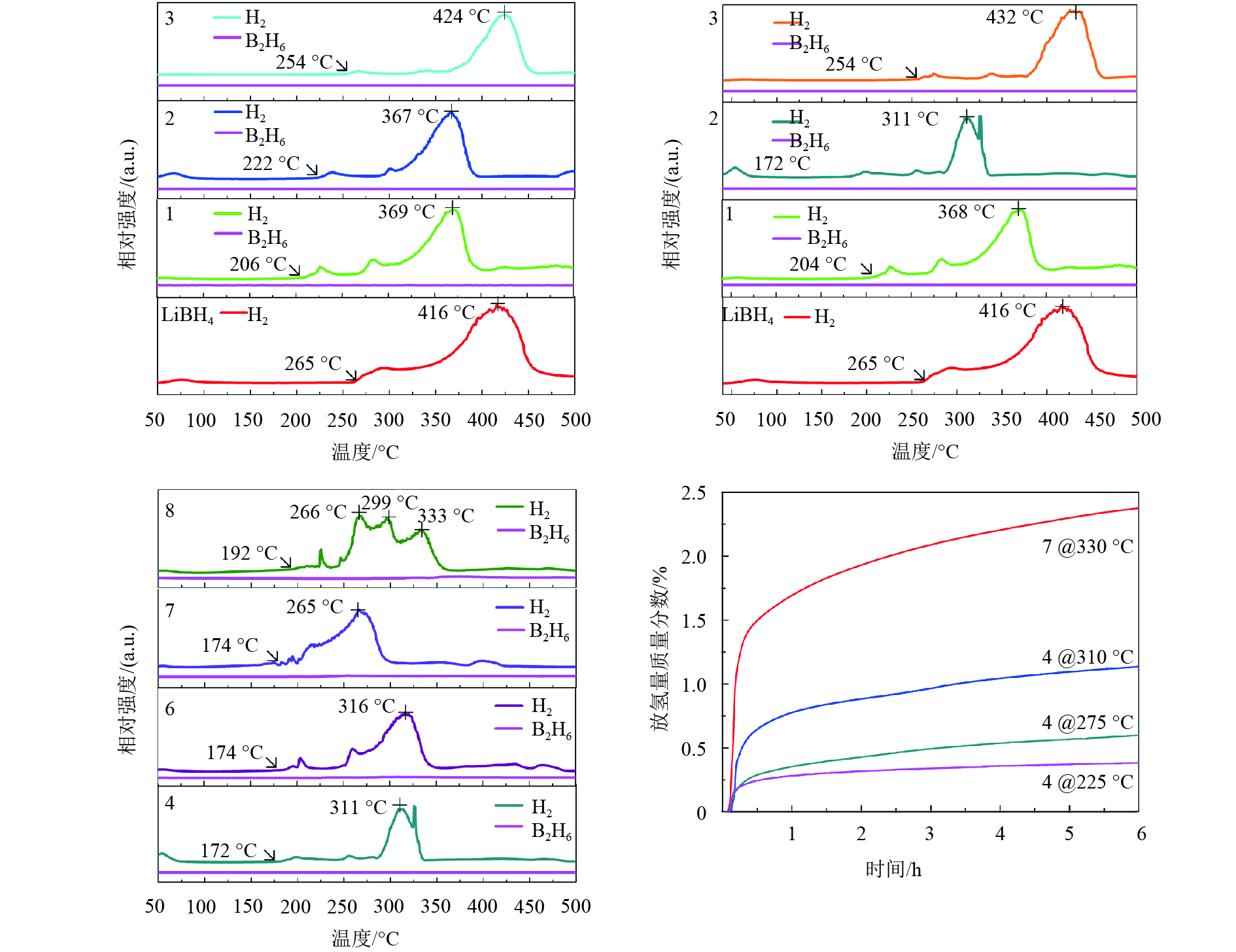
|
图 2 不同制备工艺条件下的LiBH4-NdCl3体系的变温放氢和等温放氢动力学曲线(其中NdCl3进行预球磨) Figure 2 TPD-MS and isothermal dehydrogenation kinetics of LiBH4 doped with pre-milled NdCl3 |
图3是掺杂未预球磨NdCl3的LiBH4-NdCl3体系在不同制备工艺条件下的球磨样品和等温放氢样品的XRD图谱. 由图3(a)可知,在球磨样品的XRD图谱中只能检测到LiBH4衍射峰和微弱的NdCl3衍射峰,其余未标识的微弱衍射峰是由于LiBH4样品中存在杂质导致的. 这表明LiBH4-NdCl3体系是LiBH4和NdCl3纳米晶的混合物. 由图3(b)可知,在等温放氢样品中只能检测到LiBH4的衍射峰和NdB6物相的微弱衍射峰.
