b Fujian Provincial Key Laboratory of Polymer Materials, College of Chemistry and Materials Science, Fujian Normal University, Fuzhou 350007, China
The design and synthesis of discrete metal-organic coordination capsules with specific geometries and cavities have been investigated extensively [1-5]. The interest in these materials stems not only from their remarkable structural characteristics but also from their promising applications in supramolecular chemistry and material science [6-9]. Most coordination capsules are one-pot synthesized through the self-assembly of metallic building blocks and organic ligands, generally accompanied by the annoyingly irrational and uncontrollable growth of polymetallic building vertices [10, 11]. Recent investigations on supramolecular transformations of coordination cage open the new door for the controllable synthesis of cage or capsule compounds [12, 13]. Various stimuli, including solvents, guests, light, post-modification reactions and so on, have been employed to trigger the transformation processes, which allow for the formation of new structures with specific properties and functions, as well as help elucidate stimuli-responsive structural reorganization processes in biological systems [14-18].
On the other hand, titanium-oxo coordination compounds have received significant attention in recent years due to their well-defined structure and numerous photocatalytic applications [19-26]. In contrast to extensively studied Ti-oxo clusters (TOC) and Ti-oxo MOFs, only two metal–organic coordination quasi-capsules with negligible small cavities have been constructed by polynuclear Ti–O building units [27, 28], though few Ti–ligand cages using single Ti ion vertices were reported [29-32]. One key process to form such titanium supramolecular architectures is the formation of suitable titanium-oxo building blocks providing ligand-accessible coordination sites to be assembled into the desired molecules, which is unfortunately hampered by high charge density and the easy hydrolysis attributes of titanium ions. Recently, several Ti-carboxylate MOFs have been post-synthesized through supramolecular transformations [33, 34]. Such transformation method rules out the disadvantage of easy hydrolysis of titanium ions, inspiring us to adapt similar strategies to synthesize titanium-oxo coordination capsules with new structures and specific properties. To the best of our knowledge, supramolecular transformation of titanium-oxo coordination capsule has not been reported to date.
Our previous work has demonstrated that phenol could be an effective solvent to synthesize highly stable TOCs through reprocessing the titanium-oxo clusters presynthesized in isopropanol [35]. During the process, the Ti-O cores containing 5-coordinated TiⅣ centers would undergo reconstruction to give new cores fully comprising 6-coordinated TiⅣ centers. Considering the easy reconstruction of titanium-oxo coordination compounds induced by phenol, we decided to utilize them to realize the supramolecular transformations of titanium–organic coordination capsules (Scheme 1). In this work, two titanium-oxo coordination compounds [Ti12O4(OPri)32(DBC)4] (PTC-74, HOPri = isopropanol, DBC = 1, 4-dicarboxybenzene) and [Ti18O6(HPO3)6(OPri)42(BPDC)3] (PTC-261, H3PO3 = phosphorous acid, BPDC = 4, 4'-biphenyldicarboxylic acid) both with 5-coordinated TiⅣ centers obtained in isopropanol were chosen as precursors to realize the supramolecular transformations induced by phenol. The reaction of coordination rectangle PTC-74 with H3PO3 in phenol obtained a coordination capsule of [Ti18O6(HPO3)6(DBC)3(PhO)42]⊃2PhOH (PTC-262, PhOH=phenol). Meanwhile, the quasi-capsule like titanium-oxo coordination compound PTC-261 could also be reprocessed in phenol to give another coordination capsule of [Ti18O6(HPO3)6(BPDC)3(PhO)42]⊃5PhOH (PTC-263) with much larger cavity, which owns the identical titanium oxo building blocks but different bridging ligands with that in PTC-262. Phenol guest molecules can be quantitative identified inside the post-synthesized capsules depending on the cavity dimension. In addition, they display much better visible light absorption and air stability than the isopropanol stabilized one.

