b Renewable Energy Conversion and Storage Center, Nankai University, Tianjin 300071, China
The increasing demand for energy has led to the consumption of large quantities of fossil fuels, which in turn causes increased anthropogenic CO2 emission. This excess production of CO2 is credited with causing adverse impact on climate such as global warming [1]. Therefore, reduction of CO2 concentration while maintaining fossil fuel energy supply at present is crucial from both technological and environmental perspectives. In contrast to CO2 storage, transformation of CO2 to value-added chemicals is more attractive since it can offer higher efficiency of energy conversion and storage [2]. Among various reactions involving CO2, the synthesis of carboxylic acids and their derivatives attracts much attention because high value-added carboxylic acids are important products or intermediates in medical chemistry (e.g., aspirin) and organic synthesis [3-9]. The global market value associated with carboxylic acids will possibly increase to €16 billion by 2024 [10]. Although there are diverse protocols for the preparation of carboxylic acids such as carbonylation of organic halides with toxic carbon monoxide, the one-step carboxylation of carbon nucleophiles using abundant and inexpensive CO2 as C1 electrophile is the simplest and the most straightforward method. Although the efficient carboxylation of C–H bond with CO2 under green and mild conditions have been achieved [11-15], limited by the thermodynamic stability and kinetic inertness of CO2, the carboxylation of terminal alkynes with CO2 were usually performed at elevated temperatures or under high CO2 pressures which cause additional environmental issues [16-18]. Therefore, the development of efficient catalysts that can catalyze the reaction under mild condition is highly desirable.
The utilization of solar energy as a renewable energy source can not only meet the increasing global energy demand but also offer a solution to the growing environmental concerns of greenhouse gases. By making use of the photothermal effect, light source can be employed to replace conventional heating method to drive the reaction, realizing the storage and transformation of solar energy with high efficiency [19, 20]. Ag nanoparticles (NPs) are promising photothermal materials because they have strong surface plasmon resonance (SPR) effects that can induce temperature increase of the surrounding environment under light irradiation [21, 22]. Ag NPs are also active for the conversion of CO2 [18]. However, they tend to aggregate during catalytic process and are difficult to recover to small nanoparticles and hence require porous materials as matrix to keep their uniform dispersion. As a kind of porous crystalline materials with high surface areas, metal-organic frameworks (MOFs) cannot only stabilize surface-clean NPs by spatial confinement, but also provide abundant catalytically active sites for CO2 conversion [23-29]. Moreover, MOFs were found to exhibit photothermal effects [30-32]. To date, metal NPs-MOF composites have been exploited in photothermal therapy and catalytic reactions such as hydrogenation of olefins [33-36].
In photothermal CO2-involved catalytic reactions, on one hand, photothermal hydrogenation of CO2 to obtain different chemicals like CO, HCOOH, CH3OH or CH4 have been achieved using transition metal-based catalysts [37-42]. On the other hand, it is still of great significance to develop other strategies for photothermal CO2 conversion, such as photothermal CO2-involved organic synthesis. Herein, a series of Ag NPs/MIL-100(Fe) with different Ag loadings were synthesized and their catalytic performance toward carboxylation of terminal alkynes with 1 atm CO2 under visible light irradiation at room temperature were evaluated. Owing to the synergistic effect of Ag NPs and MIL-100(Fe), their catalytic performance without direct heating was comparable to that heated at 50 ℃.
MIL-100(Fe), a well-known MOF with high stability and porosity, was chosen as not only the support for Ag NPs but also the catalyst with abundant Lewis acid active sites [43, 44]. An impregnation-reduction method was introduced to load Ag NPs on MIL-100(Fe) (Scheme 1). MIL-100(Fe) was impregnated in the acetonitrile solution of AgNO3 to adsorb Ag ions on its surface. The solvent was totally removed by vacuumizing, and the residue with adsorbed Ag ions was reduced by NaBH4 to obtain Ag/MIL-100(Fe). The concentration of AgNO3 was varied to prepare Ag/MIL-100(Fe) with different Ag loadings: 5 wt%, 10 wt%, 15 wt% and 20 wt%, which were denoted herein as 5Ag/MIL-100(Fe), 10Ag/MIL-100(Fe), 15Ag/MIL-100(Fe) and 20Ag/MIL-100(Fe) respectively. The actual Ag loadings were determined by inductively coupled plasma atomic emission spectroscopy (ICP-AES) as shown in Table S1 in Supporting information, which were close to the expected loadings. The relatively low Ag loading of 16.4 wt% in 20Ag/MIL-100(Fe) was due to the loss of larger Ag NPs that could not be immobilized stably by MIL-100(Fe) during washing step.