图4是掺杂预球磨NdCl3的LiBH4-NdCl3体系在不同制备工艺条件下的球磨样品和等温放氢样品的XRD图谱. 由图4(a)可知,在球磨样品中只能检测到LiBH4的衍射峰. 由于预球磨后的NdCl3衍射峰已经极其微弱(此处未给出数据),因此可以推断球磨后的LiBH4-NdCl3体系是由LiBH4和NdCl3非晶组成的. 由图4(b)可知,在等温放氢样品中,能够检测到LiBH4的衍射峰,这表明在放氢产物中存在未分解的LiBH4. 这与等温放氢动力学的结果是一致的. 此外,还检测到NdB6和LiH物相的微弱衍射峰. 因此,LiBH4与NdCl3之间发生了去稳定化反应,从而改善了LiBH4的放氢性能,其反应方程式可能为:
12LiBH4+2NdCl3→2NdB6+6LiH+6LiCl+21H2,
其中,LiCl物相可能处于非晶态,导致在XRD图谱中无法检测到其衍射峰.
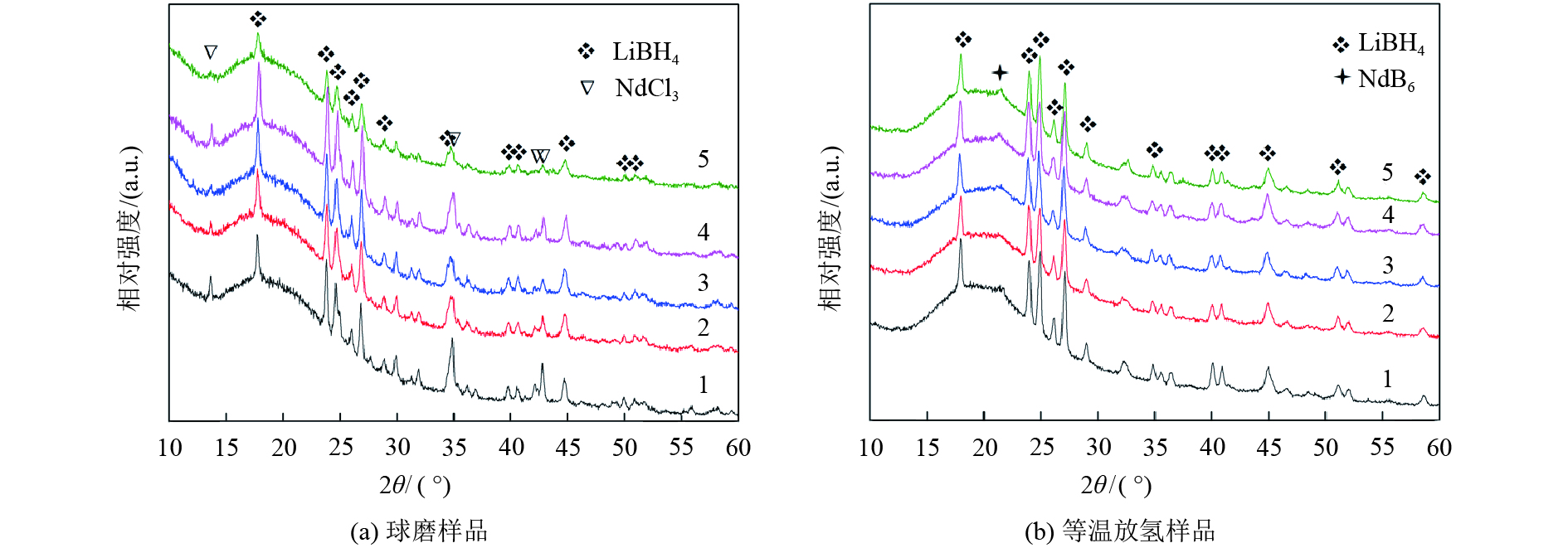
|
图 3 不同制备工艺条件下的LiBH4-NdCl3体系的XRD图谱(其中NdCl3未预球磨) Figure 3 XRD patterns of LiBH4 doped with un-milled NdCl3 |
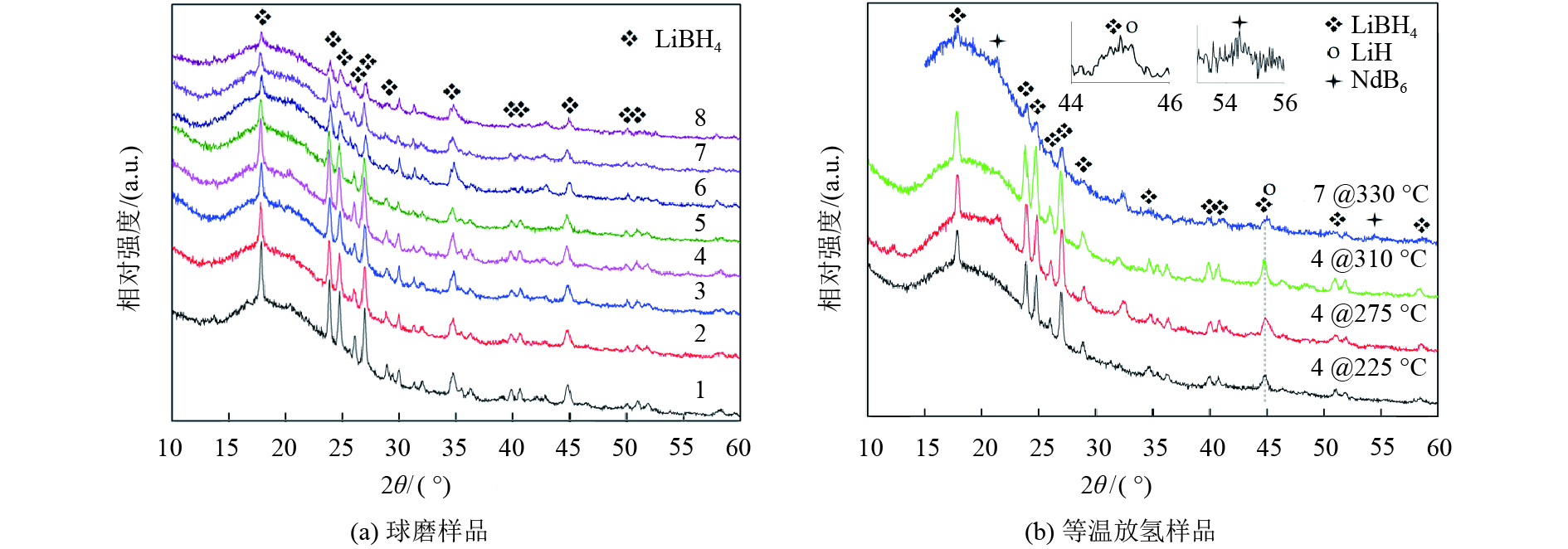
|
图 4 不同制备工艺条件下的LiBH4-NdCl3体系的XRD图谱(其中NdCl3进行预球磨) Figure 4 XRD patterns of LiBH4 doped with pre-milled NdCl3 |

|