|
Download:
|
| Scheme 1. Illustration of the phenol-triggered supramolecular transformation of titanium–organic coordination capsule. | |
The coordination rectangle PTC-74 was firstly synthesized in isopropanol according to a modified literature method reported by us [36]. It consists of four {Ti3}α units and four DBC ligands, which are alternately connected in a rectangular style (Fig. 1a). Importantly, each {Ti3}α unit in PTC-74 contains one 5-coordinated TiⅣ-center. According to our previous investigations, such 5-coordinated TiⅣ-centers might undergo reconstruction during phenol-thermal modification [35], and H3PO3 might facilitate the assembly of titanium coordination capsules [27]. Indeed, after treatingPTC-74 with phenol in the presence of H3PO3, yellow hexagonal crystals of PTC-262 were discovered beside some previous reported red prism crystals of PTC-184 (Fig. S1 in Supporting information) [35]. Full structural analysis by X-ray diffraction indicates thatPTC-262 is composed of six {Ti3}β building units, six phosphite ligands and three terephthalic acid ligands. Every three building units are alternately linked with three phosphites to form a Ti9P3 entity, and then such two entities are further combined together with three terephthalic acid ligands to form the molecular capsule (Fig. 1b). The structure of the {Ti3}β unit in PTC-262 is different from its precursor unit in PTC-74, even though they share the same composition {Ti3O13}. The {Ti3}α unit in PTC-74 comprises one μ3-O and two μ2-O atoms which are nearly coplanar with the three Ti atoms (Fig. 1c), while the {Ti3}β unit in PTC-262 contains one μ3-O and three μ2-O atoms which are located above and below the Ti-Ti-Ti plane, respectively (Fig. 1d). Such additional μ2-O atom in PTC-262 makes its Ti atoms all 6-coordinated. Alternatively, PTC-262 can be formed with pure phase and higher yield in the reaction of phenol with the dendritic crystalline precipitate produced by directly mixing of DBC, H3PO3 and Ti(OPri)4 in isopropanol at 80 ℃, though the precipitate is unsuitable for single crystal X-ray diffraction. It is worth noting that the one-pot synthesis of PTC-262 via the reaction of Ti(OPri)4 with DBC and H3PO3 in phenol failed, highlighting the advantages of post-synthesis in controllable synthesis of coordination capsules or cages.

|
Download:
|
| Fig. 1. Structures of (a) PTC-74, (b) PTC-262, (e) PTC-261 and (f) PTC-263. Illustration of (c) the {Ti3} units in PTC-74 and PTC-261, (d) the {Ti3} units in PTC-262 and PTC-263. Atom color codes: pink P; violet 5 coordinated Ti; green 6 coordinated Ti; red O; gray C. H atoms have been omitted for clarity. | |
Changing the length of the bridging ligand is an effective way to regulate the size of the cavity in capsule. We intended to synthesize the larger coordination rectangle by using BPDC instead of DBC, which might further transform into the larger capsule. Directly reaction of BPDC with Ti(OPri)4 in isopropanol at 80 ℃ results no coordination rectangle but white amorphous precipitate. However, colorless hexagonal prismatic crystals of PTC-261 could be obtained with high yield when H3PO3 was further added into the synthetic reaction system. Six {Ti3}α subunits, which are identical with the subunits in PTC-74, were assembled into the {Ti18} coordination capsule of PTC-261 by H3PO3 and BPDC in isopropanol (Fig. 1e). The structure of quasi-capsule like PTC-261 is similar to the previously reported sulfonates stabilized capsules (Fig. S3 in Supporting information) [27]. However, each {Ti3}α unit in PTC-261 contains one 5-coordinated TiⅣ-center (Fig. 1c), since carboxylic acid has one less coordinated oxygen atom than sulfonic acid. Such 5-coordinated TiⅣ-centers in PTC-261 terminated with isopropanol provide us with the fine opportunity to realize the supramolecular transformation triggered by phenol once again.