|
Download:
|
| Scheme 1. Synthetic route to Ag/MIL-100(Fe) and photothermal conversion of CO2. | |
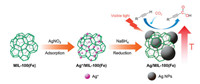
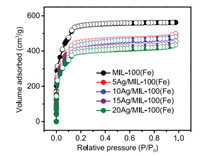
Powder X-ray diffraction (PXRD) patterns of the prepared composites confirmed their high crystallinity (Fig. 1). Weak diffraction peaks at 38.12° were attributed to Ag NPs (JCPDS No. 04-0783) and their intensities increased with increasing Ag loadings. The Brunauer-Emmett-Teller (BET) surface areas of these composites were analyzed by N2 adsorption-desorption isotherms at 77 K (Fig. 2). Although the BET surface areas decreased slightly after loading Ag NPs (Table S1), the potential catalytically active sites of MIL-100(Fe) were still accessible to the guests.

|
Download:
|
| Fig. 1. PXRD patterns of MIL-100(Fe) and Ag/MIL-100(Fe) with different Ag loadings. | |
|
Download:
|
| Fig. 2. N2 adsorption-desorption isotherms (77 K) of MIL-100(Fe) and Ag/MIL-100(Fe) with different Ag loadings. | |
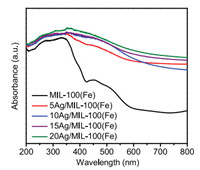
UV–vis absorption spectra of MIL-100(Fe) and Ag/MIL-100(Fe) were measured to investigate their light absorption properties. MIL-100(Fe) exhibited absorption centred at 320 and 445 nm (Fig. 3) due to ligand-to-metal charge transfer and d-d transition of metal nodes [45-48]. After loading Ag NPs, enhanced absorption of Ag/MIL-100(Fe) in the visible region was originated from SPR effects of Ag NPs [45, 49-51], which indicated high harvesting efficiency of light energy by Ag/MIL-100(Fe). To investigate the photothermal effects of Ag/MIL-100(Fe), the temperature increases of the solutions caused by MIL-100(Fe) and Ag/MIL-100(Fe) under visible light irradiation (accounts for 43% of solar energy) after 0.5 h were measured (Table S2 in Supporting information). For pristine MIL-100(Fe), the temperature increased by 17 ℃. After loading Ag NPs on MIL-100(Fe), higher temperature increases of 18−26 ℃ were observed, which were consistent with their light absorption properties. The results also revealed that both Ag NPs and MIL-100(Fe) contributed to increased temperature of the solution. For comparison, when activated carbon (AC) was used as support, 15Ag/AC showed more prominent photothermal effect with a temperature increase of 27 ℃ due to the broad absorption of both Ag NPs and AC in visible region (Fig. S1 in Supporting information).
|
Download:
|
| Fig. 3. UV–vis absorption spectra of MIL-100(Fe) and Ag/MIL-100(Fe) with different Ag loadings. | |
Transmission electron microscopy (TEM) images showed that Ag NPs with diameters of 5−7 nm were uniformly distributed on MIL-100(Fe) (Fig. 4a and Fig. S2 in Supporting information). Ag NPs with diameters > 9 nm were also formed when Ag loading was increased to 15 wt%. The diameters of most Ag NPs were larger than the pore sizes (2.5 and 2.9 nm) of MIL-100(Fe), indicating that Ag NPs were located on the surface and/or encapsulated in the framework of MIL-100(Fe) which could prevent the growth of Ag NPs [52]. Scanning electron microscopy (SEM) images and energy-dispersive X-ray spectroscopy (EDS) elemental mapping results also confirmed the uniform distributions of Ag NPs on MIL-100(Fe) (Figs. 4b-e and Figs. S3-S5 in Supporting information). For comparison, only large Ag NPs (> 15 nm) were obtained for 15Ag/AC (Fig. S6 in Supporting information).