图 5 不同制备工艺条件下的LiBH4-NdCl3体系的球磨样品SEM形貌图(其中NdCl3未预球磨) Figure 5 SEM analysis of LiBH4 doped with un-milled NdCl3 after ball milling |

|
图 6 不同制备工艺条件下的LiBH4-NdCl3体系的等温放氢样品SEM形貌图(其中NdCl3未预球磨) Figure 6 SEM analysis of LiBH4 doped with un-milled NdCl3 after isothermal dehydrogenation |

|
图 7 不同制备工艺条件下的LiBH4-NdCl3体系的球磨样品SEM形貌图(其中NdCl3进行预球磨) Figure 7 SEM analysis of LiBH4 doped with pre-milled NdCl3 after ball milling |
图5和图6分别是掺杂未预球磨NdCl3的LiBH4-NdCl3体系在不同制备工艺条件下的球磨样品和等温放氢样品的SEM形貌图. 由图5可知,NdCl3纳米颗粒弥散地分布在LiBH4基体上. 延长球磨时间或增大球粉比,可以进一步细化NdCl3颗粒,增大LiBH4与NdCl3的接触面积,进而提高LiBH4的放氢动力学性能. 然而,实验发现只有延长球磨时间才能有效降低LiBH4放氢温度,而增大球粉比无法进一步降低放氢温度,如图1所示. 本文认为这与LiBH4-NdCl3体系的热力学状态有关. 相对于增大球粉比,延长球磨时间能够提供更多的额外能量,促进去稳定化反应的进行. 由图6可知,灰色基体主要是未分解的LiBH4;白色近圆形纳米颗粒NdB4成聚集状态,并且是由大量纳米晶组成. 这与XRD图谱中NdB4物相衍射峰峰强低的结果相符.