Consequently, PTC-261 was also reprocessed in phenol, then a new capsule of PTC-263 was discovered. Single-crystal X-ray diffraction analysis reveals that PTC-263, constructed from six {Ti3}β units, six phosphorous acid ligands and three BPDC ligands, possesses analogous structure toPTC-262 (Fig. 1f). The longer bridging BPDC ligands in PTC-263 make it just like an "extended version" of PTC-262. The number of phenol ligands coordinated to PTC-263 is identical to the number of isopropanol ligands in precursor PTC-261. As mentioned above, the {Ti3}β unit in PTC-263 has the same composition {Ti3O13} but different structure with the {Ti3}α unit in PTC-261 (Fig. S4 in Supporting information). Additionally, carboxylate ligands in PTC-263 are located further away from the central axis and two sides of this capsule are more curved (Fig. S5 in Supporting information). If propyl groups in isopropanol and benzene groups in terminal phenol are eliminated, PTC-261 and PTC-263 have the same components but different geometric configurations (Fig. S6 in Supporting information).
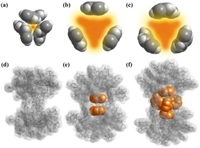
The cavities in molecular capsules can provide unique chemical environments, serving as supramolecular containers, depending on their dimension and composition. Therefore, the host–guest systems of the three synthesized coordination capsules are different. The cavity of PTC-261 is too small to hold any guest molecules for the reason that the three bridge BPDC ligands in the capsule are entangled with each other (Figs. 2a and d). However, carboxylate ligands in PTC-262 and PTC-263 are located further away from the central axis than that in PTC-261, offering the two capsules much larger cavity than that in PTC-261 (Figs. 2b and c). As a result, two phenolic molecules are located horizontally at both sides of the cavity in PTC-262 (Fig. 2e). Meanwhile, apart from two side located molecules, three other phenolic molecules are vertically situated on the waistline of the largest capsule PTC-263 (Fig. 2f). Consequently, five phenolic guest molecules are fixed in the cavity in PTC-263, presenting a triangular bipyramid configuration (Fig. 2f). These three capsules successfully show us the case of accurately controlling the quantity of guest molecules at molecular level depending on the size of the cavities.
|
Download:
|
| Fig. 2. The top view of the cavities in (a) PTC-261, (b) PTC-262 and (c) PTC-263, highlighting in orange area. Illustration of the host–guest systems of in (d) PTC-261, (e) PTC-262 and (f) PTC-263. | |
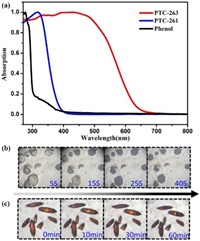
Since titanium-oxo coordination capsules PTC-261 and PTC-263 have the same components but different geometric configurations if propyl groups in isopropanol and benzene groups in terminal phenol are eliminated, the differences in their light absorption and stability properties were also studied. The phenol-triggered capsule PTC-263 represents orange color while its precursor PTC-261 terminated with isopropanol is colorless. The solid-state UV–vis absorption studies were further applied to evaluate their optical properties. Both isopropanol stabilized PTC-261 and phenol (solid state at room temperature) present no obvious absorption in the visible light range (Fig. 3a). However, PTC-263 which was transformed from PTC-261 triggered by phenol can significantly red-shift the absorption edge to ~650 nm (Fig. 3a). Therefore, the incorporation of phenol with Ti-O core could significant narrow its bandgap, which should attribute to the penetration of highest occupied phenol levels into the bandgap of the Ti-O core [37]. In addition, the crystals of PTC-263 could survive in the air for half an hour, while its precursor crystals were weathered and losing crystallinity as soon as they were taken out from isopropanol (Figs. 3b and c and Fig. S11 in Supporting information). As we know, isopropanol is much more volatile than phenol, indicating that the volatility of solvent has a great influence on the crystalline stability of discrete coordination capsules. Therefore, phenolic titanium-oxo coordination capsules display much better visible light absorption and air stability than the isopropanol stabilized ones with similar cores.
|
Download:
|
| Fig. 3. (a) The solid-state absorption spectra of phenol, PTC-263 and its precursor PTC-261. The time dependent crystal photos of (b) PTC-261 and (c) PTC-263 in the air. | |
In summary, we have successfully realized the supramolecular transformation of titanium–organic coordination capsules in the presence of phenol (and H3PO3), which is accompanied by the reconstruction of 5-coordinated Ti centers to 6-coordinated ones. Meanwhile, different amounts of encapsulated phenol guest molecules can be identified, dependent on the sizes of the obtained cavities. In addition, they display much better visible light absorption and air stability than the isopropanol stabilized ones. Therefore, the developed strategy provides an innovative approach for structural modulation of titanium–organic coordination capsules, which might also be applied to other supramolecular systems.