|
Download:
|
| Fig. 4. (a) TEM image, (b-e) SEM image and corresponding elemental mappings of 15Ag/MIL-100(Fe). Inset in (a) was size distribution of Ag NPs. | |
Encouraged by the photothermal effect of Ag/MIL-100(Fe), their catalytic activities toward photothermal carboxylation of terminal alkynes with CO2 were evaluated. 1-ethynylbenzene was employed as model substrate. The reactions were performed under 1 atm CO2 and visible light irradiation (420 nm < λ < 780 nm) at room temperature for 12 h and the corresponding yields of 3-phenylpropiolic acid were presented in Table 1. MIL-100(Fe) afforded the yield of 47% due to its accessible Lewis acid active sites [53]. When Ag NPs were loaded, Ag/MIL-100(Fe) exhibited better catalytic performance than MIL-100(Fe), proving that the SPR effect of Ag NPs was able to promote the carboxylation reaction. The yields increased from 69% to 92% as the Ag loadings increased from 5 wt% to 15 wt%. 3-Phenylpropiolic acid was formed with the highest yield of 92% for optimal 15Ag/MIL-100(Fe). However, a decreased yield of 83% could be observed when further increasing Ag loading to 20 wt% because large Ag NPs exhibited inferior catalytic activities than small Ag NPs. There were works of the carboxylation of terminal alkynes with CO2 using various catalysts including Ag/MIL-100(Fe) with good yields (> 90%) obtained (Table S3 in Supporting information). However, most of them were performed under heating conditions. Although several reactions could proceed at room temperature, prolonged reaction times were required. In this work, the yield of above 90% could be achieved without heating in less reaction time (12 h). The possible reaction mechanism had been proposed (Fig. S7 in Supporting information) [18]. The alkyne was activated by Ag NPs and Lewis acid active sites in MIL-100(Fe) and deprotonated by Cs2CO3 to form the metal acetylide. Then the insertion of CO2 into the carbon-metal bonds afforded metal propiolate intermediate. Finally, metal propiolate intermediate reacted with another terminal alkyne and Cs2CO3 to form cesium propiolate and regenerated metal acetylide for next cycle. The carboxylic acid product could be obtained by acidification of cesium propiolate.
|
|
Table 1 Synthesis of 3-phenylpropiolic acid from CO2 and 1-ethynylbenzene.a |
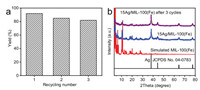
To further demonstrate the synergistic effect of Ag NPs and MIL-100(Fe) on enhanced catalytic performance, the reaction was carried out with 15Ag/AC as catalyst. Although 15Ag/AC showed strong photothermal effect, the diameters of Ag NPs in 15Ag/AC were much larger than those in Ag/MIL-100(Fe) and the AC structure did not contain catalytically active sites. As a result, the yield of 3-phenylpropiolic acid for 15Ag/AC was only 39%. This result revealed that not only the photothermal effects but also the catalytic activities of both Ag NPs and MIL-100(Fe) were important in improving catalytic efficiency. For comparison, the reaction was also performed under the same condition with light irradiation replaced by heating (50 ℃). The corresponding yield of 91% was similar to that achieved under visible light irradiation (Table 1, entry 8). This result confirmed that the carboxylation of 1-ethynylbenzene with 1 atm CO2 could be achieved effectively using visible light irradiation as an alternative to energy-consuming heating when 15Ag/MIL-100(Fe) served as the catalyst. The reactions under room temperature (without light) and simulated sunlight by using AM1.5 filter were also performed and corresponding yields were 54% and 75% (Table 1, entries 9 and 10). The results showed that although the reaction could proceed at room temperature, an elevated temperature was required to achieve high product yield. In addition, the lower light intensity (100 mW/cm2) of simulated sunlight compared with that (200 mW/cm2) of pre-set visible light led to lower temperature increase (16 ℃), and thus gave inferior performance. Furthermore, the carboxylation of other aromatic alkynes with electron-withdrawing (Cl) or electron-donating (CH3, OCH3) moieties were also investigated (Table 2). The reaction of electron-withdrawing moiety substituted aromatic alkyne exhibited 90% yield and the aromatic alkynes with electron-donating moieties could be converted to corresponding products with the yields of 87%–88%. 86% yield could also be achieved for alkyne with a heterocyclic group. Moreover, the recycling test showed that the yield could be maintained above 80% after three cycles (Fig. 5a). The decreased yield was probably due to the partial decomposition of MIL-100(Fe) in alkaline solution which could be confirmed by decreased peak intensities of MIL-100(Fe) in PXRD pattern (Fig. 5b).