图7是掺杂预球磨NdCl3的LiBH4-NdCl3体系在不同制备工艺条件下的球磨样品的SEM形貌图. 由图7可知,与掺杂未预球磨NdCl3的样品相似,延长球磨时间和增大球粉比能够进一步细化NdCl3和LiBH4颗粒,并且NdCl3纳米颗粒均匀弥散分布在LiBH4颗粒上,从而能够改善LiBH4的放氢性能. 但是,由于预球磨后的NdCl3具有更高的表面能,球磨时间过长或者球粉比过大能够促进LiBH4和NdCl3发生去稳定化反应,从而导致NdCl3失效,如图2所示.
3 结论本文采用高能球磨方法制备LiBH4-NdCl3储氢体系,研究了LiBH4-NdCl3体系的放氢机制,并考察了预球磨、球磨时间、球粉比和掺杂量等工艺参数对体系的影响规律. 研究发现,NdCl3对LiBH4放氢性能的改善作用是通过去稳定化反应进行的. 要有效地改善LiBH4的放氢性能,首先需要使NdCl3纳米化,为去稳定化反应提供额外的表面能;其次是合适的球磨时间和球粉比需要考虑NdCl3的原始状态,NdCl3是否预球磨与体系的最佳球磨时间和球粉比是相关的;再次,增大LiBH4与NdCl3的接触面积是改善LiBH4放氢性能的有效方法,这可以通过增加NdCl3掺杂量来实现.
| [1] | HE T, PACHFULE P, WU H. Hydrogen carriers[J]. Nature Reviews Materials, 2016, 1: 16059-16075. DOI: 10.1038/natrevmats.2016.59. |
| [2] |
陈军, 朱敏. 高容量储氢材料的研究进展[J].
中国材料进展, 2009, 28(5): 2-10.
CHEN J, ZHU M. Progress in research of hydrogen storage materials with high capacity[J]. Materials China, 2009, 28(5): 2-10. |
| [3] | KLEBANOFF L E, KELLER J O. 5 Years of hydrogen storage research in the U.S. DOE metal hydride center of excellence (MHCoE)[J]. International Journal of Hydrogen Energy, 2013, 38(11): 4533-4576. DOI: 10.1016/j.ijhydene.2013.01.051. |
| [4] | WANG H, LIN H J, CAI W T, et al. Tuning kinetics and thermodynamics of hydrogen storage in light metal element based systems – A review of recent progress[J]. Journal of Alloys and Compounds, 2016, 658: 280-300. DOI: 10.1016/j.jallcom.2015.10.090. |
| [5] | LU Z H, XU Q. Recent progress in boron and nitrogen based chemical hydrogen storage[J]. Functional Materials letters, 2012, 5(1): 1230001-9. DOI: 10.1142/S1793604712300010. |
| [6] |
李爱菊, 张仁元, 周晓霞. 化学储能材料开发与应用[J].
广东工业大学学报, 2002, 19(1): 81-84.
LI A J, ZHANG R Y, ZHOU X X. Progress of research on chemical energy storage materials[J]. Journal of Guangdong University of Technology, 2002, 19(1): 81-84. |
| [7] | MAURON P, BUCHTER F, FRIEDRICHS O, REMHOF A, et al. Stability and reversibility of LiBH4[J]. The Journal of Physical Chemistry B, 2007, 112(3): 906-910. |
| [8] |
孙立贤, 宋莉芳, 姜春红, 等. 新型储氢材料及其热力学与动力学问题[J].