Declaration of competing interestThe authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
AcknowledgmentsThis work is supported by the National Natural Science Foundation of China (NSFC, No. 21922111), the Strategic Priority Research Program of the Chinese Academy of Sciences (No. XDB20000000).
Appendix A. Supplementary dataSupplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cclet.2021.01.010.
| [1] |
M. Fujita, M. Tominaga, A. Hori, B. Therrien, Acc. Chem. Res. 38 (2005) 369-378. DOI:10.1021/ar040153h |
| [2] |
T.R. Cook, P.J. Stang, Chem. Rev. 115 (2015) 7001-7045. DOI:10.1021/cr5005666 |
| [3] |
D.A. Roberts, B.S. Pilgrim, J.R. Nitschke, Chem. Soc. Rev. 47 (2018) 626-644. DOI:10.3390/w10050626 |
| [4] |
K. Byrne, M. Zubair, N. Zhu, et al., Nat. Commun. 8 (2017) 15268. DOI:10.1038/ncomms15268 |
| [5] |
J. Jiao, C. Tan, Z. Li, et al., J. Am. Chem. Soc. 140 (2018) 2251-2259. DOI:10.1021/jacs.7b11679 |
| [6] |
T.S. Koblenz, J. Wassenaar, J.N. Reek, Chem. Soc. Rev. 37 (2008) 247-262. DOI:10.1039/B614961H |
| [7] |
M. Liu, W. Liao, C. Hu, S. Du, H. Zhang, Angew. Chem. Int. Ed. 51 (2012) 1585-1588. DOI:10.1002/anie.201106732 |
| [8] |
Y.T. Zhang, X.L. Wang, S.B. Li, et al., Chem. Commun. 52 (2016) 9632-9635. DOI:10.1039/C6CC04583A |
| [9] |
W.H. Fang, L. Zhang, J. Zhang, G.Y. Yang, Chem. Commun. 52 (2016) 1455-1457. DOI:10.1039/C5CC08702C |
| [10] |
J.M. Breen, W. Schmitt, Angew. Chem. Int. Ed. 47 (2008) 6904-6908. DOI:10.1002/anie.200801770 |
| [11] |
S.T. Zheng, J. Zhang, G.Y. Yang, J. Am. Chem. Soc. 132 (2010) 15102-15103. DOI:10.1021/ja105986b |
| [12] |
A.J. McConnell, C.S. Wood, P.P. Neelakandan, J.R. Nitschke, Chem. Rev. 115 (2015) 7729-7793. DOI:10.1021/cr500632f |
| [13] |
W. Wang, Y.X. Wang, H.B. Yang, Chem. Soc. Rev. 45 (2016) 2656-2693. DOI:10.1039/C5CS00301F |
| [14] |
Y. Lu, H.N. Zhang, G.X. Jin, Acc. Chem. Res. 51 (2018) 2148-2158. DOI:10.1021/acs.accounts.8b00220 |
| [15] |
I.A. Riddell, M.M.J. Smulders, J.K. Clegg, et al., Nat. Chem. 4 (2012) 860. DOI:10.1038/nchem.1456 |
| [16] |
J. Park, L.B. Sun, Y.P. Chen, Z. Perry, H.C. Zhou, Angew. Chem. Int. Ed. 53 (2014) 5842-5846. DOI:10.1002/anie.201310211 |
| [17] |
T. Murase, S. Sato, M. Fujita, Angew. Chem. Int. Ed. 46 (2007) 5133-5136. DOI:10.1002/anie.200700793 |
| [18] |