|
|
Table 2 Synthesis of propiolic acid derivatives from CO2 and alkynes.a |
|
Download:
|
| Fig. 5. (a) Catalytic recycling test of carboxylation of 1-ethynylbenzene with CO2 using 15Ag/MIL-100(Fe) catalyst. (b) PXRD pattern of 15Ag/MIL-100(Fe) after 3 cycles. | |
In summary, Ag/MIL-100(Fe) composites were prepared using MIL-100(Fe) as support through a facile solution impregnation-reduction method. The framework structure of MIL-100(Fe) stabilized Ag NPs and prevented their aggregation during the catalytic process. Both MIL-100(Fe) and Ag NPs exhibited photothermal effects and catalytic activities for carboxylation of terminal alkynes with CO2. Remarkably, benefitting from the synergistic effects of Ag NPs and MIL-100(Fe), Ag/MIL-100(Fe) catalyst could efficiently promote carboxylation of terminal alkynes with 1 atm CO2 under visible light irradiation at room temperature and the yield was comparable to that obtained by heating. This work demonstrated an efficient strategy for catalyzing photothermal organic reaction with CO2, and provided a promising approach of utilizing solar energy to drive CO2 conversion in the future.
Declaration of competing interestThe authors report no declarations of interest.
AcknowledgmentsThis work was supported by the Natural Science Foundation of Tianjin (No. 18JCJQJC47200), the Ministry of Education of China (No. B12015) and the Fundamental Research Funds for the Central Universities, Nankai University (Nos. 63201016 and 63201043).
Appendix A. Supplementary dataSupplementary material related to this article can befound, in the online version, at doi:https://doi.org/10.1016/j.cclet.2021.04.007.
| [1] |
A. Goeppert, M. Czaun, J.P. Jones, G.K. Surya Prakash, G.A. Olah, Chem. Soc. Rev. 43 (2014) 7995-8048. DOI:10.1039/C4CS00122B |
| [2] |
D.P. Schrag, Science 315 (2007) 812-813. DOI:10.1126/science.1137632 |
| [3] |
E.V. Kondratenko, G. Mul, J. Baltrusaitis, G.O. Larrazábal, J. Pérez-Ramírez, Energy Environ. Sci. 6 (2013) 3112-3135. DOI:10.1039/c3ee41272e |
| [4] |
X.B. Lu, D.J. Darensbourg, Chem. Soc. Rev. 41 (2012) 1462-1484. DOI:10.1039/C1CS15142H |
| [5] |
Q. Liu, L. Wu, R. Jackstell, M. Beller, Nat. Commun. 6 (2015) 5933. DOI:10.1038/ncomms6933 |
| [6] |
J. Liu, C. Chen, K. Zhang, L. Zhang, Chin. Chem. Lett. 32 (2021) 649-659. DOI:10.1016/j.cclet.2020.07.040 |
| [7] |
M. Aresta, A. Dibenedetto, A. Angelini, Chem. Rev. 114 (2014) 1709-1742. DOI:10.1021/cr4002758 |
| [8] |
Q.W. Song, Z.H. Zhou, L.N. He, Green Chem. 19 (2017) 3707-3728. DOI:10.1039/C7GC00199A |
| [9] |