中国科学:化学, 2010, 40(09): 1243-1252.
SUN L X, SONG L F, JIANG C H. Thermodynamics and kinetics of new hydrogen storage materials[J]. SCIENTIA SINICA Chemica, 2010, 40(09): 1243-1252. |
| [9] | SONG L F, JIANG C H, LIU S S. Progress in improving thermodynamics and kinetics of new hydrogen storage materials[J]. Frontiers of Physics, 2011, 6(2): 151-161. DOI: 10.1007/s11467-011-0175-2. |
| [10] | RUDE LH, NIELSEN T K, RAVNSBAK D B, et al. Tailoring properties of borohydrides for hydrogen storage: A review[J]. physica status solidi (a), 2011, 208(8): 1754-1773. DOI: 10.1002/pssa.v208.8. |
| [11] | VAJO J J, SKEITH S L, MERTENS F. Reversible storage of hydrogen in destabilized LiBH4[J]. The Journal of Physical Chemistry B, 2005, 109(9): 3719-3722. DOI: 10.1021/jp040769o. |
| [12] | VAJO J J, SKEITH S L, MERTENS F, et al. Hydrogen-generating solid-state hydride/hydroxide reactions[J]. Journal of Alloys and Compounds, 2005, 390(1-2): 55-61. DOI: 10.1016/j.jallcom.2004.08.042. |
| [13] | YANG J, SUDIK A, WOLVERTON C. Destabilizing LiBH4 with a metal (M = Mg, Al, Ti, V, Cr, or Sc) or metal hydride (MH2, MgH2, TiH2, or CaH2) [J]. Journal of Physical Chemistry C, 2007, 111(51): 19134-19140. DOI: 10.1021/jp076434z. |
| [14] | AU M, JURGENSEN A, ZEIGLER K. Modified lithium borohydrides for reversible hydrogen storage (2)[J]. The Journal of Physical Chemistry B, 2006, 110(51): 26482-26487. DOI: 10.1021/jp065490h. |
| [15] | AU M, WALTERS R T. Reversibility aspect of lithium borohydrides[J]. International Journal of Hydrogen Energy, 2010, 35(19): 10311-10316. DOI: 10.1016/j.ijhydene.2010.07.130. |
| [16] | LI Y, LI P, QU X H. Investigation on LiBH4-CaH2 composite and its potential for thermal energy storage [J]. Scientific Reports, 2017, 7: 1-8. DOI: 10.1038/s41598-016-0028-x. |
| [17] | LIU H Z, WANG X H, ZHOU H. Improved hydrogen desorption properties of LiBH4 by AlH3 addition [J]. International Journal Of Hydrogen Energy, 2016, 41(47): 22118-22127. DOI: 10.1016/j.ijhydene.2016.09.177. |
| [18] | LIU D M, HUANG W J, SI T Z, et al. Hydrogen storage properties of LiBH4 destabilized by SrH2[J]. Journal of Alloys and Compounds, 2013, 551: 8-11. DOI: 10.1016/j.jallcom.2012.09.138. |
| [19] | CAI W T, WANG H, SUN D L, et al. Nanosize-controlled reversibility for a destabilizing reaction in the LiBH4–NdH2+xsystem [J]. The Journal of Physical Chemistry C, 2013, 117(19): 9566-9572. DOI: 10.1021/jp402332q. |
| [20] | SHIM J H, LEE Y S, SUH J Y. Thermodynamics of the dehydrogenation of the LiBH4–YH3 composite: Experimental and theoretical studies [J]. Journal of Alloys and Compounds, 2012, 510(1): L9-L12. DOI: 10.1016/j.jallcom.2011.09.005. |
| [21] | FABIANA C G. Destabilization of LiBH4 by MH2 (M=Ce, La) for hydrogen storage: Nanostructural effects on the hydrogen sorption kinetics [J]. International Journal of Hydrogen Energy, 2011, 36(23): 15231-15238. DOI: 10.1016/j.ijhydene.2011.08.059. |
| [22] | XIA G L, TAN Y B, WU F L, et al. Graphene-wrapped reversible reaction for advanced hydrogen storage[J]. Nano Energy, 2016, 26: 488-495. DOI: 10.1016/j.nanoen.2016.06.016. |