K. Su, M. Wu, D. Yuan, M. Hong, Nat. Commun. 9 (2018) 4941. DOI:10.1038/s41467-018-07427-z |
| [19] |
P. Coppens, Y. Chen, E. Trzop, Chem. Rev. 114 (2014) 9645-9661. DOI:10.1021/cr400724e |
| [20] |
W.H. Fang, L. Zhang, J. Zhang, Chem. Soc. Rev. 47 (2018) 404-421. DOI:10.1039/C7CS00511C |
| [21] |
L. Rozes, C. Sanchez, Chem. Soc. Rev. 40 (2011) 1006-1030. DOI:10.1039/c0cs00137f |
| [22] |
P.D. Matthews, T.C. King, D.S. Wright, Chem. Commun. 50 (2014) 12815-12823. DOI:10.1039/C4CC04421E |
| [23] |
X. Fan, J. Wang, K. Wu, L. Zhang, J. Zhang, Angew. Chem. Int. Ed. 58 (2019) 1320-1323. DOI:10.1002/anie.201809961 |
| [24] |
S. Yuan, J.S. Qin, C.T. Lollar, H.C. Zhou, ACS Cent. Sci. 4 (2018) 440-450. DOI:10.1021/acscentsci.8b00073 |
| [25] |
M. Dan-Hardi, C. Serre, T. Frot, et al., J. Am. Chem. Soc. 131 (2009) 10857-10859. DOI:10.1021/ja903726m |
| [26] |
N.M. Padial, J. Castells-Gil, N. Almora-Barrios, et al., J. Am. Chem. Soc. 141 (2019) 13124-13133. DOI:10.1021/jacs.9b04915 |
| [27] |
X. Fan, N. Narayanam, M. Gao, L. Zhang, J. Zhang, Dalton Trans. 47 (2018) 663-665. |
| [28] |
Y. Huang, G.D. Zou, H.M. Li, Y. Cui, Y. Fan, New J. Chem. 42 (2018) 14079-14082. DOI:10.1039/C8NJ02992J |
| [29] |
C. Breckner, R.E. Powers, K.N. Raymond, Angew. Chem. Int. Ed. 37 (1998) 1837-1839. DOI:10.1002/(SICI)1521-3773(19980803)37:13/14<1837::AID-ANIE1837>3.0.CO;2-A |
| [30] |
A.V. Davis, K.N. Raymond, J. Am. Chem. Soc. 127 (2005) 7912-7919. DOI:10.1021/ja051037s |
| [31] |
Y.P. He, L.B. Yuan, G.H. Chen, et al., J. Am. Chem. Soc. 139 (2017) 16845-16851. DOI:10.1021/jacs.7b09463 |
| [32] |
B.C. Zhu, W.H. Fang, J. Wang, et al., Chem. Eur. J. 24 (2018) 14358-14362. DOI:10.1002/chem.201802930 |
| [33] |
Y. Keum, S. Park, Y.P. Chen, J. Park, Angew. Chem. Int. Ed. 57 (2018) 14852-14856. DOI:10.1002/anie.201809762 |
| [34] |
P. Ji, Y. Song, T. Drake, et al., J. Am. Chem. Soc. 140 (2018) 433-440. DOI:10.1021/jacs.7b11241 |
| [35] |
X. Fan, H. Fu, M.Y. Gao, L. Zhang, J. Zhang, Inorg. Chem. 58 (2019) 13353-13359. DOI:10.1021/acs.inorgchem.9b02238 |
| [36] |
M.Y. Gao, X. Fan, L. Zhang, J. Zhang, Inorg. Chem. 57 (2018) 5642-5647. DOI:10.1021/acs.inorgchem.8b00586 |
| [37] |
J.B. Benedict, P. Coppens, J. Am. Chem. Soc. 132 (2010) 2938-2944. DOI:10.1021/ja909600w |
 2021, Vol. 32
2021, Vol. 32 