D. Yu, S.P. Teong, Y. Zhang, Coord. Chem. Rev. 293-294 (2015) 279-291. DOI:10.1016/j.ccr.2014.09.002 |
| [10] |
R. Martin, A. Tortajada, F. Juliá-Hernández, M. Borjesson, T. Moragas, Angew. Chem. Int. Ed. 57 (2018) 15948-159820. DOI:10.1002/anie.201803186 |
| [11] |
I.I.F. Boogaerts, S.P. Nolan, J. Am. Chem. Soc. 132 (2010) 8858-8859. DOI:10.1021/ja103429q |
| [12] |
Q.Y. Meng, T.E. Schirmer, A.L. Berger, K. Donabauer, B. Konig, J. Am. Chem. Soc. 141 (2019) 11393-11397. DOI:10.1021/jacs.9b05360 |
| [13] |
C.S. Yeung, Angew. Chem. Int. Ed. 58 (2019) 5491-5502. DOI:10.1002/anie.201902939 |
| [14] |
M. Borjesson, D. Janssen-Muller, B. Sahoo, et al., J. Am. Chem. Soc. 142 (2020) 16234-16239. DOI:10.1021/jacs.0c08810 |
| [15] |
L. Song, D.M. Fu, L. Chen, et al., Angew. Chem. Int. Ed. 59 (2020) 21121-21128. DOI:10.1002/anie.202008630 |
| [16] |
B. Yu, J.N. Xie, C.L. Zhong, W. Li, L.N. He, ACS Catal. 5 (2015) 3940-3944. DOI:10.1021/acscatal.5b00764 |
| [17] |
P. Bhanja, A. Modak, A. Bhaumik, Chem. Eur. J. 24 (2018) 7278-7297. DOI:10.1002/chem.201800075 |
| [18] |
X.H. Liu, J.G. Ma, Z. Niu, G.M. Yang, P. Cheng, Angew. Chem. Int. Ed. 54 (2015) 988-991. DOI:10.1002/anie.201409103 |
| [19] |
S. Wu, Y. Li, Q. Zhang, et al., Adv. Energy Mater. 10 (2020) 2002602. DOI:10.1002/aenm.202002602 |
| [20] |
Y. Li, L. Wang, J. Low, et al., Chin. Chem. Lett. 31 (2020) 231-234. DOI:10.1016/j.cclet.2019.04.022 |
| [21] |
L.M. Liz-Marzan, C.J. Murphy, J. Wang, Chem. Soc. Rev. 43 (2014) 3820-3822. DOI:10.1039/c4cs90026j |
| [22] |
S. Linic, U. Aslam, C. Boerigter, M. Morabito, Nat. Mater. 14 (2015) 567-576. DOI:10.1038/nmat4281 |
| [23] |
J. Lee, O.K. Farha, J. Roberts, et al., Chem. Soc. Rev. 38 (2009) 1450-1459. DOI:10.1039/b807080f |
| [24] |
H.C. Zhou, J.R. Long, O.M. Yaghi, Chem. Rev. 112 (2012) 673-674. DOI:10.1021/cr300014x |
| [25] |
Q. Yang, Q. Xu, H.L. Jiang, Chem. Soc. Rev. 46 (2017) 4774-4808. DOI:10.1039/C6CS00724D |
| [26] |
R. Mahugo, A. Mayoral, M. Sánchez-Sánchez, I. Diaz, Front. Chem. 7 (2019) 686. DOI:10.3389/fchem.2019.00686 |
| [27] |
M. Ding, R.W. Flaig, H.L. Jiang, O.M. Yaghi, Chem. Soc. Rev. 48 (2019) 2783-2828. DOI:10.1039/c8cs00829a |
| [28] |
G. Cai, M. Ding, Q. Wu, H.L. Jiang, Natl. Sci. Rev. 7 (2019) 37-45. |
| [29] |
Y. Pan, Y. Qian, X. Zheng, et al., Natl. Sci. Rev. 8 (2021) nwaa224. DOI:10.1093/nsr/nwaa224 |
| [30] |
J. Espín, L. Garzón-Tovar, G. Boix, I. Imaz, D. Maspoch, Chem. Commun. 54 (2018) 4184-4187. DOI:10.1039/C8CC01593G |
| [31] |
J. Espín, L. Garzón-Tovar, A. Carné-Sánchez, I. Imaz, D. Maspoch, ACS Appl. Mater. Interfaces 10 (2018) 9555-9562. DOI:10.1021/acsami.8b00557 |