| [23] | LI Y T, ZHANG Q A, FANG F. Facile self-assembly of light metal borohydrides with controllable nanostructures[J]. RSC Advances, 2014, 4(2): 983-986. DOI: 10.1039/C3RA45867A. |
| [24] | SUN T, LIU J W, JIA Y. Confined LiBH4: Enabling fast hydrogen release at 100 ℃ [J]. International Journal of Hydrogen Energy, 2012, 37(24): 18920-18926. DOI: 10.1016/j.ijhydene.2012.09.119. |
| [25] | NIELSEN T K, BESENBACHER F, JENSEN T R. Nanoconfined hydrides for energy storage[J]. Nanoscale, 2011, 3(5): 2086-2098. DOI: 10.1039/c0nr00725k. |
| [26] | NIELSEN T K, BOSENBERG U, GOSALAWIT R, et al. A reversible nanoconfined chemical reaction[J]. ACS Nano, 2010, 4(7): 3903-3908. DOI: 10.1021/nn1006946. |
| [27] | GROSS A F, VAJO J J, VAN ATTA S L. Enhanced hydrogen storage kinetics of LiBH4 in nanoporous carbon scaffolds [J]. The Journal of Physical Chemistry C, 2008, 112(14): 5651-5657. DOI: 10.1021/jp711066t. |
| [28] | LIU X F, PEASLEE D, JOST C Z. Systematic pore-size effects of nanoconfinement of LiBH4: elimination of diborane release and tunable behavior for hydrogen storage applications [J]. Chemistry of Materials, 2011, 23(5): 1331-1336. DOI: 10.1021/cm103546g. |
| [29] | LIU X F, PEASLEE D, JOST C Z, et al. Controlling the decomposition pathway of LiBH4 via confinement in highly ordered nanoporous carbon [J]. Journal of Physical Chemistry C, 2010, 114(33): 14036-14041. DOI: 10.1021/jp1055045. |
| [30] | MIWA K, OHBA N, TOWATA S I. First-principles study on lithium borohydride LiBH4[J]. Physical Review B, 2004, 69(24): 245120. DOI: 10.1103/PhysRevB.69.245120. |
| [31] | NICKELS E A, JONES M O, DAVID W I F, et al. Tuning the decomposition temperature in complex hydrides: synthesis of a mixed alkali metal borohydride[J]. Angewandte Chemie International Edition, 2008, 47(15): 2817-2819. DOI: 10.1002/(ISSN)1521-3773. |
| [32] | LI H W, ORIMO S, NAKAMORI Y, et al. Materials designing of metal borohydrides: Viewpoints from thermodynamical stabilities[J]. Journal of Alloys and Compounds, 2007, 446-447: 315-318. DOI: 10.1016/j.jallcom.2007.02.156. |
| [33] | LINDEMANN I, DOMENECH FEFFER R, DUNSCH L, et al. Al3Li4(BH4)13: acomplex double-cation borohydride with a new structure [J]. Chemistry–A European Journal, 2010, 16(29): 8707-8712. DOI: 10.1002/chem.201000831. |
| [34] | YIN L C, WANG P, FANG Z Z, et al. Thermodynamically tuning LiBH4 by fluorine anion doping for hydrogen storage: A density functional study [J]. Chemical Physics Letters, 2008, 450(4-6): 318-321. DOI: 10.1016/j.cplett.2007.11.060. |
| [35] | MOSEGAARD L, MOLLER B, JORGENSEN J E, et al. Reactivity of LiBH4: In situ synchrotron radiation powder X-ray diffraction study [J]. The Journal of Physical Chemistry C, 2008, 112(4): 1299-1303. DOI: 10.1021/jp076999v. |
| [36] | RUDE L H, ZAVOROTYNSKA O, ARNBJERG L M, et al. Bromide substitution in lithium borohydride, LiBH4-LiBr [J]. International Journal of Hydrogen Energy, 2011, 36(24): 15664-15672. DOI: 10.1016/j.ijhydene.2011.08.087. |
| [37] | ZUTTEL A, WENGER P, RENTSCH S, et al. LiBH4 a new hydrogen storage material [J]. Journal of Power Sources, 2003, 118(1-2): 1-7. DOI: 10.1016/S0378-7753(03)00054-5. |
 2018, Vol. 35
2018, Vol. 35