| [32] |
J.D. Xiao, H.L. Jiang, Acc. Chem. Res. 52 (2019) 356-366. DOI:10.1021/acs.accounts.8b00521 |
| [33] |
K. Khaletskaya, J. Reboul, M. Meilikhov, et al., J. Am. Chem. Soc. 135 (2013) 10998-11005. DOI:10.1021/ja403108x |
| [34] |
X. Liu, L. He, J. Zheng, et al., Adv. Mater. 27 (2015) 3273-3277. DOI:10.1002/adma.201405583 |
| [35] |
Q. Yang, Q. Xu, S.H. Yu, H.L. Jiang, Angew. Chem. Int. Ed. 55 (2016) 3685-3689. DOI:10.1002/anie.201510655 |
| [36] |
F. Wang, Y. Huang, Z. Chai, et al., Chem. Sci. 7 (2016) 6887-6893. DOI:10.1039/C6SC03239G |
| [37] |
X. Meng, T. Wang, L. Liu, et al., Angew. Chem. Int. Ed. 53 (2014) 11478-11482. DOI:10.1002/anie.201404953 |
| [38] |
W. Zhang, L. Wang, K. Wang, et al., Small 13 (2017) 1602583. DOI:10.1002/smll.201602583 |
| [39] |
G. Chen, R. Gao, Y. Zhao, et al., Adv. Mater. 30 (2018) 1704663. DOI:10.1002/adma.201704663 |
| [40] |
A.A. Jelle, K.K. Ghuman, P.G. O'Brien, et al., Adv. Energy Mater. 8 (2018) 1702277. DOI:10.1002/aenm.201702277 |
| [41] |
Q. Yang, C.C. Yang, C.H. Lin, H.L. Jiang, Angew. Chem. Int. Ed. 58 (2019) 3511-3515. DOI:10.1002/anie.201813494 |
| [42] |
Q. Guo, S.G. Xia, X.B. Li, et al., Chem. Commun. 56 (2020) 7849-7852. DOI:10.1039/d0cc01091j |
| [43] |
M. Opanasenko, A. Dhakshinamoorthy, Y.K. Hwang, et al., ChemSusChem 6 (2013) 865-871. DOI:10.1002/cssc.201300032 |
| [44] |
J. Kim, S.N. Kim, H.G. Jang, G. Seo, W.S. Ahn, Appl. Catal. A 453 (2013) 175-180. |
| [45] |
R. Liang, S. Luo, F. Jing, et al., Appl. Catal. B 176-177 (2015) 240-248. DOI:10.1016/j.apcatb.2015.04.009 |
| [46] |
K.G.M. Laurier, F. Vermoortele, R. Ameloot, et al., J. Am. Chem. Soc. 135 (2013) 14488-14491. DOI:10.1021/ja405086e |
| [47] |
A. Dhakshinamoorthy, A.M. Asiri, H. García, Angew. Chem. Int. Ed. 55 (2016) 5414-5445. DOI:10.1002/anie.201505581 |
| [48] |
L. Zhu, M. Gao, C.K.N. Peh, G.W. Ho, Mater. Horiz. 5 (2018) 323-343. DOI:10.1039/C7MH01064H |
| [49] |
Y. Sun, Y. Yin, B.T. Mayers, T. Herricks, Y. Xia, Chem. Mater. 14 (2002) 4736-4745. DOI:10.1021/cm020587b |
| [50] |
K.L. Kelly, E. Coronado, L.L. Zhao, G.C. Schatz, J. Phys. Chem. B 107 (2003) 668-677. |
| [51] |
X. Meng, L. Liu, S. Ouyang, et al., Adv. Mater. 28 (2016) 6781-6803. DOI:10.1002/adma.201600305 |
| [52] |
H.L. Jiang, T. Akita, T. Ishida, M. Haruta, Q. Xu, J. Am. Chem. Soc. 133 (2011) 1304-1306. DOI:10.1021/ja1099006 |
| [53] |
P.L. Llewellyn, S. Bourrelly, C. Serre, et al., Langmuir 24 (2008) 7245-7250. DOI:10.1021/la800227x |
 2021, Vol. 32
2021, Vol. 32